For informational purposes only—not medical advice.
Tirzepatide vs Retatrutide: Evidence-Based Comparison for Weight Loss (2026)
Affiliate Disclosure: Weight Loss Provider Guide may earn a commission if you sign up through links on this page at no extra cost to you. This doesn't affect our ratings, our editorial process, or what we recommend. Full editorial policy.
Last Updated: February 11, 2026 · Sources: FDA.gov, PubMed, Eli Lilly · Medical Disclaimer at bottom of page
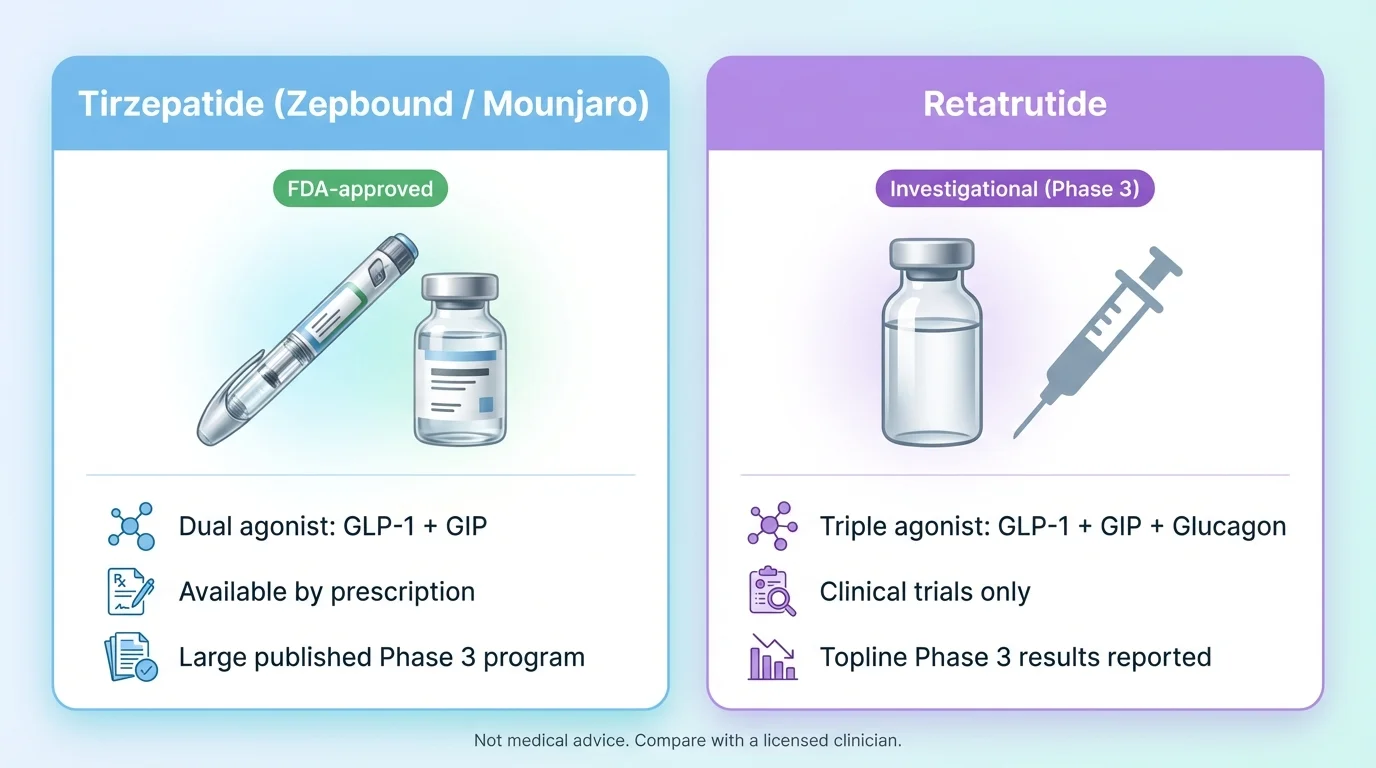
Wondering about tirzepatide vs retatrutide for weight loss? Here's the short answer: Tirzepatide (Zepbound/Mounjaro) is the proven, FDA-approved choice available right now — with mean weight loss of 20.9% at 72 weeks (15 mg, SURMOUNT-1; treatment-regimen estimand) per the FDA prescribing information. Retatrutide (LY3437943) is a next-generation triple agonist with even stronger early results — up to 28.7% body weight loss in its first Phase 3 trial — but it is not FDA-approved and is currently available only in clinical trials. Lilly has said seven additional Phase 3 trials are expected to complete in 2026; approval timing will depend on those results and FDA review. Here's the complete side-by-side, evidence-based comparison (note: no direct head-to-head clinical trial of tirzepatide vs retatrutide exists yet).
At-a-Glance Comparison Table
| Category | Tirzepatide (Zepbound / Mounjaro) | Retatrutide (LY3437943) | What This Means for You |
|---|---|---|---|
| Manufacturer | Eli Lilly | Eli Lilly | Same company, same research pipeline |
| Drug Class | Dual GLP-1/GIP agonist | Triple GLP-1/GIP/Glucagon agonist | Retatrutide adds a third target (glucagon) |
| FDA Status | ✓ FDA-approved (2022 for diabetes, 2023 for weight loss, 2024 for sleep apnea) | ✗ Not FDA-approved — Phase 3 trials ongoing | Only tirzepatide can be prescribed today |
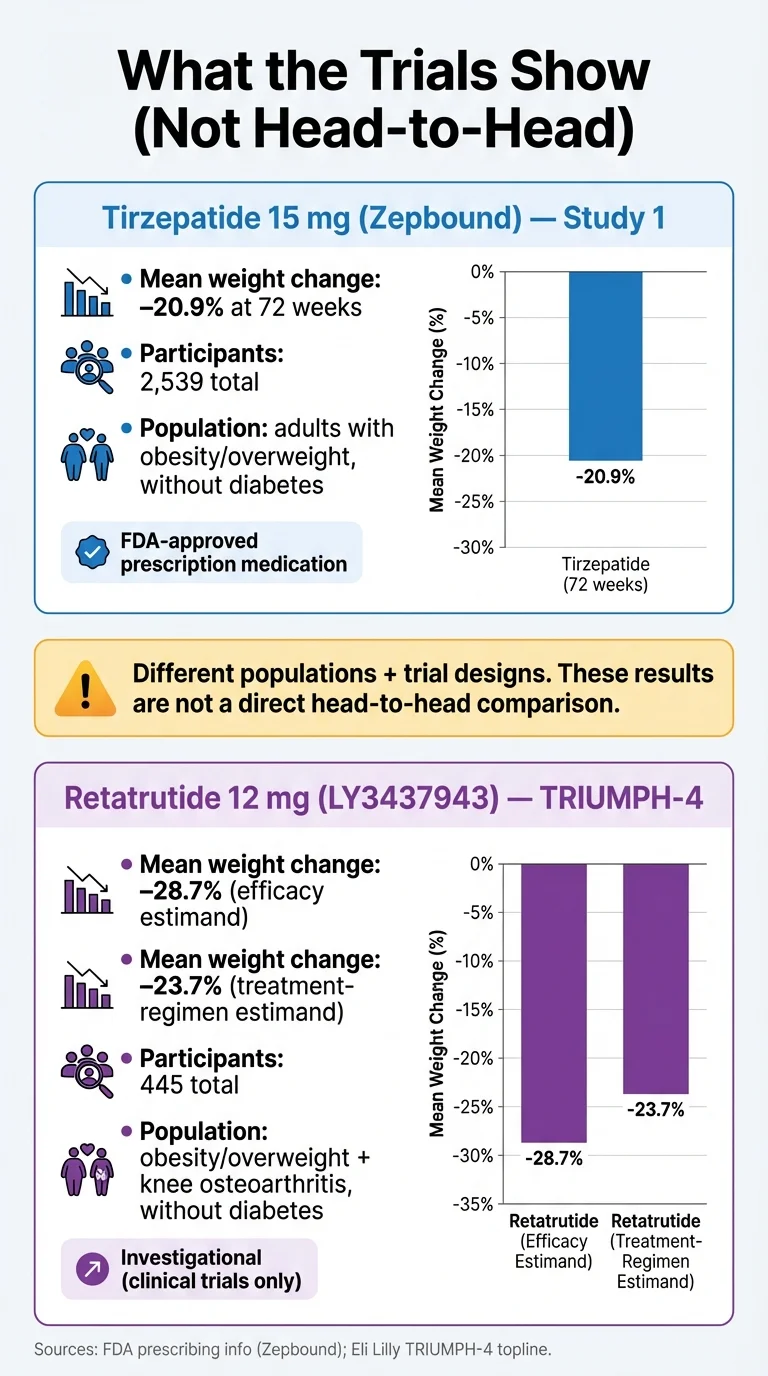
| Avg. Weight Loss (trials) | 20.9% body weight (SURMOUNT-1, 15 mg, 72 wks, treatment-regimen estimand) | 28.7% efficacy estimand / 23.7% treatment-regimen estimand (TRIUMPH-4, 12 mg, 68 wks) | Different trial designs — not a direct apples-to-apples comparison |
| Trial Size | 2,539 participants (SURMOUNT-1) | 445 participants (TRIUMPH-4) | Tirzepatide has vastly more data |
| Administration | Once-weekly injection (pre-filled pen; single-dose vials also available via LillyDirect self-pay) | Once-weekly injection (trial only) | Same convenience level |
| Dosing Range | 2.5 mg → 5 → 7.5 → 10 → 12.5 → 15 mg | 2 mg → 4 → 6 → 9 → 12 mg (trial protocol) | Both use gradual titration |
| Availability | Available now — prescription + telehealth | Not available — clinical trials only | You can start tirzepatide today |
| Monthly Cost | $25+ (insurance + savings card), $299–$449 (LillyDirect self-pay vials), ~$1,086 list price (see cost section) | N/A — not commercially available | Multiple affordable access routes for tirzepatide exist |
| Can It Be Compounded? | Sometimes available from compounding pharmacies, but compounded drugs are not FDA-approved. FDA advises compounded GLP-1 medicines should generally be used only when the FDA-approved drug is not available or cannot meet a patient's medical needs. | ✗ No — FDA explicitly prohibits compounding retatrutide | Any “retatrutide” sold online is unregulated and risky |
| Best For | Anyone ready to start proven treatment now | People willing to wait 2+ years for potentially stronger results | Most people should start now, switch later if needed |
Sources: Zepbound FDA prescribing information; Eli Lilly TRIUMPH-4 press release (Dec 11, 2025); FDA guidance on unapproved GLP-1 drugs.
What Is Tirzepatide?
Tirzepatide is a once-weekly injectable medication that activates two hormone receptors in your body: GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide). It's manufactured by Eli Lilly and sold under two brand names — Mounjaro for type 2 diabetes and Zepbound for chronic weight management and moderate-to-severe obstructive sleep apnea.
It's the most studied dual-agonist weight loss medication on the market. The SURMOUNT clinical trial program enrolled more than 5,000 participants across multiple global studies, and its results have been published in the New England Journal of Medicine — the gold standard of medical research.
Here's what those studies showed:
- SURMOUNT-1 (2,539 adults with obesity, no diabetes, 72 weeks): Average weight loss of 16.0% on 5 mg, 21.4% on 10 mg, and 22.5% on 15 mg (efficacy estimand) — compared to 2.4% on placebo. At the highest dose, 57% of participants lost at least 20% of their body weight. [^1]
- SURMOUNT-4 (670 adults, 88 weeks total): Participants who continued tirzepatide after an initial lead-in period achieved an average total weight loss of 25.3%. Those switched to placebo regained a significant portion of weight, confirming the drug's ongoing effectiveness. [^2]
- SURMOUNT-5 (head-to-head vs. semaglutide, 72 weeks): Tirzepatide produced 47% greater relative weight loss than semaglutide — 20.2% vs. 13.7%. [^3]
In practical terms, that means someone starting at 240 pounds on the highest dose of tirzepatide could reasonably expect to lose around 50 pounds over 72 weeks based on trial averages.
Tirzepatide is FDA-approved and can be prescribed through in-person doctors or licensed telehealth providers. It's available as a brand-name pre-filled pen (Zepbound/Mounjaro) and, with some restrictions, through compounding pharmacies.
What Is Retatrutide?
Retatrutide (also known by its research code LY3437943) is an investigational medication — also developed by Eli Lilly — that activates three hormone receptors: GLP-1, GIP, and glucagon. That third receptor is what makes it different from every weight loss drug currently on the market.
It is not FDA-approved. It cannot be prescribed. It cannot be legally purchased. It is only accessible through enrollment in clinical trials.
Eli Lilly is currently running the TRIUMPH Phase 3 program — the initial registrational program includes four global trials in more than 5,800 participants across multiple conditions, including obesity, type 2 diabetes, obstructive sleep apnea, and knee osteoarthritis. Lilly is also studying retatrutide in additional Phase 3 trials for other conditions, including chronic low back pain, cardiovascular and renal outcomes, and metabolic liver disease. [^4]
The first Phase 3 results were reported in December 2025:
- TRIUMPH-4 (445 adults with obesity and knee osteoarthritis, 68 weeks): Participants on the 12 mg dose lost an average of 28.7% of their body weight — approximately 71.2 pounds from a starting weight of about 248 pounds. The 9 mg dose produced 26.4% weight loss. Both doses also dramatically reduced knee pain, with some participants becoming completely pain-free. [^5]
Earlier Phase 2 data (published in the New England Journal of Medicine in 2023) showed weight loss of up to 24.2% at 48 weeks in a smaller group of 338 participants — and the weight loss curve had not plateaued, meaning participants were still losing at the end of the study. [^6]
Seven additional Phase 3 trial results are expected throughout 2026. If those results are positive and FDA review goes smoothly, the earliest possible approval would be 2027 or later. That's not a conservative estimate — it's realistic based on standard regulatory timelines.
⚠️ Important Safety Notice
Retatrutide has not been reviewed or approved by the FDA. Products sold online as “retatrutide” are unregulated research peptides, not the pharmaceutical-grade compound used in Eli Lilly's clinical trials. The FDA has issued warning letters to companies selling retatrutide APIs and has stated explicitly that “retatrutide cannot be used in compounding under federal law.” [^7] Do not purchase or self-administer any product marketed as retatrutide.
How Do Tirzepatide and Retatrutide Work Differently?
Both drugs belong to the same family — incretin-based hormone therapies. But the number and type of receptors they activate changes what they do in your body.
GLP-1 receptor (activated by both): This is the workhorse. GLP-1 reduces appetite, slows how fast food leaves your stomach, and improves blood sugar control. It's the same pathway that semaglutide (Ozempic/Wegovy) targets — and it's the reason GLP-1 drugs work for weight loss.
GIP receptor (activated by both): GIP enhances the insulin response after eating and appears to improve how your body handles fat metabolism. This is what separates tirzepatide from semaglutide — dual agonism appears to produce stronger weight loss than GLP-1 alone, and the SURMOUNT-5 head-to-head trial confirmed this.
Glucagon receptor (activated by retatrutide only): This is the differentiator. Glucagon increases your body's energy expenditure (how many calories you burn at rest), promotes the breakdown of stored fat, and may help preserve lean muscle mass during weight loss. In theory, activating the glucagon receptor means your body burns more calories even while eating less — a two-sided approach that could explain retatrutide's stronger results.
A simple way to think about it: GLP-1 reduces calories coming in. GIP improves how your body processes those calories. Glucagon increases calories going out. Tirzepatide has the first two. Retatrutide has all three.
That said, theory and trial results are not the same thing. Retatrutide's glucagon benefits are biologically plausible and consistent with its Phase 2 and Phase 3 data — but the full picture won't be clear until the complete TRIUMPH program reports out and peer-reviewed publications analyze the long-term data in detail.

Tirzepatide vs Retatrutide Weight Loss Results: What the Trials Actually Show
This is the section most people came here for, so let's be precise about what we know and what we don't.
The Headline Numbers
| Trial Detail | Tirzepatide (SURMOUNT-1) | Retatrutide (TRIUMPH-4) |
|---|---|---|
| Trial Phase | Phase 3 (gold standard for FDA approval) | Phase 3 (first readout) |
| Participants | 2,539 | 445 |
| Population | Adults with obesity, no diabetes | Adults with obesity + knee osteoarthritis, no diabetes |
| Duration | 72 weeks | 68 weeks |
| Highest Dose Tested | 15 mg | 12 mg |
| Avg. Weight Loss (efficacy estimand) | 22.5% | 28.7% |
| Avg. Weight Loss (treatment-regimen estimand) | 20.9% | 23.7% (12 mg); 20.0% (9 mg) |
| Avg. Pounds Lost | ~52 lbs (from ~231 lb avg starting weight) | ~71.2 lbs (from ~248 lb avg starting weight) |
| Discontinuation Due to AEs | 4.8%–6.7% (dose-dependent) | 12.2%–18.2% (dose-dependent) |
| Published In | NEJM (peer-reviewed, 2022) | Press release (Dec 2025); peer-reviewed publication pending |
Sources: Jastreboff et al., NEJM 2022 (SURMOUNT-1) [^1]; Eli Lilly TRIUMPH-4 press release [^5]; HCPLive TRIUMPH-4 summary [^8].
Why You Can't Directly Compare These Numbers
I want to be upfront about something most comparison articles skip over: these numbers come from different trials with different populations, and they are not an apples-to-apples comparison.
Here's why that matters:
- Different populations. TRIUMPH-4 enrolled people with both obesity and knee osteoarthritis — a group that tends to have higher starting BMI (average 40.4 in TRIUMPH-4 vs. 38.0 in SURMOUNT-1). People with higher starting BMI often lose a greater percentage of body weight.
- Different estimands. SURMOUNT-1 reported both “efficacy estimand” (22.5% — what would happen if everyone stayed on the drug) and “treatment-regimen estimand” (20.9% — what actually happened including people who dropped out). TRIUMPH-4's headline 28.7% figure is the efficacy estimand. The treatment-regimen number is 23.7% at 12 mg — still impressive, but meaningfully lower than the headline figure.
- Different trial sizes. 2,539 participants vs. 445. Larger trials generally produce more reliable averages.
- No head-to-head study. The only way to truly compare two drugs is to randomize patients to one or the other in the same trial. That hasn't been done for tirzepatide vs. retatrutide.
What We Can Fairly Say
Retatrutide's early Phase 3 results are genuinely remarkable — potentially the strongest weight loss numbers ever reported for a pharmaceutical treatment. The glucagon component appears to be providing a meaningful additional benefit.
But “the strongest early data” and “FDA-approved, extensively studied, available right now” are in fundamentally different categories. Tirzepatide has been through the full gauntlet: multiple large Phase 3 trials, FDA review, post-market safety data, and substantial real-world use since FDA approval. Retatrutide still has years of testing ahead.
The results from seven additional TRIUMPH trials expected in 2026 will tell us much more — including how retatrutide performs in populations without knee osteoarthritis, in people with type 2 diabetes, and at a maintenance dose of 4 mg.
“I've been on Zepbound since December 2023. Started at 204 pounds and I'm now at 126 pounds. It has been amazing — I added weight lifting and power walking, plus I cook my own meals now.” — User review on Drugs.com (Zepbound rated 8.8/10, 82% positive, 450+ reviews) [^9]
Side Effects and Safety: What's Similar, What's Different
The GI Side Effects Both Drugs Share
All GLP-1-class medications cause gastrointestinal side effects. This is not a flaw — it's a direct consequence of how these drugs work. Slowing gastric emptying and reducing appetite means your digestive system is processing food differently, and your body needs time to adjust.
The most common side effects for both tirzepatide and retatrutide are nausea, diarrhea, vomiting, constipation, and decreased appetite. Here's what the clinical trial data shows:
| Side Effect | Tirzepatide 15 mg (FDA label, pooled Studies 1 & 2) | Retatrutide 12 mg (TRIUMPH-4) |
|---|---|---|
| Nausea | 28% | 43.2% |
| Diarrhea | 23% | 33.1% |
| Vomiting | 13% | 20.9% |
| Constipation | 11% | 25.0% |
| Decreased appetite | Not separately reported in FDA label | 18.2% |
| Permanent discontinuation due to AEs | 6.7% (15 mg) | 18.2% (12 mg arm) |
Note: Tirzepatide rates are from the FDA-approved Zepbound prescribing information (pooled placebo-controlled data, Studies 1 & 2). TRIUMPH-4 rates are from Lilly's December 2025 press release. Direct comparison is imprecise due to different trial designs but is directionally informative.
Sources: Zepbound FDA prescribing information [^10]; TRIUMPH-4 press release [^5].
The Honest Truth About Side Effects
Let's be straightforward: GI side effects are real. If you take tirzepatide or any GLP-1-class medication, there's roughly a 1-in-4 chance you'll experience some nausea, especially in the first few weeks after starting or increasing your dose.
But here's what the data also shows: the vast majority of these side effects are mild to moderate, are often most noticeable when starting treatment or after dose escalation, and many people find they improve over time. In the FDA's pooled analysis of Zepbound clinical trials, 93.3% of participants on the 15 mg dose continued treatment despite side effects — permanent discontinuation due to adverse events was only 6.7%. [^10]
This is exactly why dose titration exists. You start at the lowest dose (2.5 mg for tirzepatide) and gradually step up over weeks to months. A good provider will work with you to find the dose that balances results with tolerability.
“For the first time, probably in my life, I don't think about food. I don't crave junk food, I get full very quickly. Even if I never lose what I want to, I am so grateful for this drug because I am eating all healthy foods.” — User review on Drugs.com [^9]
What's Different About Retatrutide's Safety Profile
TRIUMPH-4 showed notably higher rates of GI side effects and higher treatment discontinuation rates (12.2–18.2% vs. 4% on placebo) compared to tirzepatide's SURMOUNT trials. Some of this is likely driven by the higher magnitude of weight loss itself — Lilly noted that some discontinuations were due to “perceived excessive weight loss,” particularly in participants with lower baseline BMI. [^5]
There was also a new safety signal: dysesthesia (abnormal skin sensations like tingling, burning, or numbness). This was not reported in the earlier Phase 2 trial and analysts flagged it as something to watch in upcoming TRIUMPH readouts. Lilly noted it did not appear to lead to treatment discontinuation. [^11]
Safety Warnings That Apply to Both
Both medications carry a boxed warning about thyroid C-cell tumors observed in animal studies. Neither should be used by anyone with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Other important warnings include risk of pancreatitis, gallbladder problems, and potential interactions with other medications — particularly insulin and sulfonylureas. The Zepbound label also explicitly states: “Zepbound contains tirzepatide and should not be used with other tirzepatide-containing products or any GLP-1 receptor agonist medicines.” [^10]

What Happens When You Stop? (The Honest Answer)
This is the part most weight loss articles bury or skip entirely, so let's address it head-on.
When you stop taking a GLP-1-class medication, weight regain is common. The SURMOUNT-4 trial demonstrated this directly: participants who stopped tirzepatide after 36 weeks and switched to placebo regained a significant portion of their lost weight over the next 52 weeks. Those who continued tirzepatide maintained and even extended their weight loss — ending at 25.3% total after 88 weeks. [^2]
This is not unique to tirzepatide. It's true for virtually all obesity medications, because obesity is a chronic metabolic condition — not a temporary problem with a short-term fix. The same way you wouldn't stop taking blood pressure medication and expect your blood pressure to stay controlled, stopping a weight loss medication typically leads to some degree of reversal.
Here's the good news. This doesn't mean you're signing up for the maximum dose forever. Many people, after losing a significant amount of weight, can work with their provider to find a lower maintenance dose that preserves results with fewer side effects and lower cost. Retatrutide's TRIUMPH program is actually studying a 4 mg maintenance dose for exactly this purpose. And lifestyle habits built during treatment — better nutrition, consistent exercise, improved relationship with food — carry forward even if the medication changes.
The point isn't to discourage you. It's to set realistic expectations so you go in with a sustainable plan rather than chasing a quick fix. The people who get the best long-term results treat these medications as one part of a comprehensive approach that includes dietary changes, resistance training, adequate protein, and ongoing medical support.
Cost and Access: Tirzepatide Now vs. Retatrutide Later
What Tirzepatide Actually Costs in 2026
The “list price” for Zepbound is $1,086 per month for pre-filled pens. But very few people pay that. Here's the real landscape:
| Access Pathway | Estimated Monthly Cost | Notes |
|---|---|---|
| With commercial insurance + Zepbound Savings Card | As low as $25/month | Requires covered insurance plan; savings subject to annual limits |
| Commercial insurance without coverage + Savings Card | ~$499/month | For patients whose plan doesn't include Zepbound |
| LillyDirect (self-pay vials) | 2.5 mg: $299; 5 mg: $399; 7.5–15 mg: $449/month | Self-Pay Journey Program; 1-month supply = 4 single-dose vials; terms apply |
| Medicare (announced for as early as April 2026) | $50/month cap announced | Contingent on program rollout and FDA approval of a multi-dose pen; timelines may vary |
| Telehealth providers | Varies by provider | Typically includes medication, consults, and monitoring; check individual provider pricing |
| GoodRx coupon (brand-name pens) | ~$499–$995/month | Without insurance, pharmacy pricing with discount |
Sources: Eli Lilly pricing information [^12]; GoodRx [^13]; Eli Lilly / U.S. government access agreement (Nov 2025) [^14]; Lilly single-dose vial pricing update (Dec 2025) [^15].
The cost picture has improved significantly over the past year. Between LillyDirect vials, the Medicare access agreement, and competitive telehealth pricing, tirzepatide is more accessible than it's ever been.
What Retatrutide Costs
Nothing — because you can't buy it. Retatrutide is not commercially available and has no price. Any website claiming to sell retatrutide is selling an unregulated product.
If and when retatrutide receives FDA approval, pricing will likely be in a similar range to Zepbound — Eli Lilly will be bringing it to the same market. But that's speculation. The drug doesn't have a price because it doesn't have a market yet.
The Real Cost Calculation
The cost that matters most isn't the monthly price of medication — it's the total cost of waiting. Every month without treatment is a month of ongoing health risks from obesity, including cardiovascular disease, type 2 diabetes, joint damage, and sleep apnea.
If you're waiting for retatrutide because it might be 5–8% more effective in trials, you're paying for that wait with 2+ years of untreated weight and its consequences. Starting tirzepatide now and potentially switching later is almost always the better financial and health equation.
If cost is a concern, telehealth providers like MEDVI offer tirzepatide programs at competitive monthly rates that include the medication, medical evaluation, and ongoing provider support.
MEDVI
See pricing
When you click, you'll go to MEDVI to start an eligibility check and see current pricing and next steps.
Tirzepatide vs Retatrutide for Muscle Loss
Muscle loss during weight loss is a legitimate concern — and one that the “bodybuilding” and “muscle loss” search variants tell us a lot of people are thinking about.
Here's the reality: any significant weight loss, whether from medication, surgery, or diet alone, involves some loss of lean mass. Studies suggest that roughly 25–40% of total weight lost may come from lean tissue rather than fat. This is not unique to GLP-1 drugs; it's a biological consequence of caloric deficit.
Tirzepatide's SURMOUNT trials documented body composition changes, and the lean mass loss was consistent with what you'd expect from this magnitude of weight loss.
Retatrutide's glucagon receptor activation is theoretically promising for muscle preservation. Glucagon promotes fat oxidation (burning stored fat for fuel) and may shift the ratio of weight loss more toward fat and away from lean mass. The TRIUMPH-4 data showed impressive fat loss, but detailed body composition analysis hasn't been published yet. We'll know more when the full peer-reviewed data comes out.
Regardless of which medication you use, here's what the evidence says you should do:
- Resistance training 2–3x per week. This is the single most important thing you can do to preserve muscle during weight loss. It doesn't need to be complicated — compound movements like squats, presses, and rows performed consistently.
- Adequate protein intake. Aim for 0.7–1.0 grams per pound of body weight daily. On a GLP-1 medication, your appetite will be reduced, so you need to be intentional about protein — prioritize it at every meal.
- Don't rush the dose increases. Faster dose escalation can mean more aggressive appetite suppression, which can make it harder to hit protein targets.
- Track your strength, not just the scale. If your lifts are holding steady or progressing while the scale drops, you're likely preserving muscle well.
These strategies work for both tirzepatide and retatrutide. They're the proven variables you can actually control right now.
Tirzepatide vs Retatrutide Dosage Comparison
| Dosing Detail | Tirzepatide (Zepbound) | Retatrutide (TRIUMPH trials) |
|---|---|---|
| Starting Dose | 2.5 mg once weekly | 2 mg once weekly |
| Escalation Steps | 2.5 → 5 → 7.5 → 10 → 12.5 → 15 mg | 2 → 4 → 6 → 9 → 12 mg |
| Escalation Interval | Every 4 weeks (minimum) | Every 4 weeks (trial protocol) |
| Time to Max Dose | ~20 weeks | ~16 weeks (trial protocol) |
| Maximum Dose | 15 mg | 12 mg (highest tested in Phase 3) |
| Maintenance Dose Option | Provider-determined (many patients maintain on 10 mg) | 4 mg being studied in TRIUMPH-1 and TRIUMPH-2 |
| Injection Method | Pre-filled single-dose pen (auto-injector) | Subcutaneous injection (delivery format TBD) |
| Missed Dose | Take within 4 days; if >4 days, skip to next scheduled dose | N/A — trial protocol managed by investigators |
| Storage | Refrigerate before first use; room temp up to 21 days (single-dose pens/vials per FDA label) | N/A |
Source: Zepbound FDA prescribing information [^10]; TRIUMPH-4 trial registration, ClinicalTrials.gov [^16]; TRIUMPH program design, Giblin et al. [^4].
Tirzepatide's dosing is well-established, FDA-guided, and flexible enough for providers to adjust based on individual response and tolerability. Retatrutide's final commercial dosing won't be determined until Phase 3 results are complete and an FDA application is filed.
Can You Take Tirzepatide and Retatrutide Together?
No. There is zero clinical evidence supporting this combination, no study has tested it, and no medical authority recommends it.
The Zepbound prescribing information explicitly states that tirzepatide “should not be used with other tirzepatide-containing products or any GLP-1 receptor agonist medicines.” [^10] Retatrutide activates the same GLP-1 and GIP receptors as tirzepatide, meaning combining them would likely amplify side effects — particularly GI symptoms — without a proven safety or efficacy basis.
Beyond the medical concerns, there's a practical impossibility: retatrutide isn't available to combine with anything. If you're considering this because tirzepatide alone hasn't produced adequate results, the better conversation is with your provider about dose optimization, lifestyle factors (protein, resistance training), or whether you might be a candidate for a clinical trial.
Switching From Tirzepatide to Retatrutide
You can't switch right now because retatrutide isn't available. But it's a reasonable question for the future, so here's what to think about.
When switching might make sense (if retatrutide is eventually approved):
- You've been on tirzepatide for 12+ months and your weight loss has plateaued despite dose optimization
- You're particularly concerned about body composition (fat loss vs. lean mass preservation)
- Your provider recommends it based on your individual health profile and the full Phase 3 data
What you shouldn't do: Wait to start treatment. The data clearly shows that starting tirzepatide now produces meaningful, life-changing results. If a stronger option becomes available in 2–3 years, you and your doctor can discuss transitioning at that point. You'll be in a much better position — healthier, lighter, and with established habits — than if you'd spent those years waiting.
There is no published protocol for switching between these specific drugs, since one isn't approved. When the time comes, your prescribing provider will determine the appropriate transition plan, likely involving a careful dose adjustment period.
“Where to Buy Retatrutide” and “Is Retatrutide a Scam?”
These questions come up constantly, and this section could save you real money and real health risk.
The FDA's Position (Plain English)
The FDA has made this crystal clear: “Retatrutide and cagrilintide cannot be used in compounding under federal law. Additionally, these are not components of FDA-approved drugs and have not been found safe and effective for any condition.” [^7]
The agency has issued warning letters to companies distributing retatrutide as an active pharmaceutical ingredient. Any product sold as “retatrutide” outside of a clinical trial is, by definition, not the pharmaceutical-grade compound Eli Lilly is testing.
Red Flags: How to Spot a Scam
If you see any of the following, walk away:
- A website selling “retatrutide” for human use (it is illegal to sell)
- Labels saying “for research purposes only” or “not for human consumption” paired with dosing instructions
- No verifiable state board of pharmacy license for the dispensing pharmacy
- Payment only through cryptocurrency, wire transfer, or non-reversible methods
- No prescriber involvement — you're ordering without a medical evaluation
- Pricing that seems too good to be true ($50–$150/month for a “next-generation” peptide)
- Promises about purity or lab testing without verifiable third-party certificates
The Safe Alternative
If you want GLP-1-based weight loss results right now, the legitimate path is an FDA-approved medication — tirzepatide (Zepbound/Mounjaro) or semaglutide (Wegovy/Ozempic) — prescribed by a licensed healthcare provider and dispensed by a licensed pharmacy.
Telehealth providers have made this process significantly more accessible. You complete a health questionnaire, a licensed provider reviews your information, and if eligible, a prescription is issued and medication ships to your door.

Tirzepatide vs Retatrutide for Inflammation and Other Health Markers
Weight loss itself is one of the most powerful anti-inflammatory interventions known to medicine. When you lose significant body fat, inflammatory markers like hsCRP (high-sensitivity C-reactive protein) drop, blood pressure improves, cholesterol profiles shift favorably, and the risk of cardiovascular events decreases.
Both tirzepatide and retatrutide produce these benefits, largely driven by the weight loss they cause. But there are some differences worth noting:
Tirzepatide has demonstrated improvements across a wide range of cardiovascular and metabolic markers in the SURMOUNT trials, including reductions in blood pressure, triglycerides, and inflammatory markers. It also received an FDA indication for moderate-to-severe obstructive sleep apnea in adults with obesity in 2024 — making it the first GLP-1 medication approved for that condition. [^10]
Retatrutide showed clinically meaningful improvements in non-HDL cholesterol, hsCRP, and triglycerides in TRIUMPH-4. [^5] Eli Lilly is also studying retatrutide for metabolic dysfunction-associated steatotic liver disease (MASLD, formerly called NAFLD/NASH) — a condition driven by inflammation and fat accumulation in the liver — and for cardiovascular/renal outcomes. [^4]
The glucagon receptor component of retatrutide may provide anti-inflammatory benefits beyond weight loss alone (glucagon has known effects on liver fat metabolism), but this hasn't been isolated and proven in human studies yet.
Bottom line: If reducing inflammation is a priority for you, both drugs will help significantly through weight loss. Tirzepatide is the one you can access today with established data. Retatrutide's additional benefits are plausible but unconfirmed.
Tirzepatide vs Retatrutide vs Semaglutide: Three-Way Comparison
Since many readers are also weighing semaglutide, here's how all three compare:
| Feature | Semaglutide (Wegovy/Ozempic) | Tirzepatide (Zepbound/Mounjaro) | Retatrutide (Investigational) |
|---|---|---|---|
| Receptors | GLP-1 only | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
| Avg. Weight Loss | ~13.7% (SURMOUNT-5 head-to-head) | ~20.2% (SURMOUNT-5 head-to-head) | ~28.7% (TRIUMPH-4 efficacy estimand) |
| FDA Approved | ✓ Yes | ✓ Yes | ✗ No |
| Available Now | ✓ Yes | ✓ Yes | ✗ No |
| Manufacturer | Novo Nordisk | Eli Lilly | Eli Lilly |
| List Price (monthly) | ~$1,349 (Wegovy) | ~$1,086 (Zepbound) | N/A |
| Cardiovascular Outcome Data | ✓ Yes (SELECT trial) | ✓ Yes (SURPASS-CVOT: non-inferior to dulaglutide for MACE; SURMOUNT-MMO ongoing) | No |
| OSA Indication | ✗ No | ✓ Yes | Being studied |
Sources: SURMOUNT-5 results [^3]; Zepbound and Wegovy FDA prescribing information.
The trajectory is clear: single agonist → dual agonist → triple agonist. Each generation appears to produce stronger weight loss results. Semaglutide has the most long-term safety data and cardiovascular outcome data in adults with overweight/obesity without diabetes (SELECT trial — reduced major adverse cardiovascular events vs placebo). Tirzepatide also has published cardiovascular outcomes data in type 2 diabetes with established cardiovascular disease (SURPASS-CVOT — met noninferiority vs dulaglutide for MACE). These studies use different populations and comparators, so they are not directly comparable, but both drugs have cardiovascular safety evidence. Tirzepatide sits in the sweet spot of strong efficacy with substantial real-world data and current availability.
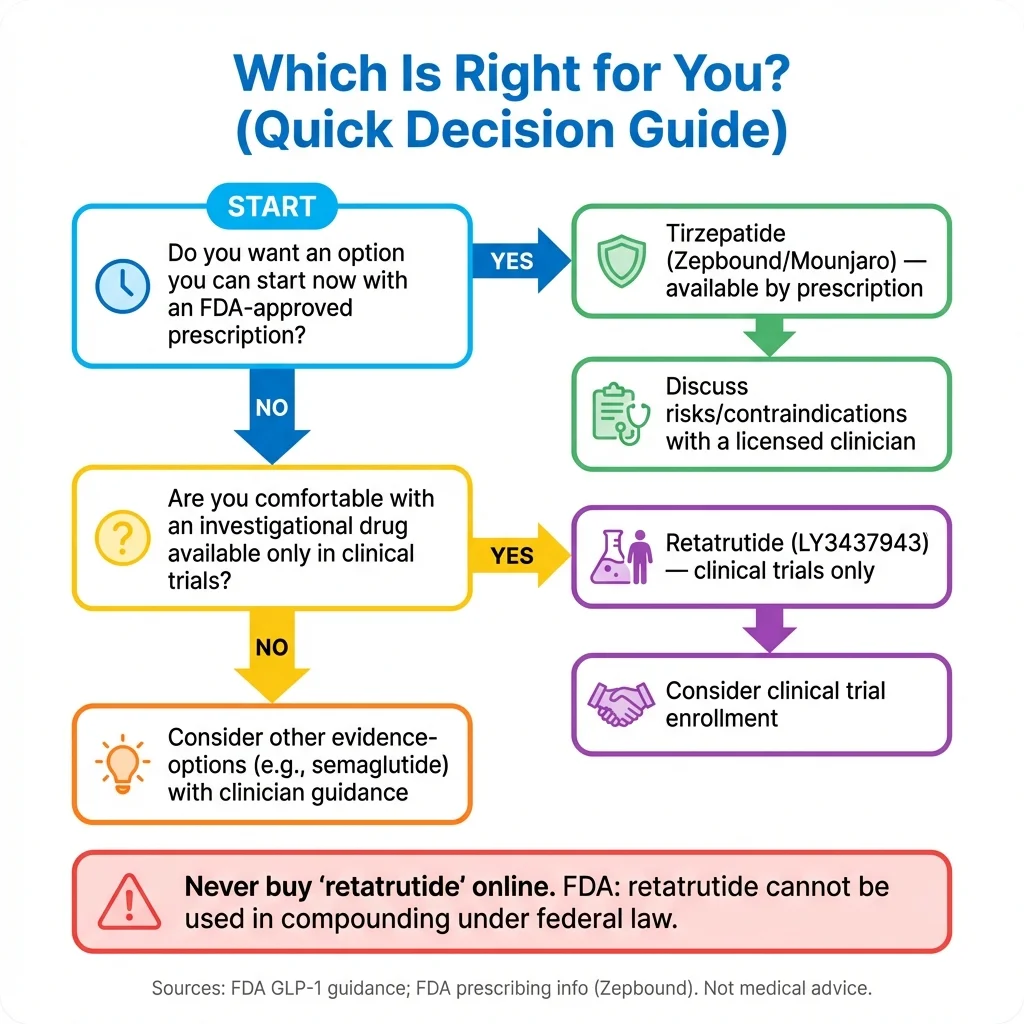
Decision Framework: Which Is Right for You?
This is where we stop comparing data points and start helping you make a decision.
Choose Tirzepatide Now If:
- ✓ You want to start treatment today, not in 2+ years
- ✓ You want an FDA-approved medication with extensive safety data and substantial real-world use since approval
- ✓ You have 20+ pounds to lose and want the strongest available treatment
- ✓ You want access through insurance, LillyDirect, or telehealth providers
- ✓ You value proven outcomes over theoretical improvements
- ✓ You have obesity-related conditions (sleep apnea, pre-diabetes, joint pain) that benefit from starting treatment now
- ✓ You're comfortable starting with what's proven and switching later if something better becomes available
Consider Waiting for Retatrutide If:
- ⌛ You are not in a rush and genuinely comfortable waiting 2+ years
- ⌛ You've already tried tirzepatide at maximum dose and plateaued
- ⌛ You're specifically interested in enrolling in a clinical trial (check clinicaltrials.gov for TRIUMPH trials near you)
- ⌛ You have no urgent obesity-related health conditions that need treatment now
Decision Framework Summary
| If You… | Consider… | Why |
|---|---|---|
| Are ready to start now | Tirzepatide | FDA-approved, available today through insurance, LillyDirect, or telehealth — no waiting |
| Want to wait for the strongest option | Retatrutide (2027+) | Potentially stronger results, but 2+ years away and still unproven in full Phase 3 |
| Tried semaglutide and want more | Tirzepatide | 47% greater weight loss vs. semaglutide in head-to-head SURMOUNT-5 trial |
| Are concerned about side effects | Tirzepatide | Lower GI side effect rates and lower discontinuation rate vs. retatrutide in trials |
| Are focused on body composition | Either (with resistance training) | Retatrutide's glucagon component may favor fat loss, but protein + lifting matters more |
| Are motivated by data | Tirzepatide now, switch later if warranted | Tirzepatide has peer-reviewed data, FDA review, and substantial real-world use since approval; retatrutide has one press release |
The Reality Most People Need to Hear
Every month you wait to start treatment is a month of compounding health consequences. Obesity is a progressive condition — it generally gets harder to treat over time, not easier. The risks of cardiovascular disease, type 2 diabetes, joint deterioration, and metabolic syndrome don't pause while you wait for a slightly better medication.
Starting tirzepatide now and potentially transitioning to retatrutide later (if it's approved and shows clear superiority in head-to-head data) is almost certainly a better strategy than doing nothing for 2–3 years.
Many adults who meet the FDA indication criteria (BMI ≥30, or BMI ≥27 with at least one weight-related condition) may be eligible, but final eligibility is determined by a licensed clinician after reviewing your medical history. Telehealth providers have streamlined the process — you answer a health questionnaire, a licensed provider reviews your information, and if you're eligible, a prescription is issued and medication ships to your door.
MEDVI
How to Start Tirzepatide Safely
Who Qualifies
Per the FDA-approved indication, Zepbound is for adults with:
- A BMI of 30 or greater (obesity), OR
- A BMI of 27 or greater with at least one weight-related condition (such as high blood pressure, type 2 diabetes, high cholesterol, or obstructive sleep apnea)
It's also approved for adults with moderate-to-severe obstructive sleep apnea and obesity. [^10]
Your Options for Getting Started
In-person physician: Your primary care doctor, endocrinologist, or an obesity medicine specialist can prescribe brand-name Zepbound or Mounjaro. This route gives you the best chance of insurance coverage and may be necessary for certain insurance prior authorizations.
Telehealth provider: The fastest, most accessible route for most people. The typical process:
- Complete a brief online health questionnaire
- A licensed provider reviews your medical history
- If eligible, a prescription is issued
- Medication ships directly to your door (cold-chain, as required)
- Follow-up check-ins are scheduled to monitor progress and adjust dosing
| Provider | What's Included | Key Advantage |
|---|---|---|
| MEDVI | Medical evaluation, prescription, medication, ongoing monitoring | Streamlined online process with licensed providers |
| Eden | Medical evaluation, prescription, medication, provider access | Established platform with comprehensive support |
Both are legitimate, licensed telehealth providers. Research any provider before signing up — confirm they use licensed prescribers and state-licensed pharmacies.
Questions to Ask Before You Pay
Before starting with any provider, online or in-person, ask:
- “Is this FDA-approved Zepbound/Mounjaro, or is it compounded tirzepatide?” Both are legal options, but you should know which you're getting.
- “Which pharmacy fills the prescription? Is it state-licensed?” Verify independently if you want.
- “What's the follow-up plan?” Good providers check in regularly, monitor for side effects, and adjust dosing. A provider who writes a prescription and disappears is a red flag.
- “What happens if I have side effects or need to pause?” You want clear protocols, not a runaround.
- “Are there any additional fees beyond the monthly cost?” Watch for hidden enrollment fees, shipping charges, or consult fees.
“I was nervous about doing it online, but the whole process was incredibly easy. I had my prescription within 24 hours and started a few days later. I've lost 42 pounds in about 6 months.” — User review on WebMD [^17]
Tirzepatide vs Retatrutide for Bodybuilding and Athletic Performance
This sub-topic gets a lot of search volume, so let's address it directly.
If you're a bodybuilder or athlete searching “tirzepatide vs retatrutide bodybuilding,” you're probably asking one of two things: Can I use these drugs to cut body fat while preserving muscle? Or is retatrutide better for body composition than tirzepatide because of the glucagon component?
What We Know
GLP-1 class medications are not anabolic. They don't build muscle. They primarily work by reducing appetite and slowing digestion, which creates a caloric deficit. The weight you lose will be a mix of fat and lean tissue, just as with any deficit — though the ratio varies depending on your protein intake, training, and genetics.
Tirzepatide has been used by many people who train seriously. The main challenge isn't the drug itself — it's maintaining adequate caloric intake and protein when your appetite is dramatically suppressed. People who lift heavy report that the “food noise” disappears, which is life-changing for most users but can be a challenge for athletes who need to hit specific macros.
Retatrutide's glucagon component is where it gets interesting. Glucagon promotes lipolysis (breakdown of stored fat) and increases thermogenesis (calorie burning). In theory, this could shift weight loss more toward fat and away from lean mass. The Phase 2 trial showed body fat percentage decreasing substantially, but detailed lean mass data hasn't been published with full granularity yet.
Practical Advice for Training on GLP-1 Medications
Whether you're on tirzepatide now or considering retatrutide in the future, the protocol is essentially the same:
Protein is non-negotiable. Aim for 1 gram per pound of lean body mass daily. This is harder than it sounds when your appetite is dramatically reduced. Strategies that work: protein shakes, Greek yogurt, cottage cheese, lean meats eaten early in the day, and setting protein targets before filling up on other foods.
Resistance training preserves muscle. Compound lifts (squats, deadlifts, bench, rows, overhead press) 3–4 times per week. Don't abandon heavy training just because you're losing weight — that's when it matters most. Progressive overload signals your body to keep muscle even in a deficit.
Track strength, not just weight. If your squat is holding at 225 while your bodyweight drops from 240 to 200, you're doing extremely well. The scale will go down; the goal is keeping the bar where it is.
Don't cut calories on top of the drug. This is the biggest mistake athletes make. The medication already creates a significant deficit through appetite suppression. Stacking additional restriction on top increases muscle loss risk and tanks performance. Eat to your protein target and let the drug handle the deficit.
Creatine, electrolytes, and hydration. GLP-1 drugs can affect hydration and electrolyte balance. Creatine monohydrate (3–5g daily) supports muscle retention and training performance. Stay on top of sodium, potassium, and magnesium — especially if you're experiencing GI side effects.
Time your injection strategically. Some users find appetite suppression peaks 1–2 days after injection. If you have demanding training sessions or need to eat more on certain days, timing your injection around your schedule can help.
The bottom line for athletes: these drugs are tools for fat loss, not muscle building. Use them alongside a solid training and nutrition program. Retatrutide may eventually prove superior for body composition, but tirzepatide with proper training and nutrition already produces excellent results for people who take the protocol seriously.
What to Expect: Tirzepatide Week by Week
Since many people considering treatment want to know what the journey actually looks like, here's a realistic timeline based on clinical trial data and real user experiences:
Weeks 1–4 (2.5 mg Starting Dose)
This is the adjustment period. You're on the lowest dose, and the primary goal is letting your body acclimate. Most people notice a moderate reduction in appetite and the beginning of what users call “food noise” quieting down — the constant mental chatter about food starts to fade.
This is primarily an adjustment period. Many people notice appetite changes early, but the scale response varies widely. Focus on tolerability, hydration, and smaller meals.
Side effects, if they occur, are most common during the first 1–2 weeks: mild nausea, some changes in bowel habits, and occasional fatigue. Eating smaller meals, staying hydrated, and avoiding greasy or heavy foods helps significantly.
Weeks 5–8 (5 mg Dose)
The first dose increase. Appetite suppression may become more noticeable. Most users report eating significantly less without effort — not through willpower, but because hunger signals genuinely change. Weight change varies person to person — prioritize protein, resistance training, and side effect management.
If you experienced GI side effects on the starter dose, they may briefly return after the increase before settling within a week or two.
Weeks 9–20 (Titrating Through 7.5 → 10 → 12.5 → 15 mg)
This is where the transformation becomes visible. Dose increases happen every 4 weeks at minimum, and your provider guides the pace based on your tolerance and response. Not everyone needs the maximum 15 mg dose — some people achieve excellent results at 10 mg or 12.5 mg with fewer side effects.
By week 12, the SURMOUNT-1 data shows that 82% of participants had already achieved at least 5% body weight loss. In real-world terms, someone starting at 230 pounds would have lost about 12+ pounds by this point. [^18]
This is also when other health markers start to shift: blood pressure improves, blood sugar stabilizes, energy levels increase, joint pain decreases, and sleep quality gets noticeably better.
Weeks 20–52
You're on your maintenance dose and in a steady state. Weight loss continues but typically at a gradually slower pace as you approach a new equilibrium. The SURMOUNT-1 trial showed weight loss continuing through week 72 at all doses — participants were still losing at the one-year mark.
Side effects have typically stabilized or resolved. Most users report that GI symptoms peak during dose escalation and are minimal on a stable dose.
This is when the lifestyle habits you've built — better nutrition, consistent exercise, improved relationship with food — become the foundation for long-term success.
Beyond Week 52
Long-term data from SURMOUNT-4 shows that continuing treatment maintains weight loss and can produce additional reduction (25.3% total at 88 weeks). The conversation with your provider shifts to long-term management: finding the right maintenance dose, monitoring health markers, and planning for sustainable weight management. [^2]
Retatrutide Clinical Trial Timeline: What to Watch For
Understanding where retatrutide is in its development helps you plan realistically. Here's the timeline:
| Milestone | Status / Expected Date |
|---|---|
| Phase 2 results published | ✓ Complete — NEJM, August 2023 |
| Phase 3 TRIUMPH program launched | ✓ Complete — Enrollment began 2023; initial registrational program: 4 global trials, 5,800+ participants |
| TRIUMPH-4 topline results (obesity + knee OA) | ✓ Complete — Dec 11, 2025 (positive) |
| Seven additional TRIUMPH readouts | Expected throughout 2026 |
| Peer-reviewed TRIUMPH-4 publication | Expected 2026 (at a medical congress + journal) |
| FDA application filing (NDA) | Estimated 2026–2027 (if all trials succeed) |
| FDA review period | Typically 10–12 months after filing |
| Earliest possible commercial availability | 2027 at earliest — realistically could be 2028 |
Source: Eli Lilly press releases [^5]; TRIUMPH program design paper [^4].
The seven Phase 3 readouts expected in 2026 are critical. They'll tell us how retatrutide performs in populations without knee osteoarthritis, in people with type 2 diabetes, at the 4 mg maintenance dose, and for conditions like sleep apnea and metabolic liver disease. If any of those trials show unexpected safety concerns or fail to meet endpoints, the timeline could extend significantly.
We'll update this article as each readout is announced. Bookmark this page if you want to stay current.
Frequently Asked Questions
Is retatrutide FDA approved?▾
No. Retatrutide is an investigational drug currently in Phase 3 clinical trials. It has not been reviewed or approved by the FDA for any condition. The first Phase 3 results (TRIUMPH-4) were reported in December 2025, with seven additional trials expected to report in 2026.
When will retatrutide be available?▾
If Phase 3 trials succeed and FDA review proceeds smoothly, the earliest possible commercial availability would be 2027 or later. This is not a conservative estimate — it’s based on typical regulatory timelines for a new drug application.
Is retatrutide stronger than tirzepatide?▾
Early data suggests retatrutide produces greater average weight loss (28.7% in TRIUMPH-4 vs. 20.9–22.5% for tirzepatide in SURMOUNT-1). However, these come from different trials with different populations and cannot be directly compared. No head-to-head study exists.
Can I buy retatrutide online?▾
Not legally. Any product sold as “retatrutide” is an unregulated research chemical, not the pharmaceutical compound used in clinical trials. The FDA has explicitly stated that retatrutide cannot be used in compounding under federal law.
Can you get retatrutide from a compounding pharmacy?▾
No. The FDA specifically prohibits retatrutide from being compounded. Any compounding pharmacy offering retatrutide is operating illegally.
Can you take tirzepatide and retatrutide together?▾
No. There is no evidence supporting this combination, and combining GLP-1 class drugs increases adverse effect risk. The Zepbound label explicitly warns against co-administration with other GLP-1 receptor agonists.
Does retatrutide preserve muscle better than tirzepatide?▾
Theoretically, the glucagon receptor activation may shift weight loss more toward fat and away from lean mass. However, this has not been definitively proven in large-scale published data. Resistance training and adequate protein intake remain the most evidence-based strategies for muscle preservation on any GLP-1 medication.
Which causes more nausea — tirzepatide or retatrutide?▾
Based on available data, retatrutide at the highest doses appears to cause more frequent GI side effects. TRIUMPH-4 reported nausea in 43.2% of the 12 mg group, compared to 28% in the FDA’s pooled Zepbound data for the 15 mg tirzepatide group.
What is the new safety signal with retatrutide?▾
TRIUMPH-4 identified dysesthesia (abnormal skin sensations like tingling or numbness) as a new adverse event not seen in earlier Phase 2 trials. Analysts are monitoring this signal, though Lilly noted it did not appear to lead to treatment discontinuation.
How much does tirzepatide cost per month?▾
It depends on your access pathway. With commercial insurance and a savings card, as low as $25/month. Through LillyDirect self-pay vials, $299–$449/month depending on dose (2.5 mg $299, 5 mg $399, 7.5–15 mg $449). Through telehealth providers, pricing varies. The brand-name list price is $1,086/month, but few people pay that.
Will Medicare cover tirzepatide for weight loss?▾
Lilly has announced that starting as early as April 2026, Medicare beneficiaries will pay no more than $50 per month for Zepbound under an agreement with the U.S. government. This is contingent on program rollout and FDA approval of a new multi-dose pen format; timelines may vary.
Is tirzepatide better than semaglutide?▾
In the head-to-head SURMOUNT-5 trial, tirzepatide produced 47% greater relative weight loss than semaglutide — 20.2% vs. 13.7% at 72 weeks. However, semaglutide has longer-term safety data and published cardiovascular outcome results from the SELECT trial.
What happens if I stop taking tirzepatide?▾
The SURMOUNT-4 trial showed that stopping tirzepatide leads to significant weight regain, while continuing treatment maintains and extends weight loss (25.3% total at 88 weeks). Obesity is a chronic condition that typically requires ongoing management.
Who should NOT take tirzepatide?▾
Tirzepatide is contraindicated in anyone with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). It should be used with caution in people with a history of pancreatitis, gallbladder disease, or severe GI conditions. It is not approved for use in children. Pregnant women or those planning pregnancy should not use it.
What about tirzepatide and pregnancy?▾
Tirzepatide is not recommended during pregnancy. Based on animal studies, there may be risks to the fetus. If pregnancy is recognized, the prescribing information advises discontinuing tirzepatide and discussing risks and alternatives with a healthcare provider. Important for women using oral contraceptives: Tirzepatide delays gastric emptying, which may affect absorption of birth control pills. The FDA prescribing information advises patients using oral contraceptives to switch to a non-oral method or add a barrier method for 4 weeks after starting tirzepatide and for 4 weeks after each dose increase.
Are the weight loss results permanent?▾
The medication produces weight loss for as long as you take it, and stopping typically leads to some degree of regain. However, lifestyle changes made during treatment — diet, exercise, behavioral habits — can help sustain a meaningful portion of results. Many people work with their provider to find a lower maintenance dose for long-term management.
Is tirzepatide the same as semaglutide?▾
No. Tirzepatide is a dual GLP-1/GIP receptor agonist (two targets). Semaglutide is a GLP-1 receptor agonist only (one target). They are different drugs made by different companies (Eli Lilly vs. Novo Nordisk) with different clinical profiles. Tirzepatide produces more weight loss in head-to-head comparison.
What does "triple agonist" mean for retatrutide?▾
It means retatrutide activates three hormone receptors: GLP-1, GIP, and glucagon. Each receptor contributes a different mechanism — appetite suppression, metabolic optimization, and increased energy expenditure. This is why retatrutide is sometimes called the “next generation” beyond dual agonists like tirzepatide.
Does tirzepatide help with sleep apnea?▾
Yes. Tirzepatide (Zepbound) received FDA approval in December 2024 for moderate-to-severe obstructive sleep apnea in adults with obesity. It is the first GLP-1 medication approved for this condition.
How fast do you lose weight on tirzepatide?▾
Most people begin noticing weight loss within the first 4–8 weeks. A post-hoc analysis of SURMOUNT-1 found that 82% of participants were “early responders” (achieving ≥5% weight loss by week 12), and even 90% of “late responders” eventually achieved clinically meaningful weight loss by week 72 if they continued treatment.
What if I can’t afford brand-name Zepbound?▾
You have options: LillyDirect self-pay vials ($299–$449/month depending on dose), the Zepbound Savings Card (for commercially insured patients), telehealth providers offering competitive pricing, or discussing alternatives with your doctor. Lilly has also announced a $50/month Medicare cap, contingent on program rollout.
Is retatrutide being studied for conditions other than weight loss?▾
Yes. The TRIUMPH program includes trials for type 2 diabetes, knee osteoarthritis, obstructive sleep apnea, chronic low back pain, cardiovascular and renal outcomes, and metabolic liver disease (MASLD).
How does the glucagon receptor in retatrutide increase fat burning?▾
Glucagon activates pathways in the liver and fat tissue that break down stored triglycerides (fat) and convert them into usable energy. It also increases thermogenesis — the process by which your body generates heat and burns calories. In a weight loss context, this means your body is actively burning more stored fat even while you’re eating less from the GLP-1 and GIP components. Think of it as the third leg of a stool: GLP-1 reduces intake, GIP improves metabolic processing, and glucagon increases output.
How many TRIUMPH trials are there?▾
The initial TRIUMPH Phase 3 registrational program includes four global trials (TRIUMPH-1 through TRIUMPH-4) that together enrolled over 5,800 participants across multiple conditions. Lilly is also studying retatrutide in additional Phase 3 trials for type 2 diabetes, chronic low back pain, cardiovascular/renal outcomes, and metabolic liver disease. Seven additional Phase 3 readouts are expected in 2026.
What is the "food noise" people talk about with GLP-1 drugs?▾
“Food noise” is not a medical term — it’s a widely used patient-reported description of the persistent, intrusive thoughts about food that many people with obesity experience. Things like constantly thinking about your next meal, craving specific foods, or feeling pulled toward eating even when you’re not physically hungry. Many tirzepatide users report that this food noise dramatically decreases or disappears within the first few weeks of treatment, which they describe as one of the most transformative aspects of the medication.
What is the difference between Mounjaro and Zepbound?▾
Mounjaro and Zepbound are both tirzepatide — the exact same active ingredient at the same doses, made by the same manufacturer (Eli Lilly). The difference is the FDA-approved indication: Mounjaro is approved for type 2 diabetes, and Zepbound is approved for chronic weight management and obstructive sleep apnea. Which one you’re prescribed depends on your condition, and insurance coverage may differ between the two.
What is the difference between the efficacy estimand and treatment-regimen estimand?▾
These are two ways of analyzing clinical trial results. The efficacy estimand estimates what would happen if all participants stayed on the medication for the full trial — essentially modeling perfect adherence. The treatment-regimen estimand reflects what actually happened, including people who stopped early. The efficacy estimand always shows higher weight loss numbers. For SURMOUNT-1 (tirzepatide 15 mg): efficacy estimand = 22.5%, treatment-regimen estimand = 20.9%. For TRIUMPH-4 (retatrutide 12 mg): efficacy estimand = 28.7%, treatment-regimen estimand = 23.7%.
How long do tirzepatide side effects last?▾
For most people, GI side effects (nausea, diarrhea, constipation) peak during the first 1–2 weeks of each dose increase and significantly decrease as the body adjusts. By the time you’ve been on a stable dose for 3–4 weeks, side effects are typically mild or absent. In the FDA’s pooled clinical trial data, only 6.7% of participants on 15 mg discontinued due to adverse events, and the majority of adverse events were categorized as mild to moderate.
Is compounded tirzepatide safe?▾
Compounded tirzepatide from a state-licensed compounding pharmacy with a valid prescription is a legal option. However, the FDA has raised concerns about quality control with some compounders, including improper storage during shipping, dosing errors, and even fraudulent products. If you go the compounded route, verify that the pharmacy is state-licensed, ask about their quality testing, and ensure cold-chain shipping is maintained.
What does "investigational" mean?▾
When a drug is described as “investigational,” it means it is being studied in clinical trials but has not yet been approved by the FDA. Investigational drugs have not been determined to be safe and effective by the FDA, even if early results look promising. You cannot legally purchase an investigational drug — the only way to access one is through enrollment in a clinical trial. Retatrutide is currently investigational.
Are there alternatives if I can’t afford or access tirzepatide?▾
Yes. Semaglutide (Wegovy for weight loss, Ozempic for diabetes) is another GLP-1 medication with strong weight loss data. It produces less weight loss than tirzepatide on average (about 13.7% vs. 20.2% in the head-to-head SURMOUNT-5 trial), but it’s still a significant and meaningful amount. Discuss all available options — including lifestyle interventions, other medications, and insurance pathways — with your healthcare provider.
How do I enroll in a retatrutide clinical trial?▾
Visit ClinicalTrials.gov and search for “retatrutide” or “TRIUMPH” to find active trials near you. Each trial has specific eligibility criteria (age, BMI, health conditions, etc.). Enrollment in a clinical trial means you might receive retatrutide or a placebo — there’s no guarantee you’ll get the active drug. Talk to your doctor about whether clinical trial participation makes sense for your situation.
The Bottom Line
If you came to this page asking “tirzepatide vs retatrutide — which should I choose?” the answer is more straightforward than most comparison articles make it seem.
Retatrutide's early data is remarkable. A 28.7% average weight loss in TRIUMPH-4 is the strongest result ever reported for a weight loss medication. The triple-agonist mechanism is scientifically elegant, and Eli Lilly's decision to study it across multiple major conditions reflects genuine confidence in the compound. There's a real chance retatrutide becomes the gold standard of obesity treatment within a few years.
But “a real chance it becomes the gold standard in a few years” is fundamentally different from “an FDA-approved medication you can start taking this week.”
Tirzepatide has been through the full gauntlet. Multiple Phase 3 trials with over 5,000 participants. FDA review and approval. A head-to-head trial proving superiority over semaglutide. Published cardiovascular outcomes data (SURPASS-CVOT). Substantial real-world use since FDA approval. Post-market safety surveillance. An expanding set of indications including sleep apnea. An increasingly accessible cost landscape with options from $25 to $449 per month depending on your pathway.
For most people reading this, the math is simple: start what's proven now, and switch to something better later if the science supports it. That's not settling — it's smart medicine. Every month of treatment produces real health benefits. Every month of waiting does not.
“I started at 204 pounds and now at 126 pounds. It has been amazing for me. I added weight lifting and power walking, plus I cook my own meals now. Must take lots of FIBER! I have five more pounds to go.” — User review on Drugs.com [^9]
If you're ready to take the next step, a licensed telehealth provider can help you find out if you qualify for tirzepatide in minutes.
MEDVI
Sources and Methodology
How We Wrote This Article
Medical and regulatory statements in this article are based on primary sources (FDA prescribing information, peer-reviewed publications in the New England Journal of Medicine and other indexed journals, official Eli Lilly press releases and pipeline disclosures, and FDA safety communications). We also include clearly labeled patient-reported experience quotes from third-party review platforms for context. Trial data is referenced with specific study names and registry identifiers.
Where data comes from press releases (such as TRIUMPH-4 topline results), we have labeled it as such. Press release data has not undergone peer review and may be presented differently in eventual publications. We do not provide dosing guidance for retatrutide because it is an investigational drug not approved for human use outside clinical trials.
We do not provide guidance on obtaining unregulated peptides, grey-market products, or any substance not dispensed by a licensed pharmacy with a valid prescription.
This article will be updated as new TRIUMPH trial results are released in 2026 and as FDA regulatory decisions progress.
References
- [^1]: Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(3):205-216. doi:10.1056/NEJMoa2206038 — PubMed
- [^2]: Aronne LJ, Sattar N, Horn DB, et al. Continued treatment with tirzepatide for maintenance of weight reduction in adults with obesity: The SURMOUNT-4 randomized clinical trial. JAMA. 2024;331(1):38-48. — PubMed
- [^3]: Aronne LJ, Bade Horn D, le Roux CW, et al. Tirzepatide as compared with semaglutide for the treatment of obesity. N Engl J Med. 2025;393(1). doi:10.1056/NEJMoa2416394 — PubMed
- [^4]: Giblin K, Kaplan LM, Somers VK, et al. Retatrutide for the treatment of obesity, obstructive sleep apnea and knee osteoarthritis: Rationale and design of the TRIUMPH registrational clinical trials. Diabetes Obes Metab. 2025. — PubMed
- [^5]: Eli Lilly. Lilly's triple agonist, retatrutide, delivered weight loss of up to an average of 71.2 lbs along with substantial relief from osteoarthritis pain in first successful Phase 3 trial. Press release, Dec 11, 2025. — PR Newswire
- [^6]: Jastreboff AM, Kaplan LM, Frías JP, et al. Triple–hormone-receptor agonist retatrutide for obesity — A phase 2 trial. N Engl J Med. 2023;389(6):514-526. doi:10.1056/NEJMoa2301972 — PubMed
- [^7]: FDA. FDA's concerns with unapproved GLP-1 drugs used for weight loss. Updated February 2026. — FDA.gov
- [^8]: HCPLive. TRIUMPH-4: Retatrutide delivers weight loss, knee osteoarthritis pain relief. Dec 2025. — HCPLive
- [^9]: Drugs.com. Zepbound user reviews for weight loss. Zepbound rated 8.8/10, 82% positive, 450+ reviews. — Drugs.com
- [^10]: FDA. Zepbound (tirzepatide) prescribing information. Revised 2025. — FDA AccessData
- [^11]: BioSpace. Lilly's retatrutide scores triple trial triumph with 26% weight loss, but new safety signal emerges. Dec 12, 2025. — BioSpace
- [^12]: Eli Lilly. Zepbound cost information. — pricinginfo.lilly.com/zepbound
- [^13]: GoodRx. Zepbound pricing and coupons. — GoodRx
- [^14]: Eli Lilly. Lilly and U.S. government expand access to obesity medicines. Nov 6, 2025. — Lilly Investor Relations
- [^15]: Eli Lilly. Lilly lowers the price of Zepbound single-dose vials. Dec 1, 2025. — Lilly Investor Relations
- [^16]: ClinicalTrials.gov. TRIUMPH-4: A Study of Retatrutide (LY3437943) in Participants With Obesity and Knee Osteoarthritis. NCT05931367. — ClinicalTrials.gov (trial registry)
- [^17]: WebMD. Zepbound (tirzepatide) patient reviews and ratings. — WebMD Reviews
- [^18]: Aronne LJ, et al. Weight reduction over time in tirzepatide-treated participants by early weight loss response: Post hoc analysis of SURMOUNT-1. — PubMed
Update Log
We commit to keeping this article current as the science evolves. Major updates:
- February 11, 2026 — Article published with Phase 3 TRIUMPH-4 data (Dec 2025), updated Zepbound pricing (including LillyDirect vials and Medicare access agreement), current FDA guidance on retatrutide compounding prohibition, and verified user reviews.
- Next planned update: When the next TRIUMPH Phase 3 readout is announced (expected H1 2026).
Have we missed something or gotten a fact wrong? Contact us at [email protected] — we take accuracy seriously, especially on health topics.
This content is for informational purposes only and does not constitute medical advice. Always consult a licensed healthcare provider before starting, stopping, or changing any medication. Tirzepatide and retatrutide carry specific risks and contraindications — review the full prescribing information and discuss your individual health situation with your doctor.
Affiliate Disclosure: Some links in this article are affiliate links to telehealth providers. If you use these links to sign up, we may earn a commission at no additional cost to you. We only recommend providers we believe offer legitimate, safe access to FDA-approved medications.
Full Medical Disclaimer · Advertising Disclosure · Editorial Standards
Related Articles
Retatrutide
Everything known about retatrutide, the triple-agonist GLP-1 drug in clinical development.
GLP-3 Retatrutide
How retatrutide's triple receptor mechanism differs from current GLP-1 and GIP agonists.
GLP-3 Retatrutide Alternatives: 4 Legal Options (2026)
Retatrutide isn't FDA-approved — but 4 legal alternatives are. Compare Zepbound, Wegovy pill, Foundayo, prices & how to start in 2026.
What Is GLP-1?
A complete beginner's guide to GLP-1 medications, how they work, and what to expect.
Best GLP-1 Telehealth Providers
Ranked and reviewed telehealth providers offering GLP-1 prescriptions for weight loss online.
GLP-1 Cost With & Without Insurance
Complete guide to GLP-1 medication costs including cash prices, insurance coverage, and savings tips.