Retatrutide: What It Is, Trial Results, Side Effects & How to Get It
Retatrutide is an investigational "triple-G" weight loss drug developed by Eli Lilly showing up to 29% body weight reduction in clinical trials—more than any currently approved obesity medication. This guide covers everything you need to know about retatrutide: FDA status, TRIUMPH-4 results, side effects, and how to access it safely.

Medical Disclaimer: This content is for informational purposes only and does not constitute medical advice. Retatrutide is an investigational medication not approved by the FDA. Always consult a healthcare provider before starting any treatment.
The Quick Answer
Retatrutide is an investigational weight loss medication developed by Eli Lilly that targets three hormone receptors (GLP-1, GIP, and glucagon) instead of one or two. In the first Phase 3 readout (TRIUMPH-4, studying obesity with knee osteoarthritis), Eli Lilly reported mean weight loss of 28.7% (71.2 lbs) at 68 weeks with 12 mg and 26.4% (64.2 lbs) with 9 mg, compared to 2.1% with placebo.
However, retatrutide is NOT FDA approved and not commercially available. For most people, the only legitimate way to access it today is through clinical trial enrollment. (In rare cases, investigational drugs may be available through regulated expanded access programs, but this is uncommon.)
Any "retatrutide peptide" sold online is unregulated and potentially dangerous. No official FDA approval date has been announced; Lilly expects additional Phase 3 readouts in 2026.

Infographic summary: In TRIUMPH-4 topline results (68 weeks), retatrutide 12 mg showed -28.7% mean weight loss (71.2 lbs) and 9 mg showed -26.4% (64.2 lbs) vs placebo -2.1% (4.6 lbs). TRIUMPH-4 results are company-reported; peer-reviewed publication pending. Source: PRNewswire
At a Glance: What We Know Today
| Factor | Current Status | Confidence | Source |
|---|---|---|---|
| FDA Status | Not approved (investigational) | Confirmed | FDA |
| Developer | Eli Lilly and Company | Confirmed | Eli Lilly |
| Mechanism | GIP + GLP-1 + Glucagon receptor agonist | Confirmed | NEJM |
| Administration | Once-weekly injection (in trials) | Confirmed | Clinical trial protocols |
| Legal Access | Clinical trials only | Confirmed | FDA, Eli Lilly |
| Compounding | Cannot be used in compounding | Confirmed | FDA |
| Expected Approval | Unknown (no announced date; Phase 3 readouts expected 2026) | Projected | Eli Lilly |
| Estimated Cost | $1,000–$1,500/month | Projected | Based on comparable drugs |
Retatrutide in One Minute
If you're short on time, here's what matters:
- 1What it is: An investigational injectable medication that mimics three hormones to reduce appetite, slow digestion, and increase metabolism.
- 2Who it's being studied for: Adults with obesity (BMI ≥30) or overweight with health conditions, with or without Type 2 diabetes.
- 3What's exciting: Trial participants lost 24–29% of their body weight—more than any approved medication—with additional benefits for blood pressure, cholesterol, and joint pain.
- 4What's still unknown: Long-term safety beyond 68 weeks, rare side effects, and how it performs in larger, more diverse populations.
- 5What to do next: If you want to try retatrutide, search for recruiting clinical trials at ClinicalTrials.gov. If you need treatment now, talk to your doctor about FDA-approved alternatives like Wegovy or Zepbound.
If you're searching "where to buy retatrutide": You cannot legally buy it anywhere. Products sold online as "retatrutide peptide" or "research chemicals" are unregulated, potentially counterfeit, and illegal to sell for human use. The FDA has explicitly warned against these products.
Is Retatrutide FDA Approved?
No. Retatrutide is not FDA approved.
As of January 2026, retatrutide remains an investigational medication. It cannot be legally prescribed by any doctor, dispensed by any pharmacy, or sold by any retailer in the United States or elsewhere. The only way to access retatrutide legally is by enrolling in a clinical trial.
What "Investigational" Actually Means
When a drug is investigational, it means:
- It has not completed the full regulatory review process
- The FDA has not confirmed it is safe and effective for general use
- No official prescribing information, dosing guidelines, or drug label exists
- Healthcare providers cannot legally prescribe it outside of research settings
- Insurance cannot cover it (because there's no approved indication)
Retatrutide is currently in Phase 3 clinical trials—the final stage before a company can apply for FDA approval. While Phase 3 results so far have been positive, more trials are ongoing, and the full safety and efficacy picture is still emerging.
Can Retatrutide Be Compounded?
No. The FDA has explicitly stated that retatrutide cannot be used in compounding under federal law and notes it is not a component of any FDA-approved drug.
Compounding pharmacies can sometimes create customized versions of FDA-approved medications. However, because retatrutide has never been approved, it does not qualify for compounding. Any pharmacy or clinic claiming to offer "compounded retatrutide" is operating outside FDA regulations.
When Could Retatrutide Be Available?
Here's the realistic timeline based on current trial completion dates and standard FDA review processes:
| Milestone | Status / Expected Timing |
|---|---|
| TRIUMPH-4 Phase 3 results | December 2025 ✓ (Completed - Positive topline; peer-reviewed publication pending) |
| Remaining Phase 3 trials complete | Expected throughout 2026 |
| New Drug Application (NDA) submission | Timing depends on trial results |
| FDA review period | Typically 6–10 months after submission |
| Potential FDA approval | No official date announced |
| Commercial availability | Unknown (depends on approval) |
Important: This timeline reflects current information from Eli Lilly press releases. Retatrutide is still investigational. No official FDA approval date has been announced. Timing depends on trial completion, regulatory review, and manufacturing readiness.
What Is Retatrutide?
Retatrutide (research code: LY3437943) is an investigational medication being developed by Eli Lilly and Company for the treatment of obesity, Type 2 diabetes, and related conditions. It belongs to a new class of drugs called "triple agonists" because it activates three different hormone receptors in the body.
The Basics
- Developer: Eli Lilly and Company (also makers of Mounjaro and Zepbound)
- Research Code: LY3437943
- Brand Name: Not yet assigned (will be determined if/when FDA approved)
- Common Nicknames: "Triple-G" drug, "Triple agonist"
- Administration: Once-weekly subcutaneous injection (under the skin)
- Drug Class: GIP/GLP-1/Glucagon receptor agonist
Why Retatrutide Is Getting Attention
Retatrutide has generated significant interest because early clinical trials suggest it may produce greater weight loss than any currently approved medication:
| Medication | Mechanism | Avg. Weight Loss | Timeframe |
|---|---|---|---|
| Retatrutide | Triple agonist (GLP-1 + GIP + Glucagon) | 24–29% | 48–68 weeks |
| Tirzepatide (Zepbound) | Dual agonist (GLP-1 + GIP) | 20–22% | 72 weeks |
| Semaglutide (Wegovy) | Single agonist (GLP-1) | 15–17% | 68 weeks |
The key difference is the glucagon receptor activation, which may increase metabolism and promote additional fat burning beyond what GLP-1 alone can achieve.
What Conditions Is Retatrutide Being Studied For?
Eli Lilly is running multiple Phase 3 trials (the TRIUMPH program) studying retatrutide for:
- Obesity and overweight (with or without weight-related health conditions)
- Type 2 diabetes (with obesity/overweight)
- Knee osteoarthritis (in people with obesity)
- Obstructive sleep apnea (in people with obesity)
- Metabolic dysfunction-associated steatotic liver disease (formerly NAFLD/NASH)
- Cardiovascular and kidney outcomes (long-term risk reduction)
- Chronic low back pain (in people with obesity)
How Does Retatrutide Work?
Retatrutide works by mimicking three natural hormones that regulate appetite, blood sugar, and metabolism. This "triple agonist" approach is what sets it apart from existing medications.
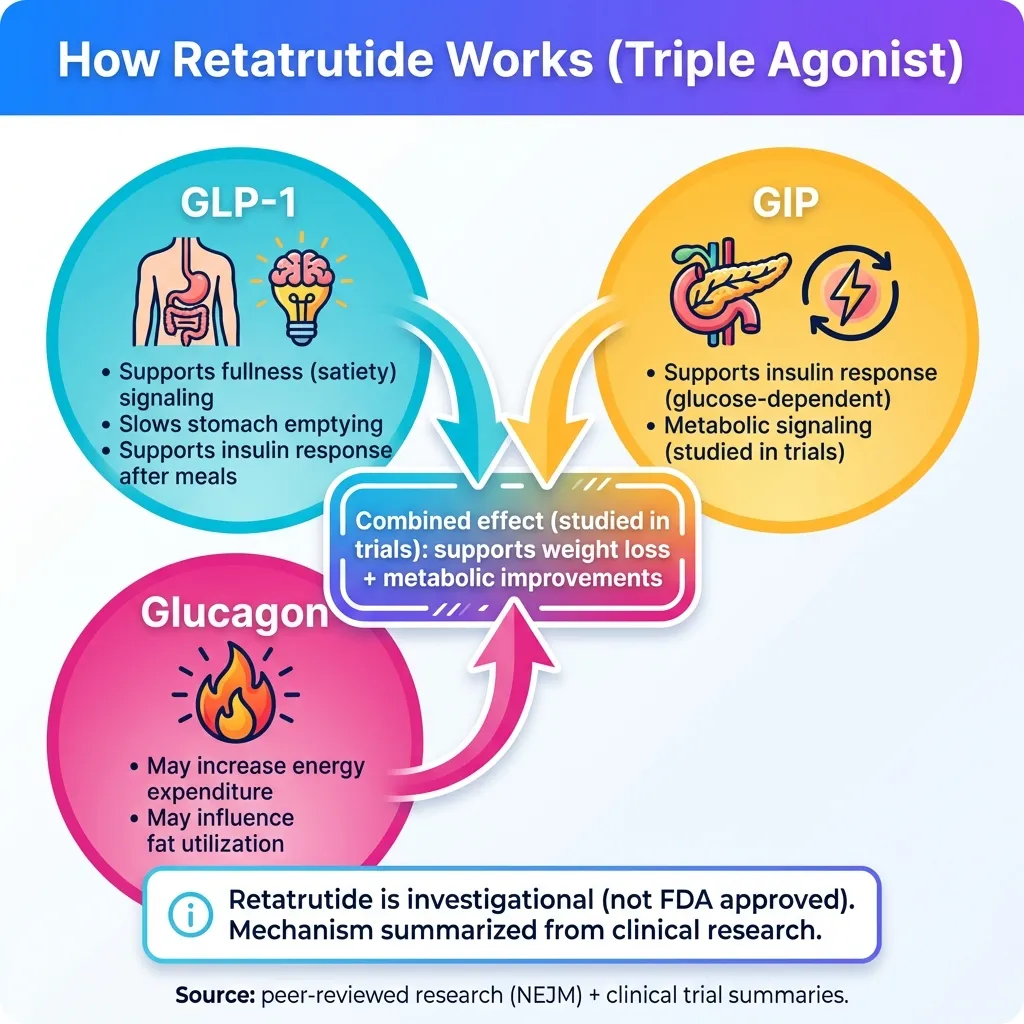
Visual representation of retatrutide's triple-agonist mechanism. Educational only.
The Three Hormone Receptors
1GLP-1 (Glucagon-Like Peptide-1)
GLP-1 is a gut hormone released after eating. When retatrutide activates GLP-1 receptors, it:
- Slows stomach emptying (so you feel full longer)
- Reduces appetite signals in the brain
- Increases insulin release after meals
- Decreases glucagon release (which normally raises blood sugar)
This is the same receptor targeted by Ozempic, Wegovy, and Rybelsus (semaglutide). It's the foundation of most current GLP-1 weight loss medications.
2GIP (Glucose-Dependent Insulinotropic Polypeptide)
GIP is another gut hormone that works alongside GLP-1. When retatrutide activates GIP receptors, it:
- Enhances insulin secretion
- Improves fat metabolism
- May have direct effects on fat tissue
Tirzepatide (Mounjaro, Zepbound) was the first approved medication to combine GLP-1 and GIP activity. Adding GIP appears to boost weight loss beyond GLP-1 alone.
3Glucagon
Here's where retatrutide differs from everything else. Glucagon is a hormone that normally tells your liver to release stored sugar. But research shows glucagon also:
- Increases energy expenditure (you burn more calories at rest)
- Promotes the breakdown of fat for energy (lipolysis)
- Affects satiety signaling in the brain
The combination of GLP-1's appetite suppression with glucagon's metabolism boost may explain why retatrutide appears to produce greater weight loss than dual agonists like tirzepatide.
Why Triple May Be Better Than Dual or Single
Think of it this way:
- SingleSemaglutide: Reduces appetite and slows digestion
- DualTirzepatide: Reduces appetite + improves insulin/fat metabolism
- TripleRetatrutide: Reduces appetite + improves metabolism + increases calorie burning
Each additional mechanism addresses a different aspect of weight regulation. The clinical trial data suggests these effects are additive—more targets, more weight loss. However, retatrutide has not been directly compared to tirzepatide or semaglutide in head-to-head trials. For a detailed comparison between the two, see our tirzepatide vs retatrutide guide.
Retatrutide Clinical Trial Results
The evidence for retatrutide comes from Phase 2 and Phase 3 clinical trials. Here's what the data shows.
Phase 2 Trial (NEJM, 2023)
The landmark Phase 2 trial was published in the New England Journal of Medicine in August 2023. This study established the foundation for retatrutide's development.
Study Details: 338 adults with obesity (BMI ≥30 or ≥27 with health condition), no Type 2 diabetes, 48-week treatment period, randomized to retatrutide (1mg, 4mg, 8mg, or 12mg weekly) or placebo.
Phase 2 Weight Loss Results
| Dose | Weight Loss at 24 Weeks | Weight Loss at 48 Weeks |
|---|---|---|
| 1 mg | -7.2% | -8.7% |
| 4 mg | -12.9% | -17.1% |
| 8 mg | -17.3% | -22.8% |
| 12 mg | -17.5% | -24.2% |
| Placebo | -1.6% | -2.1% |
Key Findings:
- At the highest dose (12mg), participants lost an average of 24.2% of their body weight—about 58 pounds
- 100% of participants on the 8mg and 12mg doses lost at least 5% of their body weight
- 83% of participants on the 12mg dose lost at least 15% of their body weight
- Weight loss was still continuing at 48 weeks (no plateau reached)
Phase 3 TRIUMPH-4 Results (December 2025)
Note: These are company-reported topline results; full peer-reviewed publication is pending.
Study Details: 445 adults with obesity/overweight AND knee osteoarthritis, no Type 2 diabetes, 68-week treatment period, average starting weight: 248.5 lbs (BMI 40.4), randomized to retatrutide 9mg, 12mg, or placebo.
TRIUMPH-4 Weight Loss Results
| Dose | Weight Loss at 68 Weeks | Pounds Lost (Average) |
|---|---|---|
| 9 mg | -26.4% | 64.2 lbs |
| 12 mg | -28.7% | 71.2 lbs |
| Placebo | -2.1% | 4.6 lbs |
Knee Pain Improvement
- Pain scores reduced by up to 75.8%
- 12–14% on retatrutide were completely pain-free (vs. 4.2% placebo)
- Significant improvements in physical function
Cardiovascular Benefits
- Systolic BP reduced by 14 mmHg (12mg)
- Improvements in non-HDL cholesterol
- Reduced triglycerides and inflammation markers
Ongoing TRIUMPH Trials
| Trial | NCT Number | Population | Doses | Completion | Status |
|---|---|---|---|---|---|
| TRIUMPH-1 | NCT05929066 | Obesity without T2D | 4, 9, 12 mg | 2026 | Recruiting |
| TRIUMPH-2 | NCT05928793 | T2D + obesity | 4, 9, 12 mg | 2026 | Recruiting |
| TRIUMPH-3 | NCT06147336 | Obesity + CVD | 9, 12 mg | 2026 | Recruiting |
| TRIUMPH-4 | NCT05931367 | Obesity + knee OA | 9, 12 mg | Dec 2025 | Completed |
Source: ClinicalTrials.gov. Additional trials studying sleep apnea, MASH, chronic low back pain, and long-term outcomes are also underway.
What We Still Don't Know
Questions Remaining:
- • Long-term efficacy beyond 68 weeks
- • Durability after stopping
- • Real-world effectiveness
- • Rare adverse events in larger populations
- • Data for special populations (elderly, kidney disease)
- • Head-to-head comparisons with existing meds
What Results Show:
- • Substantial weight loss in controlled settings
- • Dose-dependent effects (higher = more loss)
- • Metabolic health markers improve
- • Side effects similar to other GLP-1s
- • Weight loss continues beyond 48 weeks
- • Benefits for joint pain observed
Retatrutide Side Effects and Safety
Like other GLP-1 medications, retatrutide's most common side effects are gastrointestinal. Here's what the clinical trials have shown.
Most Common Side Effects (TRIUMPH-4)
| Side Effect | 9mg Dose | 12mg Dose | Placebo |
|---|---|---|---|
| Nausea | 38.1% | 43.2% | 10.7% |
| Diarrhea | 34.7% | 33.1% | 13.4% |
| Constipation | 21.8% | 25.0% | 8.7% |
| Vomiting | 20.4% | 20.9% | 0.0% |
| Decreased appetite | 19.0% | 18.2% | 9.4% |
| Dysesthesia (tingling/numbness)* | 8.8% | 20.9% | 0.7% |
*Dysesthesia (tingling/numbness) is a new finding in Phase 3 trials that was not prominently reported in Phase 2. These events were generally mild and rarely led to stopping treatment. Source: PharmExec
For tips on managing GI side effects, see our GLP-1 side effect relief guide.
When Side Effects Typically Occur
- Are most common during the first few weeks of treatment
- Occur or worsen during dose increases (titration)
- Are rated as mild to moderate in severity
- Improve as the body adjusts to the medication
- Can be minimized by starting at a low dose and increasing slowly
Serious Side Effects (Rare)
- Pancreatitis — Rare but serious; stop medication and seek care for severe abdominal pain
- Gallbladder problems — Including gallstones, may be more common during rapid weight loss
- Hypoglycemia — Mainly a concern if combined with insulin or sulfonylureas
- Allergic reactions — Rare; seek immediate care for severe symptoms
Thyroid tumors: In animal studies, GLP-1 medications have caused thyroid tumors. It's unknown whether this applies to humans. Retatrutide should not be used by anyone with a personal or family history of medullary thyroid cancer or MEN 2.
Discontinuation Rates
In TRIUMPH-4:
- 12.2% on 9mg stopped due to adverse events
- 18.2% on 12mg stopped due to adverse events
- 4.0% on placebo stopped due to adverse events
Some discontinuations were due to "perceived excessive weight loss"—meaning some participants lost weight faster than they wanted.
Dosing Information
Important: Retatrutide is not FDA approved, so there is no official prescribing information. This information is based on clinical trial protocols and may change.
Trial Dosing Schedule (TRIUMPH-4 Protocol)
Because retatrutide is investigational, there is no FDA-approved dose. In TRIUMPH-4, participants started at 2 mg once weekly and were escalated in 4-week intervals to their assigned maintenance dose (9 mg or 12 mg), per protocol.
| Week | Dose | Notes |
|---|---|---|
| Weeks 1-4 | 2 mg weekly | Starting dose (TRIUMPH-4 protocol) |
| Every 4 weeks | Stepwise escalation | Per protocol based on tolerability |
| Maintenance | 9 or 12 mg weekly | Assigned maintenance dose (trial-dependent) |
The gradual escalation helps minimize side effects. Clinical trials typically took 16-20 weeks to reach the target maintenance dose.
Note: Exact titration steps and timing can vary by study and participant tolerability. Refer to the specific trial protocol for full details. Source: PharmExec
Where to Get Retatrutide Safely
Retatrutide is not FDA approved and is not commercially available. For most people, the only legitimate way to access it today is through clinical trial enrollment. (In rare cases, investigational drugs may be available through regulated expanded access programs, but this depends on sponsor and regulator criteria.)
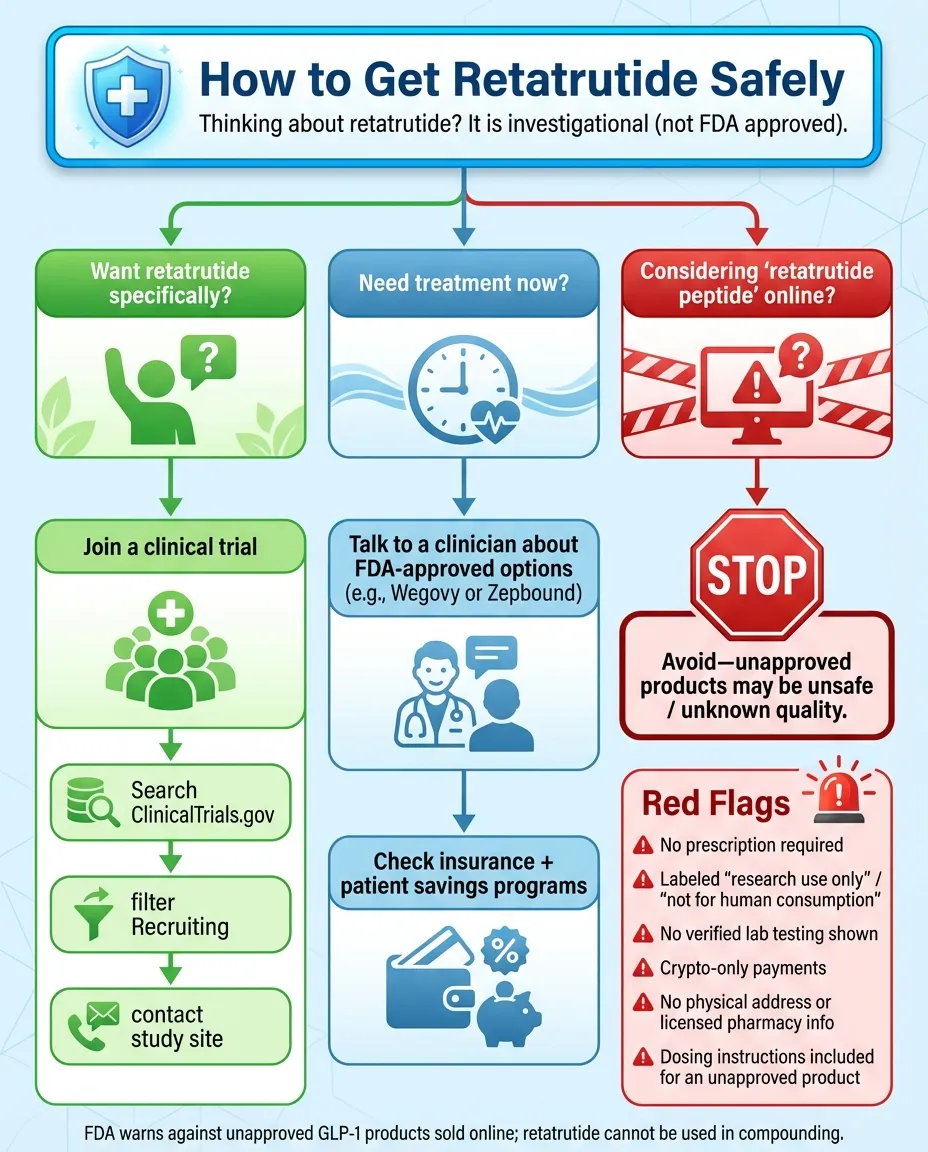
Infographic summary: The only legitimate pathway to retatrutide is through clinical trial enrollment at ClinicalTrials.gov. "Retatrutide peptide" sold online is unregulated, potentially contaminated, and illegal to sell for human use.
How to Find Clinical Trials
- 1Visit ClinicalTrials.gov
- 2Search "retatrutide" in the search bar
- 3Filter by "Recruiting" status and your location
- 4Review eligibility criteria for each trial
- 5Contact the study coordinator via the provided information
Clinical Trial Eligibility Checklist
Based on typical trial criteria, candidates usually have:
| Criteria | Description |
|---|---|
| BMI ≥30 (or ≥27 with health conditions) | Most trials require obesity or overweight with comorbidities |
| Age 18-75 | Adult participants; pediatric trials not yet recruiting |
| No history of pancreatitis | GLP-1 medications may affect pancreas |
| No medullary thyroid cancer or MEN 2 | Black box warning for thyroid tumors in animals |
| No recent bariatric surgery | Usually excluded within 1-2 years |
| Stable weight | Not currently on weight loss medications |
| Willing to attend study visits | Regular clinic visits and monitoring required |
| Able to self-inject | Weekly subcutaneous injections at home |
If You're Not Eligible for a Trial
If you don't qualify for a clinical trial or prefer not to enroll, talk to your doctor about FDA-approved alternatives:
- Wegovy (semaglutide) — Single GLP-1 agonist, ~15-17% weight loss
- Zepbound (tirzepatide) — Dual GLP-1/GIP agonist, ~20-22% weight loss
- Saxenda (liraglutide) — Daily injection GLP-1 option
Compare GLP-1 providers to find the best option for your needs.
"Retatrutide Peptide" Online Warning
These Products Are Dangerous and Illegal
If you search for "retatrutide" online, you'll find products marketed as "retatrutide peptide" or "research chemicals." These are NOT legitimate medications.
- They are not FDA approved or regulated
- They may be counterfeit, contaminated, or mislabeled
- Purity and dosing cannot be verified
- They are illegal to sell for human use
- There is no medical oversight or monitoring
- The FDA has explicitly warned against them
Red Flag Checklist: Signs of Unsafe Sources
| Red Flag | Why It's Dangerous |
|---|---|
| "Retatrutide peptide" or "research chemical" | Not legitimate medication; unregulated |
| "Not for human consumption" disclaimer | Illegal workaround to avoid FDA oversight |
| No prescription required | All legitimate GLP-1 medications require Rx |
| Ships from overseas/China | Cannot verify purity, potency, or safety |
| Significantly lower than expected cost | Likely counterfeit or contaminated |
| No contact information or physical address | Cannot hold seller accountable |
| Payment via cryptocurrency only | Untraceable; no recourse if problems |
| Testimonials from bodybuilding/fitness forums | Not medical evidence; high-risk behavior |
Real harm has occurred: People have been hospitalized after using unregulated "peptides" purchased online. Without knowing what's actually in these products, you're taking a serious health risk with no recourse if something goes wrong.
Retatrutide vs. Tirzepatide vs. Semaglutide
Here's how retatrutide compares to the currently approved GLP-1 medications. For a deeper comparison of approved options, see our semaglutide vs. tirzepatide guide.
| Feature | Retatrutide | Tirzepatide | Semaglutide |
|---|---|---|---|
| Mechanism | GLP-1 + GIP + Glucagon | GLP-1 + GIP | GLP-1 |
| FDA Status | Investigational | Yes (Mounjaro 2022; Zepbound 2023) | Yes (Ozempic 2017; Wegovy 2021; Rybelsus 2019) |
| Brand Names | None yet | Mounjaro, Zepbound | Ozempic, Wegovy, Rybelsus |
| Weight Loss (trials) | 24-29% | 20-22% | 15-17% |
| Dosing | Weekly injection | Weekly injection | Weekly injection or daily oral |
| Access | Clinical trials only | Prescription | Prescription |
| Compounding | Not permitted | Available (shortage) | Available (shortage) |
| Insurance | N/A | Variable | Variable |
| Estimated Cost | $1,000-1,500/mo (projected) | $1,000-1,100/mo | $900-1,400/mo |
Important caveat: These comparisons are based on results from separate studies with different populations. Retatrutide has not been directly compared to tirzepatide or semaglutide in head-to-head trials.
Decision Framework: Which Is Right for You?
| Your Situation | Recommendation | Best For |
|---|---|---|
| Need treatment now + eligible for Rx | Start with FDA-approved options (Wegovy, Zepbound) | Those with BMI criteria and insurance/budget |
| Want retatrutide specifically | Search clinical trials at ClinicalTrials.gov | Motivated participants willing to commit to trial requirements |
| Previous GLP-1 not effective | Try different approved medication or wait for retatrutide | Those who responded poorly to single or dual agonists |
| Cost is primary barrier | Compare compounded options for approved meds | Those seeking lower-cost alternatives to brand-name |
| Want to avoid injection | Ask about Rybelsus (oral semaglutide, approved for T2D). Wegovy is injectable. Oral weight loss options are limited. | Those with needle phobia or preference for oral |
Retatrutide Cost and Insurance
Since retatrutide isn't FDA approved, there's no official pricing yet. However, we can estimate based on similar medications:
Estimated List Price
$1,000–$1,500/month
Based on Zepbound (~$1,060) and Wegovy (~$1,349)
Insurance Coverage
Not yet determined
Will depend on FDA-approved indications
For clinical trial participants: Study medication is provided free of charge during the trial period.
Should You Wait or Start Treatment Now?
This is a question many people are asking. Here's our honest take:
Consider Starting Now If:
- • You have obesity-related health conditions (diabetes, sleep apnea, joint pain)
- • You meet BMI criteria for treatment
- • You have access to approved medications (insurance or budget for self-pay)
- • You want to start building healthy habits during treatment
- • Waiting would mean continued health decline
Consider Waiting If:
- • You specifically want retatrutide's triple mechanism
- • You're eligible for a clinical trial
- • Previous GLP-1 medications haven't worked well for you
- • Cost is a barrier and you can wait for generic options
- • You have no urgent health conditions requiring treatment
The bottom line: If you need treatment now and qualify for approved medications, there's no reason to wait. You can always switch to retatrutide later if it's approved and offers clear advantages for your situation. Obesity is a chronic condition that benefits from earlier intervention.
What Happens When You Stop Treatment?
Based on studies of similar medications, weight typically begins to return after stopping treatment. The extent of weight regain varies, but clinical trials show most people regain significant weight within a year.
For detailed information on what to expect after stopping GLP-1 medications, see our comprehensive guide: What happens when you stop taking GLP-1.
Key takeaway: Weight loss medications typically need to be continued to maintain results—similar to how blood pressure medications manage hypertension. They work best when combined with lifestyle changes (improved nutrition, increased physical activity).
Frequently Asked Questions About Retatrutide
Sources and References
Primary Sources
- New England Journal of Medicine (2023): Jastreboff AM, et al. "Triple–Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial." N Engl J Med 2023;389:514-526. Read study
- Eli Lilly and Company (December 2025): "Lilly's triple agonist, retatrutide, delivered weight loss of up to an average of 71.2 lbs along with substantial relief from osteoarthritis pain in first successful Phase 3 trial." Press release | PRNewswire
- U.S. Food and Drug Administration: "FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss." FDA Warning
- ClinicalTrials.gov: Retatrutide clinical trial registry entries. View trials
Additional Clinical References
- The Lancet (2023): Rosenstock J, et al. "Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes." Lancet 2023;402:529-544.
- Nature Medicine (2024): Sanyal AJ, et al. "Triple hormone receptor agonist retatrutide for metabolic dysfunction-associated steatotic liver disease."
- Eli Lilly Official: "What to know about retatrutide." Read more
Comparative Clinical Trials
- SURMOUNT-1 (Tirzepatide): Jastreboff AM, et al. N Engl J Med 2022;387:205-216.
- STEP 1 (Semaglutide): Wilding JPH, et al. N Engl J Med 2021;384:989-1002.
- STEP 1 Extension (Semaglutide withdrawal): Wilding JPH, et al. "Weight regain and cardiometabolic effects after withdrawal of semaglutide." Diabetes Obes Metab 2022.
Editorial Standards
How we verify information:
- • We cite primary sources (peer-reviewed journals, FDA communications, manufacturer announcements)
- • We label "company-stated" claims vs. "peer-reviewed" evidence
- • We clearly indicate when information is projected vs. confirmed
How we handle conflicts:
- • Affiliate relationships do not influence our clinical conclusions
- • We recommend FDA-approved treatments over unapproved options
- • We prioritize patient safety in all recommendations
Read more about our editorial standards.
Update Log
- January 24, 2026: Updated dosing titration table to reflect TRIUMPH-4 protocol (2 mg start, 4-week escalation). Fixed placebo weight loss to 4.6 lbs per PRNewswire. Added inline citations. Clarified FDA approval dates for tirzepatide and semaglutide brands.
- January 23, 2026: Initial publication with TRIUMPH-4 topline results and comprehensive safety summary.
Have questions about retatrutide?
Check if you qualify for a clinical trial at ClinicalTrials.gov or talk to your healthcare provider about FDA-approved weight loss options available today.