For informational purposes only—not medical advice.
Medically reviewed by Dr. Diana Isaacs, PharmD, BCACP, BC-ADM, CDCES, FCCP, FADCES
Oral Semaglutide vs Injectable: Which Form Is Right for You?
If you are comparing oral semaglutide vs injectable semaglutide — often searched as oral semaglutide vs injection — you are asking one of the most practical questions in weight loss and diabetes treatment right now: pill or shot?
Both forms use the same active ingredient — semaglutide, a GLP-1 receptor agonist that curbs appetite, slows digestion, and helps control blood sugar. Injectable semaglutide has been available since 2017 (Ozempic for type 2 diabetes) and 2021 (Wegovy for weight management), and these injections are taken once a week. The oral versions (Rybelsus, and now the Wegovy pill) are taken once a day.
What changed everything: on December 22, 2025, the FDA approved the Wegovy pill — oral semaglutide at 25 mg — specifically for weight loss. It launched in January 2026. In clinical trials, it produced roughly 13.6% body weight loss on average (and up to 16.6% among those who stayed on treatment), which is comparable to the roughly 15% seen with injectable Wegovy. For the first time, the pill can go toe-to-toe with the shot on weight loss results.
But “comparable” does not mean “identical in every way.” Dosing rules, cost, daily routine, side effects, and your personal medical situation all shape which form actually works better for you. This page covers all of it — every product, every tradeoff, every number — cited from FDA prescribing information and published clinical trials, so you can walk into your next doctor's appointment ready to have the right conversation.
Affiliate Disclosure: This page may contain affiliate links. We may earn a commission at no cost to you. Full disclosure.
Medical Disclaimer: This content is for informational purposes only and is not medical advice. Always consult a licensed healthcare provider before starting any medication. Full medical disclaimer.

Oral vs Injectable Semaglutide at a Glance
Before we go deeper, here are the main semaglutide products people mean when comparing pill vs shot — plus a note on Ozempic tablets, which are FDA-approved but not yet widely available.
| Rybelsus | Wegovy Pill | Ozempic | Wegovy Injection | |
|---|---|---|---|---|
| Type | Oral (daily pill) | Oral (daily pill) | Injectable (weekly shot) | Injectable (weekly shot) |
| FDA-approved for | Type 2 diabetes (glycemic control) | Weight loss; CV risk reduction | Type 2 diabetes; CV risk reduction in T2D; CKD risk reduction in T2D | Weight loss; CV risk reduction; MASH |
| Max dose | 14 mg/day (R1) or 9 mg/day (R2) | 25 mg/day | 2 mg/week | 2.4 mg/week |
| Avg weight loss | ~8–9 lbs over 26 wks (off-label) | ~13.6% (ITT) / 16.6% (on-treatment) over 64 wks | ~10 lbs over 30 wks (diabetes patients) | ~15% body weight over 68 wks |
| Needle required | No | No | Yes | Yes |
| Refrigeration | No | No | Yes (before first use) | Yes (before first use) |
| Empty stomach required | Yes — 30 min fast | Yes — 30 min fast | No | No |
| Self-pay cost/month | ~$997 list | $149–$299 (NovoCare) | $1,027 list; $199–$499 (w/ savings) | $199–$349 (NovoCare) |
| Insurance coverage | Common for diabetes | Varies for weight loss | Common for diabetes | Varies for weight loss |
| Bioavailability | 0.4–1% (R1); 1–2% (R2) | ~1–2% | ~89% | ~89% |
Sources: FDA prescribing information for Wegovy (Dec 2025), Ozempic, and Rybelsus (2025 labels). Pricing from NovoCare.com and NovoPricing.com, verified February 2026.
Note: Rybelsus does not have an FDA-approved cardiovascular risk-reduction indication. Ozempic also has an FDA-approved indication for CKD risk reduction in T2D (FLOW trial, Jan 2025).
You probably noticed the pill doses are dramatically higher than the injection doses. That is not a typo. Oral semaglutide has very low bioavailability — only about 1–2% of the drug survives your stomach acid and reaches your bloodstream. Injectable semaglutide bypasses digestion entirely, so roughly 89% reaches your blood. A 25 mg daily pill delivers a similar amount of semaglutide into your system as a 2.4 mg weekly injection. Different math, similar result.
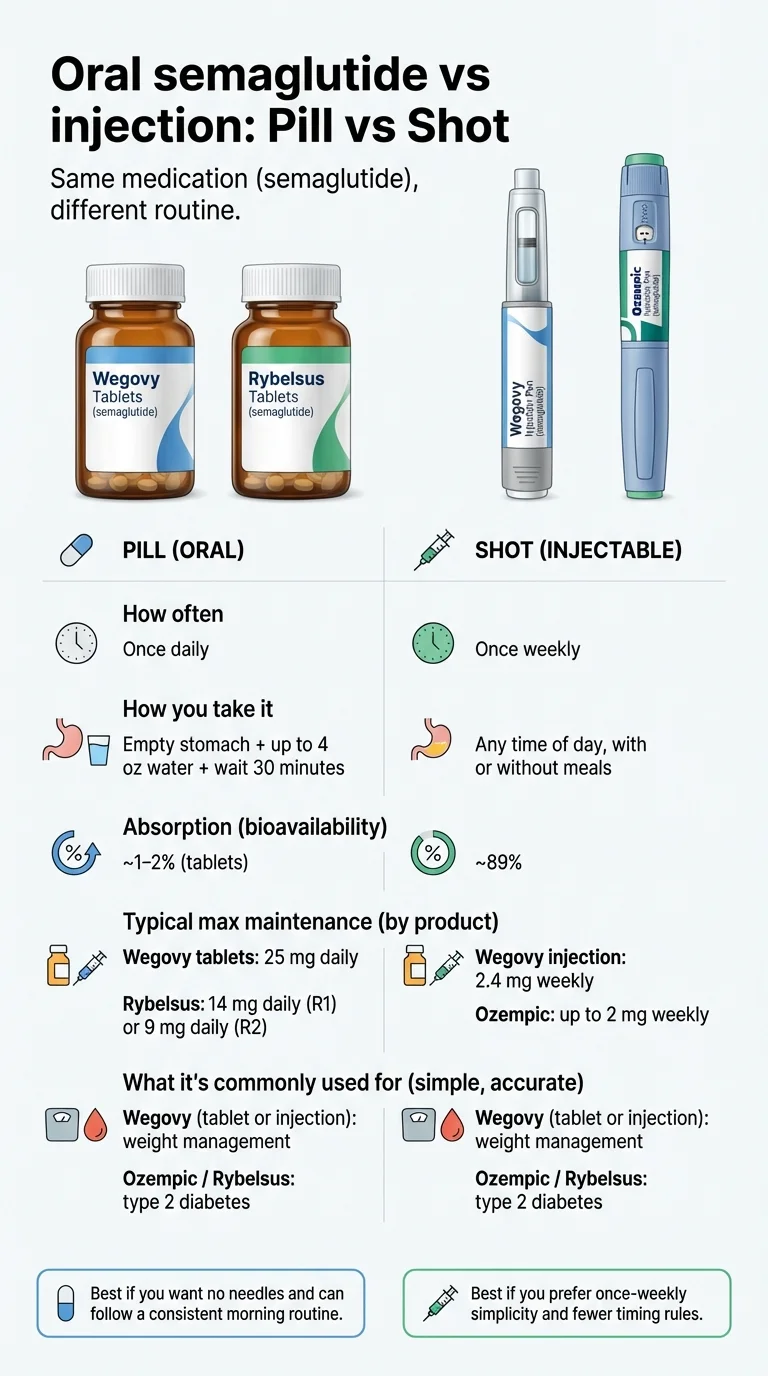
What Counts as “Oral Semaglutide” and “Injectable Semaglutide”?
This is the first confusion to clear up. There are now five FDA-approved semaglutide products (with Ozempic tablets approved in February 2026 but not yet in pharmacies), and they are not interchangeable. Here are the four that are currently available — knowing which is which matters because the names get tangled constantly online.
Oral semaglutide (the pills)
Rybelsus is approved for type 2 diabetes. It now comes in two formulations that are not the same milligram-for-milligram. Formulation R1 has 3 mg, 7 mg, and 14 mg tablets. Formulation R2, which is newer, has 1.5 mg, 4 mg, and 9 mg tablets. The R2 formulation absorbs better (1–2% bioavailability vs. 0.4–1% for R1), which is why the doses are lower but the effect is similar. This matters if your pharmacy switches your formulation — ask your doctor first. Rybelsus is not FDA-approved for weight loss, though it is commonly prescribed off-label for that purpose. [Source: FDA Rybelsus prescribing information, 2025]
Wegovy pill was approved December 22, 2025 for weight loss and cardiovascular risk reduction. Dose strengths: 1.5 mg, 4 mg, 9 mg, and 25 mg. This is the game-changer — the first oral GLP-1 approved specifically for weight management. At its maximum dose of 25 mg, it achieves weight loss comparable to injectable Wegovy. Launched in the U.S. in January 2026. [Source: FDA, Novo Nordisk press release, Dec 22, 2025]
Injectable semaglutide (the shots)
Ozempic is approved for type 2 diabetes, cardiovascular risk reduction, and reducing the risk of worsening kidney disease in adults with T2D and chronic kidney disease (based on the FLOW trial, Jan 2025). It is a once-weekly injection using a pre-filled pen, available in 0.25 mg, 0.5 mg, 1 mg, and 2 mg doses. Like Rybelsus, it is widely prescribed off-label for weight loss. [Source: FDA Ozempic prescribing information]
Wegovy injection is approved for weight loss, cardiovascular risk reduction, and MASH (a liver condition formerly known as NASH). Once-weekly injection, maximum dose 2.4 mg. This has been the benchmark for GLP-1-based weight loss since 2021. [Source: FDA Wegovy prescribing information, Dec 2025]
What does NOT count as FDA-approved oral semaglutide
This matters for your safety. “Sublingual semaglutide drops,” “compounded oral semaglutide,” “semaglutide lozenges,” and similar products marketed online are not FDA-approved. The FDA does not review compounded drugs for safety, effectiveness, or quality before they are sold. We have a full section on this below, but the short version: if someone is selling you “oral semaglutide” and it is not Rybelsus or the Wegovy pill, be very cautious. Read more about whether compounded semaglutide is safe.
Also worth noting: “Ozempic pill” is not a product on pharmacy shelves right now. Novo Nordisk received FDA approval for oral Ozempic tablets (1.5 mg, 4 mg, 9 mg) for diabetes on February 4, 2026. U.S. pharmacy availability is expected in Q2 2026 — but they are not yet available. [Source: Drugs.com, Feb 2026]
Does Oral Semaglutide Work as Well as Injectable for Weight Loss?
This is what most people reading this page actually want to know. And the honest answer depends on which oral product you are talking about.
At diabetes doses: injectable has a modest edge
When researchers compare Rybelsus (the oral diabetes pill, max 14 mg) against Ozempic or Wegovy injections, the injection generally produces more weight loss — but the gap is smaller than most people expect.
The PIONEER clinical trials tested Rybelsus 14 mg and found modest weight loss of roughly 8–9 pounds over 26 weeks. The SUSTAIN trials tested injectable Ozempic at 1.0 mg weekly and saw slightly more weight loss, around 10 pounds over 30 weeks in diabetes patients. Neither of these trials was specifically designed for weight loss — they were studying blood sugar control in people with type 2 diabetes, so the weight loss was a welcome secondary effect. [Source: Frontiers in Endocrinology, 2021]
Real-world data tells a similar story. A 2024 study in the Journal of Pharmacy Technology compared oral and injectable semaglutide in 105 patients with type 2 diabetes. The injectable group lost more weight numerically (5.26 kg vs. 3.64 kg), but the difference was not statistically significant. A separate study in Croatia found that identical proportions — 56.7% in both groups — achieved at least 5% weight loss. [Sources: J Pharm Technol, 2024 (PMC); MDPI Diabetology, 2024]
At weight-loss doses: the Wegovy pill closes the gap
Now for the data that changes the conversation.
The OASIS 4 trial, published in the New England Journal of Medicine in September 2025, tested the Wegovy pill (oral semaglutide 25 mg daily) against placebo in 307 adults with obesity or overweight. The results were striking:
- Participants who stayed on treatment lost an average of 16.6% of their body weight over 64 weeks
- On an intent-to-treat basis (counting everyone, including dropouts), the average was 13.6%
- 76.3% of pill participants lost at least 5% of their body weight, compared to 31.3% on placebo
- About one in three participants lost 20% or more of their body weight
- Serious adverse events actually occurred less frequently with the pill (3.9%) than with placebo (8.8%)
For context, the STEP 1 trial of injectable Wegovy (2.4 mg weekly) found roughly 15% body weight loss over 68 weeks.
So the Wegovy pill (25 mg daily) now produces weight loss that is comparable to — and in some analyses slightly better than — the Wegovy injection (2.4 mg weekly). This is the first time an oral GLP-1 has matched the injectable at weight-loss doses.
[Sources: Wharton S, et al. NEJM 2025;393(11):1077-1087 (OASIS 4); Wilding JPH, et al. NEJM 2021;384:989-1002 (STEP 1)]
The bottom line on effectiveness
Key takeaway:
The insight most articles miss: the dose matters more than the delivery method. At lower, diabetes-focused doses, the injection has a modest edge. At the higher weight-loss doses now available in the Wegovy pill, that gap essentially disappears. More drug in your system = more weight loss, regardless of whether it arrives via your stomach or your skin.
Does Oral Semaglutide Work as Well as Injectable for Blood Sugar?
If you are managing type 2 diabetes, both forms are effective. Injectable semaglutide has shown slightly greater HbA1c reductions in most head-to-head comparisons, but the clinical significance of that difference varies.
The clinical trial numbers:
- PIONEER trials (oral, Rybelsus 14 mg): HbA1c reduced by about 1.0–1.4% over 26 weeks
- SUSTAIN trials (injectable, Ozempic 1.0 mg): HbA1c reduced by about 1.5–1.8% over 30–56 weeks
- UK ABCD audit (2025, real-world, 2,120 patients): Injectable showed statistically greater HbA1c reduction compared to oral
- Croatia study (2024, real-world): No significant difference between oral and injectable (−1.4% vs −1.1%, p = 0.126)
Both forms significantly outperform many other common diabetes medications. Cardiovascular risk reduction is product-specific: Ozempic (injectable) and Wegovy (tablets and injection) have FDA-approved indications to reduce major adverse cardiovascular events in certain populations, while Rybelsus is indicated for glycemic control in type 2 diabetes and does not have a CV risk-reduction indication. Injectable semaglutide (Ozempic) has the strongest evidence for cardiovascular risk reduction in T2D, backed by the SUSTAIN-6 trial. The Wegovy brand (both pill and injection) also has FDA approval for CV risk reduction based on the SELECT trial, which demonstrated a 20% reduction in major cardiovascular events in adults with established heart disease and obesity or overweight. [Sources: FDA prescribing information; Lincoff AM, et al. NEJM 2023;389:2221-2232]
[Sources: PIONEER program; SUSTAIN program; ABCD UK audit, Diabetes 2025;74(Supp 1):756-P; MDPI Diabetology 2024;5(1):5]
Dosing and Titration: Exact Schedules for Every Product
All semaglutide products start low and increase gradually. This slow ramp-up reduces the severity of side effects — particularly nausea — and helps you stay on treatment. Here are the exact schedules from the FDA labels.
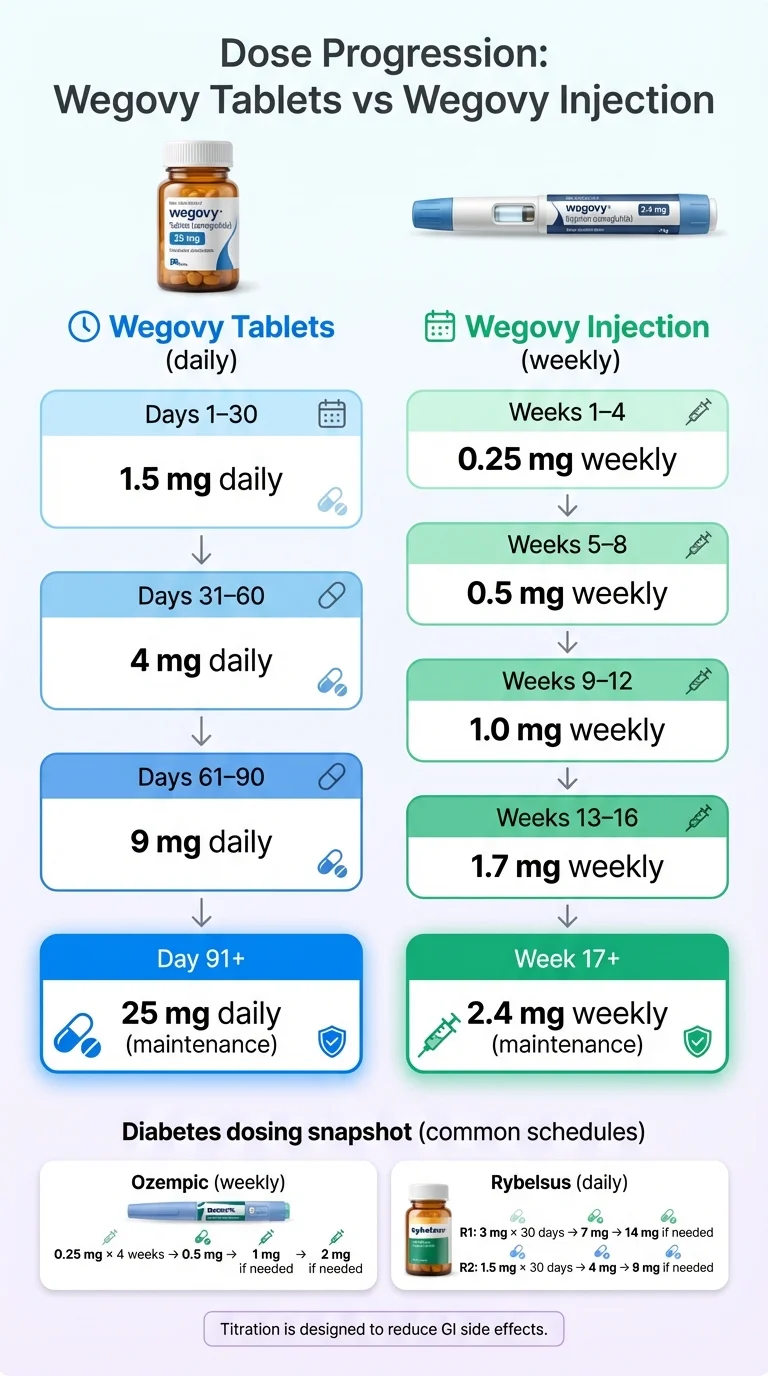
Wegovy Pill (oral — for weight loss)
| Period | Daily Dose | Notes |
|---|---|---|
| Days 1–30 | 1.5 mg | Initiation — not a therapeutic dose |
| Days 31–60 | 4 mg | Building up |
| Days 61–90 | 9 mg | Building up |
| Day 91 onward | 25 mg | Maintenance dose |
Wegovy Injection (weekly — for weight loss)
| Period | Weekly Dose |
|---|---|
| Weeks 1–4 | 0.25 mg |
| Weeks 5–8 | 0.5 mg |
| Weeks 9–12 | 1.0 mg |
| Weeks 13–16 | 1.7 mg |
| Week 17 onward | 2.4 mg |
Rybelsus (oral — for diabetes)
Rybelsus now comes in two formulations. They are not interchangeable milligram-for-milligram. Your pharmacist cannot swap one for the other without your doctor's guidance.
Formulation R1 (original): 3 mg for 30 days (initiation) → 7 mg → 14 mg if needed
Formulation R2 (newer, better absorption): 1.5 mg for 30 days (initiation) → 4 mg → 9 mg if needed
Despite the lower numbers on R2, clinical studies showed no significant differences in steady-state drug levels between 14 mg R1 and 9 mg R2, or between 7 mg R1 and 4 mg R2. The R2 formulation achieves equivalent drug exposure with lower milligram doses because its bioavailability is roughly double that of R1. Do not switch between them during the first 30 days. [Source: FDA Rybelsus prescribing information, 2025]
Ozempic (injectable — for diabetes)
0.25 mg weekly for 4 weeks (initiation) → 0.5 mg → 1 mg if needed → 2 mg if needed
[Sources: FDA prescribing information for all products listed above]
Why you cannot compare milligrams across forms
A 25 mg Wegovy pill and a 2.4 mg Wegovy injection sound wildly different. But they reach similar blood levels of semaglutide because of the bioavailability gap. Oral semaglutide: about 1–2% of the drug actually makes it through your stomach into your bloodstream. Injectable: about 89%. The pill compensates by using a much higher dose. This is also why the pill has strict dosing rules — anything that interferes with absorption shrinks an already-tiny absorption window.
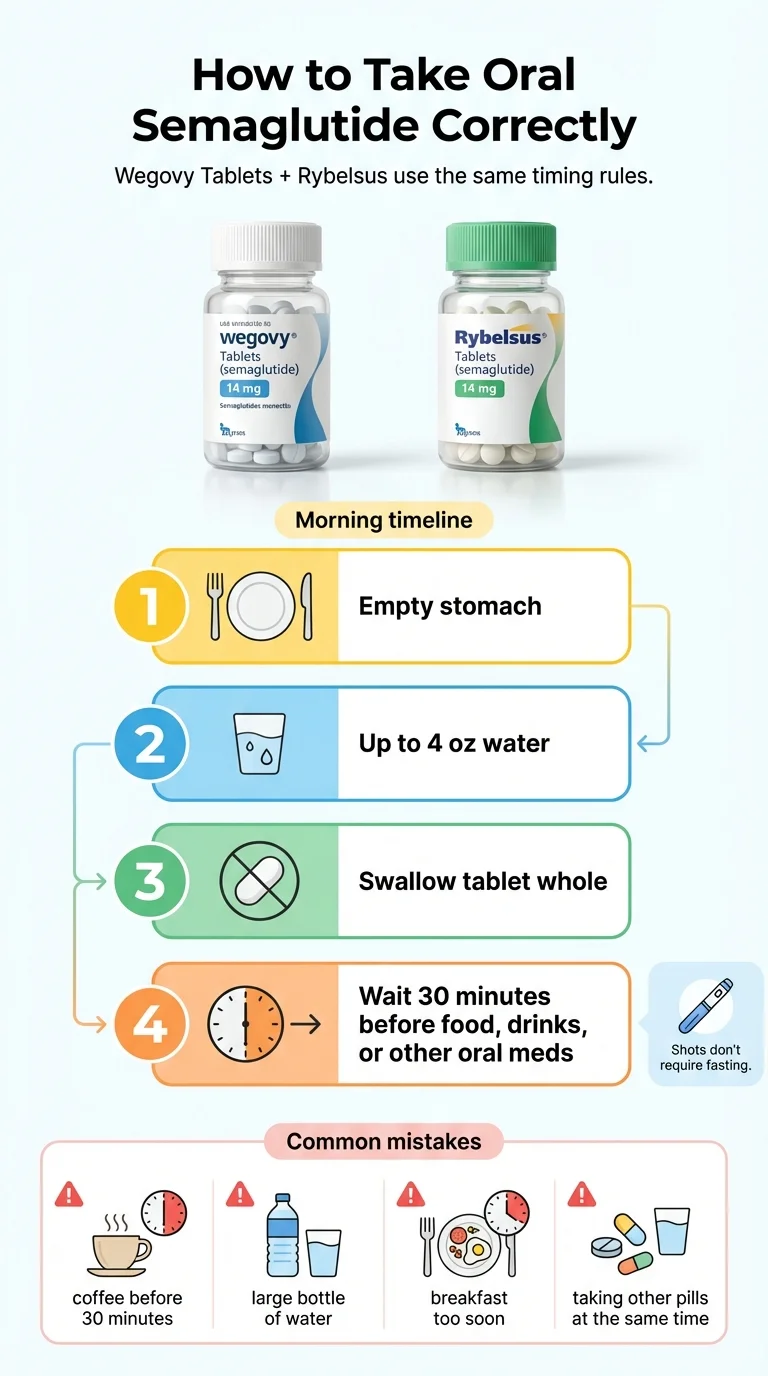
How to Take Oral Semaglutide Correctly
Getting this right is not optional. Unlike most medications, oral semaglutide has strict administration rules because its absorption rate is so low. Taken wrong, the pill may barely work. Every person who has said “the pill didn't do anything for me” should answer these five questions first.
The rules (from the FDA label)
- Take it first thing in the morning on a completely empty stomach. No food, no coffee, no juice — nothing in your stomach.
- Use only plain water, and no more than about 4 ounces (roughly half a standard glass). More water dilutes the absorption enhancer (SNAC) in the tablet.
- Swallow the tablet whole. Do not crush, chew, or split it.
- Wait at least 30 minutes before eating, drinking anything else, or taking other oral medications.
- Do not take more than one tablet per day.
The 5 mistakes that make the pill feel “weak”
Taking it with coffee or another beverage. This is the most common mistake. The tablet must be taken with water only. Coffee, tea, juice, and protein shakes all interfere with absorption.
Using too much water. The SNAC absorption enhancer in the tablet works best in a concentrated environment. Flooding your stomach with a full glass of water dilutes it. Stick to 4 ounces or less.
Eating too soon. Thirty minutes is the minimum. Research on the Wegovy label shows that longer post-dose fasting increases how much semaglutide reaches your blood. If you eat at 20 minutes, you may be cutting the drug's effectiveness significantly.
Taking other medications at the same time. Other oral medications compete with semaglutide during that critical absorption window. Take semaglutide first, wait the full 30 minutes, then take your other morning pills.
Inconsistent timing. Taking it at different times, or sometimes with food and sometimes without, creates unpredictable drug levels. Consistency is more important with the oral form than the injectable.
If you take morning medications (a practical schedule)
This is a common challenge, especially for people who also take thyroid medication (levothyroxine) or blood pressure pills on an empty stomach. Here is one approach — but confirm it with your pharmacist for your specific combination:
- 6:00 AM — Wake up. Take semaglutide tablet with a small sip of water.
- 6:30 AM — Take other morning medications with water.
- 6:45 AM or later — Eat breakfast.
If you take multiple fasting-dependent medications, ask your pharmacist to design a schedule. They do this all the time.
How to Take Injectable Semaglutide Correctly
The injection is simpler in many ways. No fasting, no water restrictions, no timing constraints around food.
Once per week, on the same day each week. Pick any day. Any time of day. With or without food.
Inject under the skin (subcutaneous) in your abdomen, upper thigh, or upper arm. Rotate your injection site each week — using the same spot repeatedly can cause tissue changes that affect absorption.
Use the pre-filled pen as directed. No mixing, no measuring. It clicks into place.
Storage differences worth knowing
| Oral (Rybelsus / Wegovy Pill) | Injectable (Ozempic / Wegovy Injection) | |
|---|---|---|
| Storage | Room temperature, in original container | Refrigerate before first use; room temp OK for limited period after first use |
| Travel | Easy — just bring the pill bottle | Need cold pack or insulin travel case |
| TSA/Airport | No issues | Allowed with prescription; can declare medical supplies |
If needles make you anxious
You are not alone — needle anxiety is one of the top reasons people search for the oral option. But if the injection is clinically the better fit for you, know that the needle on semaglutide pens is very thin. Most people describe it as a brief pinch, not a sharp pain. Letting the pen sit at room temperature before injecting can help. Some people find the abdomen less sensitive than the thigh. And plenty of people inject while distracted — watching TV, listening to a podcast, talking to someone.
Side Effects: Oral vs Injectable
Both forms share the same core side effects because they deliver the same active ingredient. The most common effects are gastrointestinal, and they tend to be worst during dose escalation before improving over time. Learn more about cons of GLP-1 medications.
What to expect (both forms)
Nausea is the most frequently reported side effect. At weight-loss doses, it is more common than many people expect: in clinical trials, nausea occurred in roughly 44% of Wegovy injection patients and 46.6% of Wegovy tablet patients (vs. 16–19% on placebo). At lower diabetes doses (Ozempic, Rybelsus), rates are generally lower — closer to 15–20%. Nausea is usually worst in the first few weeks of a new dose and gets better as your body adjusts. Diarrhea and constipation are also common during titration. Vomiting and abdominal pain occur less often. Decreased appetite is both a side effect and the mechanism of action — it is how the drug helps with weight loss.
In the OASIS 4 trial of the Wegovy pill, side effects were consistent with what injection trials had shown — no new safety signals. Interestingly, serious adverse events occurred less frequently in the oral group (3.9%) than in the placebo group (8.8%). [Source: OASIS 4, NEJM 2025]
What differs by route
Injections can cause mild injection-site reactions — some redness, itching, or a small bump. These are usually minor and temporary.
Oral tablets do not cause injection-site reactions, but GI side effects can sometimes feel more variable because absorption fluctuates depending on how well you follow the fasting and water rules. If you take the pill wrong one day and right the next, your drug levels bounce around, which can trigger more nausea.
Behavioral strategies that help (for either form)
Eat smaller meals. Eat more slowly. Stay hydrated — especially during dose escalation when GI symptoms are most common. Some people find that bland, lower-fat foods sit better during the first few weeks of a new dose. Ginger tea or ginger chews can help with mild nausea. These are not treatments — they are practical adjustments that many patients find useful.
Serious warnings (both forms — from the FDA label)
These apply regardless of pill or shot:
FDA Safety Warnings
Thyroid C-cell tumors (boxed warning). In animal studies, semaglutide caused thyroid tumors. Whether this happens in humans is unknown. Semaglutide is contraindicated if you have a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Pancreatitis. There have been reports of acute pancreatitis. Seek immediate medical attention if you develop severe, persistent abdominal pain that may radiate to your back, with or without vomiting.
Gallbladder problems. Semaglutide use has been associated with gallstones and gallbladder inflammation, especially with rapid weight loss.
Acute kidney injury. Rare, usually tied to dehydration from severe vomiting or diarrhea. Stay hydrated.
Hypoglycemia risk. Mainly a concern if you also take insulin or a sulfonylurea. Your doctor may need to reduce those doses.
Pulmonary aspiration during anesthesia. Semaglutide slows stomach emptying, which may increase aspiration risk under anesthesia. If you are scheduled for surgery under general anesthesia or deep sedation, tell your anesthesiologist that you take semaglutide. Multi-society guidance (Oct 2024) indicates that most patients can continue GLP-1 drugs before elective procedures, while people at higher risk for GI side effects may be asked to follow a liquid-only diet for 24 hours beforehand or take other precautions. Your surgical team will determine the right approach for your situation.
Pregnancy. Semaglutide should be discontinued before a planned pregnancy. The Wegovy injection label recommends stopping at least 2 months before conception due to the drug's long half-life. The oral form contains SNAC, an absorption enhancer that crosses the placenta in animal studies — an additional consideration for the pill specifically.
[Sources: FDA prescribing information for Wegovy (tablets and injection), Ozempic, and Rybelsus, 2025]
Who Should NOT Take Semaglutide (Oral or Injectable)
Regardless of form, semaglutide is not appropriate for everyone.
Do not take semaglutide if you have:
- A personal or family history of medullary thyroid carcinoma (MTC)
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- A known allergic reaction to semaglutide or any of its ingredients
Use with extra caution and medical supervision if you have:
- A history of pancreatitis
- Gallbladder disease or a history of gallstones
- Diabetic retinopathy (especially relevant when blood sugar changes rapidly)
- Severe kidney disease
- A history of suicidal thoughts or behavior (the FDA monitors for this)
Special situations:
- Pregnancy and breastfeeding: Not recommended. Discontinue before planned pregnancy. Oral form has the additional SNAC concern.
- Type 1 diabetes: Semaglutide is not approved and not appropriate.
- Combining GLP-1 medications: Never take two semaglutide products simultaneously, and do not combine semaglutide with any other GLP-1 receptor agonist.
[Source: FDA prescribing information for all semaglutide products]
Cost: What You Will Actually Pay (February 2026)
Cost is often the deciding factor. The pricing landscape shifted meaningfully with the Wegovy pill launch. For more details, see our Wegovy pill cost guide.
The numbers
| Product | List Price/Month | Self-Pay/Cash | With Insurance + Savings Card |
|---|---|---|---|
| Rybelsus (oral, diabetes) | ~$997 | ~$997 | As low as $10–$25/month |
| Wegovy pill (oral, weight loss) | ~$1,349 | $149–$299 (via NovoCare) | As low as ~$25/month |
| Ozempic (injectable, diabetes) | ~$1,027 | $199–$499 (w/ savings) | As low as $25/month |
| Wegovy injection (weekly, weight loss) | ~$1,349 | $199–$349 (via NovoCare) | As low as ~$25/month |
Prices verified February 2026. Sources: NovoCare.com, Novo Nordisk announcements, GoodRx.
Note on promotional pricing: Some prices above reflect limited-time launch offers. The Wegovy pill's $149/month rate for the 4 mg dose applies through April 15, 2026 (then $199/month). The Wegovy injection's $199/month introductory rate for starter doses applies for the first two fills through March 31, 2026 (then $349/month). We will update this table as pricing changes. [Source: NovoCare.com, WegovyTerms.com]
The insurance reality
For type 2 diabetes (Rybelsus, Ozempic): most commercial insurance plans cover these with a prior authorization. Copays can be as low as $10–$25/month with Novo Nordisk savings cards. Medicare Part D often covers them as well.
For weight loss (Wegovy pill or injection): coverage is inconsistent. Many commercial plans do not cover Wegovy for weight loss, even though it is FDA-approved for that purpose. Some plans require documentation of failed diet/exercise attempts, specific BMI thresholds, and prior authorization. Medicare generally cannot cover medications prescribed solely for weight loss due to federal law. However, Part D plans may cover Wegovy for people who meet criteria for its cardiovascular risk-reduction indication — coverage varies by plan. This is one of the most frustrating parts of the process for many patients. Learn more about qualifying for Wegovy.
Savings programs: Novo Nordisk offers savings cards and direct pricing through NovoCare. The Wegovy pill's $149/month starting price is the most affordable entry point for any FDA-approved GLP-1 for weight loss on the market right now. Government insurance (Medicare, Medicaid, Tricare) beneficiaries are typically excluded from manufacturer savings programs.
HSA/FSA: Both oral and injectable semaglutide generally qualify for HSA/FSA reimbursement with a valid prescription.
Who wins on cost?
For self-pay weight loss patients, the Wegovy pill is currently the most affordable FDA-approved option at $149–$299/month. Injectable Wegovy runs $199–$349/month through NovoCare. For insured diabetes patients, Rybelsus and Ozempic are often comparable out-of-pocket.
Switching Between Oral and Injectable Semaglutide
People switch for all kinds of practical reasons — needle fatigue, the new pill becoming available, cost changes, lifestyle shifts, travel, side effects. The FDA labels actually provide switching guidance for several scenarios.
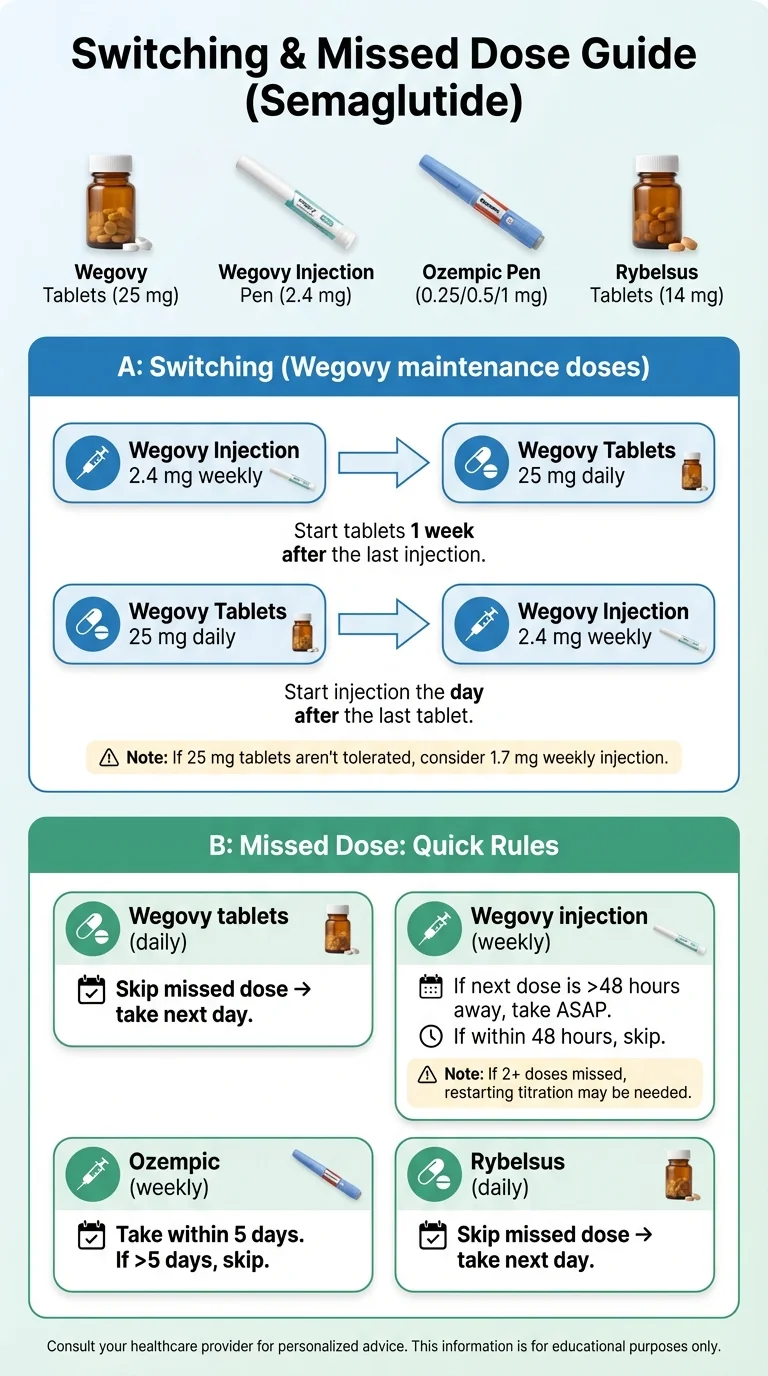
Wegovy injection → Wegovy pill
From 2.4 mg weekly injection → 25 mg daily tablets: Start the 25 mg tablet once daily one week after your last 2.4 mg injection. The label guidance is for switching between maintenance doses; switching from lower doses should be individualized by your prescriber. [Source: FDA Wegovy prescribing information, Dec 2025]
Wegovy pill → Wegovy injection
From 25 mg daily tablets → 2.4 mg weekly injection: Start the 2.4 mg injection once weekly the day after you stop taking the tablets. If the 25 mg tablet dose was not tolerated, the label says your doctor may consider switching to the 1.7 mg injection instead. [Source: FDA Wegovy prescribing information, Dec 2025]
Ozempic → Rybelsus
The FDA label has limited guidance here. It only covers switching from Ozempic 0.5 mg weekly to Rybelsus: wait one week after your last Ozempic 0.5 mg injection, then start Rybelsus at 7 mg or 14 mg (R1) or 4 mg or 9 mg (R2). Switching recommendations for patients on Ozempic 0.25 mg, 1 mg, or 2 mg are not available in the current label. If you are on those doses, your doctor will need to make a clinical judgment. [Source: FDA Rybelsus prescribing information, 2025]
Switching between Rybelsus R1 and R2
Do not switch during the first 30 days (the initiation phase). After day 30, your doctor can switch you. Start the new formulation the day after stopping the old one. Remember: the doses are not equivalent mg-for-mg. [Source: FDA Rybelsus prescribing information, 2025]
The general rules
Never switch on your own. Always do it under your doctor's guidance. And never take two semaglutide products at the same time.
Missed Doses: What to Do
Oral (Rybelsus or Wegovy pill)
Skip the missed dose. Take your next dose the following day at your regular time. Do not double up.
Injectable (Ozempic or Wegovy injection)
Wegovy injection: Take the missed dose as soon as you remember, as long as your next scheduled dose is more than 48 hours away. If your next dose is within 48 hours, skip and take the next dose on your scheduled day. If you have missed more than 2 weeks of Wegovy, contact your doctor — you may need to restart at a lower dose to reduce GI side effects.
Ozempic: Take the missed dose as soon as possible within 5 days after the missed dose. If more than 5 days have passed, skip the missed dose and take your next dose on your regularly scheduled day. [Source: FDA Ozempic prescribing information]
[Source: FDA prescribing information for each product]
Safety Warning: Compounded, Sublingual, and “Drop” Products
This section could prevent real harm, and we include it because people who search “oral semaglutide” are frequently targeted by these products.
The FDA's position
The FDA has explicitly stated that it is concerned about non-FDA-approved GLP-1 drugs marketed for weight loss. Compounded drugs are not reviewed by the FDA for safety, effectiveness, or quality before they are sold. The FDA has taken enforcement actions — including import alerts on GLP-1 ingredients with quality concerns and warning letters to companies distributing certain active pharmaceutical ingredients. [Source: FDA, “FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss,” updated 2025]
Dosing errors with compounded products
The FDA has received multiple reports of adverse events — some requiring hospitalization — related to dosing errors with compounded injectable semaglutide. These errors resulted from patients measuring incorrect doses and from healthcare professionals miscalculating doses. Some patients were prescribed doses beyond what the FDA-approved label recommends. [Source: FDA safety alert on compounded semaglutide dosing errors]
“Semaglutide salts” are a different drug
This is critical and most people do not know it. The FDA has warned that some compounded products use “salt forms” of semaglutide — specifically semaglutide sodium or semaglutide acetate. These are different active ingredients than the semaglutide used in Wegovy, Ozempic, and Rybelsus. The FDA states that it is not aware of any lawful basis for using these salt forms in compounding. [Source: FDA, “FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss”]
The compounding shortage exception ended
During the semaglutide shortage, compounding pharmacies had a legal basis to produce compounded semaglutide. The FDA determined the semaglutide injection shortage was resolved on February 21, 2025. After that, FDA ended its temporary enforcement discretion on a timeline (including separate dates for 503A vs. 503B facilities) and reiterated that compounding “essentially copies” of commercially available semaglutide is restricted. Some compounding may still be legally permissible in limited, patient-specific scenarios, but the FDA has been clear it is targeting mass-marketed, copycat GLP-1 products for enforcement. [Source: FDA, Feb 2025; FDA policy clarification on compounders]
How to protect yourself
- Only use semaglutide prescribed by a licensed healthcare provider and dispensed by a state-licensed pharmacy.
- Verify that your product is Rybelsus, Wegovy (pill or injection), or Ozempic.
- Be skeptical of dramatically lower prices, “research use only” labels, or products marketed as “sublingual drops” or “oral semaglutide liquid.”
- If you are currently using a compounded product, talk to your doctor about transitioning to an FDA-approved option. Read our full guide on is compounded semaglutide safe.
Which Form Is Right for You? A Decision Framework
After reading everything above, here is a practical way to sort through the choice. Neither form is universally “better.” The best semaglutide is the one you will actually take consistently — because adherence is the single best predictor of success.
The 5-Factor Decision Guide
| Factor | The Pill May Be Better If... | The Shot May Be Better If... |
|---|---|---|
| Needle comfort | You have needle anxiety or a needle phobia — the pill removes this barrier entirely | You are comfortable with a weekly injection using a pre-filled pen |
| Daily routine | You can reliably take a pill first thing every morning on an empty stomach and wait 30 min before eating | You prefer the simplicity of once-a-week dosing with no food restrictions |
| Budget (self-pay) | You want the most affordable entry point — Wegovy pill starts at $149/month | You can afford $199–$349/month for injectable Wegovy |
| Weight loss goals | You want significant weight loss — Wegovy pill achieves ~16.6% at the 25 mg dose | You want the most established option — injectable Wegovy has the longest track record |
| Medical situation | You have no GI conditions affecting absorption; you do not take many fasting-sensitive morning meds | You have unpredictable mornings, eat on irregular schedules, or need the strongest cardiovascular risk reduction evidence |
Four types of people and what usually works best
Based on patterns from clinical trial adherence data and prescriber experience:
The “Routine Person.” You wake up at the same time every day, you have a structured morning, and you are disciplined about habits. The pill is often a great fit for you. You will follow the fasting rules without thinking about it.
The “Chaotic Morning” Person. Your mornings are unpredictable — you eat when you can, rush out the door, or your schedule changes constantly. The weekly injection is probably a better match. No fasting window, no timing constraints. One shot on Sunday evening and you are done for the week.
The “Needle Avoider.” You would rather not start treatment at all than deal with a needle. This is more common than people admit, and it is a legitimate barrier to treatment. The Wegovy pill exists specifically for you. Do not let needle anxiety keep you from medication that could significantly improve your health.
The “Weekly Set-It-and-Forget-It” Person. You do not love daily anything — daily vitamins pile up, daily medications get skipped. But you can absolutely remember (or set a phone alarm for) one thing per week. The injection's once-weekly schedule fits your wiring.
The honest bottom line
Bottom line:
Both forms work. The clinical data now supports this more strongly than ever. If needle anxiety is the only thing holding you back, the Wegovy pill removes that barrier. If you know you will forget a daily pill or cannot manage the fasting window, the shot is the simpler path. Talk to your healthcare provider about which form fits your actual life — not which sounds better in theory.
Ready to explore your options?
See if you qualify for GLP-1 treatment with a quick eligibility check.
Check Your GLP-1 EligibilityWhat the Research Says: Key Studies at a Glance
For those who want the evidence in one place, here is a summary of the major studies comparing oral and injectable semaglutide.
| Study | Year | Type | Key Finding |
|---|---|---|---|
| PIONEER program | 2019 | Phase III (oral semaglutide, diabetes) | Rybelsus 14 mg: HbA1c −1.0 to −1.4%, modest weight loss |
| SUSTAIN program | 2017–2019 | Phase III (injectable semaglutide, diabetes) | Ozempic 1.0 mg: HbA1c −1.5 to −1.8%, greater weight loss |
| STEP 1 | 2021 | Phase III (Wegovy injection, weight loss) | 2.4 mg weekly: ~15% body weight loss over 68 weeks |
| SELECT | 2023 | CV outcomes (injectable semaglutide 2.4 mg) | 20% reduction in major CV events in adults with obesity + CVD |
| OASIS 4 | 2025 | Phase III (Wegovy pill, weight loss) | 25 mg daily: 16.6% body weight loss over 64 weeks |
| PMC real-world study | 2024 | Retrospective (T2D patients) | No statistically significant weight loss difference between oral and injectable |
| Croatia study | 2024 | Retrospective (T2D patients) | Identical proportion (56.7%) achieved ≥5% weight loss in both groups |
| ABCD UK audit | 2025 | Real-world (2,120 patients) | Injectable showed greater HbA1c reduction; other outcomes comparable |
| Cureus meta-analysis | 2025 | 4 studies, 559 patients | Injectable may offer slightly better glycemic control |
[Full citations with DOI links are listed in the Sources section at the end of this page.]
Drug Interactions and Timing Considerations
Semaglutide — in either form — can interact with other medications, primarily through its effect on stomach emptying. Because semaglutide slows gastric emptying, it can change how quickly other oral medications are absorbed into your bloodstream.
Why this matters more for the pill
When you take oral semaglutide, you must wait at least 30 minutes before taking any other oral medications. This is not just about semaglutide's own absorption — it is about preventing your other medications from interfering with semaglutide, and vice versa. If you take a blood pressure pill or a thyroid medication at the same time as your semaglutide tablet, both drugs may absorb differently than expected.
Medications to discuss with your doctor
Levothyroxine (thyroid medication): Also requires an empty stomach. Your pharmacist can help you sequence these two medications — typically semaglutide first, then levothyroxine 30 minutes later, then food 30–60 minutes after that.
Insulin and sulfonylureas: Semaglutide lowers blood sugar on its own. If you also take insulin or a sulfonylurea (like glipizide or glyburide), your doctor may need to reduce those doses to avoid low blood sugar. This applies to both oral and injectable forms.
Blood thinners (warfarin): Because semaglutide slows gastric emptying, absorption of warfarin could theoretically be affected. Your doctor may want to monitor your INR more closely when starting semaglutide.
Oral contraceptives: There is a theoretical concern that delayed gastric emptying could reduce absorption of birth control pills. The clinical significance is unclear, but discuss it with your provider, particularly during dose escalation when GI symptoms are most active.
Acetaminophen and other pain medications: Studies showed delayed absorption of acetaminophen with semaglutide, though the total amount absorbed was not significantly changed. The pain reliever may take longer to kick in, but it still works.
For the injectable form, interaction concerns are simpler because the injection bypasses your stomach entirely. The main interactions to watch are the blood-sugar-lowering ones (insulin, sulfonylureas) and the gastric emptying effect on other oral medications you take.
[Source: FDA prescribing information for Wegovy, Ozempic, and Rybelsus]
What Happens If You Stop Taking Semaglutide?
This is one of the most common follow-up questions people ask after starting treatment, and the answer matters for your long-term planning regardless of which form you take. For a deeper dive, see our guide on what happens when you stop taking GLP-1.
The short version: weight tends to come back after stopping. In clinical trials, participants who discontinued semaglutide after 68 weeks regained roughly two-thirds of the weight they had lost within a year of stopping. Appetite typically returns within 1–2 weeks as drug levels fall, and the metabolic and hormonal changes that drove weight loss gradually reverse.
This is not a failure of the drug — it reflects the biology of obesity. Semaglutide works by mimicking a hormone (GLP-1) that regulates hunger and metabolism. When the medication stops, those signals return to their pre-treatment baseline. This is similar to how blood pressure rises again when someone stops taking blood pressure medication.
What this means for your decision between oral and injectable: adherence and long-term sustainability matter more than the initial choice of form. If the fasting requirement of the pill will eventually cause you to skip doses or stop entirely, the weekly injection may produce better long-term results — and vice versa. The best form is the one you can realistically take for as long as your doctor recommends.
There is no FDA-required tapering schedule for semaglutide. If you stop, drug levels decline gradually over several weeks due to semaglutide's long half-life. Some providers choose a step-down plan to make appetite rebound easier to manage, but the best approach is individualized — follow your prescriber's guidance. Maintaining the diet, exercise, and behavioral changes you built during treatment can help slow weight regain.
[Sources: Wilding JPH, et al. STEP 1 Extension, NEJM 2022;387:1349-1360; FDA prescribing information]
Alternatives If Semaglutide Is Not Right for You
Semaglutide is not the only option. If it does not fit — due to contraindications, cost, side effects, or preference — other treatments exist.
Tirzepatide (Mounjaro/Zepbound) is a dual GIP/GLP-1 receptor agonist, currently available as a weekly injection. In trials, higher-dose tirzepatide has shown even greater weight loss than semaglutide (up to roughly 22.5% body weight). Eli Lilly's oral non-peptide GLP-1, orforglipron, was FDA-approved on April 1, 2026 and is sold as Foundayo. Compare in our semaglutide vs tirzepatide guide.
Liraglutide (Saxenda) is an older GLP-1 daily injection for weight loss. Less effective than semaglutide but still useful for some.
Other medications include phentermine-topiramate (Qsymia), naltrexone-bupropion (Contrave), and other FDA-approved options that work through different mechanisms.
Lifestyle intervention — diet, exercise, behavioral support, working with a registered dietitian — remains the foundation. GLP-1 medications for weight loss work best when combined with healthy changes, not as a replacement for them.
Talk to your provider about the full range of options. Explore best GLP-1 online programs for telehealth options.
How to Get Started With Semaglutide
Every form of semaglutide requires a prescription from a licensed healthcare provider.
Step 1: Check eligibility
For weight loss (Wegovy pill or injection): you generally need a BMI of 30 or higher, or a BMI of 27 or higher with at least one weight-related condition — high blood pressure, type 2 diabetes, high cholesterol, or sleep apnea.
For type 2 diabetes (Rybelsus, Ozempic): your doctor will determine if a GLP-1 fits your treatment plan based on your current A1c, other medications, and overall health.
Step 2: See a provider
You can see your primary care doctor, an endocrinologist, an obesity medicine specialist, or a licensed telehealth provider. A thorough evaluation should include a review of your medical history, current medications, contraindications, relevant lab work, and a real conversation about your goals and preferences — including whether oral or injectable is the better fit.
Several telehealth providers offer quick eligibility assessments with licensed clinicians and can help you access semaglutide at competitive pricing. If you are interested, you can check your eligibility here.
Step 3: Start low and stick with the titration
Whichever form you choose, begin at the lowest dose and increase gradually. Do not skip ahead. The slow ramp-up exists to reduce side effects and help you stay on treatment long-term. Most people who quit do so in the first few weeks because of nausea — and slow titration is the best defense against that.
Take the first step
Find out if you qualify for GLP-1 weight loss treatment in minutes.
Check Your GLP-1 EligibilityFrequently Asked Questions
Sources and Methodology
Every clinical data point, dosing instruction, and safety statement in this article is drawn from one or more of the following sources. This is not medical advice — it is carefully cited medical information designed to help you have a better-informed conversation with your healthcare provider.
FDA Prescribing Information:
- Wegovy (semaglutide) tablets and injection prescribing information. Novo Nordisk. Updated December 2025. [FDA AccessData]
- Ozempic (semaglutide) injection prescribing information. Novo Nordisk. Updated 2025 (includes Jan 2025 CKD/FLOW trial indication). [FDA AccessData]
- Rybelsus (semaglutide) tablets prescribing information. Novo Nordisk. Updated 2025 (includes R1 and R2 formulations). [FDA AccessData]
FDA Safety Communications:
- “FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss.” FDA.gov, updated 2025.
- “FDA alerts health care providers, compounders and patients of dosing errors associated with compounded injectable semaglutide products.” FDA.gov.
Clinical Trials:
- Wharton S, Lingvay I, Bogdanski P, et al. Oral semaglutide 25 mg in adults with overweight or obesity (OASIS 4). NEJM. 2025;393(11):1077-1087. doi:10.1056/NEJMoa2500969
- Wilding JPH, et al. Once-weekly semaglutide in adults with overweight or obesity (STEP 1). NEJM. 2021;384:989-1002. doi:10.1056/NEJMoa2032183
- Lincoff AM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes (SELECT). NEJM. 2023;389:2221-2232. doi:10.1056/NEJMoa2307563
- Pratley RE, et al. Efficacy of semaglutide in subcutaneous and oral formulations (PIONEER/SUSTAIN review). Frontiers in Endocrinology. 2021. PMC8269445.
Real-World Studies:
- Real-world comparison of oral versus injectable semaglutide. J Pharm Technol. 2024. PMC11559783.
- Effectiveness of oral versus injectable semaglutide in Croatia. MDPI Diabetology. 2024;5(1):5.
- Su Khin K, et al. Real-world data comparing injectable vs. oral semaglutide — ABCD national audit. Diabetes. 2025;74(Supp 1):756-P.
- Comparative effectiveness and safety of oral versus subcutaneous semaglutide (meta-analysis). Cureus. April 2025.
Pricing:
- NovoCare.com (Novo Nordisk patient pricing). Verified February 2026.
- Novo Nordisk press release: FDA approval of Wegovy tablets. December 22, 2025.
Update Log
2026-02-12: Accuracy audit applied: corrected Rybelsus CV indication (removed — not FDA-approved for CV risk reduction), added Ozempic CKD indication (FLOW trial, Jan 2025), updated Ozempic tablets timeline to Feb 4 2026 approval with Q2 2026 launch, corrected intro weight loss figure to lead with 13.6% ITT, added promotional pricing expiry dates, updated product count to five, corrected taper language, added medical reviewer attribution. All changes verified against FDA prescribing information.
2026-02-11: Initial publication. All dosing, pricing, safety, and clinical data verified against current FDA labels and manufacturer sources. Pricing verified via NovoCare.com and Novo Nordisk announcements.
This article will be updated when new clinical data, FDA label changes, or significant pricing changes occur. Bookmark this page and check the date at the top.
Medical Disclaimer: This content is for informational purposes only and is not medical advice. Always consult a licensed healthcare provider before starting any medication. Full medical disclaimer.
Related Articles
What Is GLP-1?
A complete beginner's guide to GLP-1 medications, how they work, and what to expect.
GLP-1s for Weight Loss
How GLP-1 receptor agonists promote weight loss through appetite reduction and metabolic changes.
Learn About GLP-1
Educational hub covering GLP-1 science, medications, eligibility, and treatment options.
History of GLP-1 Drugs
The complete timeline of GLP-1 drug development from discovery to modern weight loss treatments.
Semaglutide vs Tirzepatide
Head-to-head comparison of semaglutide and tirzepatide for weight loss effectiveness and cost.
Retatrutide vs Tirzepatide
Triple agonist vs dual agonist: trial data, side effects, availability, and what you can actually do today.