Disclosure: Some links on this site are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you. Thank you for supporting our site.·For informational purposes only—not medical advice.
Long-Term Effects of GLP-1 Drugs: Benefits, Risks, and What Happens Over Time
The Bottom Line (Read This First)
If you’re searching for the long-term effects of GLP-1 drugs — whether you’re already on Ozempic, Wegovy, Mounjaro, or Zepbound, or you’re weighing whether to start — here’s what the evidence actually shows as of early 2026:
The benefits are real.
Semaglutide 2.4 mg (Wegovy) reduced major cardiovascular events by 20% in the SELECT trial (17,604 people with overweight/obesity and existing heart disease). GLP-1 drugs as a class improve blood sugar control, protect kidneys in certain populations, and may even lower dementia risk. These aren’t small claims — they’re backed by some of the largest drug trials ever conducted.
The risks are real too, but mostly manageable.
Gastrointestinal side effects hit roughly 40% of users early on but usually improve. Serious risks — pancreatitis, gallbladder disease, kidney injury from dehydration — are uncommon but documented. The thyroid cancer warning is based on animal data, and human evidence remains inconclusive after nearly 20 years of use.
The biggest thing most people don’t realize:
A 2026 BMJ systematic review of 37 studies found that, on average, people who stop semaglutide or tirzepatide regain weight at roughly 0.8 kg per month, with projections suggesting return to baseline weight in roughly 1.5–2 years for many patients. This isn’t a failure of willpower — it reflects that obesity operates like a chronic condition requiring ongoing treatment.
And there are things we genuinely don’t know yet.
We have limited controlled data beyond four years for weight-loss use. Long-term thyroid cancer risk in humans remains an open question. A newly confirmed rare side effect — a type of sudden vision loss called NAION — was added to regulatory guidance in 2025.
We built this guide to be the page we wish existed when we started researching this topic: every long-term effect organized by body system, graded by evidence quality, with honest unknowns clearly marked. Below, we’ll walk through all of it — what’s proven, what’s emerging, and what’s still a question mark — so you can have a real conversation with your doctor instead of guessing.
Jump to what matters most to you:

Images are for illustrative purposes only. Actual product packaging and pen design may vary. Trademarks belong to their respective owners.
Long-Term Effects Dashboard
Before we dive deep, here’s the full picture at a glance. We graded each effect by the strength of evidence behind it — because a headline and a clinical trial are not the same thing.
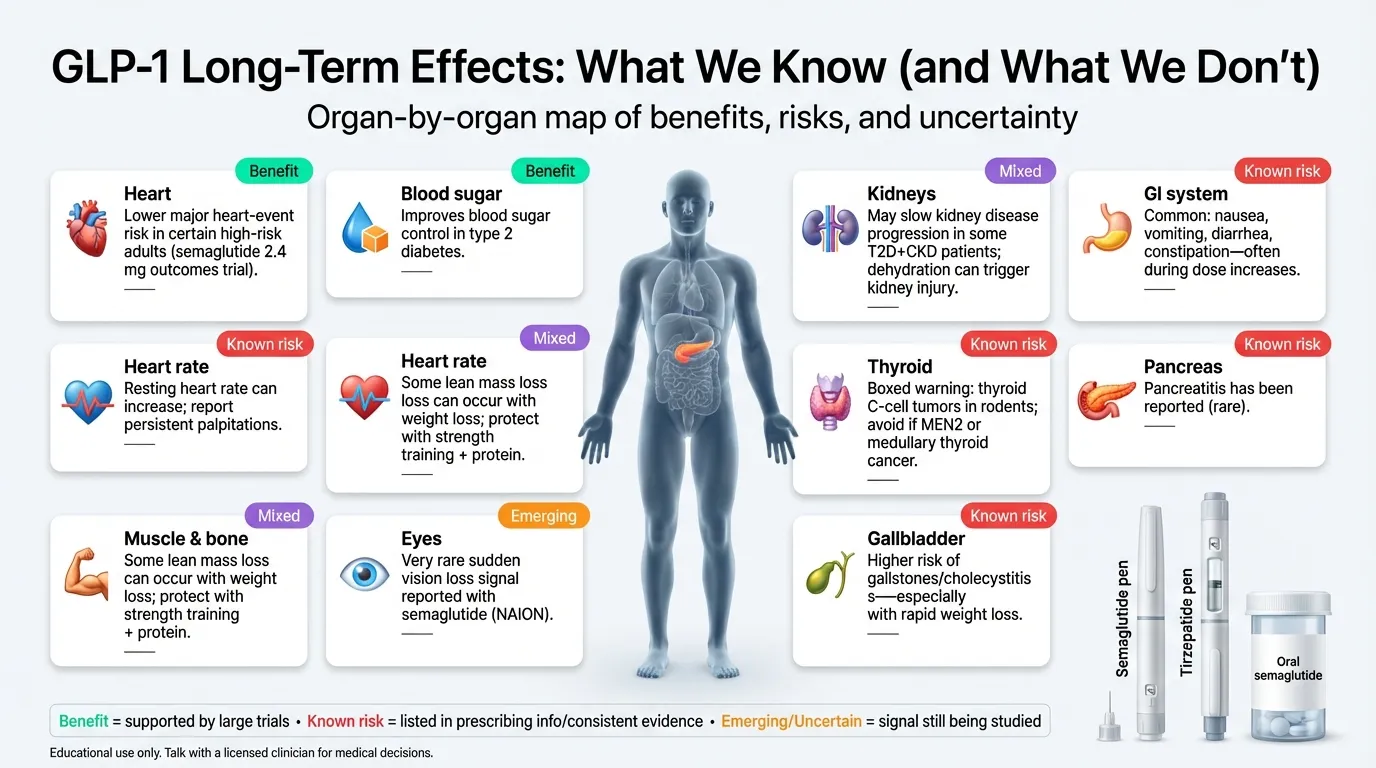
| Body System | Long-Term Effect | Benefit or Risk? | Evidence Strength | Time Studied | Key Source |
|---|---|---|---|---|---|
| Heart & blood vessels | ~20% reduced risk of heart attack, stroke, CV death (semaglutide) | ✓ Benefit | Strong (SELECT trial, 17,604 patients) | Mean follow-up 39.8 months | Lincoff et al., NEJM 2023 |
| Heart rate | Resting heart rate can increase (labeled warning) | ⚠ Risk | Moderate (FDA labeling) | Ongoing | Wegovy prescribing information |
| Blood sugar | Sustained HbA1c reduction of 1.5–2.0% | ✓ Benefit | Strong (multiple large RCTs) | 2+ years | ADA; STEP/SUSTAIN/SURPASS trials |
| Kidneys | Reduced CKD progression in T2D; but dehydration-related injury possible | ⚠ Mixed | Moderate | 2+ years | FDA label (Jan 2025); Xie et al., Nature Medicine 2025 |
| Liver | Reduced fatty liver / NASH resolution | ✓ Benefit | Moderate (Phase II/III data) | 72 weeks | Newsome et al., NEJM 2021 |
| GI system | Nausea, vomiting, constipation, gastroparesis risk | ⚠ Risk | Strong (confirmed across all major trials) | Ongoing | Jalleh et al., JCI 2026 |
| Pancreas | Pancreatitis has been reported; evidence is mixed (small/uncertain increase in meta-analyses) | ⚠ Risk | Mixed | 1–5 years | Xie et al., Nature Medicine 2025; Jalleh et al., JCI 2026 |
| Gallbladder | Increased gallstone/cholecystitis risk | ⚠ Risk | Moderate | 6+ months | FDA prescribing info; multiple RCTs |
| Thyroid | C-cell tumor warning (boxed); human cancer risk inconclusive | ⚠ Uncertain | Limited (animal data + signals) | Ongoing | FDA boxed warning; JCI 2026 |
| Eyes / vision | NAION (rare sudden vision loss) — newly confirmed | ⚠ Risk (very rare) | Emerging | Postmarketing | EMA PRAC 2025; MHRA 2026; JAMA Ophthalmology 2024 |
| Muscle & body composition | ~20–25% of weight lost is lean mass | ⚠ Risk | Moderate | 1–2 years | UC Davis 2025; Metabolism 2025 |
| Bones | Possible bone density loss (rapid weight loss + nutrient gaps) | ⚠ Emerging | Limited | 1–2 years | JCI 2026; UC Davis 2025 |
| Brain & cognition | Reduced dementia and Alzheimer’s risk; food noise reduction | ✓ Benefit | Emerging | Observational | Xie et al., Nature Medicine 2025 |
| Addiction & cravings | Reduced alcohol, nicotine, and substance cravings | ✓ Emerging benefit | Early | Observational | Nature Medicine mapping studies |
| Mental health | No confirmed link to suicidality (FDA removed warning Jan 2026) | Neutral | Moderate | Postmarketing | FDA Jan 2026 |
| Weight after stopping | ~2/3 of lost weight regained within 1–2 years | ⚠ Risk | Strong | 1–2 years post-stop | STEP 1 extension; BMJ 2026 systematic review |
✓ = Benefit supported by strong evidence · ⚠ = Risk or emerging signal · Evidence grading based on study design, sample size, and replication.
What Are GLP-1 Drugs, and Which Ones Are We Talking About?
GLP-1 stands for glucagon-like peptide-1. It’s a hormone your gut naturally releases after you eat. It tells your pancreas to make insulin, signals your brain that you’re full, and slows how fast food leaves your stomach.
GLP-1 drugs are synthetic versions of this hormone — engineered to last much longer than the natural version (hours or days instead of minutes). That amplified effect is what drives both the benefits and the side effects.
Here are the most commonly used FDA-approved GLP-1–based medications discussed in this guide (availability can change; some older products have been discontinued):
| Brand Name | Generic | Type | Approved For | How Taken |
|---|---|---|---|---|
| Ozempic | Semaglutide | GLP-1 RA | Type 2 diabetes | Weekly injection |
| Wegovy | Semaglutide | GLP-1 RA | Obesity; CV risk reduction; MASH | Weekly injection |
| Wegovy tablets | Semaglutide | GLP-1 RA | Obesity; CV risk reduction | Daily pill (approved Dec 2025) |
| Rybelsus | Semaglutide | GLP-1 RA | Type 2 diabetes | Daily pill |
| Mounjaro | Tirzepatide | GLP-1/GIP dual | Type 2 diabetes | Weekly injection |
| Zepbound | Tirzepatide | GLP-1/GIP dual | Obesity | Weekly injection |
| Saxenda | Liraglutide | GLP-1 RA | Obesity | Daily injection |
| Victoza | Liraglutide | GLP-1 RA | Type 2 diabetes | Daily injection |
| Trulicity | Dulaglutide | GLP-1 RA | Type 2 diabetes | Weekly injection |
Note: Some older GLP-1 drugs (Byetta, Bydureon BCise, Adlyxin) have been discontinued in the US.
One important note: Tirzepatide (Mounjaro/Zepbound) technically hits two receptors — GLP-1 and GIP — making it a “dual incretin.” It shows up in GLP-1 conversations because the effects overlap substantially, but the long-term profile may differ in ways we’re still learning.
Dosing matters for long-term effects. Weight-loss doses are typically higher than diabetes doses. Wegovy uses semaglutide at 2.4 mg weekly; Ozempic maxes at 2.0 mg. Higher doses generally mean more pronounced effects — both the good ones and the GI side effects, especially during dose escalation.
How GLP-1 Drugs Work (and Why the Same Mechanism Causes Both Benefits and Side Effects)
Understanding the mechanism helps demystify why these drugs do so many things at once — and why side effects aren’t just “bugs” but features of the same biology.
In the gut: GLP-1 drugs slow gastric emptying — food sits in your stomach longer, so you feel full longer. This is also why nausea, constipation, and in rare cases gastroparesis happen. It’s the same lever.
In the pancreas: They boost insulin release when blood sugar is high and suppress glucagon (a hormone that raises blood sugar). This is the original diabetes application — and why hypoglycemia risk increases if combined with insulin or sulfonylureas.
In the brain: GLP-1 receptors exist in the hypothalamus (hunger regulation), brainstem (nausea center), and reward pathways. This is why people report reduced “food noise” — that constant background hum of thinking about food — and why emerging research suggests effects on alcohol and nicotine cravings.
In the heart, kidneys, and beyond: GLP-1 receptors aren’t limited to metabolism. They’re on immune cells, in the heart, in the kidneys. This explains the cardiovascular benefits, the anti-inflammatory effects, and the kidney protection — but also why effects can show up in unexpected places.
Why this matters for long-term thinking: Because GLP-1 receptors are distributed across so many organ systems, these drugs don’t just “do one thing.” They create a cascade of effects — some intentional, some incidental, some we’re still discovering. The cardiovascular benefits, the brain effects on appetite and cravings, the kidney protection, the liver improvements — these all trace back to the same receptor system. And so do the side effects: the nausea, the gastroparesis risk, the gallbladder issues. Understanding this helps you see why both the benefits and risks are real and interconnected — not random.
What “tolerance” looks like over time: The GI side effects (especially nausea) are worst during the first 4–8 weeks and during dose increases. In the liraglutide pivotal trial, nausea dropped from 25% at week 4 to about 5.5% at week 56. Your body adapts to the GI effects. The metabolic benefits, on the other hand, tend to persist as long as you continue treatment — which is precisely why stopping often leads to regain.
The “evidence bucket” problem you should know about: When you read about GLP-1 long-term effects, the claims come from very different types of evidence, and they don’t all carry the same weight. FDA label warnings are based on the strongest evidence the regulators had at approval. Large randomized controlled trials (like SELECT or STEP) are the gold standard but typically last 1–3 years. Observational studies (like the WashU atlas) can spot patterns in millions of patients but can’t prove causation. And postmarketing reports (individual adverse events reported to the FDA) can surface rare problems but are vulnerable to reporting bias. Throughout this guide, we’ve tried to be clear about which bucket each claim comes from.
What Are the Proven Long-Term Benefits?
To be specific about what “proven” means here: supported by large randomized controlled trials or confirmed by regulatory approval based on outcomes data.
Heart and Cardiovascular Protection
This is the strongest long-term benefit in the evidence base.
The SELECT trial enrolled 17,604 adults with overweight or obesity (without diabetes) who had existing cardiovascular disease. Semaglutide 2.4 mg reduced the risk of major cardiovascular events — heart attack, stroke, or cardiovascular death — by 20% over a mean follow-up of 39.8 months. This was significant enough that the FDA approved Wegovy specifically for cardiovascular risk reduction in March 2024.
A large observational study using the TriNetX Global Network (published 2024) propensity-matched 12,123 individuals with obesity (without diabetes) who started GLP-1 drugs against non-users. GLP-1 use was associated with significantly lower rates of all-cause mortality, ischemic heart disease, heart failure, arrhythmias, stroke, and atrial fibrillation. However, this is observational data with inherent limitations — the effect sizes were very large (HR 0.23 for mortality), which likely reflects residual confounding in addition to real benefit. The direction is consistent with trial data, but the magnitude should be interpreted cautiously.
Notably, the cardiovascular benefit in SELECT emerged early — within months — before participants achieved maximal weight loss. This suggests that some of the heart protection may come from anti-inflammatory mechanisms rather than weight loss alone, which is why researchers are studying GLP-1 drugs for heart protection even in people who aren’t overweight.
Kidney Protection
In January 2025, the FDA approved semaglutide (Ozempic) for reducing the risk of kidney disease progression, kidney failure, and cardiovascular death in adults with type 2 diabetes and chronic kidney disease. This was based on the FLOW trial.
A meta-analysis of GLP-1RA trials found semaglutide was associated with a lower risk of composite kidney outcomes (RR ~0.78), confirming the renal benefit seen in individual trials.
The caveat: This benefit is best established in people with type 2 diabetes. In people using GLP-1 drugs purely for weight loss, the kidney picture is more nuanced — dehydration from vomiting or diarrhea can actually cause kidney injury. More on that in the risks section.
Blood Sugar Control
For people with type 2 diabetes, the benefits are well-documented: sustained HbA1c reductions of 1.5–2.0%, reduced progression from prediabetes to diabetes, and improved insulin sensitivity. These effects persist as long as treatment continues.
Liver Health
Semaglutide showed the ability to resolve non-alcoholic steatohepatitis (NASH) without worsening liver fibrosis in a 72-week phase II trial. It reduces liver fat content significantly. For people with fatty liver disease — which often coexists with obesity — this is a meaningful benefit that doesn’t get enough attention.
Brain and Cognitive Health
This is newer and should be labeled as emerging, but the signal is consistent.
The landmark 2025 study from Washington University (published in Nature Medicine) systematically evaluated 175 health outcomes among ~216,000 GLP-1 receptor agonist initiators compared against large comparator groups using the VA database. It found decreased risks of dementia and Alzheimer’s disease. The effect was modest — about 10–20% risk reduction — but notable because few effective treatments exist for these conditions. Because this is observational, it shows associations, not proof of causation.
The mechanism likely involves a combination of reduced brain inflammation, weight loss (which independently improves cognitive function), and direct GLP-1 receptor activation in brain regions involved in memory and cognition.
Other Benefits Under Active Investigation
- Sleep apnea: Reduced severity documented in clinical trials
- Knee osteoarthritis: Reduced pain, likely weight-mediated
- Respiratory disease: A meta-analysis of 77,485 participants showed a 14% lower risk
- Substance use disorders: Emerging data on reduced alcohol, nicotine, and opioid cravings — still observational, not yet proven in RCTs
- Longevity: Nature Biotechnology (November 2025) explored GLP-1 drugs as potential longevity candidates. Interesting but highly speculative at this stage.
Long-Term Side Effects That Are Common (and Usually Manageable)
GI Symptoms: The Price of Admission
Gastrointestinal effects are by far the most common side effect across every GLP-1 drug. The numbers from clinical trials:
| Symptom | Incidence (liraglutide trial) | Timing | Trend Over Time |
|---|---|---|---|
| Nausea | ~40% | Peaks weeks 1–8 | Usually improves significantly by 3–6 months |
| Diarrhea | ~21% | Variable | Often improves |
| Constipation | ~20% | Ongoing for some | May persist |
| Vomiting | ~16% | Peaks during dose escalation | Usually improves |
About 6.5% of trial participants discontinued GLP-1 drugs due to side effects (vs. 3.6% on placebo). Real-world discontinuation rates are higher — up to 50–75% within a year — though that includes people who stop for cost, access, or other reasons, not just side effects.
Practical management that actually helps:
- Eat slowly and stop when satisfied (not full)
- Smaller, more frequent meals
- Stay well hydrated — dehydration makes everything worse and creates real kidney risk
- Avoid lying down right after eating
- Slow dose escalation (discuss with your doctor if you’re miserable during titration)
Hair Loss and “Ozempic Face”
Both of these are primarily caused by rapid weight loss, not the drug itself. Any method that produces significant weight loss — surgery, calorie restriction, or medication — can cause temporary hair shedding (telogen effluvium) and facial volume loss.
The faster the weight loss, the more noticeable these changes tend to be. They’re cosmetic, not dangerous, but they’re worth knowing about. Adequate protein intake (discussed in the muscle section below) may help.
Fatigue, Headache, Dizziness
Common during the first weeks, especially during dose increases. Usually transient. If persistent, it’s worth checking whether you’re eating enough — reduced appetite can lead to under-fueling, which causes these symptoms independently.
Heart Rate Increase
The Wegovy prescribing information notes that semaglutide can increase resting heart rate — typically by 1–4 beats per minute in clinical trials. For most people, this is clinically insignificant. But it’s worth monitoring, especially if you have a pre-existing heart rhythm condition. Mention it to your provider if you notice persistent palpitations or a resting heart rate consistently above your baseline.
Serious Long-Term Risks (Uncommon but Real)
This section is where we want to be extremely careful with language, because the difference between “this might happen” and “this will probably happen to you” matters enormously. We’ll give you the actual numbers where they exist.
Pancreatitis
Pancreatitis is a labeled concern for all GLP-1 drugs and has been reported in clinical use. The evidence on the magnitude of risk is mixed: some observational studies (including the 2025 Nature Medicine atlas) have flagged associations with increased pancreatitis, while meta-analyses of randomized trials show a small, uncertain increase that may not remain significant in all subgroup analyses. Some real-world studies — including in patients with prior pancreatitis — have not found higher recurrence with GLP-1 use.
Context that matters: Pancreatitis was already more common in people with obesity and type 2 diabetes before GLP-1 drugs existed. The baseline risk is low, so even if there is an increase, the absolute number of additional cases is small. But pancreatitis is serious, and patients should know the warning signs.
What about pancreatic cancer? A multi-center analysis of over 1.5 million adults with type 2 diabetes found no increase in pancreatic cancer risk with GLP-1 use. Several sensitivity analyses suggested a modest, non-significant reduction. The FDA and EMA issued a joint statement concluding no confirmed causal link. This remains an area of active surveillance, but the cancer fear appears less supported than the pancreatitis risk.
Warning signs:
Severe abdominal pain (often radiating to the back) with or without vomiting. If you experience this, seek medical attention immediately. Don’t wait.
Gallbladder Disease
GLP-1 drugs increase the risk of gallstones and cholecystitis (gallbladder inflammation). This ranges from asymptomatic stones found incidentally to severe cases requiring surgery.
Why it happens: Two factors converge. First, GLP-1 drugs slow gallbladder motility. Second, rapid weight loss itself changes bile composition and increases stone formation — this happens with any rapid weight-loss method, including bariatric surgery.
Who’s more at risk: People losing weight rapidly, women, those with a history of gallstones.
Kidney Injury
Here’s where the picture gets complicated. On one hand, semaglutide has FDA-approved kidney protection for people with T2D and CKD. On the other hand, the WashU atlas study identified increased kidney problems as a novel finding in some GLP-1 users.
The likely explanation: The kidney risk appears to be largely mediated by dehydration. When GLP-1 drugs cause significant vomiting or diarrhea, and the person doesn’t adequately hydrate, it can lead to acute kidney injury. The FDA label explicitly warns about monitoring renal function in patients with volume depletion.
The takeaway: Stay hydrated. If you can’t keep fluids down, contact your provider. Kidney problems can develop without symptoms until they’re advanced.
Gastroparesis and Severe GI Events
GLP-1 drugs slow gastric emptying by design. In most people, this is temporary and manageable. But in some cases, it can progress to gastroparesis — a condition where the stomach essentially stops emptying properly.
In a JAMA analysis using a large U.S. insurance claims database, the analytic cohorts (thousands of patients using GLP-1s for weight loss, drawn from approximately 16 million records) showed a higher rate of gastroparesis (HR ~3.7) compared with bupropion–naltrexone users. Bowel obstruction, while rare, has also been reported.
The FDA label for semaglutide states the drug is “not recommended” in patients with pre-existing severe gastroparesis.
Surgery and anesthesia note: Because GLP-1 drugs slow stomach emptying, there’s an increased risk of pulmonary aspiration during procedures requiring sedation or general anesthesia. If you’re scheduled for any procedure, tell your surgical team you’re on a GLP-1 drug. Multi-society clinical guidance (October 2024) from ASA, AGA, and three other organizations states that most patients can continue GLP-1 medications before elective surgery, with individualized risk assessment. Patients at higher risk for delayed gastric emptying may be asked to follow a 24-hour liquid diet or other precautions before the procedure. Always coordinate with your anesthesiology and procedural team.
Hypoglycemia
GLP-1 drugs alone rarely cause low blood sugar. The risk increases significantly when combined with insulin or sulfonylureas. If you’re on combination therapy, this is something to monitor carefully and discuss with your prescriber.
Do GLP-1 Drugs Cause Cancer?
This question drives a huge amount of the anxiety behind “long-term effects of GLP-1” searches. Here’s an honest breakdown.
Thyroid Cancer: What the Boxed Warning Actually Means
Every GLP-1 drug carries an FDA boxed warning — the most serious type — about thyroid C-cell tumors. This warning exists because of animal studies: rats and mice treated with liraglutide developed C-cell hyperplasia, adenomas, and carcinomas at doses relevant to human use.
The critical context: Rodent thyroid biology is meaningfully different from human thyroid biology. Rodent C-cells have far more GLP-1 receptors and are much more responsive to GLP-1 stimulation than human C-cells. After nearly 20 years of GLP-1 drug use in humans, we have not seen a clear, confirmed increase in thyroid cancer cases in the data.
However — and this is where honesty matters — the JCI’s comprehensive 2026 safety review (Jalleh et al.) noted that GLP-1 drugs “may confer an increased risk for thyroid cancer” in humans, based on postmarketing signals. The signal is there. It’s just not definitive.
Who must not take GLP-1 drugs because of this: Anyone with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN2). This is an absolute contraindication.
For everyone else: The risk, if it exists in humans, appears to be very small. But it’s a genuine unknown for multi-decade use, and it would be dishonest to pretend otherwise.
Pancreatic Cancer
As mentioned above, large-scale evidence does not support an increased risk. A meta-analysis of 25 studies showed no significant increase with exenatide or liraglutide use. The FDA and EMA concluded no confirmed causal association.
What We Can’t Rule Out Over Decades
GLP-1 drugs at weight-loss doses have been widely used for roughly five years. That’s enough time to establish a safety profile for common events, but not for very rare cancers with long latency periods. Ongoing pharmacovigilance is essential, and the honest answer is: we’ll know more in 10 years than we know today.
Eyes and Vision: The NAION Question
This is one of the newest developments in GLP-1 safety — and one that most existing guides either miss entirely or handle poorly.
What Happened
In July 2024, a study published in JAMA Ophthalmology reported a higher rate of non-arteritic anterior ischemic optic neuropathy (NAION) — sometimes called an “eye stroke” — in patients taking semaglutide compared to non-users. NAION causes sudden, painless vision loss in one eye.
What Regulators Concluded
The European Medicines Agency (EMA) reviewed the evidence and in 2025 concluded that NAION is a very rare side effect of semaglutide medicines (Ozempic, Rybelsus, Wegovy). They advise that patients who develop NAION should stop semaglutide.
In February 2026, the UK’s MHRA issued a drug safety update specifically about this risk, providing patient-action guidance on sudden vision loss.
What This Means for You
NAION is rare in the general population and remains rare even with GLP-1 use. But you should know the warning sign: sudden, painless loss of vision in one eye. If this happens, seek urgent medical care immediately. Don’t wait to see if it improves.
Diabetic Retinopathy Worsening
Separately, the FDA label for semaglutide notes a risk of diabetic retinopathy complications, particularly in patients who achieve rapid glucose improvement. This applies primarily to people with pre-existing diabetic eye disease. If you have diabetes and retinopathy, discuss eye monitoring with your doctor before starting.
What Happens to Your Muscles and Bones?
This section addresses one of the most under-discussed concerns — and one that matters a lot for long-term health.
Muscle Loss: Real, but Needs Context
Here’s the number that circulates in headlines: “Up to 40% of weight lost on GLP-1 drugs is lean mass, not fat.”
Here’s the context that’s usually missing:
First, approximately 20–25% lean mass loss is more typical in most studies. A 2025 review in Metabolism confirmed this range — and noted it’s similar to lean mass loss from any calorie-restricted weight-loss method, including dieting.
Second, as UC Davis exercise physiologist Keith Baar explained in a 2025 analysis: “Much of the reported 40% lean mass loss with GLP-1 use is coming from the liver” — meaning reductions in liver fat mass, not skeletal muscle. Liver fat loss is actually a health benefit, not a problem.
Third, a 2025 review in the European Journal of Pharmacology found that relative lean mass — the proportion of lean mass to total body weight — may actually stay the same or improve during GLP-1 treatment.
That said, muscle preservation matters. Losing muscle lowers your metabolic rate and physical strength, which is especially concerning for older adults. The evidence-based mitigation strategy is straightforward:
- Resistance training 2–3 times per week (the most important intervention)
- Protein intake of 1.2–1.6 grams per kilogram of body weight per day
- Adequate total calories — under-eating defeats the purpose
Learn more: How to Prevent Muscle Loss on GLP-1 Medications
Bone Density
Bone responds to both incretin hormones and mechanical loading (how much weight your skeleton carries). Some evidence suggests GLP-1 receptor activation may be directly protective of bone. But rapid weight loss reduces the mechanical load on bones, and decreased appetite can lead to inadequate intake of bone-building nutrients: calcium, vitamin D, and magnesium.
Semaglutide specifically has shown bone density loss in some studies. Older adults and postmenopausal women face the highest risk.
Practical steps: Ensure adequate calcium and vitamin D intake, do weight-bearing exercise, and discuss bone density screening (DEXA scan) with your doctor if you’re in a higher-risk group.
Mental Health, Mood, and the FDA Update
Suicidal Thoughts: The Question That’s Now Been Answered
In 2023, concerns emerged about whether GLP-1 drugs might increase suicidal ideation. The FDA launched a formal investigation.
In January 2026, the FDA concluded its evaluation and requested the removal of the suicidal behavior and ideation warning from certain GLP-1 receptor agonist labels. Their preliminary evaluation found no evidence that these drugs cause suicidal thoughts or actions.
This doesn’t mean mental health effects don’t exist — it means the specific suicide link wasn’t supported by the evidence.
Depression and Anxiety
Rapid body changes, altered eating patterns, and shifting identity can all affect mood. Some people feel better psychologically after weight loss; others struggle with the adjustment. These are real human experiences, but they’re confounded — meaning it’s hard to separate what the drug does from what the life change does.
The WashU atlas study actually found net improvements in cognitive and behavioral health outcomes across GLP-1 users. But individual experiences vary, and mental health monitoring during any significant medical treatment is good practice.
Addiction and Cravings: An Unexpected Finding
Emerging research suggests GLP-1 drugs may reduce cravings for alcohol, nicotine, and even gambling. The mechanism appears to involve GLP-1 receptors in brain regions responsible for impulse control and reward processing.
This is based primarily on observational data and patient reports — not yet confirmed in large randomized trials. It’s promising, but we’d call it “interesting and worth watching” rather than “proven benefit.” Clinical trials are underway.
What Happens When You Stop Taking GLP-1 Drugs?
This might be the most important section on this page, because it’s the question that determines whether GLP-1 treatment is a short-term tool or a long-term commitment.
The Weight Regain Data
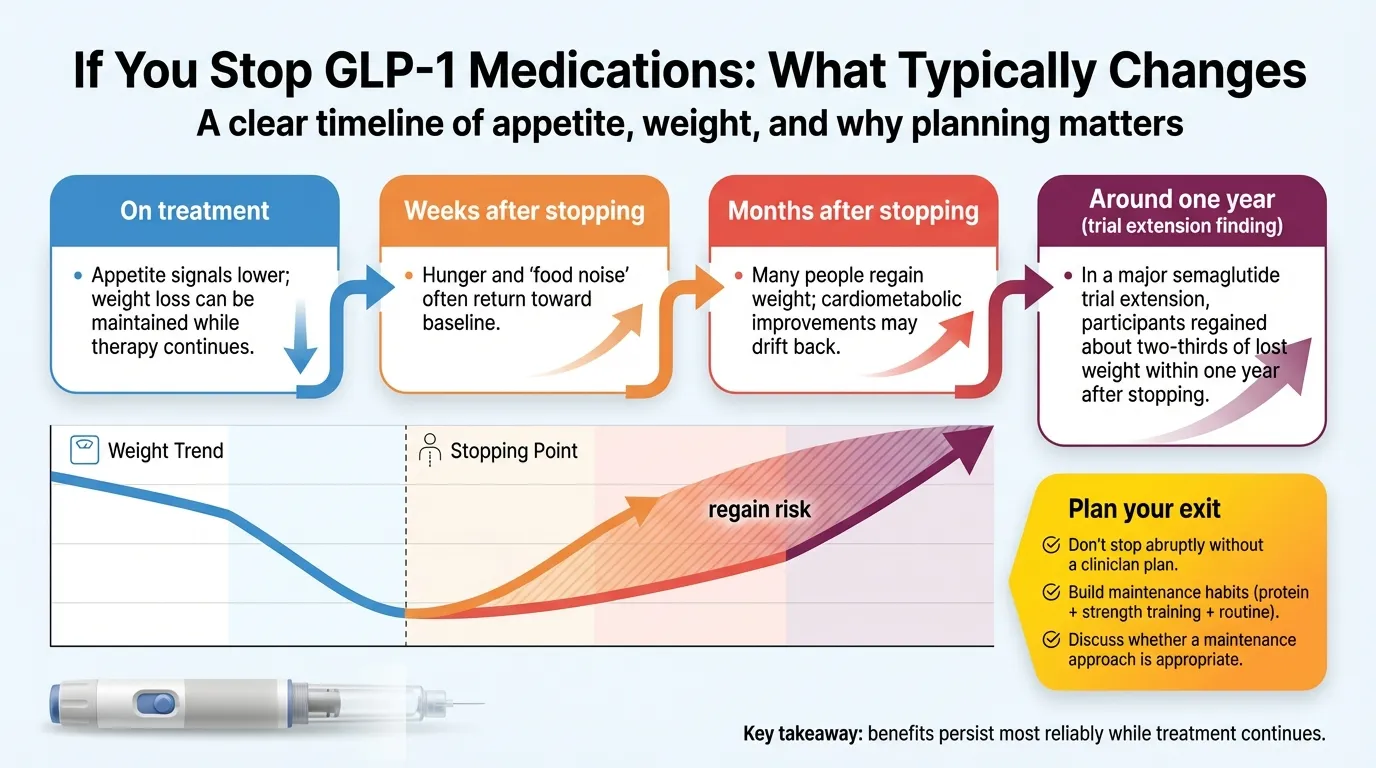
STEP 1 trial extension: One year after stopping semaglutide 2.4 mg, participants regained approximately two-thirds of their prior weight loss. Cardiometabolic improvements (blood pressure, cholesterol, blood sugar) reversed in parallel.
SURMOUNT-4 trial: In SURMOUNT-4, withdrawing tirzepatide led to weight regain in most participants; post hoc analyses report that most regained ≥25% of their initial weight reduction within one year, with greater reversal of cardiometabolic improvements in those who regained more.
BMJ 2026 systematic review (the most comprehensive to date, 37 studies, 9,341 adults): Weight increased by an average of 0.4 kg per month after stopping any weight management medication. For newer drugs like semaglutide and tirzepatide, regain averaged 0.8 kg per month. At that rate, researchers projected return to pre-treatment weight within approximately 1.5–2 years for many patients.
Lancet eClinicalMedicine 2025 meta-analysis (18 RCTs, 3,771 participants): Confirmed significant metabolic rebound — not just weight, but HbA1c, blood pressure, and lipid markers also worsened after discontinuation.

A Real-World Nuance Worth Knowing
Not everyone regains. A Cleveland Clinic real-world study (published in AJMC, 2025) found that 42% of patients who stopped GLP-1 drugs did not regain significant weight at one year. Real-world regain appears to be somewhat slower than clinical trial data suggests, possibly because real-world patients had more modest weight loss to begin with.
Why Regain Happens
This isn’t about willpower. When you lose weight, your body mounts a biological defense: hunger hormones increase, metabolic rate decreases, and appetite-regulating circuits recalibrate to push you back toward your previous weight. GLP-1 drugs override this defense. When you remove the drug, the defense comes back.
The Oxford researchers who conducted the BMJ analysis put it directly: “This isn’t a failing of the medicines — it reflects the nature of obesity as a chronic, relapsing condition.”
What This Means Practically
- Don’t stop abruptly without talking to your doctor. A supervised taper or transition plan is important.
- Lifestyle habits (exercise, nutrition) matter regardless — they support health benefits independent of weight, and may modestly slow regain
- The “maintenance dose” conversation: Some doctors are exploring lower doses or longer dosing intervals for weight maintenance. This is an active area of clinical discussion, not yet well-studied in formal trials.
- The honest framing: For many people, GLP-1 treatment may be long-term — similar to blood pressure medication or statins. This is a conversation to have with your doctor, factoring in benefits, risks, cost, and your personal goals.
Who Should NOT Take GLP-1 Drugs?
Absolute Contraindications
- Personal or family history of medullary thyroid carcinoma (MTC) — the boxed warning
- Multiple Endocrine Neoplasia syndrome type 2 (MEN2)
- Known hypersensitivity to the specific drug or its components
Strong Cautions (Discuss Thoroughly with Your Doctor)
- History of pancreatitis — GLP-1 drugs increase this risk
- Severe gastroparesis or GI motility disorders — these drugs slow gastric emptying further
- Severe kidney disease with active volume depletion risk
- Pregnancy or planning pregnancy — GLP-1 drugs are not recommended during pregnancy. Stop at least 2 months before planned conception (for semaglutide; timing varies by drug — discuss with your prescriber).
- Diabetic retinopathy — discuss eye monitoring before starting, especially with rapid glucose improvement expected
- Birth control interaction (tirzepatide): The FDA label for Zepbound/Mounjaro notes that tirzepatide may reduce the effectiveness of oral contraceptives. Women using oral birth control should discuss alternative or backup contraception with their provider.
Full guide: GLP-1 Contraindications
Are GLP-1 Drugs Safe Long-Term for Non-Diabetics?
This is a specific and important question because most of the longest safety data comes from diabetic populations. People using GLP-1 drugs purely for weight loss deserve a separate answer.
The strongest non-diabetic evidence: An observational cohort study using the TriNetX Network (published 2024) propensity-matched 12,123 individuals with obesity without Type 2 diabetes who started GLP-1 drugs against non-users. GLP-1 treatment was associated with significantly lower all-cause mortality and reduced cardiovascular complications. As observational data, the effect sizes should be interpreted cautiously — but the direction of benefit was consistent across subgroups and regions.
The SELECT trial — the one that got Wegovy its CV risk reduction label — specifically enrolled people with overweight/obesity without diabetes.
What’s different for non-diabetics:
- Higher doses are typically used (weight-loss doses vs. diabetes doses)
- The risk-benefit calculus shifts: diabetes itself causes organ damage, so GLP-1 benefits in diabetics partly come from glucose control. In non-diabetics, the benefits are more about weight reduction, cardiovascular protection, and anti-inflammatory effects.
- Controlled data beyond ~3 years in non-diabetic obesity populations is still limited
Bottom line: The evidence is reassuring so far, but shorter in duration than the diabetes data. Ongoing monitoring and regular check-ins with your provider are important.
What About Compounded GLP-1 Drugs?
Compounded GLP-1 drugs are a growing market — and a growing concern.
The FDA has issued specific warnings about unapproved GLP-1 drugs used for weight loss. Key issues include:
Dosing errors: The FDA received multiple reports of hospitalizations from patients incorrectly measuring and self-administering compounded semaglutide. Some providers also miscalculated doses.
Salt form concerns: Some compounders sell semaglutide sodium or semaglutide acetate — these are different active ingredients than what’s in the FDA-approved drugs. The FDA states they are “not aware of any lawful basis for their use in compounding” and doesn’t have information on whether they have the same properties.
Refrigeration problems: Reports of compounded injectables arriving warm or without adequate cold packing, which can compromise drug quality.
No long-term safety data: All of the studies cited throughout this guide used FDA-approved formulations. No long-term studies exist on compounded versions specifically.
If you’re considering compounded GLP-1 drugs: Ask whether the pharmacy is state-licensed, what formulation they use, how they handle cold-chain shipping, and whether the specific compound has any FDA-reviewed safety data. If a deal seems too good to be true, it might be.
Read more: Is Compounded Semaglutide Safe?
The Long-Term Cost and Access Reality
Cost is one of the top reasons people stop GLP-1 drugs — and stopping, as we’ve covered, usually means regain. So this isn’t a side issue. It’s central to the long-term effects conversation.
What “Long-Term” Costs Look Like
Brand-name GLP-1 drugs have list prices ranging from roughly $900 to $1,500 per month without insurance. Over five years, that’s $54,000 to $90,000. Insurance coverage varies enormously — some plans cover GLP-1 drugs for obesity, many don’t, and prior authorization hoops are common.
If your insurance covers it, your out-of-pocket cost might be reasonable. If it doesn’t, the cost becomes a major factor in whether long-term treatment is realistic.
The “Am I Paying Forever?” Question
If GLP-1 drugs function like chronic disease management (and the weight regain data suggests they do for many people), then yes — ongoing treatment is the current model. This is similar to statins for cholesterol or antihypertensives for blood pressure: the benefit persists while you take the medication.
The pharmaceutical landscape is shifting, though. The Wegovy pill (oral semaglutide 25 mg) was approved in December 2025 and launched in January 2026, offering a daily pill option. The manufacturer’s list price is currently the same for the pill and pen versions; out-of-pocket cost varies by insurance and eligibility for savings programs. Generic and biosimilar competition may eventually lower costs further. And some providers offer dose-reduction maintenance plans that cost less than full treatment doses.
Shortages and Supply Issues
GLP-1 drug shortages have been an ongoing issue since 2022. Demand has outstripped supply, particularly for semaglutide. Shortages can force treatment interruptions, which is concerning given the regain data. If you’re starting treatment, ask your provider about their plan if your medication becomes unavailable.
A Note on “Telehealth-Only” Providers
The explosion of online GLP-1 prescribing has created a wide range of quality. Some telehealth providers offer excellent care with proper monitoring and follow-up. Others are essentially prescription mills — minimal evaluation, no follow-up, no lab work.
For long-term treatment, you need a provider who will monitor you over time, not just one who’ll write a script. We cover what to look for (and what to avoid) in our provider comparison guide.
Your Long-Term Monitoring Plan
If you’re going to be on a GLP-1 drug for months or years, proactive monitoring is the difference between catching problems early and discovering them late. Here’s what good long-term care looks like:
Before Starting (Baseline)
- Complete medical history review (pancreatitis, gallbladder, kidney disease, thyroid conditions, eating disorder history)
- Baseline labs: metabolic panel (kidney function), lipid panel, HbA1c, thyroid function
- Blood pressure, resting heart rate, weight, waist circumference
- Eye exam if you have diabetes with retinopathy
- Discussion of pregnancy planning timeline if applicable
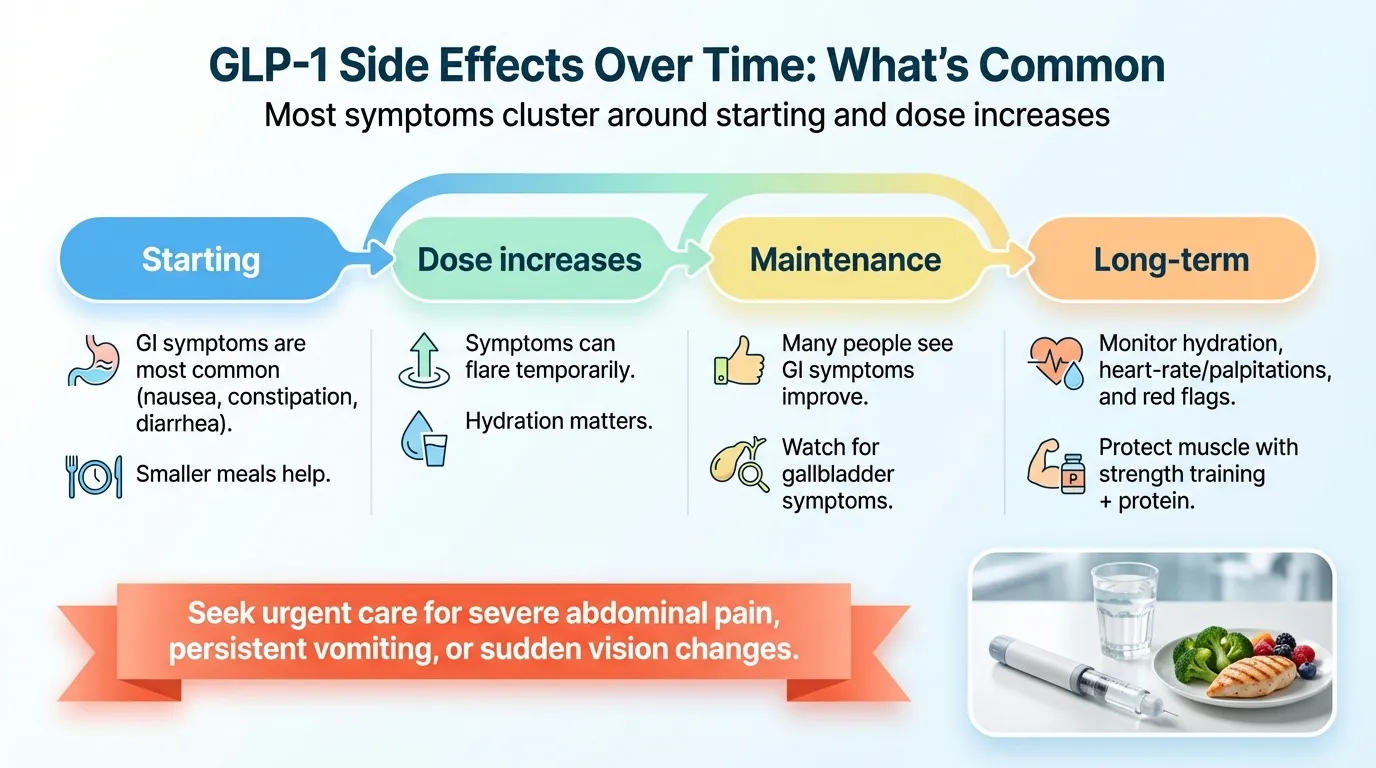
First 3 Months (Titration Phase)
- Check-ins during each dose increase
- Monitor GI symptoms and hydration status
- Watch for red-flag symptoms (severe abdominal pain, persistent vomiting, sudden vision changes)
- Adjust dose escalation pace if side effects are severe
Months 3–12 (Stabilization)
- Lab work: kidney function, metabolic panel (every 3–6 months per provider guidance)
- Weight and body composition tracking
- Assess protein intake and nutritional adequacy
- Begin or maintain resistance training program
- Screen for mental health changes
Year 1+ (Long-Term Maintenance)
- Annual comprehensive labs (metabolic panel, lipid panel, thyroid function, HbA1c)
- Periodic bone density screening if at risk (discuss DEXA scan with your doctor)
- Review whether dose, drug, or treatment plan should be adjusted
- Update monitoring plan based on any new safety data
Red Flag Symptoms: When to Call Your Doctor
| Symptom | Possible Concern | Action |
|---|---|---|
| Severe abdominal pain (especially radiating to back) | Pancreatitis | Seek medical care today |
| Persistent vomiting / can’t keep fluids down | Dehydration → kidney risk | Contact provider same day |
| Sudden painless vision loss in one eye | NAION | Urgent / emergency care |
| Lump or swelling in neck, hoarseness, difficulty swallowing | Thyroid concern | Schedule appointment promptly |
| Yellowing skin/eyes, dark urine | Gallbladder / liver issue | Contact provider same day |
| Signs of severe allergic reaction (swelling, difficulty breathing) | Allergic reaction | Emergency care immediately |
Questions to Bring to Your Next Appointment
- What monitoring should I be getting, and how often?
- Am I at higher risk for any specific long-term effect based on my health history?
- What’s our plan if I want to stop or reduce my dose?
- Should I be taking any supplements (calcium, vitamin D, protein)?
- What exercise program do you recommend to protect muscle and bone?
- Should we track my resting heart rate?
- What symptoms should prompt an immediate call?
- When should we re-evaluate whether this medication is still the right choice?
What We Honestly Don’t Know Yet
This section is here because we believe transparency builds more trust than reassurance. Here are the genuine open questions in GLP-1 long-term safety as of early 2026:

- Effects beyond 4–5 years for weight-loss use. The diabetes data goes back nearly 20 years. The weight-loss-at-high-doses data does not. We’re in the “first generation” of long-term users at obesity doses.
- Lifetime safety. If these are truly “forever medications” for many people, we need 10-20 year data that doesn’t exist yet.
- Thyroid cancer in humans over decades. The animal signal is concerning. The human data is reassuring so far. But “so far” covers a limited window.
- Pregnancy outcomes. GLP-1 drugs are contraindicated in pregnancy, but data on optimal timing for stopping before conception is limited. Animal reproduction studies have shown adverse effects.
- Long-term gut microbiome changes. GLP-1 drugs alter gut bacteria composition. The implications of sustained microbiome changes over years are unknown.
- Effects in adolescents over time. Some GLP-1 drugs are approved for ages 12+, but long-term developmental data in growing bodies is sparse.
- Compounding nutritional deficiencies. Reduced calorie intake over years may cumulatively deplete calcium, vitamin D, B vitamins, iron, and protein — but we don’t have long-term nutritional tracking data specific to GLP-1 users.
- Why response varies so much. Some people lose 20%+ of body weight; others lose 5%. Pharmacogenomic research into who responds best is just beginning.
- Next-generation drugs. Triple agonists like retatrutide (targeting GLP-1, GIP, and glucagon receptors) are in clinical trials. They may offer greater efficacy, but their long-term profile is even less established.
- Whether the cardiovascular benefits persist after stopping. We know the weight comes back. Do the heart benefits reverse too? The BMJ 2026 meta-analysis suggests cardiometabolic markers return toward baseline, but hard cardiovascular outcomes after discontinuation haven’t been well-studied.
What to Do Next
If you’ve read this far, you’re better informed about the long-term effects of GLP-1 drugs than most people — including, frankly, some of the providers prescribing them. Here’s how to use this information.
If You’re Currently on a GLP-1 Drug
Print or bookmark the monitoring checklist and red-flag symptoms above. Bring the “questions for your doctor” list to your next appointment. Make sure you’re doing resistance training and eating adequate protein. These two habits aren’t optional — they’re the difference between healthy weight loss and problematic weight loss.
If You’re Considering Starting
The evidence supports a favorable risk-benefit profile for most people with obesity or Type 2 diabetes, especially those with cardiovascular risk factors. But “most people” isn’t the same as “you specifically.” The right move is a thorough conversation with a provider who will do baseline labs, discuss your medical history, and set up a monitoring plan — not just hand you a prescription.
If You’re Choosing a Provider
Not all GLP-1 prescribers are created equal. A good provider includes regular follow-up appointments, lab monitoring, nutrition guidance, and a clear plan for dose management. A red flag is a provider who prescribes without follow-up, doesn’t order labs, or can’t answer basic questions about long-term monitoring.
We’ve compared the top GLP-1 telehealth providers on pricing, monitoring practices, medication sourcing, and cancellation policies. If you’re evaluating your options, our provider comparison guide can help you find one that takes long-term care seriously.
See all options: Best GLP-1 Online Programs
How We Built This Guide
Sources: This guide draws primarily from FDA prescribing information, peer-reviewed studies published in Nature Medicine, The New England Journal of Medicine, JAMA, The Lancet, BMJ, JCI, and JAMA Ophthalmology, Cochrane systematic reviews, and official regulatory communications from the FDA, EMA, and UK MHRA.
Evidence grading: We categorize evidence as Strong (large RCTs or meta-analyses with consistent findings), Moderate (smaller RCTs, large observational studies), Emerging (observational signals, early-phase data), or Unknown (insufficient data). Observational studies are presented as associations, not causation.
What we don’t do: We don’t treat animal data as human proof. We don’t cite single case reports as evidence of trends. We don’t suppress negative findings to make GLP-1 drugs look better (or worse) than the data supports.
Update schedule: We review this page when major regulatory updates or large studies are released, and we update the “Last Evidence Review” date when the key claims and tables are re-verified against current sources.
Affiliate relationship: We earn commissions from some provider links on this site. This never influences our evaluations. If we thought a provider was unsafe, we wouldn’t list them regardless of commission. Full disclosure is available on our editorial standards page.
Update Log
- February 2026: Added UK MHRA drug safety update on NAION/semaglutide. Updated weight regain section with BMJ 2026 meta-analysis. Updated oral semaglutide approval status.
- January 2026: Added FDA’s removal of suicidal ideation warning from GLP-1 labels. Updated compounded drug section with latest FDA salt-form guidance.
- December 2025: Added UC Davis systemic impact findings. Updated muscle loss section with European Journal of Pharmacology review.
- November 2025: Added NAION confirmation from EMA PRAC review. Added Nature Biotechnology longevity discussion.
FAQ
Are GLP-1 drugs safe to take long-term?
For most people, yes — the evidence supports a favorable safety profile over 2–4 years, with cardiovascular benefits strong enough to earn FDA approval for heart risk reduction. Serious side effects exist but are uncommon. The honest caveat: we don’t have controlled data beyond about 4 years for weight-loss doses in non-diabetic populations. Ongoing monitoring is important.
What are the most serious long-term side effects of GLP-1 drugs?
Pancreatitis (evidence is mixed on magnitude, but it is a labeled concern), gallbladder disease, kidney injury from dehydration, gastroparesis, and a very rare risk of NAION (sudden vision loss). The thyroid cancer boxed warning is based on animal data; human evidence remains inconclusive.
Do GLP-1 drugs cause cancer?
The boxed thyroid cancer warning comes from rodent studies. After nearly 20 years of human use, no definitive increase in thyroid or pancreatic cancer has been confirmed in large trials. However, very long-term data (10–20+ years) doesn’t exist yet. If you have a personal or family history of medullary thyroid carcinoma or MEN2, GLP-1 drugs are contraindicated.
What happens when you stop GLP-1 drugs?
Most people regain a significant portion of lost weight. The BMJ 2026 systematic review projects return to pre-treatment weight within about 1.5–2 years for semaglutide/tirzepatide users, with cardiometabolic markers also reverting. However, real-world data shows about 42% of people maintain their weight loss at one year post-discontinuation.
Do you have to take GLP-1 drugs forever?
Not necessarily "forever," but emerging evidence frames obesity as a chronic condition where ongoing treatment produces the best outcomes — similar to blood pressure medication. Discuss your specific situation and goals with your doctor. Some are exploring maintenance doses or lifestyle-supported transitions.
Can GLP-1 drugs cause muscle loss?
Yes, about 20–25% of weight lost is lean mass — similar to other weight-loss methods. Resistance training and adequate protein intake (1.2–1.6 g/kg/day) are the evidence-based strategies to minimize this. Much of the "40% lean mass loss" figure cited in some reports comes from liver fat reduction, not skeletal muscle.
Is "Ozempic face" permanent?
It’s caused by facial fat and volume loss from rapid weight loss, not the drug specifically. It can improve with weight stabilization. The faster you lose weight, the more pronounced it tends to be. Slower weight loss may minimize it.
Do GLP-1 drugs affect fertility or pregnancy?
GLP-1 drugs are not recommended during pregnancy. Animal studies have shown adverse reproductive effects. Interestingly, weight loss itself can improve fertility, and there have been anecdotal reports of increased conception rates during treatment — but stop the medication before becoming pregnant. Discuss timing with your doctor (at least 2 months before planned conception for semaglutide).
Are long-term effects different for women vs. men?
The major clinical trials included both sexes, and the core safety profile appears similar. However, women face some specific considerations: gallstone risk is higher, bone density loss may be more significant (especially postmenopausal), and tirzepatide may reduce oral contraceptive effectiveness.
Can Ozempic or Wegovy cause vision loss?
In rare cases, yes. The EMA confirmed in 2025 that NAION (non-arteritic anterior ischemic optic neuropathy) is a very rare side effect of semaglutide. The UK MHRA issued safety guidance in February 2026. Watch for sudden, painless vision loss in one eye and seek urgent care immediately.
Do GLP-1 drugs cause depression or suicidal thoughts?
The FDA investigated this concern and in January 2026 requested the removal of the suicidal ideation warning from GLP-1 labels, finding no evidence of a causal link. Depression and mood changes can occur during significant weight loss and life changes from any cause, so mental health awareness remains important.
Are long-term effects different in non-diabetics?
The core safety profile appears similar, but most long-term data comes from diabetic populations. Non-diabetics typically use higher doses (weight-loss doses), which may produce more GI side effects. A large 2024 global cohort study of non-diabetic obese individuals showed protective cardiovascular and mortality benefits. Ongoing monitoring is especially important since this population has a shorter evidence track record.
Is GLP-1 microdosing safe long-term?
Very little formal evidence exists on microdosing. It’s not an FDA-approved protocol, and no large trials have studied it. Some providers offer low-dose maintenance, but the long-term safety and efficacy of sub-therapeutic doses is genuinely unknown.
How long does GLP-1 nausea last?
For most people, nausea peaks in the first 4–8 weeks and during dose increases, then significantly improves. In the liraglutide trial, nausea dropped from 25% at week 4 to about 5.5% at week 56. Slow dose escalation, smaller meals, and staying hydrated help.
Can you take GLP-1 drugs with other medications?
Generally yes, but there are important interactions. GLP-1 drugs slow gastric emptying, which can affect absorption of oral medications. Tirzepatide specifically may reduce oral contraceptive effectiveness. Combining GLP-1 drugs with insulin or sulfonylureas increases hypoglycemia risk. Always review your full medication list with your prescriber.
What's the difference between semaglutide and tirzepatide long-term?
Tirzepatide is a dual GLP-1/GIP agonist, while semaglutide targets only GLP-1. Tirzepatide produces greater weight loss on average (~20% vs ~15%) and may have slightly different GI side effect profiles. Long-term safety data is more extensive for semaglutide (longer on the market). Both have strong cardiovascular evidence, though head-to-head long-term comparisons are limited.
Can GLP-1 drugs cause hair loss?
Yes, but it's caused by rapid weight loss (telogen effluvium), not the drug itself. This happens with any method of significant weight loss. It's usually temporary and resolves as weight stabilizes. Adequate protein and nutrition may help.
Sources Cited in This Guide
- Lincoff AM et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. NEJM. 2023. (SELECT trial)
- Xie Y, Choi T, Al-Aly Z. Mapping the effectiveness and risks of GLP-1 receptor agonists (~216,000 GLP-1RA initiators, 175 outcomes). Nature Medicine. January 2025.
- Jalleh RJ, Talley NJ, Horowitz M, Nauck MA. The science of safety: adverse effects of GLP-1 receptor agonists. JCI. 2026.
- Wilding JPH et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: STEP 1 trial extension. Diabetes Obes Metab. 2022.
- West S et al. Weight regain after cessation of medication for weight management: systematic review and meta-analysis. BMJ. 2026.
- Metabolic rebound after GLP-1 receptor agonist discontinuation. Lancet eClinicalMedicine. 2025.
- EMA PRAC. NAION confirmed as very rare side effect of semaglutide medicines. 2025.
- UK MHRA Drug Safety Update. Semaglutide: risk of NAION. February 2026.
- Marso SP et al. Semaglutide and cardiovascular outcomes. NEJM. 2016.
- FDA prescribing information: Wegovy (semaglutide), Ozempic (semaglutide), Zepbound (tirzepatide).
- FDA. Concerns with unapproved GLP-1 drugs used for weight loss. Updated 2025.
- FDA. Requests removal of suicidal behavior warning from GLP-1 labels. January 2026.
- FDA. Approval of semaglutide for CKD risk reduction. January 2025.
- FDA. Approval of Wegovy for cardiovascular risk reduction. March 2024.
- Hathaway JT et al. Risk of NAION with GLP-1 receptor agonist use. JAMA Ophthalmology. 2024.
- Cochrane Review. GLP-1 drugs effective for weight loss. 2025.
- UC Davis Health. Systemic impact of GLP-1-based therapies. December 2025.
- Gasoyan H et al. Weight trajectories after GLP-1 discontinuation: real-world data. AJMC. 2025.
- Newsome PN et al. Semaglutide and NASH. NEJM. 2021.
- Global cohort study: long-term safety of GLP-1 RAs in obesity without T2D (TriNetX, 12,123 matched individuals). Diabetes Obes Metab. 2024.
- Kindel TL et al. Multi-society clinical practice guidance for perioperative use of GLP-1RAs. Surg Endosc. October 2024.
- Wegovy tablets (oral semaglutide 25 mg) FDA prescribing information. December 2025.
Medical Disclaimer: This content is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before starting, stopping, or changing any medication. Individual medical decisions should be made in consultation with your doctor based on your specific health circumstances.
This guide is updated regularly as new evidence becomes available. If you found it helpful, you can bookmark this page — we’ll keep it current.