Retatrutide vs Orforglipron: Which New GLP‑1 Is Better for Weight Loss?
Last updated: March 4, 2026 | Evidence last verified: March 2026
Written by the WPG Research Team | Editorial standards | Affiliate disclosure
Affiliate Disclosure: Weight Loss Provider Guide may earn a commission if you sign up through links on this page at no extra cost to you. This doesn't affect our ratings, our editorial process, or what we recommend. Full editorial policy.
Last Updated: March 4, 2026 · Sources: PubMed/NEJM, Eli Lilly, ClinicalTrials.gov, FDA.gov · Medical Disclaimer at bottom of page
Retatrutide vs orforglipron — here's what you actually need to know. Both drugs are from Eli Lilly. Both are next-generation weight loss medications. Neither is available yet. But the differences between them are significant, and what you decide to do today matters more than which one wins on paper.
Retatrutide is a once-weekly injection that targets three hormone receptors simultaneously (GLP-1, GIP, and glucagon). In its first Phase 3 trial (TRIUMPH-4, December 2025), participants lost an average of 28.7% of their body weight — roughly 71 lbs — at the highest dose over 68 weeks. That rivals bariatric surgery results. It likely won't be available until 2028.
Orforglipron is a once-daily pill that targets one receptor (GLP-1 only). In its Phase 3 obesity trial (ATTAIN-1, published in the New England Journal of Medicine, September 2025), the highest dose produced 12.4% average weight loss (~27.3 lbs) over 72 weeks using the efficacy estimand (the more conservative treatment-regimen analysis showed 11.2% / ~25 lbs). Less dramatic than retatrutide, but still clinically meaningful. And it could reach the market by mid-2026, after Lilly submitted its FDA application with a National Priority Voucher fast-tracking the review.
So: retatrutide is the power play. Orforglipron is the convenience play. One is a weekly shot that could produce the most weight loss we've ever seen from a medication. The other is a daily pill you can take any time, no needles, no food restrictions — and it's arriving first.
But here's what nobody else is telling you: if your health is being affected by your weight right now, waiting years for either drug while effective treatment exists today is a decision with real consequences. Current GLP-1 medications (semaglutide, tirzepatide) are already helping millions of people lose 15–22% of their body weight. We'll help you figure out whether waiting, starting now, or joining a clinical trial makes the most sense — based on your actual situation.
Below, we break down every detail: mechanism, clinical data, side effects, timelines, expected pricing, and what to do next.

Quick Comparison: Retatrutide vs Orforglipron at a Glance

| Retatrutide | Orforglipron | |
|---|---|---|
| Drug type | Triple agonist (GLP-1 + GIP + Glucagon) | GLP-1 receptor agonist (single target) |
| How you take it | Once-weekly subcutaneous injection | Once-daily oral pill |
| Phase 3 weight loss | 28.7% at 12 mg over 68 weeks (TRIUMPH-4)† | 12.4% at 36 mg over 72 weeks (ATTAIN-1)† |
| Average pounds lost | ~71.2 lbs | ~27.3 lbs |
| ≥25% weight loss achieved | 58.6% of participants (12 mg) | Not reported at this threshold |
| ≥15% weight loss achieved | Not yet reported for TRIUMPH-4 | 36.0% of participants (36 mg) |
| Developer | Eli Lilly | Eli Lilly (licensed from Chugai Pharmaceutical) |
| Phase | Phase 3 (7 more readouts in 2026) | NDA submitted to FDA (late 2025) |
| Expected FDA approval | ~2027–2028 | Q2 2026 (National Priority Voucher) |
| Expected self-pay cost | Unknown (est. $350–$1,000+/mo based on comparable drugs) | $149–$399/mo (announced by Lilly via LillyDirect) |
| Food/water restrictions | N/A (injection) | None — take any time of day |
| Key advantage | Strongest weight loss of any drug ever tested | First oral non-peptide GLP-1 for obesity |
| Key limitation | Requires injection; furthest from market | Lower efficacy than injectable alternatives |
| Available now? | ❌ Clinical trials only | ❌ Awaiting FDA decision |
Evidence confidence: Retatrutide Phase 3 data comes from a single trial (TRIUMPH-4) in a knee osteoarthritis subpopulation. The broader obesity trial (TRIUMPH-1) has not yet reported. Orforglipron Phase 3 data is published in a peer-reviewed journal (NEJM). Both datasets come from Eli Lilly–sponsored trials.
†Efficacy estimand (assumes participants stayed on treatment). The more conservative treatment-regimen estimand (includes all participants regardless of adherence) showed: retatrutide 12 mg: 23.7% / ~60.0 lbs; orforglipron 36 mg: 11.2% / ~25.0 lbs. We use the efficacy estimand in this table because both companies lead with it in their press releases, and it better represents what happens when you stay on the medication.
Sources: Eli Lilly TRIUMPH-4 press release (Dec 11, 2025); Wharton et al., NEJM 2025 (ATTAIN-1); Eli Lilly ATTAIN-1 press release (Sep 16, 2025).
Neither drug is available yet. If you're ready to explore GLP-1 treatment now, see current provider options → or take our provider match quiz →
Why Are People Comparing Retatrutide and Orforglipron?
These two drugs keep showing up together because they represent Eli Lilly's two biggest bets on the future of weight loss treatment — and they couldn't be more different.
Eli Lilly already dominates the GLP-1 market with tirzepatide (sold as Mounjaro for type 2 diabetes and Zepbound for weight loss). Now they're developing two next-generation drugs that attack the problem from opposite angles:
Retatrutide is the brute-force approach. It hits three hormone receptors at once — more than any obesity drug ever tested. Early results suggest it could produce the largest average weight loss of any medication in history. It's a weekly injection, and it's still years from pharmacies.
Orforglipron is the accessibility play. It's a daily pill with no food or water restrictions — you take it whenever you want. It won't produce the jaw-dropping weight loss numbers of the injectables, but it removes the two biggest barriers to treatment: needles and complicated dosing schedules.
The timing is driving the interest. In December 2025, retatrutide posted its first Phase 3 results showing 28.7% body weight reduction — numbers that shook the entire obesity treatment world. Around the same time, Lilly submitted orforglipron to the FDA with an expedited review voucher, potentially putting it on pharmacy shelves by mid-2026. Clarivate's Drugs to Watch 2026 report projects $16 billion in orforglipron sales and $30 billion in retatrutide sales by 2031.
People searching “retatrutide vs orforglipron” aren't doing academic research. They're asking a very human question: Which one matters to me, and should I wait for either one — or start treatment now?
We'll answer all of it.
What Exactly Is Retatrutide?
Retatrutide (development code: LY3437943) is an investigational once-weekly injectable medication from Eli Lilly. It's the first “triple agonist” — meaning it activates three different hormone receptors simultaneously:
- GLP-1 receptor — slows stomach emptying, reduces appetite, improves insulin response. This is the same target that semaglutide (Ozempic, Wegovy) hits.
- GIP receptor — enhances insulin sensitivity and glucose-dependent insulin release. This is the second target in tirzepatide (Mounjaro, Zepbound).
- Glucagon receptor — and this is the new one. Glucagon activation increases energy expenditure and promotes fat oxidation. In plain English: it tells your body to burn more stored fat for energy, not just eat less.
Think of it this way. Semaglutide has one engine. Tirzepatide has two. Retatrutide has three. That third engine — glucagon — appears to be why the weight loss numbers are so much higher. Your body isn't just eating less; it's actively burning more.
Retatrutide is administered as a subcutaneous injection once per week. In clinical trials, participants started at 2 mg and gradually increased their dose every four weeks until reaching 9 mg or 12 mg.
Names you'll see online: Retatrutide, LY3437943, “triple agonist,” “tri-agonist,” “Lilly triple G.”
What it's being studied for: Obesity and overweight, type 2 diabetes, knee osteoarthritis, obstructive sleep apnea, chronic low back pain, cardiovascular and kidney outcomes, and metabolic liver disease (MASLD/MASH). The full Phase 3 program is called TRIUMPH. Learn more in our complete retatrutide guide.
What Exactly Is Orforglipron?
Orforglipron (development code: LY3502970) is an investigational once-daily oral pill, also from Eli Lilly (originally discovered by Chugai Pharmaceutical in Japan and licensed by Lilly in 2018).
It works as a GLP-1 receptor agonist — the same class as semaglutide — but with a crucial structural difference: it's a non-peptide, small-molecule drug. That's a mouthful, but it matters a lot practically.
Here's why. The existing oral GLP-1 on the market, Rybelsus (and the newly approved Wegovy pill), are peptide-based semaglutide pills. Peptides are fragile. Your stomach acid wants to destroy them before they can work. So those pills require strict rules: take on an empty stomach with no more than 4 oz of water, then wait 30 minutes before eating, drinking, or taking other medications. According to the FDA-approved Rybelsus label, failing to follow these instructions reduces absorption.
Orforglipron sidesteps this entirely. It's a small molecule, not a peptide. It survives digestion naturally. According to its trial protocol and Eli Lilly, orforglipron can be taken any time of day, with or without food, with no water restrictions. That's a significant convenience upgrade.
It targets only the GLP-1 receptor — not GIP, not glucagon. That's why its weight loss numbers are lower than the dual and triple agonists. But for millions of people who won't consider an injection, a daily pill they can take like any other medication is exactly what they need to actually start and stick with treatment.
Names you'll see online: Orforglipron, LY3502970, “GLP-1 pill,” “oral GLP-1,” “Lilly weight loss pill.”
What it's being studied for: Obesity and overweight, type 2 diabetes, obstructive sleep apnea, knee osteoarthritis pain, stress urinary incontinence, and hypertension.
How Do They Work — Triple Agonist vs GLP-1 Only?
To understand the difference, it helps to know the basic biology. Don't worry — we'll keep it practical.
When you eat, your gut releases hormones that help regulate appetite, blood sugar, and metabolism. Three of the most important are GLP-1, GIP, and glucagon. Think of them as three different control knobs:
GLP-1 (glucagon-like peptide-1): This hormone slows how fast food moves through your stomach, makes you feel full sooner, and helps your pancreas release insulin when blood sugar rises. When scientists create drugs that mimic GLP-1 (like semaglutide), people eat less because their brain receives stronger “I'm full” signals. This is also why nausea is the most common side effect — the same mechanism that reduces appetite can upset your stomach.
GIP (glucose-dependent insulinotropic polypeptide): This hormone enhances insulin sensitivity and supports glucose regulation. Adding GIP activation to GLP-1 (which is what tirzepatide does) appears to boost weight loss beyond GLP-1 alone and may improve how the body processes fat.
Glucagon: This is the interesting one. Glucagon tells your liver to release stored glucose and ramps up energy expenditure — essentially shifting your metabolism toward burning stored fat. It's been harder to harness therapeutically because too much glucagon can raise blood sugar. Retatrutide appears to have found a balance that keeps the metabolic benefits while the GLP-1 and GIP components manage blood sugar.
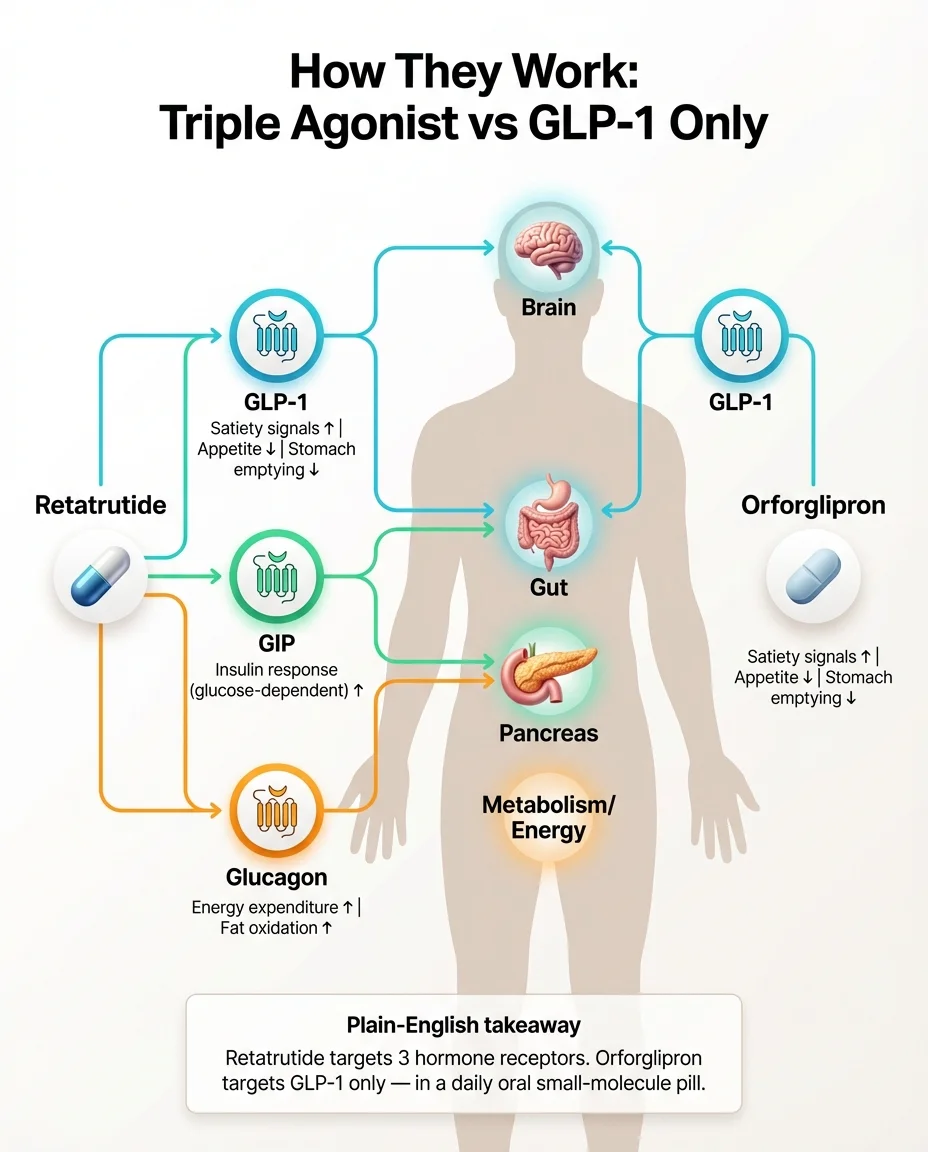
Retatrutide's three-receptor approach
Retatrutide activates all three simultaneously. The result: appetite suppression (GLP-1), improved insulin response (GIP), and increased energy expenditure and fat burning (glucagon). This “triple agonist” approach is likely why Phase 3 results are showing weight loss numbers that exceed what any single- or dual-agonist has achieved.
Orforglipron's single-receptor approach
Orforglipron activates only GLP-1 receptors. Mechanistically, it's in the same class as semaglutide — appetite reduction and slower gastric emptying. The innovation isn't in a new mechanism. It's in the delivery: a small-molecule pill that doesn't require the elaborate stomach-protection chemistry that oral semaglutide needs.
What this means practically
More receptor targets generally means more weight loss — but also more potential for side effects. Retatrutide's triple mechanism is why it's producing ~28% weight loss and why its discontinuation rates are higher. Orforglipron's single mechanism is why its weight loss is more moderate (~11%) but its side effect profile may be more manageable. Neither approach is objectively “better.” They're solving different problems for different people.
Which Produces More Weight Loss — Retatrutide or Orforglipron?
Let's look at the actual numbers. This is the section most people came here for, so we'll be direct — then honest about what the data can and can't tell us.
The headline numbers
Retatrutide (TRIUMPH-4, Phase 3, December 2025):
- Participants: 445 adults with obesity/overweight and knee osteoarthritis (no diabetes)
- Duration: 68 weeks
- Average baseline weight: 248.5 lbs (BMI 40.4)
- Weight loss at 12 mg: 28.7% of body weight (average 71.2 lbs lost)
- Weight loss at 9 mg: 26.4% of body weight (average 64.2 lbs lost)
- Placebo: 2.1%
- 58.6% of participants on the highest dose lost ≥25% of their body weight
- Nearly half exceeded 25%, and a meaningful subset surpassed 30% and 35%
Orforglipron (ATTAIN-1, Phase 3, NEJM September 2025):
- Participants: 3,127 adults with obesity/overweight (no diabetes)
- Duration: 72 weeks
- Average baseline BMI: 37.0
- Weight loss at 36 mg (efficacy estimand): 12.4% (~27.3 lbs lost)
- Weight loss at 36 mg (treatment-regimen estimand, primary endpoint): 11.2% (~25.0 lbs lost)
- Weight loss at 12 mg: 8.4% (treatment-regimen)
- Weight loss at 6 mg: 7.5% (treatment-regimen)
- Placebo: 2.1%
- At the highest dose (efficacy estimand): 59.6% lost ≥10%, 39.6% lost ≥15%, 22.0% lost ≥20%
What these numbers mean in real terms
For someone starting at 250 lbs:
- Retatrutide (28.7%): ~72 lbs lost → ending around 178 lbs
- Orforglipron (12.4%): ~31 lbs lost → ending around 219 lbs
That's a massive difference. But before you draw conclusions, there's something important we need to be upfront about.
The honesty check (why direct comparison is tricky)
These results come from different trials with different populations, different durations, and different study designs. No trial has ever tested retatrutide directly against orforglipron in the same patients under the same conditions. Key differences:
- TRIUMPH-4 enrolled people with both obesity and knee osteoarthritis — a specific subpopulation. ATTAIN-1 enrolled a broader obesity population.
- TRIUMPH-4 was 68 weeks; ATTAIN-1 was 72 weeks.
- TRIUMPH-4 had 445 participants; ATTAIN-1 had 3,127.
- TRIUMPH-4's baseline BMI was higher (40.4 vs 37.0). People with higher starting BMIs often lose a larger percentage.
- The primary TRIUMPH Phase 3 obesity trial (TRIUMPH-1) has not yet reported results. That trial runs for 80 weeks and includes a broader population. Results are expected in 2026.
We report both sets of numbers so you can see the landscape, but please treat these as separate data points, not a head-to-head result. A published network meta-analysis (PubMed, 2024) ranked retatrutide as producing the most weight loss among all GLP-1 and polyagonist therapies studied — but the authors noted the same cross-trial comparison limitations.
Beyond weight: blood sugar and cardiometabolic effects
Both drugs show meaningful benefits beyond the scale:
Orforglipron reduced blood pressure, triglycerides, non-HDL cholesterol, waist circumference, and inflammatory markers (hsCRP). Among participants with prediabetes, 91% achieved near-normal blood sugar levels compared to 42% on placebo. In the separate ACHIEVE-3 trial (published in The Lancet, February 2026), orforglipron demonstrated superior blood sugar control compared to oral semaglutide in people with type 2 diabetes.
Retatrutide improved cardiovascular risk markers in TRIUMPH-4, including non-HDL cholesterol, triglycerides, and hsCRP. It also dramatically reduced knee osteoarthritis pain — WOMAC pain scores dropped 75.8% (compared to 40.3% with placebo), and about 1 in 8 retatrutide-treated patients were completely free of knee pain by the end of the trial.
Side Effects: Retatrutide vs Orforglipron
Both drugs share the gastrointestinal side effect profile that's standard across GLP-1 medications. The difference is in degree — and in a few signals unique to each drug.

Common side effects (both drugs)
Nausea, diarrhea, vomiting, constipation, and decreased appetite. These occur most frequently during dose escalation (when the dose is being gradually increased) and tend to be mild to moderate. They usually improve as your body adjusts. This isn't unique to these drugs. Every GLP-1 medication — semaglutide, tirzepatide, liraglutide — causes these effects because the same mechanism that reduces appetite (slower gastric emptying, stronger satiety signals) can irritate the GI tract.
Orforglipron-specific findings
According to the ATTAIN-1 trial published in NEJM:
- Nausea: 28.9%–35.9% (vs 10.4% placebo)
- Constipation: 21.7%–29.8% (vs 9.3% placebo)
- Diarrhea: 21.0%–23.1% (vs 9.6% placebo)
- Vomiting: 13.0%–24.0% (vs 3.8% placebo)
- Treatment discontinuation due to adverse events: 5.3%–10.3% (vs 2.7% placebo)
- A small pulse rate increase was observed in the obesity trial, consistent with effects seen with other GLP-1 agonists
- In a separate diabetes head-to-head trial (ACHIEVE-3), the highest orforglipron dose showed more GI side effects and discontinuations compared to oral semaglutide, according to Reuters reporting
Retatrutide-specific findings
According to the TRIUMPH-4 press release from Eli Lilly:
- Nausea: 43% (vs placebo — exact rate not disclosed in topline data)
- Diarrhea: 33%
- Vomiting: 21%
- Treatment discontinuation due to adverse events: 12.2% (9 mg) and 18.2% (12 mg) vs 4.0% placebo
- Some discontinuations were specifically due to “perceived excessive weight loss” — meaning people were losing weight faster than they wanted to
New safety signal — dysesthesia: TRIUMPH-4 reported cases of dysesthesia, which is an abnormal sensation like tingling, numbness, or a “pins and needles” feeling. This was not reported in retatrutide's earlier Phase 2 trial. Eli Lilly stated that dysesthesia events did not appear to lead to treatment discontinuation. More data from the remaining seven Phase 3 trials will clarify whether this is a consistent concern or an artifact of this specific trial population.
Retatrutide's stronger results come with higher side effect rates
This is a genuine tradeoff. More receptor activation means more powerful effects — on both weight loss and side effects. The 18.2% discontinuation rate at the 12 mg dose is notably higher than what we see with most GLP-1 medications. The gradual dose titration schedule (starting at 2 mg and increasing every 4 weeks) is specifically designed to help manage this.
GLP-1 class safety warnings worth knowing
Retatrutide remains investigational. Orforglipron was FDA-approved on April 1, 2026 (sold as Foundayo), but its full real-world safety profile is still emerging. Based on approved GLP-1 medication labels (Wegovy, Zepbound, Foundayo), the following class-level warnings exist:
- Thyroid C-cell tumors: A boxed warning on semaglutide and tirzepatide labels notes that these drugs caused thyroid C-cell tumors in rodent studies. Whether this applies to humans is unknown. People with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN2) should not use these drugs.
- Pancreatitis: Cases have been reported with GLP-1 medications. Symptoms include severe abdominal pain.
- Gallbladder problems: Rapid weight loss from any cause increases gallstone risk.
Who should be particularly cautious
Without giving medical advice (talk to your clinician), the following groups warrant extra discussion with a healthcare provider before considering any GLP-1 medication:
- People who are pregnant, breastfeeding, or planning to conceive
- Those with a personal or family history of medullary thyroid cancer or MEN2
- People with a history of pancreatitis
- Those at risk for gallbladder disease
- People with concerns about muscle and lean mass loss (a topic gaining attention as very rapid weight loss becomes possible)
- Anyone currently taking medications that affect blood sugar (risk of hypoglycemia when combined)
What we still don't know about safety
Neither drug has long-term safety data beyond approximately 72 weeks. The real-world safety profile that emerges after FDA approval and widespread use will be more comprehensive than clinical trial data alone. This is true for every new medication, not just these two. For more on long-term considerations, see our guide on long-term effects of GLP-1 medications.
When Will Orforglipron Be Available? When Will Retatrutide Be Available?
This is the most practical question, and the timelines are very different.
Orforglipron: FDA-Approved (April 1, 2026)
Orforglipron is now commercially available as Foundayo. Here's the current status:
- FDA approval: Approved April 1, 2026 for chronic weight management under the brand name Foundayo.
- Availability: Available now through Ro, LillyDirect, GoodRx Care Direct, and Weight Watchers. Self-pay pricing starts at $149/month for the lowest dose.
- Lilly savings card: Eligible insured patients can pay as low as $25/month.
- Medicare Part D: Expected $50/month starting July 1, 2026.
- Key advantage: Once-daily oral tablet with no food or water restrictions — simpler dosing protocol than oral semaglutide.
- Diabetes indication: A separate submission for type 2 diabetes is planned for 2026.
Retatrutide: Likely 2027–2028
Retatrutide has a longer road:
- Phase 3 trials: TRIUMPH-4 (knee osteoarthritis) reported positive topline results in December 2025 — the first Phase 3 readout. But Lilly has stated that seven additional Phase 3 trials are expected to report throughout 2026, covering obesity (TRIUMPH-1, the big one), type 2 diabetes, sleep apnea, chronic low back pain, cardiovascular/kidney outcomes, and liver disease.
- FDA submission: Not yet announced. The NDA can't be submitted until the pivotal trials are complete and data is analyzed. Most likely timing: late 2026 or 2027.
- FDA decision expected: If submitted in late 2026/early 2027, a decision could come in 2027–2028. Clarivate's Drugs to Watch 2026 report projects a 2028 launch.
- The important trial to watch: TRIUMPH-1 is the primary obesity trial — 80 weeks long. Citi analysts have projected it could show more than 30% weight loss given the longer treatment duration. When this reports (expected 2026), we'll have a much clearer picture of retatrutide's full potential.
Timeline Summary
| Milestone | Orforglipron | Retatrutide |
|---|---|---|
| Phase 2 complete | ✅ 2023 | ✅ 2023 |
| Phase 3 started | ✅ 2023 | ✅ 2023 |
| First Phase 3 results | ✅ Aug 2025 (ATTAIN-1) | ✅ Dec 2025 (TRIUMPH-4) |
| Full Phase 3 program | ✅ Essentially complete | ⏳ 7 more trials reporting 2026 |
| FDA application submitted | ✅ Late 2025 (obesity) | ❌ Not yet |
| Expected FDA decision | 🔜 Q2 2026 | ⏳ 2027–2028 |
| Expected market launch | 🔜 Mid-to-late 2026 | ⏳ ~2028 |
Sources: Reuters (March 2, 2026); Clarivate Drugs to Watch 2026 via PharmExec; Eli Lilly investor press releases; FDA CNPV program.
How Much Will They Cost?
Pricing is one of the biggest practical concerns, and the picture is clearer for orforglipron than retatrutide.
Orforglipron: $149–$399/month (announced)
Eli Lilly has publicly committed to the following pricing through LillyDirect (their direct-to-patient pharmacy):
- Self-pay patients with obesity: Starting at $149/month for the lowest dose, up to $399/month for higher doses
- Medicare beneficiaries: $50/month starting April 2026, per Lilly's agreement with the U.S. government
- Commercial insurance: Coverage and copay amounts will depend on individual plans, PBM (pharmacy benefit manager) negotiations, and formulary placement — none of which are known yet
For context, the oral Wegovy pill (approved December 2025) launched at $149/month for the starting dose as a cash-pay option. Orforglipron's pricing appears designed to match or beat that benchmark.
Retatrutide: Unknown (but we can estimate)
No pricing has been announced, and it won't be until closer to launch (~2028). But we can look at comparable injectable GLP-1 medications for context:
- Zepbound (tirzepatide) via LillyDirect vials: $299/month (2.5 mg starting dose), $399/month (5 mg), $449/month (all higher doses) — prices reduced December 2025
- Zepbound list price: ~$1,000/month
- Wegovy (semaglutide) injection list price: $1,349.02/month
- Wegovy via NovoCare pharmacy: $349/month (self-pay, after intro offer)
Given that retatrutide is expected to be Lilly's premium offering — a first-in-class triple agonist — pricing will likely be at or above tirzepatide levels. Clarivate projects $30 billion in retatrutide revenue by 2031, which assumes premium pricing.
What you can access right now (for comparison)
If cost is a major factor in your decision, here's what's currently available:
- Compounded semaglutide through telehealth providers: Typically $149–$299/month (availability may change based on FDA shortage status)
- Zepbound vials via LillyDirect: $299–$449/month (as of Dec 2025)
- Wegovy injection via NovoCare: $199/month intro for first two fills, then $349/month (offer terms apply)
- With qualifying insurance: Copays vary widely, often $25–$150/month
We track current pricing across verified providers. See our updated cost comparison →
How Are They Taken — Daily Pill vs Weekly Injection?
The delivery method difference sounds simple, but it shapes the entire treatment experience.
Orforglipron: daily pill, zero restrictions
In the ATTAIN-1 trial, orforglipron was taken once daily. Participants started at a low dose and escalated every four weeks:
- Started at 1 mg daily
- Titrated up every 4 weeks: 1 mg → 3 mg → 6 mg → 12 mg → up to 36 mg (depending on assigned dose group)
- Participants reached 6 mg at 8 weeks, 12 mg at 12 weeks, and 36 mg at 24 weeks
- No food, liquid, or timing restrictions whatsoever
That last point is the standout. Unlike Rybelsus (oral semaglutide), which the FDA label requires to be taken on an empty stomach with no more than 4 ounces of water followed by a 30-minute fast, orforglipron works like a regular daily pill. You take it whenever it fits your routine. For a detailed comparison of oral vs injectable GLP-1 options, see our dedicated guide.
Retatrutide: once-weekly injection
Retatrutide is administered as a subcutaneous injection (under the skin, typically in the abdomen, thigh, or upper arm) once per week. In TRIUMPH-4:
- Started at 2 mg once weekly
- Escalated by 2–3 mg every four weeks
- Target doses: 9 mg or 12 mg weekly
This is the same general approach as Wegovy and Zepbound — a weekly shot using a pen device. People familiar with those medications will recognize the routine.
Real-life tradeoffs to consider
| Factor | Orforglipron (Daily Pill) | Retatrutide (Weekly Injection) |
|---|---|---|
| Needle required | No | Yes |
| Dosing frequency | Every day | Once a week |
| Missed dose impact | Miss one day = less disruptive | Miss one week = bigger gap |
| Travel/storage | Easy — room temp pill | Requires refrigeration, pen device |
| Habit formation | Adding to daily pill routine | Remembering a weekly event |
| Privacy | Looks like any pill | Injection requires some privacy |
| Adherence challenge | Daily consistency | Needle aversion |
For some people, a daily pill is effortless. For others, once a week and done is actually easier because there's less daily cognitive load. There's no universally “better” option here — it depends entirely on your preferences and lifestyle.
Should I Wait for Retatrutide or Orforglipron — or Start Treatment Now?
This is the question under the question. It's the real reason most people are comparing these drugs. And the answer depends on your situation.
We built a simple framework to help you think it through:
Starting GLP-1 treatment NOW probably makes sense if:
- Your weight is actively affecting your health today (blood sugar, blood pressure, joint pain, sleep apnea, mobility)
- Your doctor has recommended treatment
- You have type 2 diabetes that needs better management today
- You're open to injectable options (semaglutide or tirzepatide)
- Waiting 6 months to 2+ years carries meaningful health risk for you
Current GLP-1 medications already produce 15–22% weight loss on average. For most people, that's life-changing. And the ATTAIN-MAINTAIN trial showed that people can switch from injectables to orforglipron and maintain their results — so starting now doesn't lock you in forever.
Waiting for orforglipron (mid-2026) may make sense if:
- You refuse to consider injections, period
- You're in a lower BMI range where 10–12% weight loss would be sufficient
- Your health situation isn't urgent — you can safely wait 3–6 months
- You prefer the convenience of a no-restriction daily pill
- You want to be among the first on a new oral option
Waiting for retatrutide (~2028) may make sense if:
- You need maximum weight loss and have significant obesity (BMI 40+)
- You have obesity-related complications like severe knee osteoarthritis or sleep apnea
- You're comfortable with weekly injections
- You can wait 2+ years without your health deteriorating
- You're willing to monitor the remaining Phase 3 results as they come
Consider a clinical trial if:
- You want access to these drugs before they're commercially available
- You meet the enrollment criteria
- You understand trial participation involves placebo-controlled conditions
- You can visit a trial site regularly
A word of perspective
We understand the temptation to wait for “the best” option. But medications that produce 15–22% weight loss are available today, and they're already transforming lives. Waiting years for a drug that might produce 28% while your health is declining isn't always the smart play. Talk to a clinician about your specific situation.
Not sure where to start? Take our 2-minute provider match quiz → — we'll help you find a verified provider based on your preferences, budget, and insurance status.
How Do Retatrutide and Orforglipron Compare to Current GLP-1 Medications?
Context matters. Here's how these investigational drugs fit into the broader treatment landscape:
| Drug | Type | Delivery | Avg Weight Loss | Status | Est. Monthly Cost |
|---|---|---|---|---|---|
| Retatrutide | Triple agonist | Weekly injection | ~28.7% (Phase 3*)† | Phase 3 | Unknown (~2028) |
| Tirzepatide (Zepbound) | Dual agonist (GLP-1/GIP) | Weekly injection | ~22.5% | ✅ FDA approved | $299–$1,000 |
| Semaglutide 2.4mg (Wegovy injection) | GLP-1 RA | Weekly injection | ~14.9% | ✅ FDA approved | $349–$1,349 |
| Semaglutide 25mg (Wegovy pill) | GLP-1 RA | Daily pill | ~16.6% | ✅ FDA approved (Dec 2025) | $149+ |
| Orforglipron | GLP-1 RA (small molecule) | Daily pill | ~12.4% (Phase 3)† | NDA submitted | $149–$399 (est.) |
| Compounded semaglutide | GLP-1 RA | Weekly injection | Varies | Available (via telehealth) | $149–$299 |
*TRIUMPH-4 enrolled a knee osteoarthritis subpopulation. The primary obesity trial (TRIUMPH-1) has not yet reported.
†Efficacy estimand numbers shown. Treatment-regimen estimand: retatrutide 23.7%, orforglipron 11.2%.
Important: These percentages come from separate trials with different designs and cannot be compared directly. They provide general context for the range of results each medication has demonstrated.
A published network meta-analysis of seven GLP-1 and polyagonist therapies (PubMed, 2024) ranked overall efficacy for weight reduction as: retatrutide 12 mg > retatrutide 8 mg > tirzepatide 15 mg > tirzepatide 10 mg > semaglutide 2.4 mg > orforglipron. However, the authors emphasized that dual and triple receptor agonists consistently outperform single-target GLP-1 agonists, which is mechanistically expected.
Can I Get Retatrutide or Orforglipron Safely Right Now?
We need to be direct about this.
The rule
Orforglipron is now FDA-approved and commercially available as Foundayo (approved April 1, 2026). Retatrutide is not FDA-approved or commercially available. As of April 2026, the only legitimate way to access retatrutide is through enrollment in a clinical trial.
How to find legitimate clinical trials
- Go to ClinicalTrials.gov
- Search “retatrutide” or “orforglipron”
- Filter by “Recruiting” status and your geographic location
- Look for the NCT number (the trial's unique identifier)
- Contact the listed trial coordinator to discuss eligibility
Key questions to ask a trial coordinator:
- Is there a possibility I'll receive a placebo?
- What monitoring and support is provided?
- What happens after the trial ends?
- Are there any costs to participants?
Lilly's TRIUMPH program for retatrutide is enrolling across multiple trial sites. The ATTAIN program for orforglipron has largely completed enrollment, but you can check ClinicalTrials.gov for any remaining open trials.
The scam warning (please read this)
We're including this section because it matters for your safety. Retatrutide is not FDA-approved and is not available from any pharmacy, telehealth provider, or online seller. However, a quick search reveals websites selling “retatrutide peptides” and “research-grade retatrutide pens.”
These products are not regulated, not verified, and potentially dangerous. You have no way of knowing what's actually in them — the concentration, purity, sterility, or even whether the active ingredient is what the label claims. Verywell Health has reported on the risks of purchasing unregulated peptides marketed for weight loss.
🔴 Red flags that indicate a scam or unsafe product:
- Any website selling retatrutide for “research purposes only” but clearly marketing it for human weight loss
- Claims of pharmaceutical-grade quality without FDA oversight
- No prescription required
- Prices that seem too good to be true
- Shipping from unregulated international sources
- No clinical oversight or monitoring included
🟢 Green flags for legitimate access:
- Clinical trial listed on ClinicalTrials.gov with an NCT number
- Conducted at a recognized medical institution or research site
- Supervised by licensed physicians
- Includes regular safety monitoring
The same caution applies to compounded or gray-market orforglipron — if anyone is selling it before FDA approval, it's not the real, regulated product. For guidance on identifying safe compounded GLP-1 medications, see our safety guide.
Orforglipron vs Rybelsus, Ozempic, Wegovy, Zepbound, and Mounjaro
Since people searching for retatrutide and orforglipron are often already familiar with current GLP-1 medications, let's address the most common cross-comparisons.
Orforglipron vs Rybelsus (oral semaglutide)
This is the most natural comparison — both are GLP-1 pills. Rybelsus is a peptide-based semaglutide pill approved for type 2 diabetes. Per its FDA label, it must be taken on an empty stomach with no more than 4 oz of water, followed by a 30-minute wait before eating, drinking, or taking other medications. Many patients find this burdensome.
Orforglipron is a non-peptide small molecule with no food, water, or timing restrictions. In the ACHIEVE-3 head-to-head diabetes trial (published in The Lancet, February 2026), orforglipron demonstrated superior blood sugar reduction (A1C) compared to oral semaglutide — though the highest orforglipron dose also showed higher rates of GI side effects and discontinuations, per Reuters.
Orforglipron vs Wegovy pill (oral semaglutide 25 mg)
The Wegovy pill was FDA-approved in December 2025 and launched in early January 2026 at $149/month starting dose. It produced about 16.6% average weight loss — more than orforglipron's 12.4%. However, the Wegovy pill still requires the empty-stomach/30-minute-wait protocol. Orforglipron's no-restrictions dosing may be more practical for many people, even with less weight loss. Read our best oral semaglutide guide for more.
Retatrutide vs Zepbound (tirzepatide)
Zepbound is currently the most effective FDA-approved weight loss drug, producing roughly 22.5% average weight loss. Retatrutide's Phase 3 data (28.7%) exceeds this — but remember, this came from a smaller trial in a specific subpopulation, and the primary obesity trial hasn't reported yet. If TRIUMPH-1 confirms similar or greater weight loss in a broader population, retatrutide would represent a clear step above tirzepatide in raw efficacy. Both are injectable, and both are from Lilly. The eventual positioning will likely depend on whether retatrutide is reserved for more severe obesity or used broadly. See our full tirzepatide vs retatrutide comparison.
If you're already on a GLP-1 — should you plan to switch?
The ATTAIN-MAINTAIN trial specifically addressed this. People who had been on Wegovy or Zepbound for 72 weeks were switched to orforglipron. The results: they maintained the vast majority of their previously achieved weight loss on the oral pill.
This is clinically significant. It suggests a treatment pathway where people could start with an injectable for aggressive initial weight loss, then transition to a daily pill for long-term maintenance. That's a compelling model — and it's one more reason why starting treatment now doesn't mean you're committing to injections forever. Learn more about what happens when you stop taking GLP-1 medications.
What We Don't Know Yet (Honest Unknowns)
One of the biggest differences between this page and what you'll find elsewhere is this section. We think transparency about uncertainty builds more trust than pretending everything is settled. Here's what remains genuinely unknown as of March 2026:
- No head-to-head trial exists. Retatrutide and orforglipron have never been directly compared in the same clinical trial. Every comparison (including ours) is cross-trial, which has real scientific limitations.
- Retatrutide's primary obesity trial hasn't reported yet. TRIUMPH-4 was a knee osteoarthritis trial. TRIUMPH-1 — the real obesity trial, running 80 weeks — hasn't posted results. The 28.7% number is real, but it's from a specific subpopulation. TRIUMPH-1 could confirm or adjust that figure significantly.
- Long-term safety data doesn't exist. The longest published data for either drug is approximately 68–72 weeks. We don't have 2-year, 5-year, or 10-year safety profiles. Every approved GLP-1 medication continues to gather post-market safety data.
- The retatrutide dysesthesia signal is unclear. Tingling/numbness was reported in TRIUMPH-4 but not in the Phase 2 trial. Is it dose-dependent? Transient? Related to the knee OA population? Related to the speed of weight loss? We need data from the remaining seven Phase 3 readouts.
- Weight regain after stopping is unknown for both drugs. We know from studies of semaglutide and tirzepatide that people regain weight after discontinuation. Neither retatrutide nor orforglipron has published long-term discontinuation data. ATTAIN-MAINTAIN showed orforglipron can maintain weight lost on injectables, but we don't know what happens when orforglipron itself is stopped.
- Real-world pricing and insurance coverage are uncertain. Lilly announced self-pay pricing for orforglipron, but commercial insurance coverage, PBM negotiations, and formulary tier placement are all unresolved. Retatrutide pricing is completely unknown.
- Body composition data is limited. Rapid weight loss raises concerns about lean muscle mass loss. Neither drug has published detailed body composition (fat vs muscle) data from Phase 3 trials. This is an active area of concern across the entire GLP-1 class.
- How these drugs will be used in practice is uncertain. Will orforglipron be a first-line treatment? A maintenance option? Will retatrutide be reserved for severe obesity? These clinical positioning decisions haven't been made yet.
We'll update this section as new data emerges. The seven remaining Phase 3 trials for retatrutide are expected to report throughout 2026, and orforglipron's FDA decision should come mid-2026.
How We Researched This Comparison
We believe in showing our work.
What we used:
- Peer-reviewed publications: New England Journal of Medicine (ATTAIN-1), The Lancet (ATTAIN-2, ACHIEVE-3)
- Official Eli Lilly press releases and investor communications (via PR Newswire and investor.lilly.com)
- ClinicalTrials.gov trial registries (NCT numbers verified)
- FDA regulatory documents and drug labels (Rybelsus, Wegovy, Zepbound)
- Reuters reporting on regulatory timelines and clinical data
- Clarivate Drugs to Watch 2026 report (via PharmExec)
- PubMed network meta-analysis of GLP-1 therapies
What we didn't use:
- Social media posts, TikTok claims, or Reddit comments as clinical evidence
- Unverified press aggregators or AI-generated summaries
- Data from unregulated peptide sellers
- Manufacturer marketing materials presented as independent analysis
How we handle uncertainty:
- When data comes from a peer-reviewed journal, we say so
- When data comes from a company press release (not yet peer-reviewed), we note “topline results” or “per Eli Lilly press release”
- When timelines are projections, we identify the source and label them as estimates
- When we compare across trials, we explicitly state the limitations
Conflict of interest disclosure: Weight Loss Provider Guide earns affiliate commissions from select telehealth provider partnerships. These relationships do not influence our clinical data reporting, drug comparisons, or editorial conclusions. We apply the same standards regardless of whether a drug or provider is affiliated with us. Read our full editorial standards →
Evidence last verified: March 2026
Frequently Asked Questions
What to Do Next
You've made it through a lot of information. Here's where it all leads.
If you want to start GLP-1 treatment now
Millions of people are already seeing real results with currently available medications. Telehealth has made access significantly easier — many providers offer consultations within days, and pricing has become more competitive.
We've vetted the major telehealth GLP-1 providers for pricing transparency, clinical quality, and real patient experience. Compare current providers and pricing →
Not sure which type of treatment fits your situation? Take our 2-minute eligibility and provider match quiz → We'll ask a few questions about your preferences (pill vs injection, budget, insurance status) and recommend verified options.
If you want to wait for orforglipron or retatrutide
That's a completely valid choice — especially if your situation isn't urgent. Here's what to track:
- Orforglipron: Watch for the FDA decision (expected Q2 2026). We'll update this page when it happens.
- Retatrutide: Watch for TRIUMPH-1 results (the primary obesity trial, expected 2026) and remaining Phase 3 readouts. We'll cover each one.
Bookmark this page. We update it when significant new data publishes, FDA decisions occur, or pricing information changes.
If you want to explore clinical trial access
Visit ClinicalTrials.gov and search for retatrutide or orforglipron. Filter by recruiting status and your location. This is the only legitimate path to accessing these drugs before FDA approval.
Sources
- Wharton S, Aronne LJ, Stefanski A, et al. Orforglipron, an Oral Small-Molecule GLP-1 Receptor Agonist for Obesity Treatment. New England Journal of Medicine. 2025;393(18):1796-1806. Published September 16, 2025.
- Eli Lilly and Company. Lilly's triple agonist, retatrutide, delivered weight loss of up to an average of 71.2 lbs along with substantial relief from osteoarthritis pain in first successful Phase 3 trial. Press release. December 11, 2025.
- Eli Lilly and Company. Lilly's oral GLP-1, orforglipron, demonstrated meaningful weight loss and cardiometabolic improvements in complete ATTAIN-1 results published in The New England Journal of Medicine. Press release. September 16, 2025.
- Eli Lilly and Company. Lilly's orforglipron helped people maintain weight loss after switching from injectable incretins to oral GLP-1 therapy in first-of-its-kind Phase 3 trial (ATTAIN-MAINTAIN). Press release. December 18, 2025.
- Rosenstock J, Manghi FP, et al. Efficacy and safety of once-daily oral orforglipron compared with oral semaglutide in adults with type 2 diabetes (ACHIEVE-3). The Lancet. Published February 26, 2026.
- Clarivate Drugs to Watch 2026 report, as reported by Pharmaceutical Executive. March 2026.
- Reuters. Eli Lilly on track to launch oral obesity drug in second quarter pending U.S. approval. March 2, 2026.
- Reuters. Eli Lilly reports additional data on weight loss pill from diabetes trial (ACHIEVE-3). February 26, 2026.
- Sinha B, Ghosal S, et al. Efficacy and Safety of GLP-1 Receptor Agonists, Dual Agonists, and Retatrutide for Weight Loss in Adults With Overweight or Obesity: A Bayesian Network Meta-analysis. Obesity. 2025. PubMed PMID: 40685589. DOI: 10.1002/oby.24360.
- FDA. Rybelsus (semaglutide) prescribing information. Revised 2024.
- FDA. Wegovy (semaglutide) prescribing information. Includes boxed warning regarding thyroid C-cell tumors.
- Eli Lilly and Company. What to Know About Orforglipron. Updated 2026.
- Giblin K, Kaplan LM, et al. Retatrutide for the treatment of obesity, obstructive sleep apnea and knee osteoarthritis: Rationale and design of the TRIUMPH registrational clinical trials. Diabetes, Obesity and Metabolism. 2026;28(1):83-93.
- U.S. Food and Drug Administration. Commissioner's National Priority Voucher program. November 2025.
Affiliate Disclosure
Weight Loss Provider Guide is an independent resource. We earn commissions from select telehealth partner links (such as MEDVi) when you sign up through our referral links. This financial relationship does not influence our clinical data reporting, drug analysis, editorial recommendations, or the content of our comparison tables. We recommend starting with whatever treatment option is safest and most appropriate for your individual health situation — whether or not it involves one of our affiliate partners.
This page does not provide medical advice. The information is for educational purposes and should not replace consultation with a qualified healthcare provider. Retatrutide is an investigational drug that has not been approved by the FDA. Orforglipron (Foundayo) was FDA-approved on April 1, 2026. Always consult your doctor before starting, stopping, or changing any medication.
Last updated: April 10, 2026 | Next review: When TRIUMPH-1 results publish