
Retatrutide (LY3437943): The investigational triple-agonist targeting GLP-1, GIP, and glucagon receptors
GLP 3 Retatrutide: What It Is, How It Works, and What We Actually Know in 2026
For informational purposes only—not medical advice.
If you searched glp 3 retatrutide, here's the short answer: "GLP-3" is not a real hormone. It's a nickname for retatrutide (LY3437943), an investigational triple-agonist drug from Eli Lilly that targets three hormone receptors at once — GLP-1, GIP, and glucagon. In December 2025, Lilly reported Phase 3 trial results showing participants lost an average of 28.7% of their body weight (~71 lbs) over 68 weeks — among the largest average weight-loss results ever reported in a Phase 3 obesity drug trial.
Retatrutide is not FDA-approved. It is not legally available for prescription or purchase. The only legitimate way to access it today is through a clinical trial.
This guide covers everything known about GLP-3 retatrutide as of — mechanism, trial results, side effects, dosage protocols, FDA timeline, comparisons to current medications, and how to find a clinical trial near you. Every clinical claim is sourced from peer-reviewed publications, official Eli Lilly press releases, or ClinicalTrials.gov.
Answer: GLP-3 retatrutide (LY3437943) is an investigational triple-agonist drug from Eli Lilly targeting GLP-1, GIP, and glucagon receptors. Phase 3 trials show up to 28.7% weight loss. It is not FDA-approved and cannot be legally purchased. "GLP-3" is not a real hormone — it's a nickname. The only legitimate access is through clinical trials.
Reviewed by: WPG Research Team, Health & Wellness Editor
Last Updated: | Sources: NEJM, Nature Medicine, Eli Lilly, FDA.gov, ClinicalTrials.gov
Retatrutide shows among the highest weight loss results reported in any Phase 3 obesity drug trial to date (~28.7% body weight at 68 weeks). Its triple-agonist mechanism — targeting GLP-1, GIP, and glucagon simultaneously — is genuinely novel, and the liver fat reduction data (82% at 24 weeks, per Sanyal et al., Nature Medicine 2024) is in a class of its own.
However: retatrutide is NOT FDA-approved and is NOT available for purchase — legally or safely. The only legitimate access today is through clinical trials. The dysesthesia signal (abnormal skin sensations in up to 21% of participants) is a genuinely new finding that needs monitoring across the remaining Phase 3 trials.
If you need weight loss treatment now, FDA-approved options like semaglutide and tirzepatide are available today with years of real-world safety data. Starting treatment now doesn't prevent you from switching to retatrutide later if it's approved.
GLP-3 Retatrutide at a Glance
| Category | Details |
|---|---|
| Full name | Retatrutide (code name: LY3437943) |
| What "GLP-3" means | Nickname — targets 3 receptors (GLP-1, GIP, glucagon) |
| Developer | Eli Lilly and Company |
| Type | Once-weekly subcutaneous injection |
| Phase 3 weight loss | 28.7% body weight (~71 lbs) at 68 weeks (TRIUMPH-4, 12mg; company-reported topline). Note: TRIUMPH-4 enrolled patients with knee osteoarthritis; general population results from TRIUMPH-1 are pending. |
| Phase 2 weight loss | 24.2% body weight (~58 lbs) at 48 weeks (NEJM, 12mg dose) |
| FDA status | Not approved — Phase 3 clinical trials ongoing |
| Earliest possible approval | Estimated 2027 (not confirmed by Lilly or FDA) |
| How to access now | Clinical trials only — search at ClinicalTrials.gov |
| Can you buy it online? | No. FDA has issued warnings against unapproved retatrutide products |
Sources: Jastreboff et al., NEJM 2023; Eli Lilly press release, December 11, 2025; FDA.gov
What Is "GLP-3"? (And Why the Name Is Misleading)
Let's clear this up right away: there is no human hormone called GLP-3.
Your body produces GLP-1 (glucagon-like peptide-1) and GLP-2 (glucagon-like peptide-2). That's it. There is no third one. The "GLP-3" label started circulating on social media and in news headlines as shorthand for a drug that targets three receptors — but it's not a scientific term, and you won't find it in any medical journal or FDA filing.
The drug people are actually talking about is retatrutide, sometimes called "triple-G" or "triple agonist" in clinical literature. Eli Lilly, the company developing it, has never used the term "GLP-3." Neither has the FDA or the New England Journal of Medicine.
So why does this matter? Because if you search for "GLP-3" looking for real information, you'll run into a lot of confusion. Some sites selling unregulated peptides use the term to sound legitimate. Others use it as clickbait. The correct name is retatrutide, and that's what you should look for when evaluating any source.
Quick glossary of terms you'll see online
| Term | What it actually means |
|---|---|
| GLP-3 | Not a real hormone. Online nickname for retatrutide |
| Triple agonist | A drug that activates three different hormone receptors |
| Triple-G | Slang for GLP-1 + GIP + Glucagon (the three targets) |
| LY3437943 | Retatrutide's research code name |
| GLP3RT, GLP-3RT | Informal abbreviations used on forums |
| "Reta" / "Reta peptide" | Shorthand for retatrutide, often used by peptide sellers |
If a website calls it "GLP-3 peptide" and offers to sell it to you, that's a red flag. More on that in the safety section below.
How Retatrutide Works: The Triple-Agonist Mechanism Explained
To understand why retatrutide is generating so much attention, it helps to see how weight loss medications have evolved over the past few years.
Generation 1 — Single agonist (GLP-1 only): Drugs like semaglutide (sold as Ozempic for diabetes and Wegovy for weight loss) target one receptor: GLP-1. This slows your stomach emptying, reduces appetite, and helps regulate blood sugar. It works. Wegovy showed about 15% average weight loss in its pivotal trial (STEP 1). That was a breakthrough.
Generation 2 — Dual agonist (GLP-1 + GIP): Tirzepatide (sold as Mounjaro for diabetes and Zepbound for weight loss) targets two receptors: GLP-1 and GIP. GIP enhances insulin release and works alongside GLP-1 on appetite signaling. The result was even better — about 21-22% average weight loss in the SURMOUNT-1 trial.
Generation 3 — Triple agonist (GLP-1 + GIP + Glucagon): Retatrutide targets all three. The addition of glucagon is the key differentiator, and it changes the equation in a fundamental way.
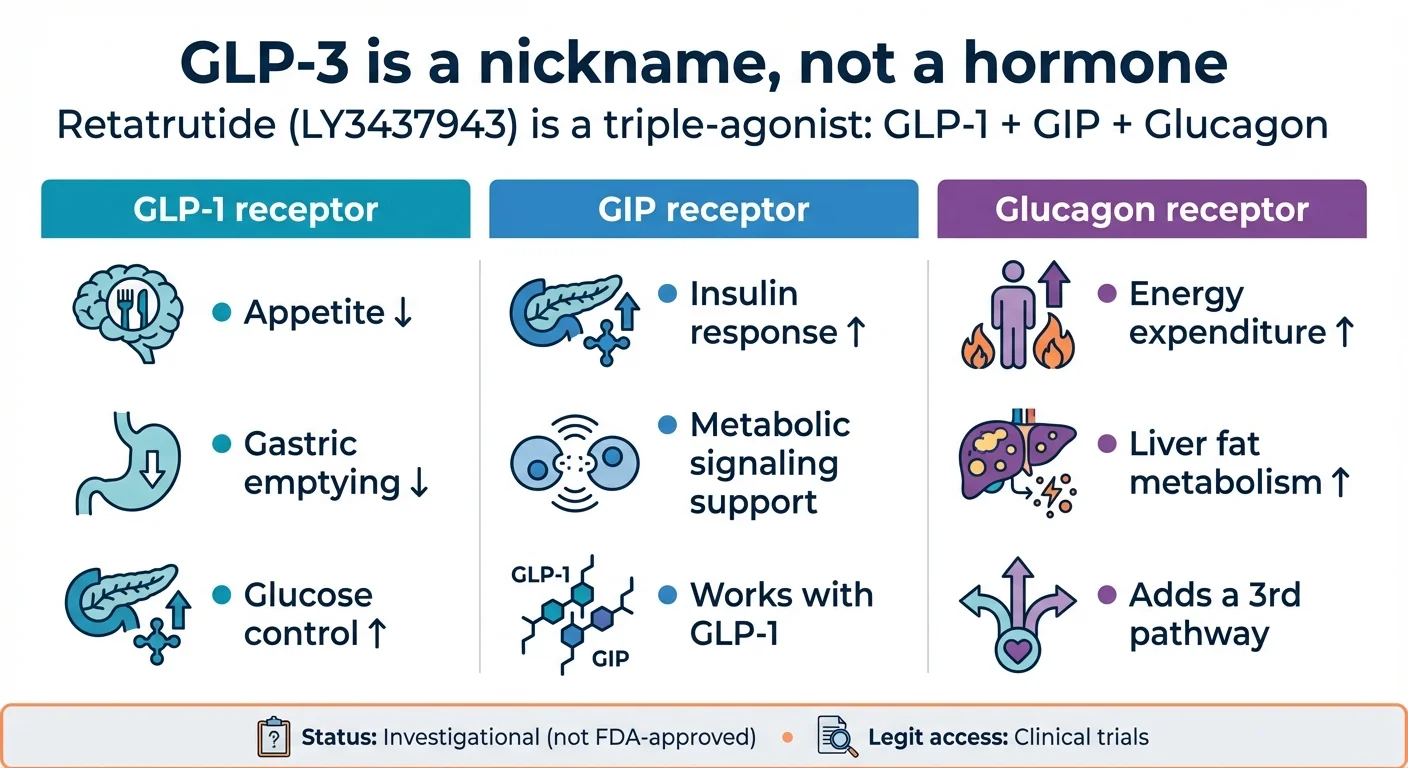
How retatrutide's triple-agonist mechanism targets three receptors simultaneously for enhanced weight loss
Here's what each receptor does and why it matters:
| Receptor | What it does | How it helps with weight loss |
|---|---|---|
| GLP-1 | Slows stomach emptying, reduces appetite, improves blood sugar control | You eat less and feel full longer |
| GIP | Enhances insulin release after eating, supports GLP-1's appetite effects | Improves how your body processes food and fat |
| Glucagon | Increases energy expenditure, promotes breakdown of stored fat, accelerates liver fat metabolism | Your body burns more calories — even at rest |
The glucagon piece is what makes retatrutide different from everything that came before it. Semaglutide and tirzepatide primarily work by reducing how much you eat. Retatrutide does that and increases how much energy your body uses. It's not just suppressing appetite — it's shifting your metabolism to burn stored fat more aggressively.
This is also why retatrutide shows dramatic results for fatty liver disease. In a Phase 2 substudy published in Nature Medicine (June 2024), the 12mg dose reduced liver fat by 82.4% at 24 weeks. For context, semaglutide achieves about 50% and tirzepatide about 47% over longer timeframes. The glucagon pathway drives fat out of the liver at a rate nothing else has matched.
The trade-off — and there is always a trade-off — is that glucagon agonism can increase heart rate and may contribute to side effects not seen with single or dual agonists. We'll cover those in detail below.
Why Glucagon Is the Breakthrough (and the Risk)
Here's an analogy that might help. Think of weight loss medications as having two levers to pull: "eat less" and "burn more."
Semaglutide pulls the "eat less" lever. It suppresses appetite and slows digestion, so you naturally consume fewer calories. Tirzepatide pulls the same lever harder, with the added help of GIP.
Retatrutide pulls both levers. It suppresses appetite and it tells your body to increase energy expenditure — to actively burn stored fat, particularly in the liver. That's the glucagon piece. Your metabolism speeds up. Your body reaches into its fat stores and converts them to fuel.
This is also why the liver fat results are so dramatic. Glucagon specifically targets hepatic fat metabolism. It tells the liver: process this stored fat, convert it, get it out. No other approved obesity drug has a direct mechanism for this.
The flip side is that glucagon is a powerful hormone. It naturally raises blood sugar (which GLP-1 and GIP counteract in retatrutide's design), it can increase heart rate, and it may contribute to the dysesthesia signal that's emerging in Phase 3. More power, more complexity, more unknowns.
For the technically curious: Retatrutide is a synthetic peptide designed for once-weekly dosing, with a half-life of approximately 6 days. It's built on a GIP peptide backbone and is most potent at the GIP receptor, moderately potent at GLP-1, and mildest at the glucagon receptor. A fatty acid conjugation extends its duration in the body. For full molecular details, see Coskun et al. in Cell Metabolism (2022).
Sources: Jastreboff et al., NEJM 2023; Sanyal et al., Nature Medicine 2024; Coskun et al., Cell Metabolism 2022; PMC review 2025
Clinical Trial Results: What the Data Actually Shows
This is the section that matters most. Not what social media says. Not what peptide sellers claim. What happened in controlled, peer-reviewed clinical trials with real patients.
Phase 2: The Study That Started the Hype (NEJM, 2023)
The Phase 2 trial was published in The New England Journal of Medicine — one of the most respected medical journals in the world. It enrolled 338 adults with obesity (BMI ≥30) or overweight (BMI ≥27 with weight-related conditions) across U.S. sites. Participants were randomized to receive various doses of retatrutide or placebo for 48 weeks, alongside lifestyle counseling.
| Weekly Dose | Starting Dose | Weight Loss at 24 Weeks | Weight Loss at 48 Weeks |
|---|---|---|---|
| 1 mg | 1 mg (no escalation) | -7.2% | -8.7% |
| 4 mg | 2 mg (escalated) | -11.8% | -16.3% |
| 4 mg | 4 mg (no escalation) | -13.9% | -17.8% |
| 8 mg | 2 mg (escalated) | -16.7% | -21.7% |
| 8 mg | 4 mg (escalated) | -17.9% | -23.9% |
| 12 mg | 2 mg (escalated) | -17.5% | -24.2% |
| Placebo | — | -1.6% | -2.1% |
Source: Jastreboff et al., NEJM 2023 (NCT04881760)
Several things stand out. First, the 12mg group lost an average of 24.2% of their body weight — roughly 58 lbs — in under a year. Second, participants were still losing weight at 48 weeks. They hadn't hit a plateau. The trial simply ended before they stopped losing. That's unusual and significant.
Third, how you start matters. Groups that began at 2mg and escalated slowly had meaningfully fewer side effects than groups that started at 4mg. The weight loss outcomes were nearly identical either way. This validated the "start low, go slow" approach that's now standard in all the Phase 3 trials.
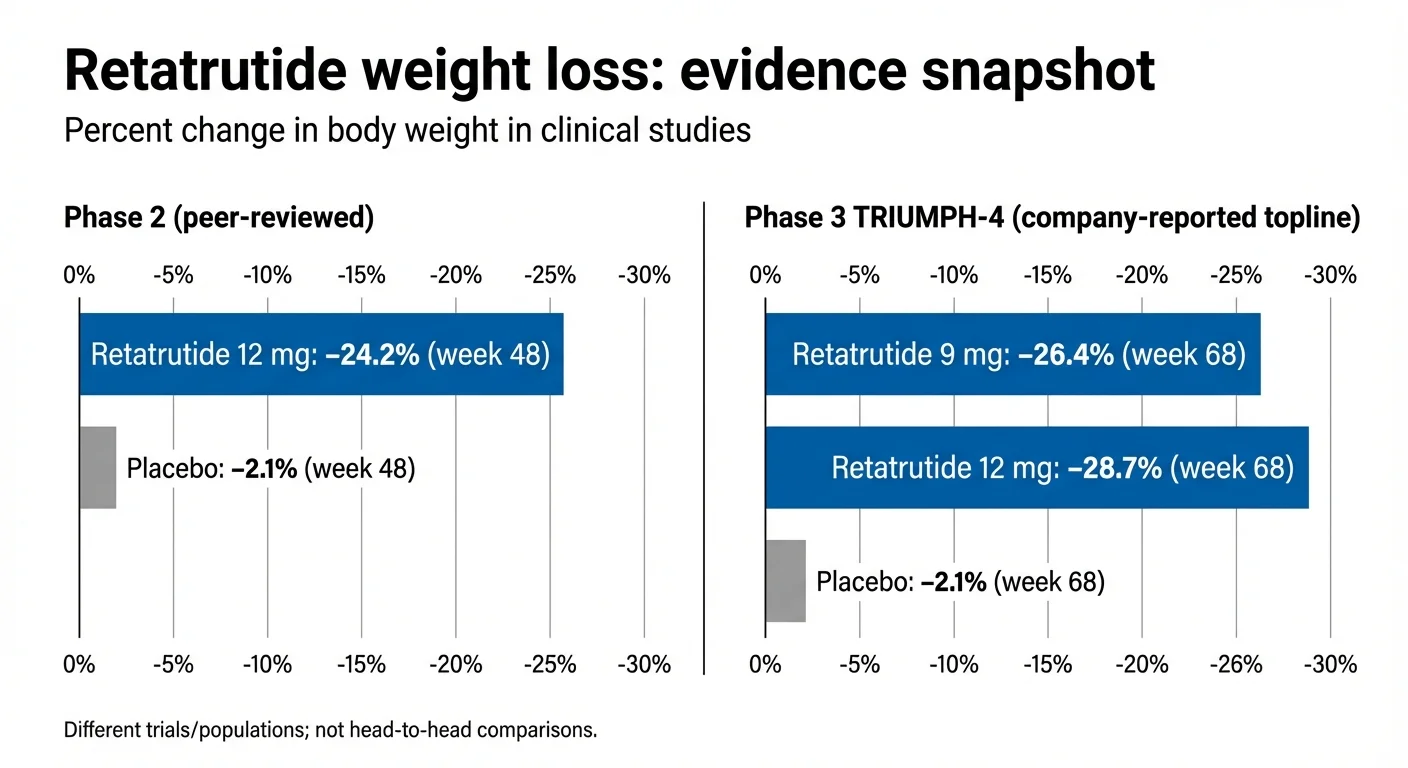
Retatrutide weight loss results: Phase 2 vs Phase 3 TRIUMPH-4 trial data
Phase 3: TRIUMPH-4 Results (December 2025)
In December 2025, Eli Lilly announced topline results from TRIUMPH-4, the first completed Phase 3 trial for retatrutide. This was a 68-week study of 445 adults with obesity or overweight who also had knee osteoarthritis.
| Outcome | Retatrutide 9mg | Retatrutide 12mg | Placebo |
|---|---|---|---|
| Weight loss (efficacy estimand) | -26.4% (-64.2 lbs) | -28.7% (-71.2 lbs) | -2.1% (-4.6 lbs) |
| Weight loss (treatment-regimen estimand) | -20.0% (-50.5 lbs) | -23.7% (-60.0 lbs) | -4.6% (-11.7 lbs) |
| Knee pain reduction (WOMAC) | -4.5 points (-75.8%) | -4.4 points (-74.3%) | -2.4 points (-40.3%) |
| Completely pain-free at end of trial | 14.1% | 12.0% | 4.2% |
| Discontinued due to side effects | 12.2% | 18.2% | 4.0% |
Source: Eli Lilly press release, December 11, 2025; company-reported topline results (NCT05931367). Full peer-reviewed publication pending.
A quick note on the two different weight loss numbers. The "efficacy estimand" shows what happened for people who stayed on the drug for the full 68 weeks — it represents what retatrutide can do under ideal conditions. The "treatment-regimen estimand" includes everyone who was randomized, even those who dropped out early. It represents what actually happened in the real study population. The truth for any individual will fall somewhere between these numbers.
Either way, 28.7% average weight loss is among the largest results ever reported in a Phase 3 obesity drug trial. For someone starting at 250 lbs, that's about 72 lbs. Some participants lost considerably more.
Important: These are company-reported topline results. Detailed data will be presented at a future medical meeting and published in a peer-reviewed journal.
The knee osteoarthritis results were also striking. Pain scores dropped by roughly 75% — and more than 1 in 8 people on retatrutide were completely free of knee pain by the end of the trial.
Phase 3: TRANSCEND-T2D-1 Results (May 2026)
In May 2026, Eli Lilly released topline results from TRANSCEND-T2D-1, the first Phase 3 trial for retatrutide in adults with type 2 diabetes. This was a 40-week study using the 12 mg dose.
| Outcome (12 mg, 40 weeks) | Retatrutide | Placebo |
|---|---|---|
| Mean weight loss | 36.6 lbs (16.8%) | ~1–2 lbs |
| A1C reduction | 1.7–2.0% | Minimal |
Source: Eli Lilly press release, May 2026; company-reported topline results (TRANSCEND-T2D-1). Full peer-reviewed publication pending. These results do not constitute FDA approval.
Liver Fat Reduction (Phase 2 Substudy)
A substudy of the Phase 2 trial looked specifically at participants with fatty liver disease (MASLD). The results, published in Nature Medicine in June 2024, showed liver fat reductions that surpassed every other drug studied:
| Dose | Liver Fat Reduction at 24 Weeks |
|---|---|
| 1 mg | -42.9% |
| 4 mg | -57.0% |
| 8 mg | -81.4% |
| 12 mg | -82.4% |
| Placebo | +0.3% |
Source: Sanyal et al., Nature Medicine 2024
An 82% reduction in liver fat in just 24 weeks is remarkable. For indirect comparison (different trials, populations, and durations), semaglutide showed approximately 50% reduction over 72 weeks, and tirzepatide approximately 47% at 52 weeks. The glucagon receptor activity in retatrutide appears to be the driver — glucagon specifically promotes fat metabolism in the liver.
What We Know vs. What We Don't (Yet)
This distinction matters. Being honest about uncertainty is part of giving you the complete picture.
What the data supports:
- Retatrutide produces substantial, dose-dependent weight loss
- The 12mg dose shows the highest efficacy but also the most side effects
- Slow dose escalation (starting at 2mg) significantly improves tolerability
- Liver fat reduction is dramatically superior to existing drugs
- Knee OA pain improves substantially with weight loss on retatrutide
- Cardiovascular risk markers (cholesterol, blood pressure, inflammation) improved in trials
What we don't know yet:
- Long-term safety beyond ~68 weeks
- What happens to weight after stopping (no retatrutide-specific data yet)
- Whether weight loss is primarily fat vs. lean mass (body composition data pending)
- Cardiovascular outcomes (TRIUMPH-Outcomes trial ongoing, results expected 2027-2028)
- How it performs in the general obesity population without osteoarthritis (TRIUMPH-1 results pending)
- Optimal maintenance dose (4mg maintenance is being studied)
- Real-world effectiveness outside controlled trial conditions
Retatrutide vs. Semaglutide vs. Tirzepatide: How They Compare
This is the comparison most people want. Just know upfront: no study has directly compared retatrutide to semaglutide or tirzepatide head-to-head. The numbers below come from different trials with different patient populations, durations, and designs. Treat them as context, not a definitive ranking.
| Semaglutide (Wegovy) | Tirzepatide (Zepbound) | Retatrutide | |
|---|---|---|---|
| Mechanism | GLP-1 only | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
| Developer | Novo Nordisk | Eli Lilly | Eli Lilly |
| FDA approved? | Yes (2021) | Yes (2023) | No — Phase 3 trials |
| Administration | Weekly injection | Weekly injection | Weekly injection |
| Max dose studied | 2.4 mg | 15 mg | 12 mg |
| Weight loss (key trial) | ~14.9% at 68 wks (STEP 1) | ~22.5% at 72 wks (SURMOUNT-1) | ~28.7% at 68 wks (TRIUMPH-4)† |
| Liver fat reduction | ~50% at 72 wks | ~47% at 52 wks | ~82% at 24 wks |
| Nausea rate | ~44% | ~31% | ~43% |
| Discontinuation (AEs) | ~7% | ~6-7% | ~18% (12mg) |
| Unique safety signal | — | — | Dysesthesia (up to 21%) |
| Monthly cost (list price) | ~$1,349 (Wegovy) | ~$1,086 (Zepbound) | Unknown |
| Monthly cost (self-pay) | $349/mo (NovoCare) | $299-449/mo (LillyDirect) | Not available |
| Available now? | Yes | Yes | No — trials only |
Sources: STEP 1 (NEJM 2021, peer-reviewed); SURMOUNT-1 (NEJM 2022, peer-reviewed); TRIUMPH-4 (Eli Lilly press release Dec 2025, company-reported topline†). Cross-trial comparison — not equivalent to head-to-head data. †TRIUMPH-4 uses "efficacy estimand" (on-treatment); STEP 1 figure is "treatment policy estimand" (ITT). Estimand definitions differ across trials.
What this tells us — and what it doesn't: Retatrutide shows the highest weight loss numbers in the group. The liver fat data is in a different league. And the triple mechanism could make it uniquely suited for people with metabolic syndrome, fatty liver disease, or those who need more weight loss than current medications deliver.
But retatrutide also has the highest discontinuation rate (18.2% on 12mg vs. ~7% for the others) and a novel side effect — dysesthesia — that hasn't been seen with semaglutide or tirzepatide. The practical reality is that a drug you stop taking because of side effects doesn't help you. For a deeper dive into these differences, see our tirzepatide vs retatrutide comparison.
Tirzepatide may offer the best balance of strong weight loss with manageable tolerability for most people right now. Semaglutide has the longest real-world safety record and the most data across different conditions (heart disease, kidney disease, sleep apnea).
Retatrutide could eventually surpass both — but it's not available, its long-term safety profile is incomplete, and the dysesthesia signal needs more investigation.
Retatrutide isn't available yet — but semaglutide and tirzepatide are. See which one fits.
Free 2-min quiz. No email required.
Side Effects and Safety: What's Known, What's Uncertain
Every effective medication has side effects. Retatrutide's side effect profile shares a lot in common with other GLP-1 medications — but it also has some signals that are unique and worth understanding.
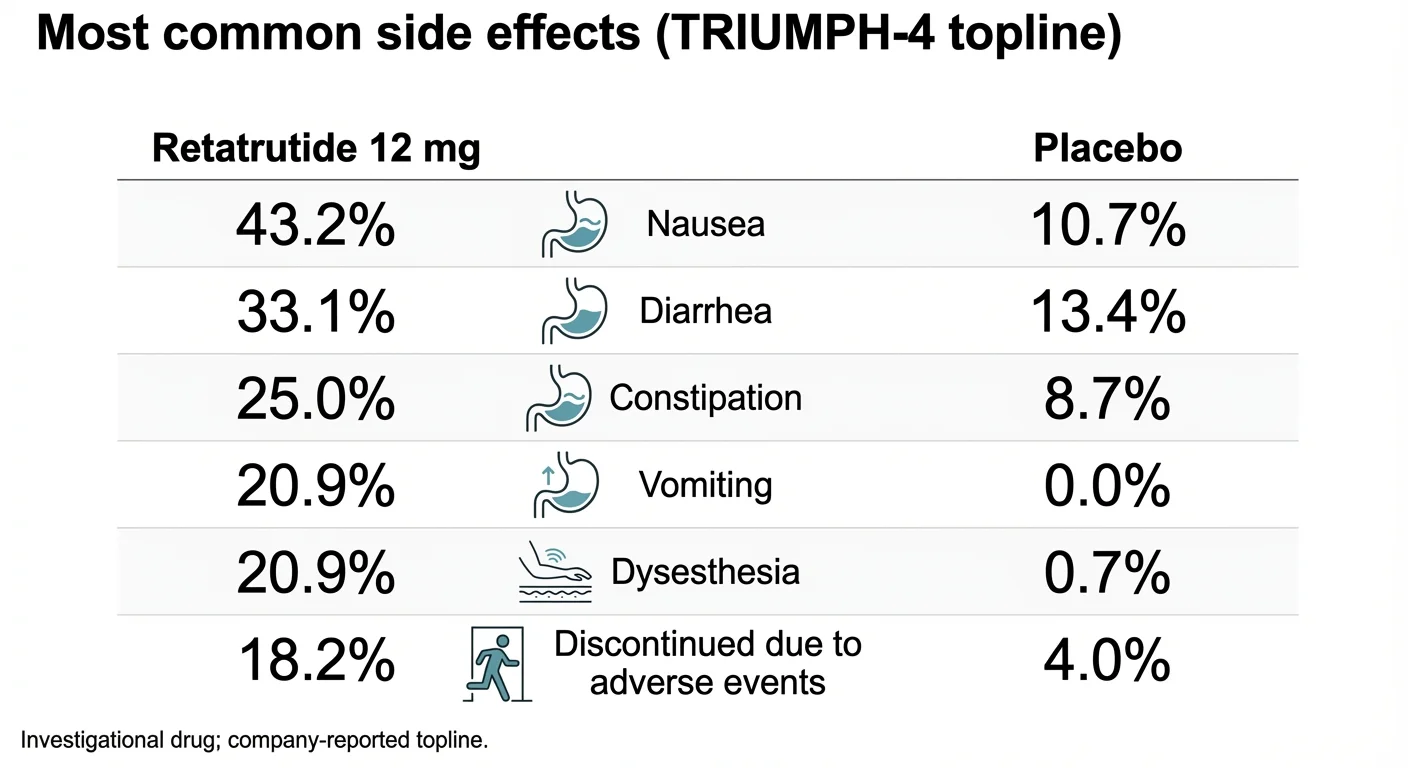
Retatrutide side effects from TRIUMPH-4 Phase 3 trial data, including the novel dysesthesia signal
Common Side Effects (from TRIUMPH-4 Phase 3)
| Side Effect | Retatrutide 9mg | Retatrutide 12mg | Placebo |
|---|---|---|---|
| Nausea | 38.1% | 43.2% | 10.7% |
| Diarrhea | 34.7% | 33.1% | 13.4% |
| Constipation | 21.8% | 25.0% | 8.7% |
| Vomiting | 20.4% | 20.9% | 0.0% |
| Decreased appetite | 19.0% | 18.2% | 9.4% |
| Dysesthesia | 8.8% | 20.9% | 0.7% |
Source: Eli Lilly press release and medical information portal (medical.lilly.com), December 11, 2025. Dose-specific safety data pending peer-reviewed publication.
The gastrointestinal side effects — nausea, diarrhea, vomiting, constipation — are consistent with what you'd see with any incretin-based therapy. They're dose-dependent (more common at higher doses), usually worst during the dose escalation period, and tend to improve over time. The Phase 2 trial showed clearly that starting at 2mg rather than 4mg roughly halves the rate of these symptoms during the first weeks.
The Dysesthesia Signal: What You Need to Know
This is the side effect that's getting the most attention from analysts and physicians, because it's new.
Dysesthesia is an abnormal sensation of touch. Normal contact with the skin feels unusual — tingly, burning, itchy, or painful. It's not the same as numbness. It's more like your sense of touch is "turned up" or distorted.
In TRIUMPH-4, 20.9% of participants on the 12mg dose reported dysesthesia, compared to just 0.7% on placebo. On the 9mg dose, the rate was 8.8%. This is a significant difference, and it was not observed in the Phase 2 trial — making it a genuinely new finding.
Here's what we know and don't know:
- Most cases were described as mild and did not lead to stopping the drug
- It's not seen with semaglutide or tirzepatide — it may be related to glucagon receptor activation, but this isn't confirmed
- The Phase 2 trial did note "altered or enhanced skin sensation" at lower rates, so it may not be entirely new — just more apparent at scale
- It needs investigation across the remaining 7 Phase 3 trials reporting in 2026
Is it a dealbreaker? That depends on how the data evolves. For now, it's a legitimate concern that warrants monitoring — not panic, but not dismissal either.
Heart Rate Effects
Retatrutide causes a modest increase in resting heart rate of about 5-10 beats per minute, peaking around 24 weeks and then declining. This is similar to what's seen with GLP-1 agonists generally. The Phase 2 trial also noted a small number of cardiac arrhythmia cases.
Discontinuation Rates: The Bigger Picture
The 18.2% discontinuation rate on the 12mg dose is notably higher than what's been seen with semaglutide (~7%) or tirzepatide (~6-7%). But there's important context:
- Discontinuation rates were correlated with baseline BMI — people with lower BMI were more likely to stop. Some stopped due to "perceived excessive weight loss." In other words, the drug worked too well for some participants.
- Among participants with BMI ≥35, the discontinuation rate dropped to about 12%.
- The 9mg dose had a lower discontinuation rate (12.2%) while still producing 26.4% weight loss.
This suggests that the 9mg dose may end up being the sweet spot for many people — strong results with better tolerability.
Long-Term Safety: Honestly Unknown
Retatrutide's longest study data is 68 weeks. We don't know what happens at 2, 3, or 5 years. We don't have data on rare but serious outcomes. The TRIUMPH-Outcomes cardiovascular trial won't report until 2027-2028. This is simply the reality of a drug still in development.
Retatrutide Dosage: What Clinical Trials Used
Retatrutide is not FDA-approved, and no official dosing guidelines exist. The information below comes exclusively from published clinical trial protocols. It is not a recommendation, prescription, or guide for self-dosing. The FDA has issued explicit warnings about "research-grade" retatrutide products sold online with dosing instructions. Those products are unapproved, their purity and dosage accuracy are unknown, and using them is both dangerous and illegal.
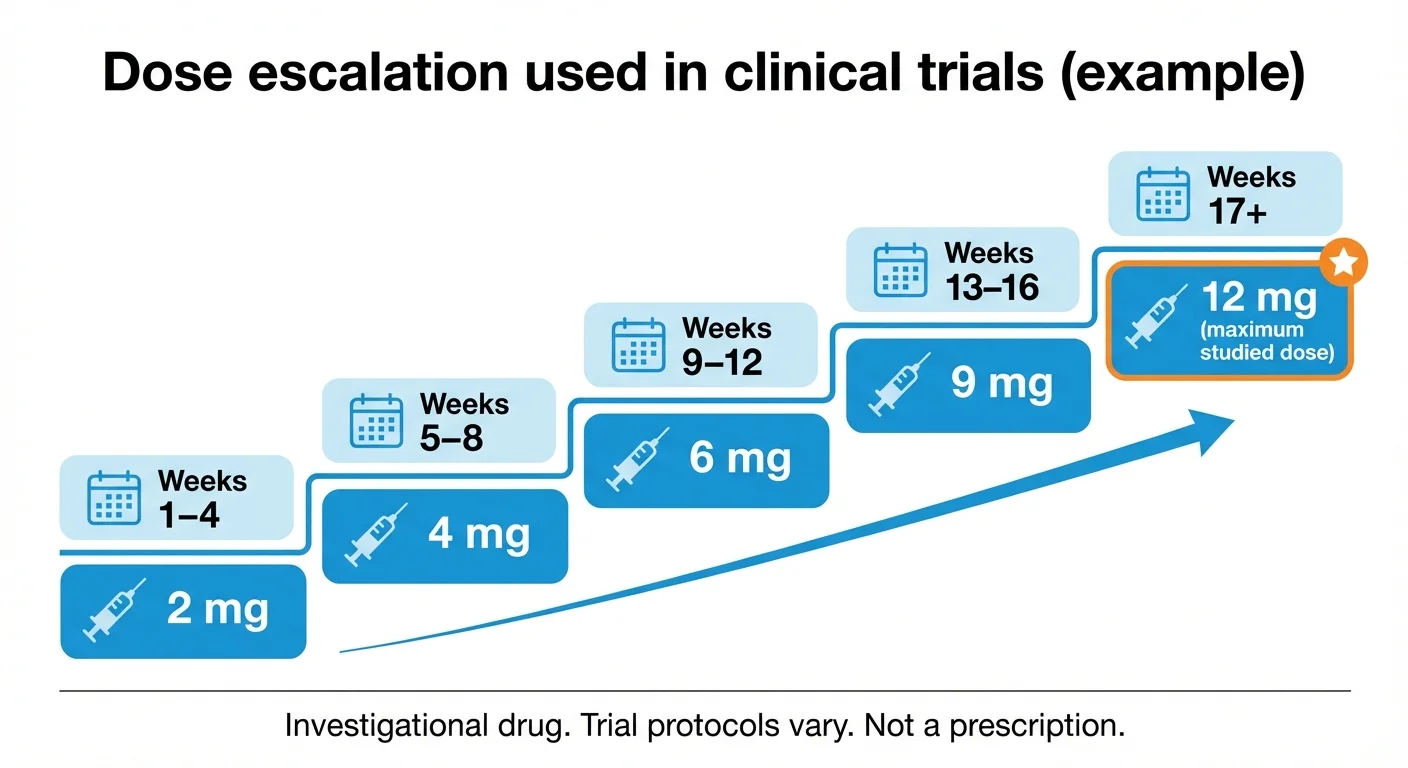
Dose escalation schedule used in retatrutide clinical trials (Phase 2 and Phase 3 TRIUMPH program)
Dose Escalation Schedule (from Clinical Trials)
In both Phase 2 and Phase 3 trials, retatrutide was administered as a once-weekly subcutaneous injection with gradual dose escalation:
| Weeks | Weekly Dose |
|---|---|
| 1-4 | 2 mg |
| 5-8 | 4 mg |
| 9-12 | 6 mg |
| 13-16 | 9 mg |
| 13+ or 17+ | 12 mg (maximum studied dose) |
Source: NEJM Phase 2 protocol; TRIUMPH-4 trial design (Eli Lilly 2025). Schedules vary by protocol; see trial records for full details.
Key principles from the trials
Start at 2mg, not higher. Phase 2 showed that participants who started at 4mg had nearly double the rate of gastrointestinal side effects compared to those who started at 2mg. The weight loss at 48 weeks was virtually the same for both groups. There's no benefit to starting high.
Escalate every 4 weeks. Faster escalation hasn't been studied and would likely increase side effects.
Holding at a dose is fine. If side effects are bothersome, staying at the current dose for an extra 2-4 weeks before escalating is common in trial protocols and doesn't blunt long-term results.
Not everyone needs 12mg. The 8-9mg range produced 22-26% weight loss in trials. The jump to 12mg adds a few more percentage points but also significantly increases side effects and discontinuation rates. The right dose is the one that produces good results at a tolerable level of side effects — and that varies by person.
Maintenance dosing is being studied. The TRIUMPH program is also testing a 4mg maintenance dose. The idea: escalate to 9-12mg for initial weight loss, then step down to 4mg for long-term maintenance. Results are expected in 2026.
Why Online Dosage Charts Are Unreliable
If you've seen retatrutide dosage charts on peptide seller websites, understand what you're looking at: marketing material for an unapproved product, often presented without context about the medical supervision, lifestyle counseling, and safety monitoring that accompanied every dose in clinical trials.
Clinical trial participants had regular check-ins with physicians, blood work monitoring, dietary counseling, and the ability to adjust or stop treatment based on side effects. A dosage chart on a website selling unregulated powder doesn't replicate any of that.
Is Retatrutide FDA Approved? When Could It Be Available?
No. Retatrutide is not FDA-approved. It cannot be legally prescribed or dispensed at any pharmacy. It is an investigational drug in Phase 3 clinical trials.
The TRIUMPH Phase 3 Program: All Current Trials
Eli Lilly's TRIUMPH program is one of the most comprehensive Phase 3 programs in obesity drug history, enrolling over 5,800 participants across multiple trials:
| Trial | What It Studies | Participants | Expected Completion | Status (Feb 2026) |
|---|---|---|---|---|
| TRIUMPH-1 (NCT05929066) | Obesity/overweight (primary registration trial) + nested OSA & OA | ~1,800 | 2026 | Ongoing — main trial for FDA submission |
| TRIUMPH-2 (NCT05929079) | Obesity/overweight + Type 2 Diabetes + nested OSA | ~1,000+ | 2026 | Ongoing |
| TRIUMPH-3 (NCT05882045) | Obesity/overweight + Cardiovascular Disease (BMI ≥35) | ~1,000+ | 2026 | Ongoing |
| TRIUMPH-4 (NCT05931367) | Obesity/overweight + Knee Osteoarthritis | 445 | Complete | Positive results (Dec 2025) |
| TRIUMPH-Outcomes (NCT06383390) | Long-term cardiovascular & renal outcomes | ~10,000 | 2027-2028+ | Ongoing |
| Additional studies | MASLD (fatty liver), chronic low back pain, maintenance dosing | Various | 2026-2027 | Various stages |
Sources: ClinicalTrials.gov; Eli Lilly investor relations; Giblin et al., Diabetes Obes Metab 2026 (trial design paper)
Note: Some media reports cite NCT05869903 for TRIUMPH-4; that is actually an orforglipron study (ATTAIN-1). The correct TRIUMPH-4 registration is NCT05931367, per Eli Lilly's own press materials and ClinicalTrials.gov.
Seven additional Phase 3 readouts are expected throughout 2026. TRIUMPH-1 is the most important — it's the primary registration trial that will form the core of any FDA submission.
Projected Timeline (Estimates, Not Confirmed)
- Phase 3 completion: Most TRIUMPH trials completing throughout 2026
- NDA submission to FDA: Estimated late 2026 (depends on results across the full program)
- FDA review period: Typically 10-12 months for a standard review
- Approval decision: Estimated 2027, possibly earlier if Lilly seeks priority review
- Commercial availability (pharmacies): Several months after approval for manufacturing scale-up
GlobalData, a pharmaceutical analytics firm, projects a 2027 approval with forecasted annual sales reaching $15.6 billion by 2031.
Important caveats: Eli Lilly has not announced an official filing timeline. These are projections, not promises. Phase 3 trial results could be delayed. The dysesthesia signal could require additional study. The FDA could request more data. Drug development timelines regularly shift.
Can You Buy GLP-3 Retatrutide? The Legality and Safety Reality
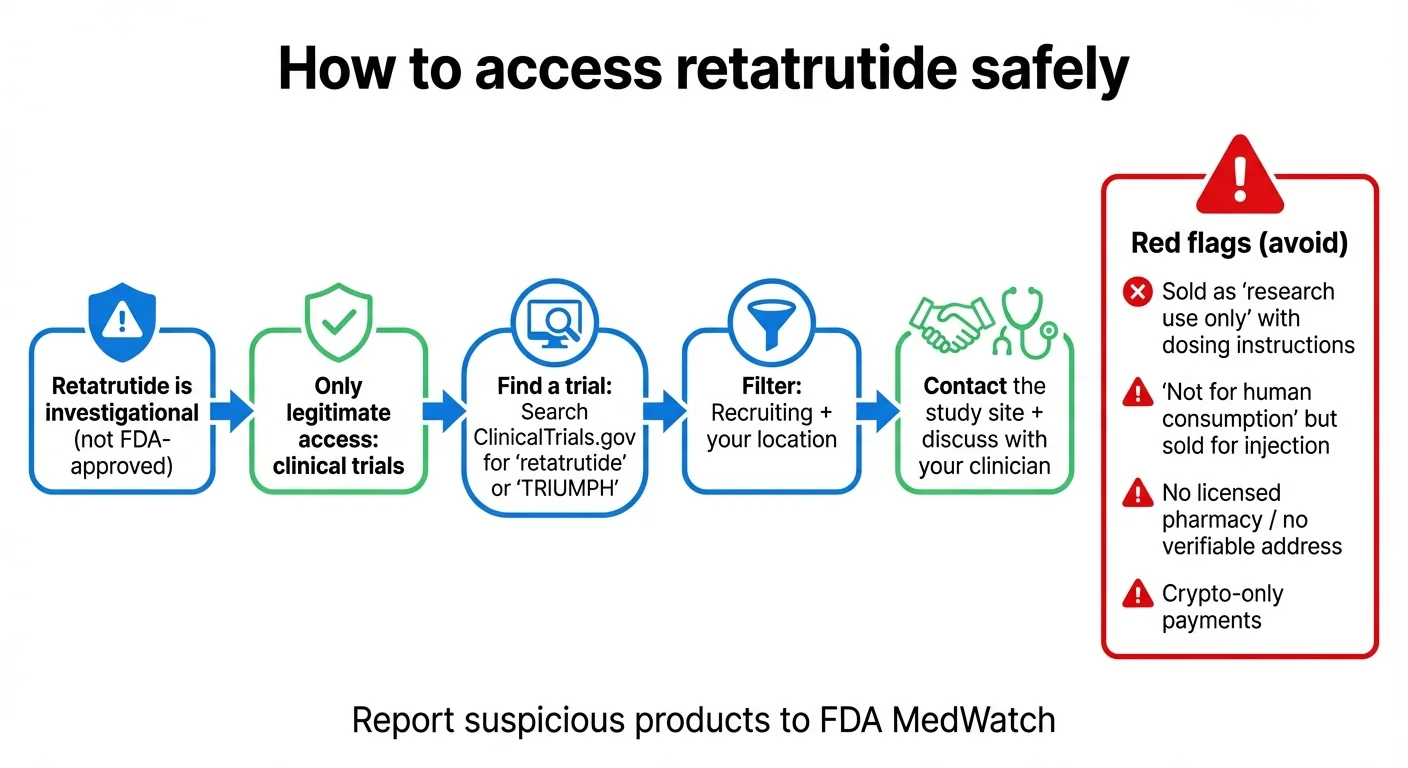
How to identify unsafe retatrutide products and access the drug legally through clinical trials
You cannot legally buy retatrutide in the United States (or most countries). Any website selling "retatrutide" is selling an unapproved, unregulated product.
The FDA has issued explicit warnings about unapproved GLP-1 drugs, including retatrutide, being sold as "research use only" products — often with dosing instructions clearly intended for human use. The FDA states these products are of "unknown quality" and "may be harmful."
The FDA has issued warning letters to companies selling compounded retatrutide, stating that these products are "unapproved new drugs" and "misbranded" when sold for human use. Retatrutide is not eligible for compounding under Section 503A or 503B of the Federal Food, Drug, and Cosmetic Act because it is not on the FDA's approved compounding list.
What you're actually buying from "research peptide" sites
- A product with no verified purity, potency, or sterility
- No manufacturing oversight by the FDA or any regulatory body
- No recourse if the product harms you
- A potential legal liability (buying unapproved drugs for personal use)
- No medical supervision for dose escalation, side effect monitoring, or drug interactions
Red Flags: How to Spot Unsafe Retatrutide Products
If you see any of the following, you're looking at an illegitimate source:
- "For research use only" — with dosing instructions for human use
- "Not for human consumption" — but sold in injectable form with reconstitution guides
- No verifiable company address, phone number, or pharmacist oversight
- Payment only via cryptocurrency or wire transfer
- Claims of "99% purity" with no independent lab verification
- No mention of FDA status or investigational nature
How to Report Suspicious Products
If you encounter retatrutide being sold illegally, you can report it to FDA MedWatch at fda.gov/medwatch or call 1-800-FDA-1088.
Sources: FDA.gov — "Concerns with Unapproved GLP-1 Drugs Used for Weight Loss"; FDA Warning Letter to GLP-1 Solution, September 9, 2025
How to Access Retatrutide Legally: Finding a Clinical Trial
The only legitimate way to get retatrutide today is by enrolling in a clinical trial. This isn't as complicated as it sounds, but it does require some effort.
Note on expanded access: In rare cases, patients with serious conditions who have exhausted other options may be eligible for expanded access (also called "compassionate use") outside of a clinical trial. This requires a physician's request to the manufacturer and FDA authorization. Eli Lilly has not publicly announced an expanded access program for retatrutide, but it is worth discussing with your doctor if you believe you may qualify.
How Clinical Trials Work (Quick Overview)
Clinical trials are research studies that test whether a drug is safe and effective before it can be approved. As a participant, you typically receive the study drug (or placebo) at no cost, get regular medical monitoring, and contribute to research that could help millions of people. You won't necessarily get the drug — some participants receive a placebo. You'll sign an informed consent form that explains the risks. You'll have regular visits with the study team. And you can withdraw at any time.
How to Find a Retatrutide Trial Near You
Step 1: Go to ClinicalTrials.gov and search for "retatrutide" or "TRIUMPH."
Step 2: Filter by status ("Recruiting"), location (your state or country), and condition (obesity, type 2 diabetes, knee osteoarthritis, etc.).
Step 3: Review the eligibility criteria. Most TRIUMPH trials require age 18+, BMI ≥27 or ≥30 (varies by trial), specific comorbidities, no recent heart attack/stroke, no history of medullary thyroid carcinoma or MEN-2, no current use of other weight loss medications, and no weight change >11 lbs in the 90 days before screening. Use our BMI calculator to check your BMI.
Step 4: Contact the study coordinator. Each trial listing includes contact information.
Step 5: Ask your doctor. Your physician can help you evaluate whether a trial is appropriate for your health situation and refer you if needed.
What to Ask the Study Coordinator
- Is this trial currently enrolling in my area?
- What are the specific eligibility criteria I need to meet?
- Is there a possibility I'll receive a placebo instead of the drug?
- How often will I need to visit the study site?
- Are there any costs to me (travel, lab work, etc.)?
- What monitoring and support does the trial include?
- How long does the study last?
- Can I continue receiving the drug after the trial ends?
Key ClinicalTrials.gov Numbers for Reference
- TRIUMPH-1 (main obesity): NCT05929066
- TRIUMPH-2 (obesity + T2 diabetes): NCT05929079
- TRIUMPH-3 (obesity + CV disease): NCT05882045
- TRIUMPH-4 (obesity + knee OA): NCT05931367
- TRIUMPH-Outcomes (CV + renal outcomes): NCT06383390
Source: ClinicalTrials.gov; Eli Lilly medical information
What Happens If You Stop? Discontinuation, Maintenance, and Weight Regain
This is a question people don't ask often enough — but it matters enormously.
Retatrutide-specific data on what happens after you stop the drug is limited because the trials measured results while on treatment. But we have strong evidence from the broader GLP-1/GIP drug class, and the pattern is clear: weight regain after stopping is common and significant.
In the STEP 1 extension study for semaglutide, participants who stopped the drug after 68 weeks regained about two-thirds of their lost weight within a year. A similar pattern was seen with tirzepatide in the SURMOUNT-4 trial — participants who switched from tirzepatide to placebo regained a substantial portion of their weight over 36 weeks.
This isn't a failure of the drugs. It reflects the biology of obesity — it's a chronic condition driven by metabolic and hormonal factors that don't change just because you lost weight. Stopping the medication removes the pharmacological support while the underlying biology remains. Your hunger hormones rebound. Your metabolic rate adjusts. The signals that were suppressed come back.
This is why the 4mg maintenance dose being studied in the TRIUMPH program is so significant. If retatrutide can produce maximum weight loss at 9-12mg and then maintain it at 4mg, the long-term side effect burden and cost could drop substantially. Published step-down maintenance data of this kind is limited across the GLP-1 drug class. The results expected in 2026 could reshape how these medications are used.
What this means for retatrutide
- Plan for long-term treatment, not a short course
- The 4mg maintenance dose being studied in TRIUMPH could be important — if a lower dose can maintain results with fewer side effects, that changes the cost and tolerability equation significantly
- Lifestyle changes (nutrition, movement, sleep) are genuinely important — not as a replacement for medication, but as a complement that may reduce the dose needed and improve results
- Discuss a long-term plan with your doctor before starting any obesity medication
Sources: Wilding et al., PubMed 2022 (STEP 1 extension); Aronne et al., JAMA 2024 (SURMOUNT-4)
Should You Wait for Retatrutide or Start Treatment Now?
This is the practical question most people reading this page are actually trying to answer. There's no universal right answer, but here's a framework to help you think about it.
Starting an available medication now probably makes more sense if:
- Your BMI is ≥30 (or ≥27 with complications like diabetes, sleep apnea, or joint pain) and your health would benefit from treatment today — not in 2027. Use our BMI calculator to check.
- You're experiencing health complications that get worse with time (uncontrolled blood sugar, worsening knee pain, sleep apnea)
- You respond well to semaglutide or tirzepatide — many people achieve 15-22% weight loss, which is life-changing
- You want a drug with a known long-term safety profile and years of real-world data
- You don't want to wait for something that may be delayed
Watching and waiting might make sense if:
- You've tried semaglutide and tirzepatide without adequate results — retatrutide's triple mechanism may work where dual or single agonists didn't
- You've plateaued on a current GLP-1/GIP medication and need more weight loss
- You have significant fatty liver disease — retatrutide's liver fat data is dramatically superior to anything available
- You're comfortable enrolling in a clinical trial
- Your doctor recommends a watchful approach based on your specific situation
What you should NOT do
- Don't wait indefinitely if you have a medical need now. An available treatment that produces 15-20% weight loss is better than an unavailable one that might produce 28%
- Don't buy "retatrutide" online from unregulated sources while you wait. The risks are real and the FDA warnings are serious
- Don't assume retatrutide will definitely be approved on any specific timeline. Phase 3 trials can produce unexpected results. Timelines shift.
The bottom line
Starting treatment now doesn't prevent you from switching to retatrutide later if and when it's approved. A year of effective treatment with an available medication is a year of improved health, reduced risk, and better quality of life that you can't get back by waiting. Explore telehealth providers or check the cheapest GLP-1 options available today.
Talk to your doctor. This is a medical decision that should be made with a clinician who knows your health history — not based on social media hype or fear of missing out.
A Framework for Thinking About Maintenance (Any GLP-1/GIP Drug)
Whether you start treatment now with an available drug or later with retatrutide, the maintenance question will come up eventually. Here's a practical framework:
Duration planning. These medications work as long as you take them. Plan for months to years, not weeks. If you're starting semaglutide or tirzepatide today, don't think of it as a short course — think of it like blood pressure medication for someone with hypertension.
Cost planning. Even with insurance coverage and savings programs, long-term use adds up. Factor this into your decision. Ask your insurer about long-term coverage policies and out-of-pocket caps.
Lifestyle foundation. Medication is the most powerful tool for weight loss, but it works best alongside consistent nutrition, movement, and sleep habits. These habits won't replace the medication's effects, but they may allow a lower maintenance dose and improve your overall health in ways the drug doesn't directly address (strength, cardiovascular fitness, mental health).
Off-ramp strategy. Discuss with your doctor: What's the plan if you want to stop? What monitoring should be in place? What's the threshold for restarting? Having this conversation upfront reduces panic if circumstances change.
Dose optimization. More isn't always better. If 8mg of retatrutide gives you 22% weight loss with manageable side effects, is the extra 6% from 12mg worth the increased nausea and dysesthesia risk? The same logic applies to current drugs. Work with your doctor to find the dose that gives you the best results at a level of side effects you can live with long-term.
Starting now doesn't lock you in — you can switch to retatrutide later. But a year of results is a year of better health you can't get back.
What Will Retatrutide Cost? (And What You Can Do About Cost Now)
Because retatrutide is not FDA-approved, there is no official price. Eli Lilly hasn't announced what it will cost. But we can make educated guesses based on the competitive landscape and Lilly's own pricing history.
What Current Medications Cost (for context)
| Medication | List Price | Self-Pay / Direct Programs | With Commercial Insurance |
|---|---|---|---|
| Wegovy (semaglutide) | ~$1,349/month | $349/mo via NovoCare Pharmacy | As low as $25/mo with savings card |
| Zepbound (tirzepatide) | ~$1,086/month | $299-449/mo via LillyDirect (vials) | As low as $25/mo with savings card |
| Mounjaro (tirzepatide, diabetes) | ~$1,023/month | Varies | Lilly savings card available |
| Retatrutide | Unknown | Not available | Not available |
Pricing as of February 2026. Self-pay programs require a valid prescription and use vials (not pre-filled pens). Pricing changes frequently — verify directly with manufacturer programs.
What Drives Launch Pricing for Obesity Drugs
Efficacy advantage. If TRIUMPH-1 confirms 25-30% weight loss in the general obesity population (not just osteoarthritis patients), Lilly has a strong argument for premium pricing. A drug that produces nearly double the weight loss of Wegovy is objectively more valuable.
Competition. By 2027, the obesity drug market will be more crowded. Novo Nordisk has its own pipeline. Amgen, Pfizer, and Viking Therapeutics are developing competitors. More competition generally pushes prices down.
Insurance coverage. The biggest cost factor for most patients isn't list price — it's insurance. Medicare currently doesn't cover anti-obesity medications (though legislation to change this has been introduced repeatedly). Private insurers are increasingly covering GLP-1s but often with prior authorization requirements. Retatrutide's coverage will depend on FDA-approved indications and how aggressively Lilly negotiates with payers.
Lilly's own savings programs. Lilly has been aggressive with patient savings programs for Zepbound. Expect similar programs for retatrutide if approved.
What You Can Do About Cost Right Now
If cost is a concern and you're considering weight loss medication today:
- Check manufacturer savings programs. Lilly's Zepbound savings card can reduce costs to as low as $25/month for commercially insured patients. Novo Nordisk has similar programs for Wegovy.
- Ask about compounded semaglutide or tirzepatide. During the FDA-declared shortage, compounded versions of these drugs were available at lower cost. The regulatory landscape for compounding changes frequently, so check current FDA guidance and work with a licensed provider.
- Explore telehealth platforms that specialize in weight management — many negotiate lower costs and handle insurance authorization.
- Talk to your insurer. Many plans cover obesity medications with proper documentation of BMI and comorbidities. Your doctor can submit prior authorization with supporting medical records.
Conditions Being Studied Beyond Weight Loss
Retatrutide's triple-agonist mechanism means it may have benefits across several obesity-related conditions. Here's what's being studied:
| Condition | What We Know | Trial Status |
|---|---|---|
| Obesity (general) | Up to 28.7% weight loss in Phase 3 | TRIUMPH-1 ongoing, results 2026 |
| Type 2 Diabetes | HbA1c reduction up to -2.02% + significant weight loss (Phase 1b) | TRIUMPH-2 ongoing, results 2026 |
| Knee Osteoarthritis | 76% pain reduction; 12% of participants pain-free (Phase 3) | TRIUMPH-4 complete, positive |
| Fatty Liver Disease (MASLD) | 82% liver fat reduction at 24 weeks (Phase 2 substudy) | Phase 3 planned |
| Obstructive Sleep Apnea | Nested study within TRIUMPH-1 and TRIUMPH-2 | Ongoing |
| Cardiovascular Outcomes | Being studied in high-risk population (BMI ≥35 + CVD) | TRIUMPH-3 and TRIUMPH-Outcomes ongoing |
| Chronic Low Back Pain | Separate study | Planned/ongoing |
Sources: ClinicalTrials.gov; Eli Lilly investor materials; Nature Medicine 2024
The fatty liver results are arguably the most medically significant finding beyond weight loss. MASLD (metabolic dysfunction-associated steatotic liver disease) affects roughly 1 in 3 adults and is becoming a leading cause of liver transplants. Current treatment options are limited — there's only one recently approved drug (resmetirom) specifically for the liver scarring stage. An 82% reduction in liver fat with retatrutide could potentially address the disease at an earlier, more reversible stage.
The knee osteoarthritis results from TRIUMPH-4 are also clinically meaningful in a way that pure weight loss numbers don't capture. Knee pain affects mobility, sleep, mental health, and independence. A 76% reduction in pain scores — with more than 1 in 8 patients becoming completely pain-free — represents a real quality-of-life improvement that goes beyond what the scale says.
For type 2 diabetes, the Phase 1b data showed HbA1c reductions of up to 2.02% — which, if confirmed in Phase 3, would make retatrutide competitive with the most effective diabetes medications available. Combined with the weight loss effects, this could position retatrutide as a comprehensive metabolic treatment.
The sleep apnea and cardiovascular studies are still ongoing. If retatrutide shows cardiovascular risk reduction (as semaglutide did in the SELECT trial), it would significantly expand its approved uses and insurance coverage.
Frequently Asked Questions
Is "GLP-3" a real hormone?
No. Humans produce GLP-1 and GLP-2, but there is no GLP-3 hormone. "GLP-3" is an informal nickname for retatrutide, which targets three hormone receptors (GLP-1, GIP, and glucagon). The term started on social media and news sites — it's not used by Eli Lilly, the FDA, or any medical journal.
What is retatrutide?
Retatrutide is an investigational once-weekly injectable drug developed by Eli Lilly. It's a triple agonist that activates GLP-1, GIP, and glucagon receptors simultaneously. Its research code name is LY3437943. It's currently in Phase 3 clinical trials and is not FDA-approved.
How much weight can you lose on retatrutide?
In Phase 3 trials (TRIUMPH-4), participants on the 12mg dose lost an average of 28.7% of their body weight — approximately 71 lbs — over 68 weeks. Phase 2 showed 24.2% at 48 weeks. Individual results vary significantly, and these numbers come from supervised clinical settings with lifestyle counseling.
What are retatrutide's side effects?
The most common side effects are gastrointestinal: nausea (43%), diarrhea (33%), vomiting (21%), and constipation (25%). A new safety signal — dysesthesia (abnormal skin sensations) — was reported in up to 21% of participants on the highest dose. Most GI side effects improve over time, especially with gradual dose escalation.
What is dysesthesia?
Dysesthesia is an abnormal sense of touch that makes normal skin contact feel unusual, tingly, burning, or painful. It was reported in the Phase 3 TRIUMPH-4 trial (up to 20.9% on 12mg dose vs. 0.7% on placebo). Most cases were mild. This side effect is not seen with semaglutide or tirzepatide and needs further investigation.
Is retatrutide FDA approved?
No. Retatrutide is investigational. Phase 3 trials (TRIUMPH program) are ongoing, with seven readouts expected in 2026. The earliest possible FDA approval is estimated for 2027, but this depends on trial results and has not been confirmed by Eli Lilly or the FDA.
Can I buy retatrutide online?
No — not legally or safely. The FDA has issued warnings about unapproved retatrutide products sold as "research use only." These products have unknown purity, dosage accuracy, and sterility. The FDA has also issued warning letters to companies selling compounded retatrutide, stating these products are unapproved and misbranded.
What is "retatrutide peptide"?
This term is typically used by unregulated online sellers marketing retatrutide as a "research peptide." These products are not FDA-approved, not manufactured under regulatory oversight, and are explicitly warned against by the FDA. "Retatrutide peptide" is technically accurate (it is a peptide), but the term is primarily used to circumvent pharmaceutical regulations.
How do I sign up for a retatrutide clinical trial?
Search ClinicalTrials.gov for "retatrutide" or "TRIUMPH." Filter by recruiting status and your location. Review eligibility criteria (generally BMI ≥27-30, age 18+, specific comorbidities). Contact the study site directly or ask your doctor for a referral.
Is retatrutide better than Ozempic?
Ozempic (semaglutide) activates one receptor — GLP-1. Retatrutide activates three — GLP-1, GIP, and glucagon. This triple mechanism has produced higher weight loss in trials (28.7% vs. ~14.9%) and dramatically better liver fat reduction (82% vs. ~50%), but with higher discontinuation rates and a novel side effect (dysesthesia).
How is retatrutide different from Mounjaro/Zepbound?
Mounjaro/Zepbound (tirzepatide) activates two receptors — GLP-1 and GIP. Retatrutide adds a third — glucagon. In trials, retatrutide showed somewhat higher weight loss (~28.7% vs ~22.5%) and much higher liver fat reduction. However, tirzepatide has lower discontinuation rates and no dysesthesia signal, and it's available now by prescription.
Do you regain weight after stopping retatrutide?
There is no retatrutide-specific discontinuation data published yet. However, studies with semaglutide and tirzepatide consistently show that weight regain is common after stopping treatment. Obesity is a chronic condition, and these medications may need to be taken long-term — a 4mg maintenance dose is being studied in the TRIUMPH program.
Is compounded retatrutide legal?
No. The FDA has issued warning letters stating that compounded retatrutide products are unapproved new drugs and misbranded. Retatrutide is not eligible for compounding under current FDA regulations (503A/503B exemptions).
How much will retatrutide cost?
Unknown. Pricing will be determined by Eli Lilly after FDA approval. For reference, Wegovy (semaglutide) lists at approximately $1,349/month but is available for $349/month through Novo Nordisk's self-pay program. Zepbound (tirzepatide) lists at approximately $1,086/month but is available for $299-449/month through LillyDirect vials. Retatrutide's pricing will likely be competitive with these products. Pricing as of February 2026.
When will retatrutide be available in pharmacies?
The most realistic estimate is 2027, assuming Phase 3 trials produce positive results throughout 2026 and Lilly files for FDA approval in late 2026. However, this timeline could shift. Commercial availability typically follows FDA approval by several months.
If you're ready to compare GLP-1 telehealth options, our quiz can help →
References, Methodology, and Update Log
How This Guide Was Researched
All clinical trial data in this guide comes from one of four source types:
- Peer-reviewed publications (New England Journal of Medicine, Nature Medicine, JAMA)
- Official Eli Lilly press releases (via investor.lilly.com and PR Newswire)
- ClinicalTrials.gov registrations (NCT numbers linked throughout)
- FDA official communications (warning letters, safety advisories)
We do not cite social media posts, unregulated seller claims, anecdotal reports, or promotional materials from non-clinical sources. When Phase 3 results are described as "topline," we label them as company-reported data that has not yet been published in a peer-reviewed journal.
Weight loss figures throughout this guide use the efficacy estimand (results from participants who remained on treatment) unless otherwise noted, as this is the standard reporting format in obesity research. We also provide treatment-regimen estimand figures where available for full transparency.
Cross-trial comparisons (retatrutide vs. semaglutide vs. tirzepatide) are clearly labeled as indirect comparisons from separate studies with different designs and populations. No head-to-head trial has compared these drugs.
Primary Sources
- Jastreboff AM, Kaplan LM, Frias JP, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. N Engl J Med. 2023;389:514-526. (NCT04881760)
- Eli Lilly and Company. "Lilly's triple agonist, retatrutide, delivered weight loss of up to an average of 71.2 lbs along with substantial relief from osteoarthritis pain in first successful Phase 3 trial." Press release. December 11, 2025.
- Sanyal AJ, Bedossa P, Engel S, et al. Triple hormone receptor agonist retatrutide for metabolic dysfunction-associated steatotic liver disease: a randomized phase 2a trial. Nat Med. 2024;30:2037-2048.
- Wilding JPH, Batterham RL, Davies M, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide. Diabetes Obes Metab. 2022;24(8):1553-1564.
- Aronne LJ, Sattar N, Horn DB, et al. Continued Treatment With Tirzepatide for Maintenance of Weight Reduction in Adults With Obesity (SURMOUNT-4). JAMA. 2024;331(1):38-48.
- U.S. Food and Drug Administration. "FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss." Updated 2025.
- U.S. Food and Drug Administration. Warning Letter to GLP-1 Solution. September 9, 2025.
- ClinicalTrials.gov. Retatrutide studies: NCT05929066 (TRIUMPH-1), NCT05929079 (TRIUMPH-2), NCT05882045 (TRIUMPH-3), NCT05931367 (TRIUMPH-4), NCT06383390 (TRIUMPH-Outcomes), NCT04881760 (Phase 2).
- GlobalData Pharmaceutical Intelligence. Retatrutide forecast report. December 2025.
- Kaur M, Misra A. "The power of three: Retatrutide's role in modern obesity and diabetes therapy." Eur J Pharmacol. 2024.
Update Log
| Date | What Changed |
|---|---|
| Initial publication | |
| — | Future updates will be logged here as new TRIUMPH results are published |
Medical Disclaimer
This content is for educational and informational purposes only. It is not medical advice, diagnosis, or treatment. Retatrutide is an investigational drug that is not approved by the FDA. Do not attempt to purchase, compound, or self-administer retatrutide outside of a supervised clinical trial.
Always consult a licensed healthcare provider before starting, stopping, or changing any medication or treatment plan. If you are considering weight loss treatment, speak with your doctor about FDA-approved options available to you today. Read our full medical disclaimer.
Affiliate Disclosure
This website may earn commissions from links to FDA-approved medications, telehealth providers, and related health services. We do not link to, promote, or earn commissions from any source selling unapproved retatrutide products. This page contains no affiliate links to retatrutide since it is not available for purchase. Our editorial content is independent of our affiliate relationships. Read our full advertising disclosure.
Related Articles
Retatrutide
Everything known about retatrutide, the triple-agonist GLP-1 drug in clinical development.
Tirzepatide vs Retatrutide
Comparing tirzepatide and retatrutide by mechanism, trial results, safety, and availability.
GLP-3 Retatrutide Alternatives: 4 Legal Options (2026)
Retatrutide isn't FDA-approved — but 4 legal alternatives are. Compare Zepbound, Wegovy pill, Foundayo, prices & how to start in 2026.
What Is GLP-1?
A complete beginner's guide to GLP-1 medications, how they work, and what to expect.
Best GLP-1 Telehealth Providers
Ranked and reviewed telehealth providers offering GLP-1 prescriptions for weight loss online.
GLP-1 Cost With & Without Insurance
Complete guide to GLP-1 medication costs including cash prices, insurance coverage, and savings tips.