Disclosure: Some links on this site are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you. Thank you for supporting our site.·For informational purposes only—not medical advice.
Best GLP-1 for Inflammation: The Right Choice by Goal
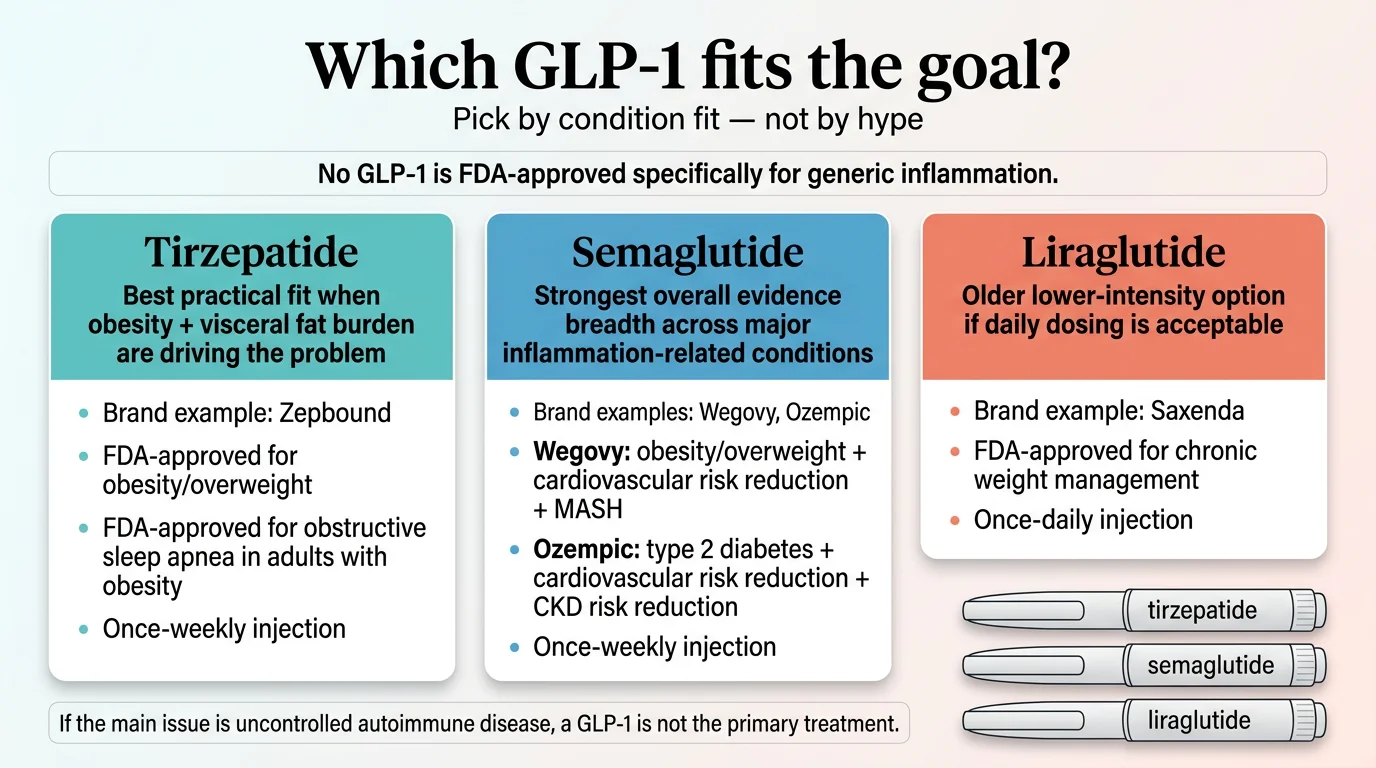
Bottom line: There is no single “best GLP-1 for inflammation” — because no GLP-1 is FDA-approved specifically for that purpose. But that headline undersells the story. Depending on what kind of inflammation you’re dealing with, one GLP-1 may be the more evidence-supported choice — though the evidence is still indirect, drawn from different trials and different conditions.
Tirzepatide is usually the strongest pick when your inflammation is driven mainly by obesity and visceral fat — it delivers the most weight loss and lowered hsCRP in sleep apnea trials. Semaglutide has the deepest direct anti-inflammatory evidence and the broadest set of inflammation-adjacent FDA approvals (MASH, cardiovascular risk, kidney disease). Generic liraglutide is the budget FDA-approved fallback if cost is the dealbreaker. And if your real issue is uncontrolled autoimmune disease like RA or lupus, a GLP-1 is not the main treatment — but it can be a useful add-on if obesity, fatty liver, or insulin resistance are making things worse.
We spent weeks reviewing every major study published through early 2026 — the JCI review, the large Nature Medicine observational study, the ACR 2025 rheumatology data, FDA label updates, and dozens of preclinical papers — to build the comparison below. This page exists to help you match the right GLP-1 to your specific inflammation problem, understand what the evidence actually supports (and where the hype gets ahead of the science), and find the most affordable access path if you decide to move forward.
We may earn a commission if you use our provider links. This does not influence our medication rankings, which are based entirely on published evidence. See our full methodology.
Quick Picks
Obesity-driven inflammation: Tirzepatide (Mounjaro/Zepbound) — strongest weight loss, hsCRP reduction in OSA trial
Broadest anti-inflammatory evidence: Semaglutide (Ozempic/Wegovy) — CV, MASH, kidney, OA, neurological data
Budget FDA-approved option: Generic liraglutide (Saxenda) — first generic GLP-1 for weight loss

Which GLP-1 Is Best for Inflammation? The Short Answer
Let’s not waste your time. Here are the three paths:
Best when obesity is driving the inflammation
Tirzepatide (Mounjaro / Zepbound). Strongest weight loss data. hsCRP reduction demonstrated in the obstructive sleep apnea trial. If visceral fat is the engine behind your inflammation, tirzepatide turns that engine off faster than anything else available.
Best for direct anti-inflammatory evidence across conditions
Semaglutide (Ozempic / Wegovy). Strongest semaglutide-specific evidence in cardiovascular inflammation, MASH (fatty liver), kidney disease, randomized trial evidence in obesity-related knee osteoarthritis, and emerging neurological data. The broadest FDA-approved indication portfolio touching inflammation-adjacent conditions.
Best budget FDA-approved option
Generic liraglutide (generic Saxenda). First generic GLP-1 approved for weight loss. Daily injection, less weight loss, shorter track record for this specific question — but FDA-approved, affordable, and real.
Important: “Best for inflammation” is not the same question as “best for weight loss” or “fewest side effects.” We cover all three questions separately below.
Best GLP-1 for Inflammation at a Glance
This table is for choosing a medication, not a provider. Provider comparison is further down the page.
| Tirzepatide | Semaglutide | Liraglutide | |
|---|---|---|---|
| Brand Names | Mounjaro (T2D), Zepbound (weight loss, OSA) | Ozempic (T2D, CV risk, CKD risk), Wegovy (weight loss, CV risk, MASH) | Victoza (T2D), Saxenda (weight loss) — generic now available |
| FDA-Approved for Inflammation? | No. Approved for T2D, obesity, OSA | No. Approved for T2D, obesity, CV risk reduction, MASH, CKD risk reduction | No. Approved for T2D, obesity |
| Strongest Inflammation Evidence | Obesity-driven systemic inflammation. hsCRP reduction in OSA trial. Potentially distinct GIP-mediated immune effects (still being studied) | Broadest evidence: CRP reduction in meta-analysis, CV event reduction (SELECT trial), MASH resolution, kidney protection (FLOW trial), knee OA trial evidence (STEP 9), emerging neurological data | Long clinical track record. Some anti-inflammatory and psoriasis-adjacent evidence, but overall human evidence base is thinner |
| Best For | Obesity-driven inflammation, sleep apnea, severe visceral fat burden, maximum weight loss priority | MASH/fatty liver, cardiovascular/kidney risk, OA knee pain w/ obesity, deepest evidence base | Budget-conscious, FDA-approved-only, people comfortable with daily injections |
| Expected Weight Loss | About 13–21% in key FDA label studies | About 10–15% in key Wegovy studies | More modest than semaglutide or tirzepatide |
| Dosing | Weekly injection | Weekly injection | Daily injection |
| Main Downside | Less inflammation-specific published data. Higher GI side effect rates in some studies. More expensive. | Not the single strongest weight loss option. May matter when obesity is the primary driver. | Significantly less weight loss. Daily injection burden. Weaker inflammation data overall. |
| Evidence Confidence | ★★★★☆ Strong, still emerging | ★★★★★ Strongest and broadest | ★★★☆☆ Established but limited |
| List Price (no insurance) | Check LillyDirect for current Zepbound self-pay pricing | Wegovy list price ~$1,349/mo; check NovoCare for current self-pay offers | Generic now available (check pharmacy pricing) |

“Lost 16 lbs in 10 weeks — no side effects. Down two sizes. I wish I’d started sooner.”
— Verified MEDVi patient on ConsumerAffairs ★★★★★
If Your Real Goal Is X, Choose Y
This is the section that matters most. Find your situation.
If Your Inflammation Is Tied to Obesity, Visceral Fat, and Metabolic Dysfunction
Likely winner: Tirzepatide
Here’s the logic. Visceral fat is not just stored energy — it’s an active inflammatory organ. It pumps out TNF-α, IL-6, and IL-1β around the clock. The more visceral fat you carry, the higher your baseline inflammation. Tirzepatide delivers approximately 13-21% body weight loss in key FDA label studies (depending on dose and population), meaningfully more than semaglutide’s 10-15%. In the SURMOUNT-OSA trial, tirzepatide also significantly reduced high-sensitivity C-reactive protein (hsCRP), a key systemic inflammation marker, in patients with obstructive sleep apnea.
If the inflammatory fire is being fueled primarily by excess body fat, tirzepatide turns the hose on harder.
Don’t choose tirzepatide if: You want the deepest inflammation-specific published evidence (semaglutide has more), you’re very concerned about GI side effects, or cost is the primary constraint.
If Your Main Concern Is Joint Pain or Knee Osteoarthritis
Likely winner: Semaglutide
The STEP 9 trial specifically examined semaglutide in knee OA with obesity and found meaningful improvements in pain and physical function. Knee OA is not an FDA-approved indication for semaglutide, but this is the strongest randomized trial evidence any GLP-1 has in joint disease. A separate ACR 2025 per-protocol analysis comparing GLP-1 agonists and SGLT2 inhibitors found that semaglutide and SGLT2 inhibitors had similar effects on OA and knee replacement outcomes — suggesting semaglutide’s OA benefit is real but not necessarily superior to other metabolic drug classes.
There’s a two-part benefit here. First, weight loss reduces mechanical load on joints — every pound lost takes roughly four pounds of pressure off your knees. Second, emerging evidence suggests GLP-1s may have direct chondroprotective (cartilage-protecting) effects independent of weight loss, likely through NF-κB pathway inhibition.
Don’t choose semaglutide if: Your OA is primarily in non-weight-bearing joints and obesity isn’t a factor (the benefit may be smaller).
If Your Concern Is Fatty Liver / MASH
Likely winner: Semaglutide
This one is straightforward. Wegovy received FDA accelerated approval for noncirrhotic MASH with moderate to advanced liver fibrosis (F2-F3), making semaglutide the only GLP-1 with a direct FDA nod for liver inflammation. Clinical trials demonstrated MASH resolution in a significant proportion of patients. Tirzepatide also showed strong liver fat reduction in the SURPASS trials and may ultimately prove comparable, but semaglutide has the label advantage today.
If Your Concern Is Sleep Apnea Plus Obesity
Likely winner: Tirzepatide
Zepbound (tirzepatide) carries an FDA-approved indication for obstructive sleep apnea in adults with obesity. The SURMOUNT-OSA trial showed significant reduction in breathing interruptions during sleep, lowered systemic inflammation markers (including hsCRP), and improved patient-reported sleep quality. Semaglutide has CV and metabolic benefits that indirectly help OSA, but tirzepatide owns this indication.
If You Have Type 2 Diabetes with Cardiovascular or Kidney Risk
Likely winner: Semaglutide
Ozempic’s label includes cardiovascular risk reduction in T2D patients with established heart disease. The FLOW trial demonstrated significant renal benefits. The SELECT trial showed a roughly 20% lower risk of major adverse cardiovascular events in people with obesity and established heart disease (without diabetes). And a large 2025 Nature Medicine observational study of VA patients with diabetes mapped 175 outcomes in GLP-1 RA users versus other diabetes drug groups — finding lower risks across several neurocognitive, cardiometabolic, and respiratory outcomes, though also higher risks of GI disorders, hypotension/syncope, and drug-induced pancreatitis. For cardiovascular and kidney inflammation in the context of diabetes, semaglutide has the deepest evidence.
If Your Biggest Issue Is Budget and You Want FDA-Approved Only
Likely winner: Generic liraglutide
In 2025, Teva launched the first generic version of Saxenda (liraglutide injection), making it the first generic GLP-1 indicated for weight loss. You give up significant weight loss potency compared to the newer agents, and you’re injecting daily instead of weekly. But it’s FDA-approved, it has a long clinical track record with some anti-inflammatory and psoriasis-adjacent evidence, and it removes the compounding question entirely.
If Your Real Goal Is Autoimmune Disease Control (RA, Lupus, Psoriasis, IBD)
We need to be straight with you here. A GLP-1 is not the primary treatment for rheumatoid arthritis, lupus, inflammatory bowel disease, or any autoimmune condition. If someone tells you to drop your DMARDs or biologics for Ozempic, walk away. Untreated inflammatory arthritis causes irreversible joint damage.
That said, the evidence for GLP-1s as an adjunct in autoimmune conditions is genuinely exciting:
- Rheumatoid arthritis: At ACR 2025, a TriNetX analysis found semaglutide users had significantly lower 30-day, 3-month, and 1-year risk of joint stiffness, pain, swelling, and synovitis compared to non-users (n=12,139 matched patients). A retrospective study of RA patients on DMARDs found SGLT2 inhibitors significantly reduced flare burden; GLP-1 agonists showed a non-significant trend toward flare reduction (p=0.15).
- Psoriatic arthritis and psoriasis: Four of five clinical studies showed significant PASI improvement with GLP-1 therapy. Some patients report skin clearing before significant weight loss occurs — a clue that the benefit isn’t purely weight-mediated. A 2024 study by Eder et al. showed less pain, lower CRP, better cholesterol and blood pressure, with benefits scaling alongside weight loss.
- Osteoarthritis: Semaglutide showed pain and function improvement in the STEP 9 randomized trial in obesity-related knee OA. A per-protocol analysis at ACR 2025 found semaglutide and SGLT2 inhibitors had similar OA/knee replacement outcomes.
- Lupus (SLE): Very early evidence suggests GLP-1s may help via metabolic improvement, but concerns about autoimmune activation persist. Only under close rheumatologist supervision.
- IBD: Population-level studies show lower incidence of new IBD in GLP-1 users. GLP-1 improves mucosal integrity. But direct clinical trials are lacking.
- Safety signal for autoimmune disease: A large ACR 2025 TriNetX study found no increased risk of new immune-mediated inflammatory diseases after GLP-1 RA initiation in patients with T2D or obesity, while also finding lower risks for mortality, heart failure, stroke, CKD, and dementia. This is reassuring — GLP-1s don’t appear to trigger autoimmune flares.
Best choice for most autoimmune patients who also have obesity or metabolic issues: Semaglutide, used alongside (never instead of) your current treatment. Discuss it with both your rheumatologist and your prescribing clinician. If you qualify based on BMI and want an affordable starting point, we cover access options including cash-pay telehealth programs later in this guide.
Do GLP-1 Drugs Actually Reduce Inflammation?
Yes — and the evidence has grown dramatically in the last two years. But the picture is more nuanced than the headlines suggest.
What We Know with Reasonable Confidence
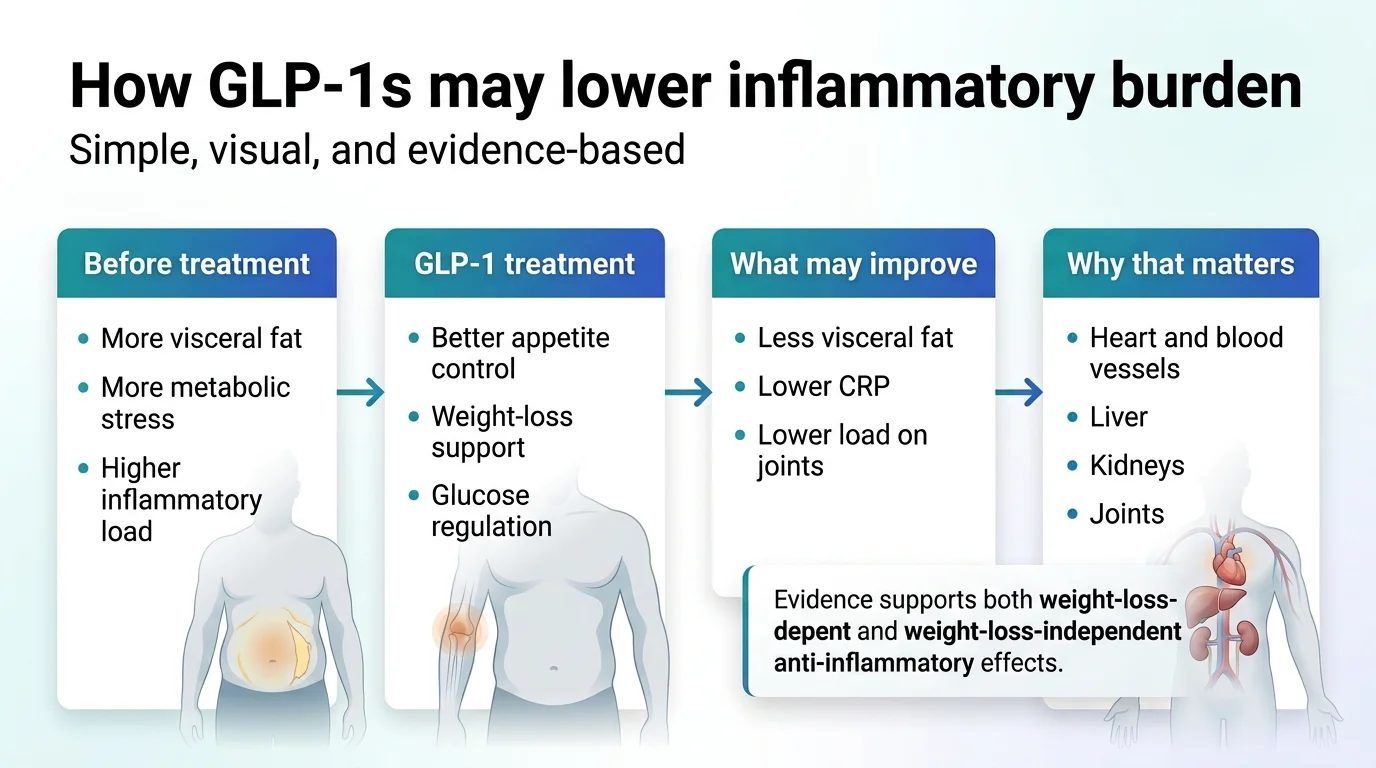
The weight-mediated pathway is well-established. Visceral fat tissue is essentially a chronic inflammation factory. It produces pro-inflammatory cytokines — TNF-α, IL-6, IL-1β — that circulate systemically and drive inflammation in joints, blood vessels, the liver, and the brain. Losing even 5-10% of body weight significantly reduces this inflammatory load. Every GLP-1 medication achieves this to varying degrees.
The direct anti-inflammatory pathway is increasingly supported. This is where the science gets genuinely exciting. A growing body of evidence — including a pivotal November 2025 review in the Journal of Clinical Investigation — demonstrates that GLP-1 medications suppress inflammation through mechanisms that are independent of weight loss:
- NF-κB pathway inhibition: GLP-1 receptor activation directly suppresses the NF-κB signaling cascade, the master control switch for inflammatory gene expression. Demonstrated in both animal models and human immune cells.
- Cytokine reduction within hours: A single dose of semaglutide or exenatide reduced circulating levels of TNF-α in mice challenged with endotoxin. Exenatide downregulated TNF and IL-1β in human peripheral blood mononuclear cells. These effects manifested within hours — well before any meaningful weight loss.
- Macrophage reprogramming: GLP-1 shifts macrophages from the M1 “attack” phenotype to the M2 “repair” phenotype. Less tissue-damaging inflammation and more healing activity.
- hsCRP reduction in clinical trials: High-sensitivity C-reactive protein has been significantly reduced by semaglutide in multiple human studies, including a 2024 systematic review and meta-analysis.
What Is Still Unsettled
No head-to-head inflammation trial exists. Nobody has run a randomized controlled trial directly comparing tirzepatide vs. semaglutide vs. liraglutide specifically for inflammatory outcomes. Our comparisons are based on indirect evidence — different trials, different populations, different endpoints.
Weight loss and direct effects are tangled. When someone loses 40 pounds on tirzepatide and their CRP drops, how much is from fat loss and how much from direct GLP-1 receptor signaling? The JCI review notes that liraglutide reduced atherosclerotic plaque size in mice even when control mice with matching weight loss were not protected — suggesting a real weight-independent effect. But the full picture remains incomplete.
Autoimmune applications are early. The autoimmune data (RA, psoriasis, lupus) is observational, not from randomized controlled trials. Promising? Absolutely. Proven? Not yet. We’re being transparent about this because your health decisions deserve honesty, not hype.
The 2025 Nature Medicine Study: Why This Matters
This study deserves its own callout because it shifted the conversation — but it also needs context. Published in Nature Medicine in 2025, researchers used VA health records to map 175 outcomes in adults with diabetes who used GLP-1 receptor agonists versus other diabetes medication groups. GLP-1 RA use was associated with lower risks across several categories: neurocognitive outcomes (including dementia), cardiometabolic outcomes (heart failure, stroke), respiratory outcomes, severe infections, and all-cause mortality.
However — and this matters for a balanced understanding — the study also found higher risks of GI disorders, hypotension/syncope, nephrolithiasis, interstitial nephritis signals, and drug-induced pancreatitis in GLP-1 RA users. This is an observational study, not a randomized trial, which means it can identify associations but not prove causation.
Why this still matters for the inflammation question: The conditions where GLP-1s showed benefit — heart failure, dementia, respiratory disease, infections — all have significant inflammatory components. The consistent direction of benefit across so many inflammation-linked outcomes, in such a large population, supports the broader hypothesis that GLP-1 medications are producing meaningful, sustained anti-inflammatory effects body-wide. But observational data has inherent limitations, and the adverse signals remind us these are real medications with real tradeoffs.
“[These findings] continue to underline the importance of inflammation in several major diseases.” — Dr. Anthony Komaroff, Harvard Health
How GLP-1 Drugs May Reduce Inflammation: Plain English
You don’t need a biochemistry degree. Here’s what’s happening in your body:
Less Visceral Fat = Less Inflammatory Fuel
Belly fat — specifically the fat around your organs (visceral fat) — isn’t just sitting there. It’s chemically active. It constantly produces inflammatory molecules that circulate through your blood and aggravate everything from joint pain to artery plaque to brain fog. When GLP-1 medications help you lose visceral fat, you’re removing a major source of chronic inflammation.
Better Insulin Sensitivity = Lower Metabolic Stress
Insulin resistance and chronic inflammation are locked in a feedback loop. Inflammation makes cells more insulin-resistant. Insulin resistance drives more inflammation. GLP-1 medications break this cycle by improving insulin sensitivity, reducing blood sugar spikes, and lowering the metabolic stress that feeds the inflammatory fire.
Direct Immune System Modulation
Independent of weight loss, GLP-1 receptor activation appears to:
- Calm the NF-κB pathway (the master inflammation switch)
- Reduce production of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β)
- Shift immune cells from “attack mode” to “repair mode”
- Reduce oxidative stress that damages tissues
- Improve gut barrier integrity (a healthier gut lining means fewer inflammatory triggers leaking into your bloodstream)
Why Your CRP May Drop
If your doctor has flagged elevated CRP (C-reactive protein), that’s a marker of systemic inflammation. Multiple studies show semaglutide significantly lowers hsCRP. If you’re starting a GLP-1 for weight loss and also want to track inflammation, ask your doctor to check your hsCRP before you begin. It gives you a real baseline to measure progress against — not just the number on the scale.
Tirzepatide for Inflammation: When It Wins
Tirzepatide is the strongest practical pick when the inflammation you care about is being driven mainly by obesity and you want the largest expected weight-loss impact.
Why Tirzepatide Is Compelling
Tirzepatide is a dual GLP-1/GIP receptor agonist — it activates two hormone pathways instead of one. The GIP receptor component may contribute additional anti-inflammatory effects through direct actions on GIP-expressing immune cells, though this mechanism is still being studied. What we know for certain is that tirzepatide produces meaningfully greater weight loss than semaglutide in head-to-head comparisons. In the SURMOUNT program, participants lost roughly 13-21% of body weight depending on dose and population.
In the SURMOUNT-OSA trial, tirzepatide significantly reduced the number of breathing interruptions during sleep, lowered hsCRP (a key systemic inflammation marker), and improved patient-reported sleep quality. Zepbound now carries an FDA indication for OSA.
Best For
- Obesity-driven systemic inflammation
- Obstructive sleep apnea with inflammatory burden
- Severe visceral fat accumulation
- Patients prioritizing maximum weight loss as the inflammation intervention
- People who haven’t responded adequately to semaglutide alone
Don’t Choose Tirzepatide If
- You want the strongest published inflammation-specific evidence base (semaglutide has more)
- You’re very sensitive to GI side effects (tirzepatide has slightly higher rates in some studies)
- Budget is the primary constraint (tirzepatide is more expensive across the board)
- You want the lowest-cost FDA-approved option (generic liraglutide wins there)
Side Effects to Discuss with Your Doctor
The most common side effects are GI-related: nausea, diarrhea, constipation, and vomiting, typically during dose escalation. These usually improve as your body adjusts. Tirzepatide carries the same class-level warnings as other GLP-1s regarding thyroid C-cell tumors (contraindicated in patients with personal/family history of medullary thyroid carcinoma or MEN2), pancreatitis risk, and gallbladder issues. Always review the full prescribing information with your clinician.
Semaglutide for Inflammation: When It Wins
Semaglutide has the strongest direct anti-inflammatory evidence of any GLP-1 medication and the broadest set of inflammation-adjacent FDA approvals.
Why Semaglutide Is Still the Evidence Leader
- Cardiovascular: The SELECT trial demonstrated roughly 20% lower risk of heart attack, stroke, or cardiovascular death in people with obesity and established heart disease. Ozempic carries a CV risk reduction indication for T2D patients with established CVD.
- Knee osteoarthritis: STEP 9 data showed pain and function improvement in OA patients with obesity. Strong randomized trial evidence, though knee OA is not currently an FDA-approved indication.
- MASH/fatty liver: Semaglutide received FDA accelerated approval for noncirrhotic MASH with moderate to advanced liver fibrosis, making it the first GLP-1 with a direct liver inflammation nod.
- Kidney protection: The FLOW trial demonstrated significant renal benefits, adding kidney inflammation to semaglutide’s portfolio.
- Neurological: Active Phase 3 clinical trials for both Alzheimer’s disease and Parkinson’s disease are underway.
- Systemic CRP reduction: A 2024 meta-analysis confirmed semaglutide’s consistent ability to lower hsCRP.
No other single GLP-1 medication touches this many inflammation-relevant conditions with this much human data.
Affordable Access
Brand-name Wegovy retails at roughly $1,349/mo without insurance. Most people don’t pay that. Options include:
- Insurance: Check if your plan covers Wegovy (coverage is expanding but still inconsistent)
- NovoCare savings programs: Manufacturer-direct offers for eligible patients
- Compounded semaglutide through telehealth: Providers like MEDVi offer compounded semaglutide programs starting at $179 for the first month (ongoing refills at $299/mo). Compounded medications are prepared by state-licensed pharmacies but are not FDA-approved or evaluated for safety, efficacy, or quality as finished products.

“Lost 16 lbs in 10 weeks — no side effects. Down two sizes. I wish I’d started sooner.”
— Verified MEDVi patient on ConsumerAffairs ★★★★★
Is Generic Liraglutide or an Older GLP-1 Ever the Better Choice?
Yes — in specific situations.
Generic Liraglutide as the Budget FDA-Approved Fallback
In 2025, Teva launched the first generic version of Saxenda (liraglutide injection), making it the first generic GLP-1 indicated for weight loss. For people who want:
- An FDA-approved medication (not compounded)
- The lowest possible cost among approved GLP-1s
- A proven track record (liraglutide has the longest history of any GLP-1 RA)
What You Give Up
- Daily injection instead of weekly
- Significantly less weight loss (~5-8% vs 15-26%)
- Less inflammation-specific data than semaglutide, especially for newer indications like MASH and knee OA
- Shorter duration of action — Cleveland Clinic researcher Dr. Abhay Singh has noted that newer, longer-acting drugs like semaglutide may provide a “constant state of anti-inflammation” that shorter-acting agents can’t match
We include generic liraglutide because pretending only two medications exist would hurt your trust in everything else we say. Some people genuinely need an FDA-approved, non-compounded option at the lowest possible cost. Generic liraglutide serves that need.
GLP-1 Anti-Inflammatory Evidence by Body System
We built this reference because no other page consolidates the evidence this way. For each body system, we’ve noted the strongest GLP-1 candidate and the confidence level of the evidence.
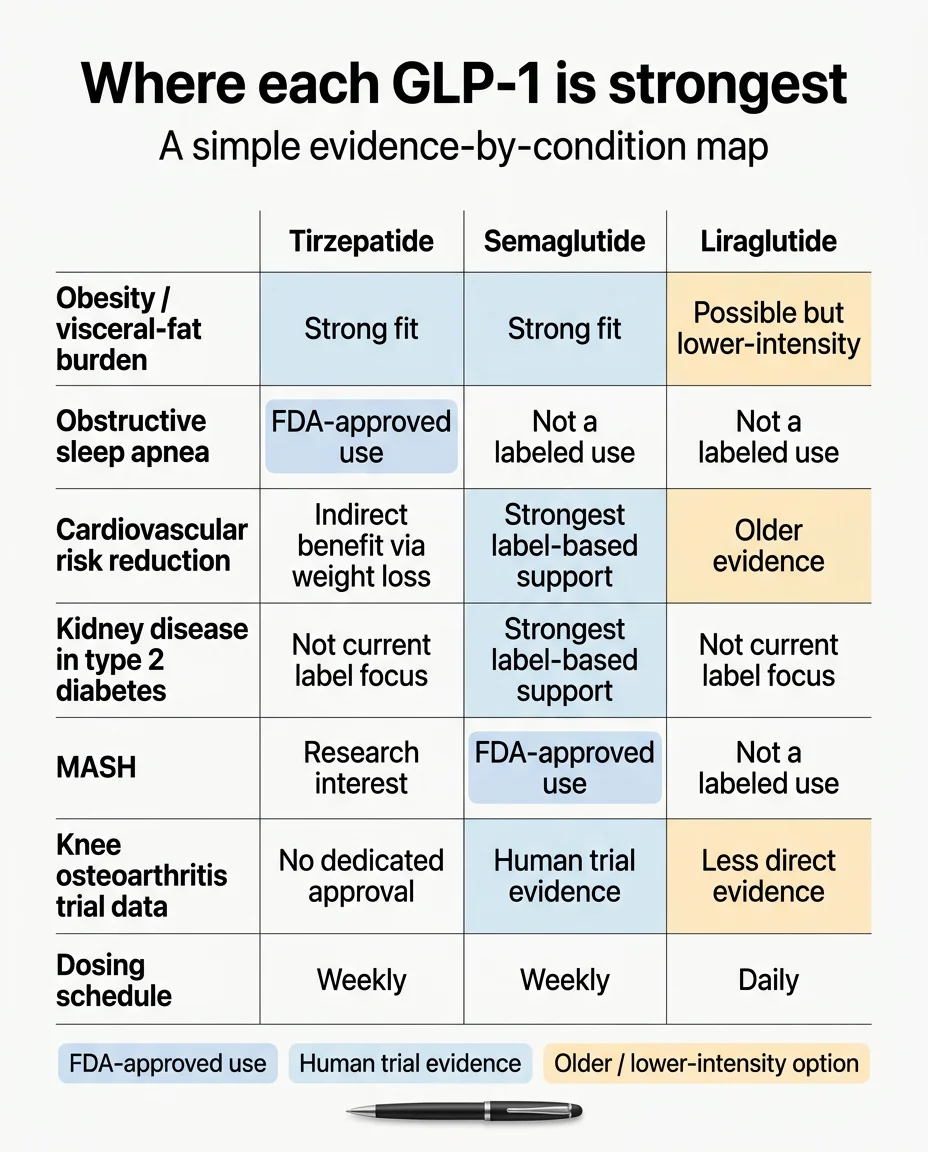
Cardiovascular Inflammation
Evidence leader: Semaglutide (strongest), Tirzepatide (strong and growing)
The most robust evidence in the entire GLP-1-inflammation landscape. Semaglutide’s SELECT trial enrolled over 17,000 participants with obesity and established cardiovascular disease and demonstrated roughly 20% lower risk of major adverse cardiovascular events. Liraglutide earlier showed plaque size reduction in animal models even when control groups had matching weight loss — one of the clearest demonstrations that GLP-1s have direct anti-inflammatory effects on blood vessels.
Confidence level: High — supported by large RCTs with hard cardiovascular endpoints.
Joint and Musculoskeletal Inflammation
Evidence leader: Semaglutide (OA and RA)
For osteoarthritis, the STEP 9 trial data and semaglutide’s randomized evidence in obesity-related knee OA make it the clear frontrunner. The benefit is two-fold: weight loss reduces mechanical joint load (each pound lost removes roughly four pounds of force from the knee), and GLP-1 receptor activation may directly protect cartilage through NF-κB pathway inhibition.
For rheumatoid arthritis, the ACR 2025 TriNetX analysis of over 12,000 semaglutide users vs. non-users found significantly lower risk of joint stiffness, pain, swelling, and synovitis at 30 days, 3 months, and 1 year.
Confidence level: Moderate to high for OA (RCT data exists). Moderate for RA and PsA (observational, but large and consistent).
Liver Inflammation (MASH/NAFLD)
Evidence leader: Semaglutide (FDA indication), Tirzepatide (strong trial data)
Semaglutide received FDA accelerated approval for MASH. In clinical trials, a significant proportion of patients achieved MASH resolution — meaning the inflammatory liver damage actually reversed. Tirzepatide showed comparable or potentially superior liver fat reduction in SURPASS trial analyses.
Confidence level: High — FDA approval based on clinical trial evidence.
Neurological Inflammation
Evidence leader: Semaglutide — still investigational
Active Phase 3 clinical trials are testing semaglutide in Alzheimer’s disease and Parkinson’s disease. The 2025 Nature Medicine observational study found GLP-1 users had lower dementia incidence. The JCI review notes that “not all neural benefits are consistent across studies” and direct attribution “remains to be conclusively demonstrated.”
Confidence level: Low to moderate — population data is suggestive, but definitive RCT results are pending. Do not start a GLP-1 specifically for neuroprotection at this time.
Kidney Inflammation
Evidence leader: Semaglutide
The FLOW trial is the landmark here. Semaglutide demonstrated significant renal benefits in patients with T2D and CKD. GLP-1 receptor signaling helps control blood pressure through effects on renal sodium handling and provides renal vasodilation.
Confidence level: High — supported by dedicated RCT (FLOW) and label updates.
Skin Inflammation (Psoriasis)
Evidence leader: Liraglutide (most published clinical data), Semaglutide (promising observational)
Liraglutide has the most clinical data specifically in psoriasis — four of five studies showed significant improvement in PASI scores. Some patients see skin clearing before any meaningful weight loss, a strong indicator of direct immune effects.
Confidence level: Moderate — multiple small studies and case series, but no large RCTs.
Gut Inflammation (IBD and Related)
Evidence leader: No clear winner — semaglutide has the most adjacent data
GLP-1 receptors are widely expressed in the GI tract. Population-level data shows lower incidence of new-onset IBD among GLP-1 users. But direct clinical trials for GLP-1s in IBD are essentially nonexistent. If you have active IBD, do not start a GLP-1 as a treatment for it.
Confidence level: Low to moderate — biologically plausible and population data is supportive, but no direct clinical trials.
How Quickly Does GLP-1 Reduce Inflammation?
This comes up constantly in forums and search. Here’s the honest timeline:
Some inflammatory biomarker changes begin quickly. The JCI review documented TNF-α reduction after a single dose. Appetite reduction and early GI effects begin within the first week. Don’t expect inflammation changes yet.
Noticeable changes in bloating, puffiness, and general “inflammatory feeling.” Early weight loss begins reducing visceral fat-driven inflammatory load.
More meaningful metabolic and inflammatory marker changes. This is typically when hsCRP starts dropping measurably. Joint pain improvements tend to emerge based on observational data.
Significant weight loss accumulates. Disease-relevant outcomes (reduced RA flares, improved liver markers, cardiovascular risk reduction) become more apparent. Longer-term studies showing organ-level protection are measured in years.
Set realistic expectations: GLP-1 medications are not ibuprofen. They don’t provide acute anti-inflammatory relief within an hour. The benefit builds over weeks to months. Give it at least 3 months of consistent use before evaluating whether it’s working.
What Dose Is Used for Inflammation?
There is no FDA-approved “inflammation dose” for any GLP-1 medication. All current evidence comes from studies using standard label-approved doses for obesity or type 2 diabetes. The anti-inflammatory benefits happen at these standard doses — there’s no reason to deviate from established titration protocols.
For semaglutide (Wegovy), the standard approach is: 0.25 mg weekly for 4 weeks → 0.5 mg → 1.0 mg → 1.7 mg → 2.4 mg maintenance. For tirzepatide (Zepbound): 2.5 mg weekly for 4 weeks → 5 mg → titrate up to 10 mg or 15 mg based on response and tolerability.
Do not try to invent a custom “anti-inflammatory protocol.” Follow the label dosing. The inflammation benefits come along for the ride.
Low-Dose GLP-1 for Inflammation: Promising or Hype?
Some clinicians have reported that even doses below the standard weight-loss titration produced noticeable reductions in pain, morning stiffness, and flare frequency. But there are no randomized controlled trials specifically examining low-dose GLP-1 for anti-inflammatory purposes. If you qualify for a standard GLP-1 prescription through approved indications, use the standard dosing. The inflammation benefits are best supported at standard doses.
Which GLP-1 Has the Fewest Side Effects?
The inflammation question and the side-effect question are connected. If side effects force you to stop, the anti-inflammatory benefit stops too.
Common Side Effects Across the Class
- Most common: Nausea (typically worst during early dose increases, then fades), diarrhea, constipation, vomiting, reduced appetite
- Less common: Headache, fatigue, dizziness, injection site reactions
- Rare but serious: Pancreatitis, gallbladder problems, thyroid concerns (MEN2 contraindication), potential kidney issues with dehydration
Comparing Tolerability
Liraglutide’s side effects are generally considered the mildest. Semaglutide is well-tolerated by most patients at standard titration. Tirzepatide has shown slightly higher rates of GI side effects in some studies, though this varies by individual and dose. Slow titration is the single biggest factor in tolerability. Good telehealth programs — including MEDVi, which uses what they call “gentle titration” — help patients find the effective dose with minimum discomfort.
“Fewest side effects” ≠ “Best for inflammation.” Don’t let side-effect fear override the evidence. Semaglutide and tirzepatide both have stronger anti-inflammatory evidence than liraglutide. Start with the evidence-supported option and manage side effects through proper titration.
Who Should NOT Use a GLP-1 for Inflammation?
This matters. GLP-1s are not for everyone.
Hard Contraindications
- Personal or family history of medullary thyroid carcinoma (MTC) or MEN2: All GLP-1 RAs carry this boxed warning. Not negotiable.
- History of severe pancreatitis: GLP-1s may increase pancreatitis risk. Discuss carefully with your doctor.
- Pregnancy or active fertility planning: GLP-1s are not approved during pregnancy. Discontinue well before conception.
Proceed with Caution
- Severe gastroparesis or GI motility disorders: GLP-1s slow gastric emptying, which could worsen symptoms.
- History of gallbladder disease: Rapid weight loss increases gallstone risk. Monitor accordingly.
- Kidney disease (without clinical guidance): Some GLP-1s are renally protective, but dose adjustments may be needed.
- Active, poorly controlled autoimmune disease: Stabilize first with proven therapies before adding a GLP-1.
- Eating disorders: GLP-1s suppress appetite dramatically. In patients with a history of anorexia or restrictive eating, this requires specialized clinical oversight.
When a GLP-1 is the wrong tool: If your inflammation is purely autoimmune (no obesity, no metabolic dysfunction, no cardiovascular risk), a GLP-1 is probably not the right tool. The evidence is strongest when metabolic factors — obesity, insulin resistance, visceral fat, fatty liver — are part of the picture. If those factors aren’t present, the benefit-to-risk calculation changes. Talk to your rheumatologist.
FDA-Approved vs. Compounded GLP-1: What Changes for Safety, Quality, and Trust?
This section might be the most important on the page for your actual decision-making, so we’re going to be thorough and honest.
What “FDA-Approved” Means
FDA-approved GLP-1 medications (Wegovy, Ozempic, Zepbound, Mounjaro, Saxenda, Victoza) have undergone rigorous clinical trials demonstrating safety, efficacy, and manufacturing quality. Each batch is produced under strict Good Manufacturing Practice (GMP) standards. The FDA monitors ongoing safety through post-market surveillance.
What “Compounded” Means
Compounded GLP-1 medications are prepared by licensed compounding pharmacies. They are prescribed by licensed physicians based on individual patient needs. Compounded medications are not FDA-approved or evaluated for safety, efficacy, or quality as finished products. They may differ in formulation, concentration, and other characteristics from the brand-name versions.
What Changed After Shortages Resolved
FDA has determined that semaglutide and tirzepatide shortages are resolved and has taken enforcement action against non-FDA-approved GLP-1 products. The regulatory landscape around compounding these specific drugs is actively shifting. Verify the current regulatory status before relying on any compounded offering.
How to Evaluate a Compounded GLP-1 Provider
If you’re going the compounded route, look for:
- Licensed, U.S.-based compounding pharmacy partners
- Clinician review before prescribing (not just a checkbox questionnaire)
- Transparent pricing including refill costs, not just the introductory rate
- Clear disclosure that compounded medications are not FDA-approved
- Easy cancellation without surprise charges
- LegitScript certification (indicates compliance monitoring)
Our perspective: We believe everyone deserves access to these medications regardless of insurance status. Compounded GLP-1 programs have made that possible for hundreds of thousands of people. But we also believe you should make that choice with full information — including the FDA’s concerns — rather than being told compounded versions are “the same thing” as brand-name drugs. They’re not the same thing. For many patients, the cost-access tradeoff is worth it. That’s a decision you should make with your eyes open.
Will Insurance Cover GLP-1 for Inflammation?
Short Answer: Probably Not Under That Label
No insurer covers GLP-1s specifically “for inflammation” because inflammation is not an approved indication. Coverage depends on qualifying through an approved use.
When Coverage Is More Likely
- Type 2 diabetes: Most plans cover at least one GLP-1
- Obesity (BMI ≥30, or ≥27 with comorbidity): Coverage is expanding but remains inconsistent. Many plans require prior authorization.
- Cardiovascular risk reduction: Some plans cover Wegovy for CV risk in patients with established heart disease
- MASH: New indication — coverage policies are still being developed
When Cash-Pay Makes More Sense
For many people — especially those without robust insurance coverage — cash-pay through a telehealth provider is faster, simpler, and sometimes cheaper than navigating insurance bureaucracy. Compounded semaglutide through providers like MEDVi starts at $179/mo (first month) with no prior authorization, no step therapy, and no waiting. Compounded GLP-1s are not FDA-approved as finished products — see our full breakdown above for what that means.
Best Online GLP-1 Providers by Situation
Once you know which medication makes sense, the next question is where to access it safely and affordably.
Best for First-Time Cash-Pay Users Who Want a Guided Start: MEDVi
MEDVi is a telehealth platform that connects patients with licensed providers (through the OpenLoop Health network) for GLP-1 prescriptions. Here’s what we’ve confirmed:
- Pricing (verified March 2026): Compounded semaglutide injections start at $179 for the first month, with refills at $299/mo. Oral tablets and tirzepatide options also available at higher price points.
- What’s included: Physician review, unlimited messaging with the care team, 24/7 support. No membership fees. No long-term contracts. Cash-pay only.
- Clinical oversight: OpenLoop Health clinicians retain the decision to prescribe. Compounded GLP-1s are not FDA-approved or evaluated for safety, efficacy, or quality as finished products.
- HSA/FSA eligible for medical weight loss prescriptions (per MEDVi’s site)
“Doctor was super helpful and supportive. Answered my many questions and we came up with a great solution.” — Source: Trustpilot, invited review, 2026
Note: This is a customer testimonial, not efficacy evidence. Individual experiences vary.
The honest tradeoff: MEDVi is cash-pay only — no insurance billing. The $179 first month price increases to $299 for ongoing refills. Some customer reviews mention billing-related frustrations. The 4.4-star Trustpilot rating alongside a lower ConsumerAffairs rating tells a mixed story: most patients are happy with the medication and medical care, but the administrative side has room to improve.
Why we still recommend them for first-timers: The medical care itself is well-regarded. The pricing is among the most transparent and affordable in the compounded GLP-1 space. The month-to-month flexibility means you’re never locked in. For someone who wants to start a GLP-1 program and see whether the anti-inflammatory benefits materialize for their specific situation, MEDVi offers one of the lowest-risk entry points available.

“Lost 16 lbs in 10 weeks — no side effects. Down two sizes. I wish I’d started sooner.”
— Verified MEDVi patient on ConsumerAffairs ★★★★★
Other Providers Worth Considering
Best Advertised Intro Pricing: Yucca Health
Yucca Health advertises compounded semaglutide starting at $146/mo on a 6-month plan. Best for price-sensitive users comfortable committing to a multi-month structure.
Best for Transparent Educational Framing: Eden
Eden offers compounded semaglutide with pricing starting from $149/first month. They clearly explain that compounded medications are not FDA-approved. Good option for people who want detailed upfront education.
Additional Options: TrimRX & SkinnyRX
Several other telehealth providers offer compounded GLP-1 programs in similar price ranges. Before using any provider, verify: current price and refill price, medication source and pharmacy, cancellation terms, and whether they’re offering compounded or FDA-approved products.
How We Ranked the Medications and Providers
Medication Ranking Methodology
- Published human clinical evidence (weighted highest) — prioritizing RCTs over observational studies over preclinical data
- FDA label support — approved indications that touch inflammation-adjacent conditions
- Condition-specific fit — which medication has the strongest evidence for each inflammatory condition
- Magnitude of weight loss — because the weight-mediated anti-inflammatory pathway is well-established
- Duration of action — longer-acting agents provide more consistent anti-inflammatory coverage
- Side-effect burden — particularly GI tolerability during titration
- Cost and access — because the best medication you can’t afford doesn’t help you
We earn affiliate commissions from some providers listed on this page. Our medication rankings are based entirely on published evidence. Our provider rankings are based on price transparency, clinical oversight, disclosure honesty, support quality, cancellation friction, pharmacy sourcing, and independent verification. If a provider pays us a commission but fails our evaluation criteria, they don’t make the list.
The One Honest Downside You Need to Hear
No GLP-1 is FDA-approved specifically for inflammation. If your real problem is uncontrolled autoimmune disease — aggressive RA, unstable lupus, active IBD — a GLP-1 is not your primary treatment and should not be used as a substitute for proven immunosuppressive therapy.
Why that should not scare you off: If obesity, visceral fat, fatty liver, insulin resistance, sleep apnea, or mechanical joint load are part of what’s driving your inflammatory burden — and for most people searching this topic, at least one of those is true — the right GLP-1 medication is one of the most rational additions you can make to your treatment plan. The evidence is real, meaningful, and accumulating.
We see people hesitate because they think “not FDA-approved for inflammation” means “doesn’t work for inflammation.” That’s a misread. It means no pharmaceutical company has run the specific clinical trials required for that exact label claim. The evidence that GLP-1s reduce inflammation is extensive, published in top-tier journals, and growing. The FDA approval gap is administrative, not scientific.
What Happens After You Start: A Realistic Timeline
We include this because unrealistic expectations are the number one reason people quit GLP-1 therapy before the anti-inflammatory benefits fully materialize.
Week 1-2: The Adjustment Phase
Appetite reduction begins quickly — often within the first few days. You may experience mild nausea, especially around meals. This is normal and typically temporary. Don’t expect inflammation changes yet.
What to do: Stay hydrated. Eat smaller meals. Focus on protein. Don’t judge the medication by week one.
Week 3-6: Early Signals
Weight loss begins in earnest. Some patients report early reductions in puffiness, bloating, and general inflammatory “heaviness.” If you deal with joint stiffness, you may notice mornings getting slightly easier.
What to do: Too early to recheck baseline labs. Stay the course.
Month 2-3: The Inflection Point
This is when the compounding effects start becoming meaningful. Your body is experiencing both direct receptor-mediated anti-inflammatory effects and the metabolic benefits of meaningful fat loss. Many patients report reduced joint pain and stiffness, less brain fog, improved energy and sleep quality, and lower need for NSAIDs.
What to do: Good time to recheck inflammatory markers if your doctor is tracking them.
Month 3-6: Meaningful Results
If GLP-1 therapy is going to produce significant anti-inflammatory benefits for your situation, you should see clear evidence by now. Continued weight loss accelerates the metabolic improvements.
What to do: Have a substantive follow-up with your clinician. Review labs. Adjust the plan as needed.
Month 6-12+: Long-Term Benefits
The longest-term studies show that sustained GLP-1 use produces ongoing anti-inflammatory benefits that build over time. The cardiovascular protection, organ-level benefits, and disease risk reductions accumulate with continued use.
The realistic expectation: GLP-1 therapy for inflammation is not a six-week experiment. It’s a long-term intervention.
Questions to Ask Your Doctor Before Starting
Before your appointment (or telehealth consultation), write down these questions:
- Is my inflammation likely obesity-driven, autoimmune, mechanical, metabolic, or a combination?
- Which outcome matters most for me: weight loss, pain reduction, CRP levels, A1c, liver function, or cardiovascular risk?
- Do I qualify for an FDA-approved GLP-1 through insurance, or is a cash-pay compounded option more realistic?
- Do I need baseline labs (hsCRP, A1c, liver panel, kidney function) before starting?
- What contraindications should I be aware of given my specific medical history?
- What’s the titration plan, and what should I do if side effects are difficult?
- How long should I try this before we reassess whether it’s working for my inflammation?
- How does this interact with my current medications (especially DMARDs, biologics, or immunosuppressants)?
Print this list or save it on your phone. Good clinicians appreciate patients who come prepared.
Frequently Asked Questions
Which GLP-1 works best for inflammation?
It depends on the type of inflammation. Semaglutide has the broadest direct anti-inflammatory evidence across the most conditions (cardiovascular, joint, liver, kidney, neurological). Tirzepatide is the strongest pick when obesity-driven systemic inflammation is the main concern, because it produces the greatest weight loss and has demonstrated hsCRP lowering in the OSA trial. Generic liraglutide is the budget FDA-approved fallback with a long safety track record. No GLP-1 is FDA-approved specifically for inflammation — the anti-inflammatory benefits come alongside approved uses for obesity, type 2 diabetes, and related conditions.
Do GLP-1 drugs really reduce inflammation?
Yes — and this isn't theoretical anymore. Multiple studies confirm GLP-1 medications lower inflammatory markers like hsCRP and reduce pro-inflammatory cytokines (TNF-α, IL-6, IL-1β). The landmark 2025 Nature Medicine study of nearly a million patients showed 10-20% reductions in inflammation-driven conditions including heart failure, dementia, and lung disease over almost a decade of follow-up. The November 2025 JCI review documented that some anti-inflammatory effects occur within hours of a single dose, before any weight loss happens — confirming a direct, weight-independent mechanism.
Is tirzepatide better than semaglutide for inflammation?
For obesity-driven systemic inflammation, tirzepatide may be superior because it produces approximately 13-21% weight loss vs semaglutide's 10-15%, and the SURMOUNT-OSA trial demonstrated hsCRP reduction. For breadth of inflammation-specific evidence (MASH, OA, CV, kidney, neurological conditions), semaglutide clearly leads with more published human studies and more FDA-label support. No head-to-head trial has directly compared their anti-inflammatory effects.
Can GLP-1 help rheumatoid arthritis?
Emerging evidence is genuinely promising. ACR 2025 TriNetX data showed semaglutide users with RA had significantly lower risk of joint stiffness, pain, swelling, and synovitis at 30 days, 3 months, and 1 year compared to matched non-users. However, GLP-1s should be used alongside standard RA treatment (DMARDs, biologics), never as a replacement. Stopping proven immunosuppressive therapy in favor of a GLP-1 risks irreversible joint damage.
Can GLP-1 help autoimmune disease?
Early evidence suggests GLP-1s may benefit several autoimmune conditions (RA, PsA, psoriasis, potentially lupus and IBD) through both weight-mediated and direct anti-inflammatory mechanisms. A large 2025 ACR TriNetX study found no increased risk of new immune-mediated inflammatory diseases after GLP-1 RA initiation — reassuring safety data. These medications should supplement, not replace, standard autoimmune treatment under specialist supervision.
How quickly does GLP-1 reduce inflammation?
Some cytokine reduction occurs within hours of a single dose in experimental settings. Most patients notice symptomatic improvement (reduced puffiness, joint stiffness, energy improvement) within 2-8 weeks. Meaningful CRP reduction and disease-specific benefits typically emerge over 1-3 months. Full benefits accumulate over 6-12 months.
What dose is used for inflammation?
There is no FDA-approved "inflammation dose." All anti-inflammatory benefits have been observed at standard approved doses for obesity or type 2 diabetes. Follow your clinician's standard titration protocol.
Is low-dose GLP-1 for inflammation real?
It's investigational. Some rheumatology clinics report benefits with microdosing, but no randomized controlled trials support this approach. If you qualify for standard dosing, use standard dosing — the evidence is much stronger there.
Will insurance cover GLP-1 for inflammation?
Not specifically for inflammation. Coverage is possible through approved indications: type 2 diabetes, obesity (BMI ≥30 or ≥27 with comorbidity), cardiovascular risk reduction, or MASH. Many people find cash-pay telehealth providers more accessible.
Are compounded GLP-1s safe?
Compounded GLP-1 medications are prescribed by licensed clinicians and prepared by licensed pharmacies, but they are not FDA-approved or evaluated for safety, efficacy, or quality as finished products. The FDA has documented adverse events associated with compounded GLP-1 products and warns against treating them as equivalent to FDA-approved medications. Quality varies by pharmacy.
What if I want FDA-approved only?
Your best options are brand-name Wegovy or Zepbound through insurance or manufacturer programs (NovoCare, LillyDirect), or generic liraglutide (the first generic GLP-1 approved for weight loss). These remove the compounding question entirely but typically cost more.
Which provider is best for beginners paying cash?
For first-time cash-pay users who want compounded semaglutide with physician oversight, MEDVi is a strong starting point at $179/first month. For the absolute lowest advertised plan pricing, Yucca Health offers $146/mo on a 6-month commitment. For people who want detailed educational transparency, Eden starts at $149/first month.
Is semaglutide anti-inflammatory?
Yes. Semaglutide has demonstrated anti-inflammatory effects through multiple mechanisms: NF-κB pathway inhibition, cytokine reduction (TNF-α, IL-6, IL-1β), macrophage repolarization from M1 to M2 phenotype, hsCRP reduction in clinical trials, and weight-loss-mediated reduction of visceral fat inflammation. A November 2025 JCI review and a 2024 meta-analysis both confirmed these findings.
Can GLP-1 help with joint pain?
Evidence supports this, especially for osteoarthritis and rheumatoid arthritis. ACR 2025 data showed GLP-1 users had improved joint outcomes. In knee OA specifically, semaglutide improved pain and physical function in patients with obesity. Joint pain from autoimmune causes may also improve if obesity or metabolic factors are contributing.
Most affordable GLP-1 for inflammation?
For FDA-approved: generic liraglutide. For compounded semaglutide (most evidence, weekly dosing): MEDVi at $179 first month, $299 ongoing — no contracts, no membership fees. For lowest possible monthly rate: Yucca Health at $146/mo on a 6-month plan.
Your Next Step
You’ve now read more about GLP-1 medications and inflammation than 99% of the people searching this topic. Here’s how to turn that knowledge into action:
If you know which medication fits your situation:
Compare providers and pricing above. For affordable compounded semaglutide access, start a free evaluation with MEDVi — $179 first month, no contracts, licensed clinician review.
If you want FDA-approved only:
Talk to your primary care doctor or endocrinologist. Check NovoCare (Wegovy) and LillyDirect (Zepbound) for manufacturer savings programs. Ask about generic liraglutide.
If you want the medication answer but aren’t ready to act:
Bookmark this page. We update it quarterly as new evidence is published. Print the doctor questions checklist and bring it to your next appointment.
If your situation is complex (autoimmune + obesity + multiple conditions):
This page gives you the evidence foundation. But your next conversation should be with both a rheumatologist (or relevant specialist) and a clinician experienced with GLP-1 prescribing. Bring this page. The comparison table alone will help frame the discussion.
Key Sources
- Journal of Clinical Investigation — “Antiinflammatory actions of GLP-1–based therapies beyond metabolic benefits” (Nov 2025). jci.org/articles/view/194751
- Nature Medicine — Large VA observational study mapping 175 outcomes in GLP-1 RA users (2025). PubMed 39833406
- Harvard Health — “Do GLP-1 drugs reduce inflammation?” (Oct 2025)
- American College of Rheumatology — ACR Convergence 2025 abstracts: Semaglutide in RA (Abstract #2107645), SGLT2i/GLP-1 RA flares (Abstract #2128553), GLP-1 vs SGLT2i OA outcomes (Abstract #0165), IMID safety (Abstract #2658)
- Nature Reviews Rheumatology — “GLP-1 receptor agonists in arthritis” (2025)
- PMC — “Anti-inflammatory benefits of semaglutide: systematic review and meta-analysis” (2024)
- PMC — “GLP-1: a multi-faceted anti-inflammatory agent” (2023)
- FDA prescribing information: Wegovy (215256s024lbl), Ozempic (209637s025lbl), Zepbound (217806Orig1s020lbl), Mounjaro, Saxenda
- FDA — “Concerns with unapproved GLP-1 drugs used for weight loss.” fda.gov/drugs/postmarket-drug-safety-information
- Teva — Generic Saxenda (liraglutide) approval announcement (2025)
Claim-level citations: Throughout this article, evidence claims are attributable to the specific sources listed above. Provider-specific claims are labeled “provider-stated” and dated. Pricing is labeled with verification dates. Where we describe study findings, we reference the specific abstract number or publication.
Affiliate Disclosure
We earn affiliate commissions from some providers listed on this page. Our medication rankings are based entirely on published evidence. Our provider rankings are based on our evaluation criteria. See our advertising disclosure and full methodology.
Medical Disclaimer
This article is for informational purposes only and does not constitute medical advice. GLP-1 medications are prescription medications requiring evaluation by a licensed clinician. The anti-inflammatory benefits discussed are based on published research. No GLP-1 medication is FDA-approved for the treatment of inflammation or any inflammatory disease. Always consult a qualified healthcare provider before starting any medication.
Compounding Disclaimer
Compounded medications referenced in this article are not FDA-approved or evaluated for safety, efficacy, or quality as finished products. Compounded products should not be presented as the same as FDA-approved brands.
Related Guides
Last Updated: March 2026 | Evidence last reviewed against published studies through early 2026 | Pricing verified: March 2026