Retatrutide vs Tirzepatide: Which Is Better Right Now?
For informational purposes only—not medical advice.
Retatrutide may eventually beat tirzepatide on weight loss. Tirzepatide is the one you can actually use today. Here's the honest comparison — with the data, the caveats, and the safe next step.
If you're comparing retatrutide vs tirzepatide, here's the short version: retatrutide is a triple hormone receptor agonist that produced 28.7% average weight loss in Eli Lilly's first Phase 3 readout — in adults with obesity and knee osteoarthritis. Tirzepatide is a dual GIP/GLP-1 agonist that produced 22.5% average weight loss at 72 weeks in SURMOUNT-1, and is FDA-approved and available now as Zepbound and Mounjaro. These are not head-to-head results.
The catch — and it's a big one — is that retatrutide is not FDA-approved. There is no FDA-approved retail prescription or pharmacy pathway today. If you see it for sale online, that's a red flag, not an opportunity.
Best practical choice right now: Tirzepatide (Zepbound / Mounjaro)
Most promising future option: Retatrutide (still in clinical trials)
The one thing you should not do: Buy "retatrutide" from an online seller
The Quick Comparison
We built this table from published clinical trial data and official FDA records — not marketing copy. Every number is cited below.
| Tirzepatide | Retatrutide | |
|---|---|---|
| What it is | Dual agonist (GLP-1 + GIP) | Triple agonist (GLP-1 + GIP + glucagon) |
| Brand names | Zepbound (weight loss), Mounjaro (T2D) | None — investigational only |
| Maker | Eli Lilly | Eli Lilly |
| FDA status | Approved (diabetes 2022, weight loss 2023, sleep apnea 2024) | Not approved — Phase 3 trials ongoing |
| Evidence grade | Peer-reviewed Phase 3, large sample, real-world use | Phase 2 peer-reviewed (NEJM); Phase 3 topline press release only |
| Best weight loss in trials | ~22.5% at 72 wks (SURMOUNT-1, n=2,539) | 28.7% at 68 wks (TRIUMPH-4, n=445) |
| Can you get it today? | Yes — by prescription | No — only through clinical trials |
| Cost today | $299–$1,086/mo depending on access route | N/A (free within clinical trials) |
| Dosing | 2.5 mg → up to 15 mg weekly | 2 mg → up to 12 mg weekly (trial protocol) |
| Unique safety signal | GI side effects; dysesthesia 0.4% at 15 mg | GI side effects + dysesthesia in ~21% at highest dose |
| Bottom line | Proven, available, life-changing results now | More powerful on paper, but you can't use it yet |
Sources: SURMOUNT-1 (Jastreboff et al., NEJM 2022); Retatrutide Phase 2 (Jastreboff et al., NEJM 2023); TRIUMPH-4 topline results (Eli Lilly press release, Dec 11, 2025); FDA prescribing information for Zepbound and Mounjaro.
Not sure which GLP-1 program is right for you? We built a short quiz that matches you to the best legitimate option based on your goals, budget, and situation.
Take the 2-Minute GLP-1 Fit QuizWhat Is the Difference Between Retatrutide and Tirzepatide?
Both are injectable weight loss medications made by the same company (Eli Lilly). Both mimic natural gut hormones to reduce appetite, improve blood sugar, and promote weight loss. The difference is how many hormones each one targets — and that single distinction drives everything else.
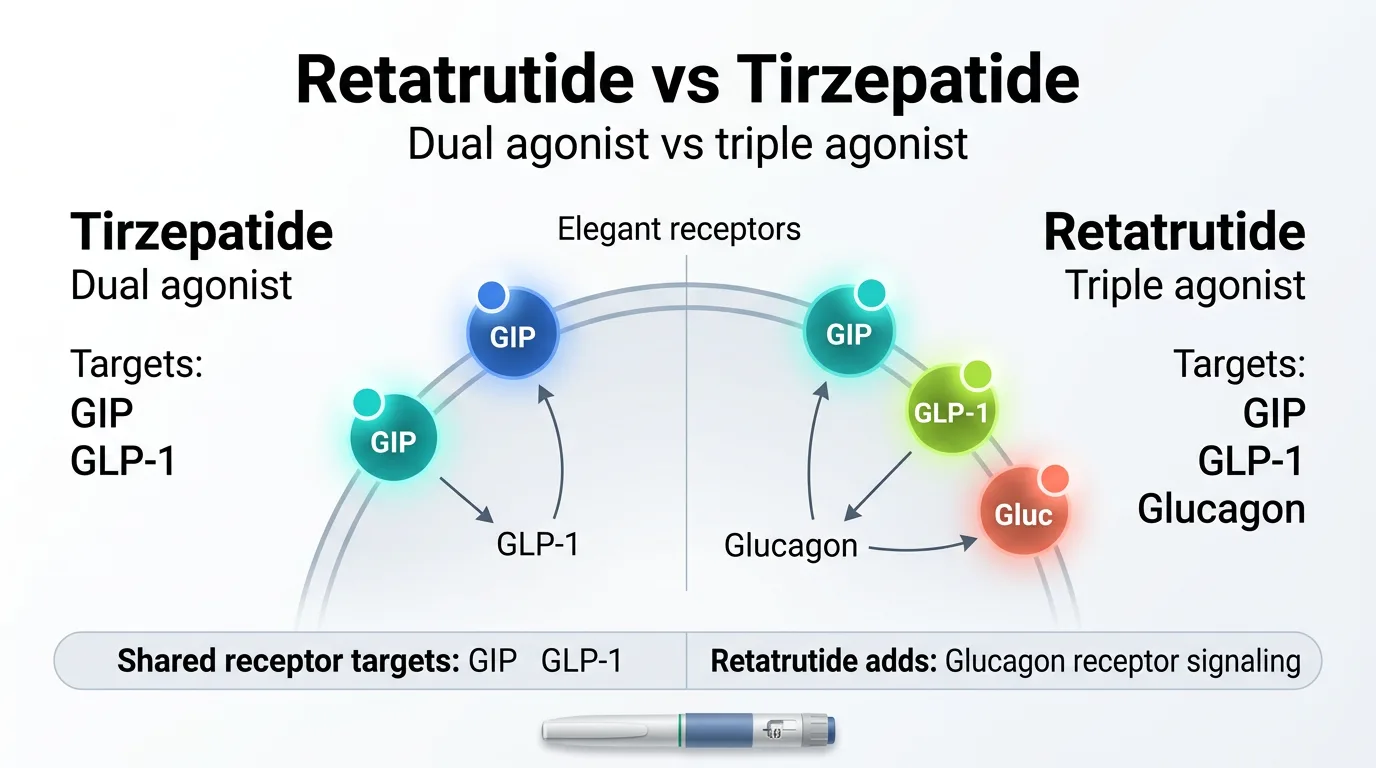
Tirzepatide: The Dual Agonist
Tirzepatide activates two hormone receptors:
- GLP-1 (glucagon-like peptide-1): Suppresses appetite, slows digestion, improves insulin response
- GIP (glucose-dependent insulinotropic polypeptide): Enhances insulin secretion, improves lipid metabolism, amplifies appetite-suppressing effect
Working together, these two pathways produce stronger appetite control and better blood sugar regulation than targeting GLP-1 alone. That's why tirzepatide outperformed semaglutide in head-to-head trials.
Tirzepatide is sold under two brand names: Mounjaro (approved for type 2 diabetes in May 2022) and Zepbound (approved for weight management in November 2023, and later for obstructive sleep apnea in 2024). Both are the same medication — different labels for different conditions.
Retatrutide: The Triple Agonist
Retatrutide does everything tirzepatide does — and adds a third target:
- GLP-1: Same appetite and digestion effects
- GIP: Same insulin and metabolic effects
- Glucagon: Increases energy expenditure, stimulates fat oxidation, ramps up your body's calorie-burning machinery
That glucagon receptor is the key differentiator. Tirzepatide helps you eat less. Retatrutide appears to help you eat less and burn more. Across separate trials, retatrutide has posted weight-loss figures roughly 3–7 percentage points higher than tirzepatide — but those are cross-trial comparisons, not head-to-head results.
But there's an important caveat. Retatrutide is still investigational. Its development code is LY3437943. It has no brand name, no FDA approval, and no legal path to a prescription outside of a clinical trial. That's not a minor footnote. It's the single most important fact in this entire comparison.
The Evolution in One Visual
Think of it as generations of the same idea:
- Semaglutide (Wegovy, Ozempic) = 1 receptor (GLP-1 only) → ~15% weight loss
- Tirzepatide (Zepbound, Mounjaro) = 2 receptors (GLP-1 + GIP) → ~21% weight loss
- Retatrutide (investigational) = 3 receptors (GLP-1 + GIP + glucagon) → ~24–29% weight loss
Each step added a hormone target. Each step increased weight loss. Each step is progressively newer — and less proven in the real world.
Is Retatrutide Better Than Tirzepatide Right Now?
For most people making a real decision today, no. Tirzepatide is the better practical choice.
Here's why:
- It's FDA-approved and available at pharmacies
- It has extensive Phase 3 data across multiple large trials
- Insurance may cover it (or you can access self-pay options)
- Doctors know how to prescribe, titrate, and manage it
- The safety profile is well-characterized after years of real-world use
Retatrutide may eventually prove to be the more effective drug for pure weight loss. The early data is remarkable. But "may eventually prove" is doing a lot of heavy lifting in that sentence. The full Phase 3 data isn't complete. The FDA hasn't reviewed it. Long-term safety is still being evaluated. And you literally cannot get it through any legitimate channel.
If you're considering GLP-1 treatment and want to start now, the conversation with your doctor should center on tirzepatide (or semaglutide, depending on your situation). If you're here because you're fascinated by what's coming next in obesity medicine, this guide will give you the full picture of where retatrutide stands — with the caveats it deserves.
How Much Weight Can You Lose on Retatrutide vs Tirzepatide?
Tirzepatide: Published Phase 3 Results
The landmark trial is SURMOUNT-1, published in the New England Journal of Medicine in 2022. It enrolled 2,539 adults with obesity (without diabetes) and ran for 72 weeks. Results at the highest approved dose (15 mg):
- Average weight loss: 22.5% of body weight
- Over 90% of participants lost at least 5% of their body weight
- Roughly 1 in 3 participants lost more than 25%
Source: Jastreboff AM, Aronne LJ, et al. NEJM 2022; 387:205-216
Retatrutide: What the Trials Show So Far
Phase 2 trial (NEJM, 2023): 338 adults with obesity, 48 weeks. The 12 mg dose produced an average of 24.2% body weight loss. This study was peer-reviewed and published in the New England Journal of Medicine.
TRIUMPH-4 (Phase 3, December 2025): 445 adults with obesity and knee osteoarthritis, 68 weeks. The 12 mg dose produced 28.7% average body weight loss — approximately 71.2 pounds from a starting weight of about 248 lbs. This is the highest weight loss figure ever reported in a Phase 3 obesity trial.
In TRIUMPH-4, nearly half of participants on the higher dose achieved at least 25% weight loss, and a meaningful portion exceeded 30%.
Important: The TRIUMPH-4 results are "topline" — meaning they come from an Eli Lilly press release, not yet a peer-reviewed journal publication. This doesn't mean the numbers are wrong, but the evidence grade is different.

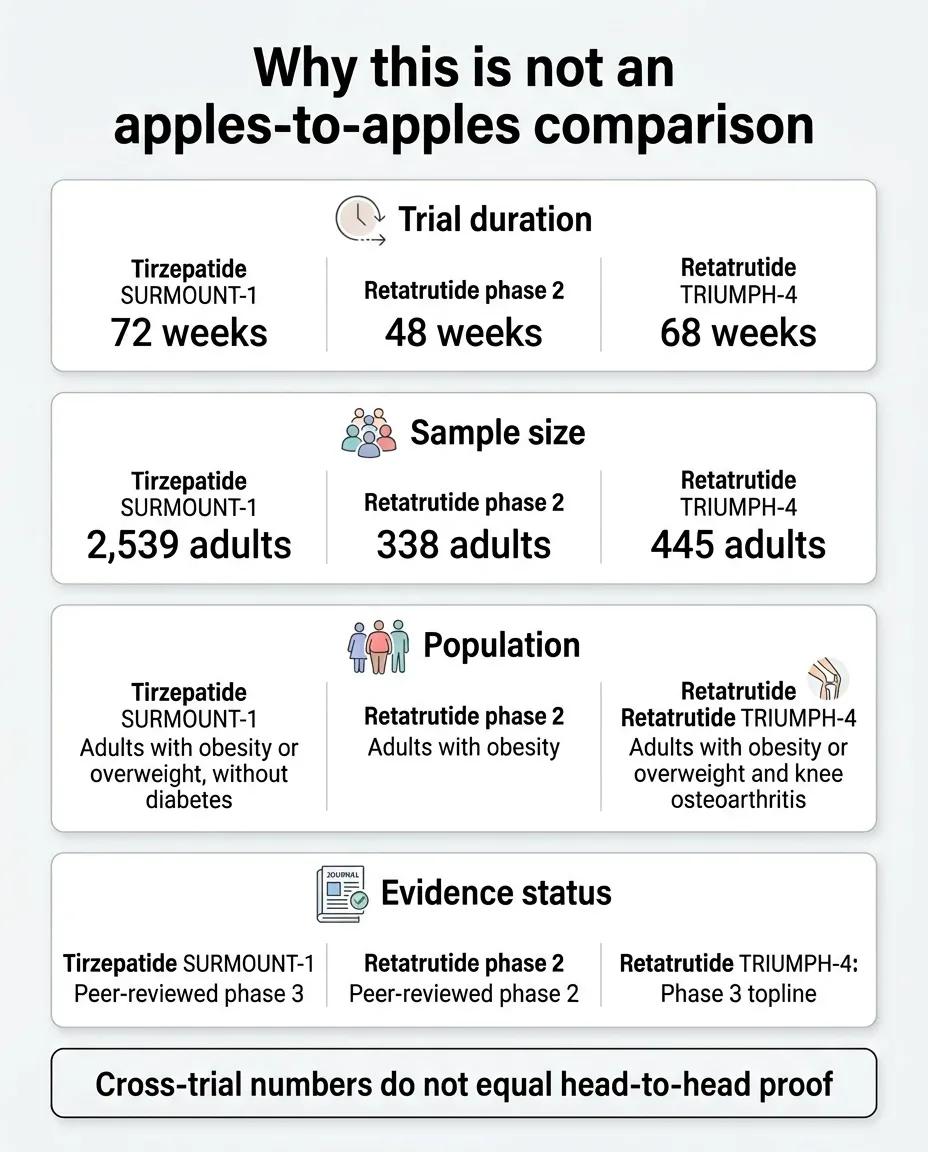
Why These Numbers Are Not Apples to Apples
Here's where most comparison pages go wrong. They put 28.7% next to 22.5% and declare a winner. We won't do that, because the data doesn't support a clean head-to-head conclusion.
| Factor | SURMOUNT-1 (Tirzepatide) | TRIUMPH-4 (Retatrutide) |
|---|---|---|
| Phase | Phase 3 | Phase 3 |
| Publication status | Peer-reviewed (NEJM) | Press release / topline only |
| Duration | 72 weeks | 68 weeks |
| Sample size | 2,539 | 445 |
| Population | Adults with obesity, no diabetes | Adults with obesity + knee osteoarthritis |
| Average baseline BMI | ~38 kg/m² | ~40.4 kg/m² |
| Max dose | 15 mg | 12 mg |
| Weight loss result | 22.5% | 28.7% |
The Reality Check: What the Difference Means in Pounds
| Your Starting Weight | Tirzepatide (~22%) | Retatrutide (~25–29%) | The Difference |
|---|---|---|---|
| 200 lbs | ~44 lbs lost | ~50–58 lbs lost | 6–14 lbs |
| 250 lbs | ~55 lbs lost | ~63–73 lbs lost | 8–18 lbs |
| 300 lbs | ~66 lbs lost | ~75–87 lbs lost | 9–21 lbs |
The difference is real and meaningful — but for most people, both of these results would be life-changing. The question isn't really "which number is bigger." It's: do you want treatment that works now, or do you want to wait 1–2+ years for something that might work slightly better?
Is There an Actual Head-to-Head Trial?
Yes — and this is a bigger deal than most pages mention.
Eli Lilly is running TRIUMPH-5 (ClinicalTrials.gov: NCT06662383), a head-to-head study comparing retatrutide directly against tirzepatide in adults with obesity. The trial enrolls approximately 800 participants and will last about 89 weeks.
This is the study that will actually answer the question everyone is asking. Until TRIUMPH-5 results are published, every comparison between these two drugs — including the one on this page — is an indirect comparison across different trials.
We don't have a confirmed date for when TRIUMPH-5 results will be available, but it's part of the broader TRIUMPH program with readouts expected through 2026 and into 2027.
What this means for you: If you're the type who wants definitive proof before making a call, the data is coming. But if you need treatment now, waiting for TRIUMPH-5 results doesn't make medical sense.
How Do Retatrutide and Tirzepatide Work Differently?
GLP-1 (both drugs): When you eat, your gut releases GLP-1 naturally. It tells your brain "you're full," slows down how fast food leaves your stomach, and helps your pancreas release insulin at the right time. Both tirzepatide and retatrutide amplify this signal dramatically. This is why appetite drops — sometimes to a degree people describe as "food noise" turning off.
GIP (both drugs): GIP works alongside GLP-1 to further improve insulin sensitivity and metabolic function. When you combine GLP-1 + GIP activation (as tirzepatide does), the appetite suppression effect is stronger than either alone.
Glucagon (retatrutide only): Here's where it gets interesting. Glucagon does something fundamentally different — instead of just reducing how much you eat, it increases how much energy your body burns. Glucagon tells the liver to mobilize stored fat for energy and cranks up your metabolic rate. That's why retatrutide is sometimes described as working on "both sides of the equation" — less input (appetite), more output (energy expenditure).
Tirzepatide delivers strong, proven appetite suppression and blood sugar control. You eat less, feel fuller, and your metabolism gets a helpful nudge. This is more than enough for life-changing results.
Retatrutide does all of that and potentially adds a metabolic "push" through glucagon. The body doesn't just eat less — it may burn more actively. This could explain the extra weight loss seen in trials.
Retatrutide vs Tirzepatide Side Effects: What's Different and What's the Same
The GI Side Effects Both Drugs Share
Nausea, diarrhea, constipation, vomiting, and decreased appetite are the most common side effects for both. These tend to be worst during dose-escalation and improve as your body adjusts.
Slow titration matters — a lot. In the retatrutide Phase 2 trial, GI side effect rates nearly doubled when patients were started at higher doses without gradual escalation.
Dysesthesia: Much More Prominent With Retatrutide So Far
Dysesthesia is an abnormal skin sensation — tingling, tenderness, or altered sensitivity to touch. It has been reported with tirzepatide at very low rates (0.2–0.4%). With retatrutide, the rate is dramatically higher.
In the TRIUMPH-4 Phase 3 trial:
- 20.9% of patients on 12 mg reported dysesthesia
- 8.8% of patients on 9 mg reported dysesthesia
- 0.7% on placebo reported it
That means roughly 1 in 5 people on the highest retatrutide dose experienced this — compared to fewer than 1 in 200 on tirzepatide. Eli Lilly described the events as "generally mild" and said they "rarely led to treatment discontinuation."
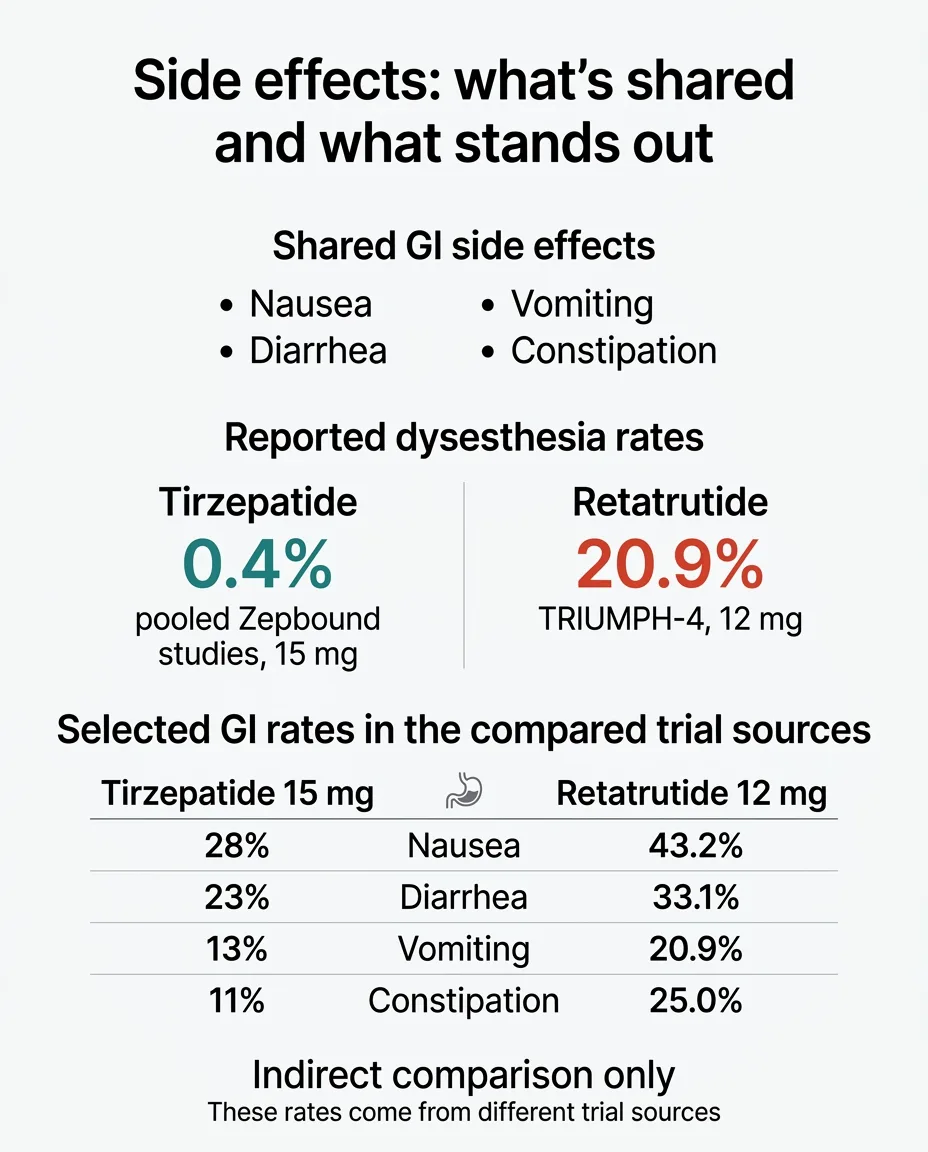
Side-by-Side: GI and Safety Comparison
| Side Effect | Tirzepatide 15 mg (Zepbound label) | Retatrutide 12 mg (TRIUMPH-4) |
|---|---|---|
| Nausea | 28% | 43.2% |
| Diarrhea | 23% | 33.1% |
| Vomiting | 13% | 20.9% |
| Constipation | 11% | 25.0% |
| Decreased appetite | 9% | 18.2% |
| Dysesthesia | 0.4% (pooled SURMOUNT) | 20.9% |
| Discontinuation due to AEs | ~5–7% | 12.2% (9 mg), 18.2% (12 mg) |
Note: Rates are from different trials with different populations. Direct comparison is approximate. Some TRIUMPH-4 discontinuations were for "perceived excessive weight loss" — not typical adverse events.
A Note on Heart Rate
Both have been associated with increases in heart rate in trials. The Zepbound label reports a mean increase of 1–3 bpm. Phase 2 retatrutide data showed dose-dependent increases that peaked around 24 weeks and then declined. Both appear safe for most people, but if you have cardiac arrhythmias or significant heart disease, this is worth discussing with a cardiologist before starting either medication.
Can You Buy Retatrutide Right Now?
No. There is no legitimate way to buy retatrutide today.
What the FDA Says
As of March 2026, retatrutide has no FDA approval. It's classified as an investigational drug. There is no FDA-approved retail prescription or pharmacy pathway today. The FDA has published explicit warnings about unapproved GLP-1 products being sold online.
What's Actually Being Sold Online
The "retatrutide" available from online peptide vendors is:
- Not manufactured by Eli Lilly — the only company with legitimate retatrutide
- Not regulated by the FDA — no quality, purity, or safety standards enforced
- Potentially counterfeit, contaminated, or incorrectly dosed
- Illegal to sell for human use — typically labeled "for research purposes only"
How to Access Retatrutide Legitimately
The only legitimate access today is through an authorized clinical trial. You can search for open trials at ClinicalTrials.gov using the keyword "retatrutide." If you qualify, the medication is provided at no cost, under strict medical supervision.
Retatrutide FDA Approval Timeline: When Will It Be Available?
No verified FDA approval date exists. Here's what we do know:
- December 2025: TRIUMPH-4 (the first Phase 3 readout) reported record-breaking results
- 2026: Eli Lilly has stated that seven additional Phase 3 readouts are expected, covering obesity, T2D, sleep apnea, chronic low back pain, fatty liver disease, and cardiovascular/renal outcomes
- Late 2026 – 2027: Any FDA filing will depend on those results and Lilly's regulatory plans
- Late 2027 – 2028: Earliest realistic commercial availability, assuming successful trials and standard FDA review timelines
The dysesthesia safety signal from TRIUMPH-4 adds a layer of uncertainty. If it shows up consistently across other TRIUMPH trials, the FDA may require additional analysis, which could affect the timeline.
Bottom line on timing: Even in the best-case scenario, retatrutide is at least a year away from being something your doctor can prescribe. For most people, that means waiting doesn't make sense when effective treatment exists today.
Retatrutide vs Tirzepatide Cost: What Can You Actually Pay Today?
Tirzepatide (Zepbound / Mounjaro): Real Pricing Paths
The list price for Zepbound is $1,086.37 for a 28-day supply. But most people don't pay list price:
| Route | Monthly Cost (Approx.) | Details |
|---|---|---|
| With insurance + savings card | As low as $25 | Must have commercial plan that covers Zepbound. Not for Medicare, Medicaid, VA, TRICARE. |
| LillyDirect self-pay (vials) | $299–$449 | Single-dose vials through Eli Lilly's direct program. Must refill within 45 days at higher doses. |
| Full list price (pens) | $1,086 | Without any savings programs. |
If you're exploring telehealth providers for tirzepatide, we maintain a comparison of verified GLP-1 providers with transparent pricing.
Why Retatrutide Has No Retail Price
Retatrutide isn't commercially available, so there's no legitimate consumer pricing today. This is worth thinking about if you're tempted to "wait for retatrutide." You'll have lost a year or more of treatment in the meantime — and there's no guarantee the eventual price will be lower than tirzepatide.
Why "Cheap Retatrutide" Is a Red Flag
If you see retatrutide advertised at $100–$200 per vial from an online peptide vendor, that's not a deal. It's a warning sign. Legitimate pharmaceutical manufacturing is expensive. Products priced dramatically below expectations are almost certainly not what they claim to be.
Does Retatrutide Cause More Muscle Loss Than Tirzepatide?
Based on current evidence, no — the lean-to-fat loss ratio appears similar for both drugs.
Tirzepatide data: In the SURMOUNT-1 DXA substudy, tirzepatide reduced body weight by 21.3%, fat mass by 33.9%, and lean mass by 10.9% — meaning most of the lost weight was fat.
Retatrutide data: A substudy published in The Lancet Diabetes & Endocrinology (2025) found that the proportion of lean mass lost relative to total weight lost was similar to what's seen with other obesity treatments.

What actually protects muscle during weight loss:
- Resistance training — the single most important factor for preserving lean mass
- Adequate protein intake — discuss specific targets with your provider
- Gradual weight loss — crash-rate loss increases muscle depletion
These strategies apply regardless of which medication you're on. For more detail, see our guide on preventing muscle loss on GLP-1 medications.
Beyond Weight Loss: What Else Do These Drugs Do?
Tirzepatide: Proven Additional Benefits
- Blood sugar control: Originally developed for T2D. Dramatic HbA1c reductions.
- Cardiovascular markers: Improved cholesterol, triglycerides, blood pressure
- Sleep apnea: FDA-approved for moderate-to-severe obstructive sleep apnea (2024)
- Real-world track record: Years of post-approval and real-world use
Retatrutide: Emerging Benefits from TRIUMPH-4
- Knee osteoarthritis pain: WOMAC pain scores reduced by up to 75.8%. Roughly 1 in 8 patients were completely free of knee pain at 68 weeks, vs ~4% on placebo.
- Cardiovascular markers: Meaningful reductions in non-HDL cholesterol, triglycerides, and hsCRP
- Blood pressure: 14 mmHg systolic blood pressure reduction at the 12 mg dose
Retatrutide is also being studied for metabolic dysfunction-associated steatotic liver disease, chronic low back pain, and cardiovascular/renal outcomes. A massive 10,000-patient trial (TRIUMPH-OUTCOMES) is investigating the impact on major cardiovascular events.
The practical implication: If you start tirzepatide now and it reduces your weight, blood pressure, blood sugar, and joint pain — that's not a consolation prize while you wait for retatrutide. That's the treatment working exactly as intended.
What About "Food Noise"?
Food noise is the mental chatter about food: thinking about your next meal while eating your current one, craving snacks you're not hungry for, obsessing over what to eat. Many people with obesity describe it as a constant background hum.
Both tirzepatide and retatrutide reduce food noise — often dramatically. Patients frequently describe it as one of the most surprising and welcome effects: the mental preoccupation with food fades, and eating becomes a calmer, more functional part of the day.
Anecdotally, some patients have reported that retatrutide's three-receptor approach produces an even more pronounced quieting of food-related thought patterns. But this is patient-reported and anecdotal — not something clinical trials have formally measured.
For many people, this psychological shift is actually the most transformative part of GLP-1 treatment — more than the number on the scale.
Managing Side Effects: Practical Tips That Apply to Both Drugs
During Dose Escalation (the Toughest Stretch)
- Eat smaller meals more frequently instead of three large ones
- Avoid high-fat, greasy, or overly rich foods — they tend to be harder to tolerate
- Stay hydrated, but sip water throughout the day rather than gulping large amounts at once
- Eat slowly and stop when you feel satisfied, not stuffed
- Ginger tea or ginger chews can help settle nausea
General Principles
- Take your injection at the same time each week. Many people find evenings or weekends work best.
- If nausea is severe, your doctor can slow the titration schedule. There's no race to maximum dose.
- Constipation is common — increase fiber and stay on top of hydration.
- If symptoms don't improve after a few weeks at a given dose, talk to your doctor before increasing again.
Can You Switch from Tirzepatide to Retatrutide?
Not right now — because retatrutide isn't available for prescribing.
If and when retatrutide becomes available, switching would be possible under medical supervision. However:
- No established dose-conversion exists between the two drugs
- You'd likely stop tirzepatide and start retatrutide at the lowest dose, titrating up from scratch
- The transition period could come with renewed GI side effects as your body adjusts
- The additional glucagon activity introduces a much higher dysesthesia risk
Reasons someone might eventually switch
- Plateaued on maximum tirzepatide dose
- Desire for additional metabolic "push" from glucagon
- Specific condition where retatrutide data is stronger (e.g., knee osteoarthritis)
Reasons someone might stay on tirzepatide
- Well-controlled and happy with current results
- Concerned about dysesthesia risk
- Insurance covers tirzepatide but may not cover retatrutide
- Preferring established long-term safety data
The practical takeaway: If tirzepatide is working for you, there is no medical reason to stop it and wait for retatrutide. You can always reassess later.
Is There a Retatrutide vs Tirzepatide Dosage Chart?
Tirzepatide has an FDA-approved dosing schedule. Retatrutide does not. There is no validated dose-conversion chart between the two, and we won't create a fake one.
Tirzepatide: Approved Titration Schedule
| Weeks | Dose | Notes |
|---|---|---|
| Weeks 1–4 | 2.5 mg weekly | Starting dose (not therapeutic — for tolerability) |
| Weeks 5–8 | 5 mg weekly | First therapeutic dose |
| Weeks 9–12 | 7.5 mg weekly | Optional increase |
| Weeks 13–16 | 10 mg weekly | Common maintenance dose |
| Weeks 17–20 | 12.5 mg weekly | Optional increase |
| Week 21+ | 15 mg weekly | Maximum approved dose |
Source: FDA prescribing information for Zepbound (tirzepatide).
Retatrutide: Trial Protocol Dosing (NOT an Approved Schedule)
| Weeks | Dose | Notes |
|---|---|---|
| Weeks 1–4 | 2 mg weekly | Starting dose |
| Weeks 5–8 | 4 mg weekly | Step 2 |
| Weeks 9–12 | 6 mg weekly | Step 3 |
| Weeks 13–16 | 9 mg weekly | Lower target dose arm |
| Week 17+ | 12 mg weekly | Higher target dose arm |
This is clinical trial protocol information, not prescribing guidance. No one should attempt to follow this schedule outside of a supervised clinical trial. There is no validated dose conversion between the two drugs.
Retatrutide vs Tirzepatide vs Semaglutide: The Full Picture
Since many people are also considering semaglutide, here's where all three fit in the landscape.
| Semaglutide | Tirzepatide | Retatrutide | |
|---|---|---|---|
| Mechanism | GLP-1 only (single agonist) | GLP-1 + GIP (dual agonist) | GLP-1 + GIP + Glucagon (triple agonist) |
| Brand names | Ozempic, Wegovy, Rybelsus | Mounjaro, Zepbound | None (investigational) |
| FDA approved | Yes | Yes | No |
| Best published weight loss | ~15% at 68 wks | ~22.5% at 72 wks | ~28.7% at 68 wks |
| Administration | Weekly injection or daily oral tablet | Weekly injection | Weekly injection |
| Evidence grade | Phase 3 + extensive real-world data | Phase 3 + growing real-world data | Phase 2 peer-reviewed + Phase 3 topline only |
| Cost without insurance | ~$900–$1,350/mo | ~$299–$1,086/mo | N/A |
| Unique strength | Most real-world data; oral option | Best efficacy + availability balance | Highest weight loss; glucagon fat burning |
The trend is clear: Each generation targets more receptors and produces more weight loss. But each new generation also has less real-world data and more unknowns. Semaglutide is the most battle-tested. Tirzepatide is the current sweet spot. Retatrutide is the frontier.
For a detailed head-to-head comparison of the two FDA-approved options, see our semaglutide vs tirzepatide guide.
Who Should Choose Tirzepatide Today?
You want legitimate treatment you can start now. Tirzepatide is available by prescription. You can have it in hand within days of a telehealth consultation or doctor visit.
You value proven safety and established protocols. Tirzepatide has years of post-approval and real-world use. The side effect profile is well-understood. The dysesthesia signal with retatrutide adds a question mark that won't be answered until more trials report.
Insurance, self-pay options, or flexible pricing matters to you. Between manufacturer savings cards, LillyDirect vials, and telehealth providers, there are multiple ways to access tirzepatide at different price points.
You've already tried semaglutide and want more. Tirzepatide is the natural next step. It targets an additional receptor and consistently outperforms semaglutide in head-to-head data.
You have type 2 diabetes alongside obesity. Tirzepatide (as Mounjaro) is specifically approved for T2D and produces excellent HbA1c reductions alongside weight loss.
You're nervous about an untested drug. That's not irrational — it's prudent. Choosing the medication with more evidence behind it is the conservative, medically sound decision.
Who Should Watch Retatrutide — Instead of Chasing It?
Follow it if you're tracking next-generation obesity medicine. The TRIUMPH program will release multiple Phase 3 readouts in 2026. Each one will tell us more about efficacy, safety, and which patients benefit most.
Watch it if you need more weight loss than tirzepatide provides. For people who max out on tirzepatide and still have significant weight to lose, retatrutide may eventually be the answer.
Ask your doctor about clinical trial eligibility. If retatrutide genuinely interests you and you meet the criteria, enrolling in a TRIUMPH trial is the only safe and legal way to access it.
Do not buy it online. The "retatrutide" being sold by peptide vendors is not manufactured by Eli Lilly, not regulated by the FDA, and not guaranteed to contain what the label says.
What Should You Do Next?
You've read the data. You understand the difference. Here's the decision tree.
"I want treatment that works, and I want to start now." → Talk to your doctor about tirzepatide (or semaglutide). Both are effective, FDA-approved, and available today. Every month of untreated obesity is a month of continued metabolic damage. Starting now doesn't prevent you from switching later.
"I'm on tirzepatide already and it's working well." → Stay on it. The incremental benefit of retatrutide doesn't justify stopping a medication that's producing real results for you.
"I'm on tirzepatide and it's NOT working well enough." → Before looking to retatrutide, make sure you've optimized what you have. Are you on the maximum dose? Pairing with adequate protein and resistance training? If you've genuinely maxed out, ask about clinical trial eligibility.
"I'm mainly researching what's coming next." → Bookmark this page. The head-to-head TRIUMPH-5 trial results will be the most important data point. We'll update this page as new data drops.
"I saw retatrutide for sale online." → It's not legitimate. Walk away. The only safe path is through a clinical trial at ClinicalTrials.gov.
Still not sure which GLP-1 program is right for you? Our free 60-second matching quiz helps you find the best legitimate option based on your budget, goals, and medical situation.
Take the GLP-1 Fit QuizFrequently Asked Questions
How We Researched and Verified This Comparison
We take accuracy seriously — especially on health topics where bad information can lead to real harm.
Primary Sources
- Jastreboff AM, et al. "Tirzepatide Once Weekly for the Treatment of Obesity." New England Journal of Medicine 2022; 387:205-216 (SURMOUNT-1 trial)
- Jastreboff AM, et al. "Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial." New England Journal of Medicine 2023
- Eli Lilly and Company. TRIUMPH-4 topline results press release, December 11, 2025
- FDA Prescribing Information: Zepbound (tirzepatide) and Mounjaro (tirzepatide)
- FDA.gov: Warnings on unapproved GLP-1 drugs
- ClinicalTrials.gov: TRIUMPH-5 head-to-head trial (NCT06662383) and TRIUMPH program listings
- PMC Network Meta-Analysis: "Comparative Efficacy and Safety of Tirzepatide vs Retatrutide"
- Coskun T, et al. "Effects of retatrutide on body composition." Lancet Diabetes & Endocrinology 2025
- Eli Lilly Investor Relations: TRIUMPH program descriptions and timelines
What We Do Not Do
- We don't treat press releases as equivalent to peer-reviewed data (we label the evidence grade)
- We don't publish unofficial dosing guidance for investigational drugs
- We don't recommend gray-market sourcing under any circumstances
- We don't present indirect trial comparisons as head-to-head proof
Verification Table (March 2026)
| Fact | Status | Verified Against |
|---|---|---|
| Retatrutide FDA approval | Not approved | FDA.gov, Drugs.com |
| TRIUMPH-4 results | 28.7% weight loss (12 mg) | Eli Lilly press release, Dec 11, 2025 |
| Tirzepatide availability | Available by prescription | FDA-approved label |
| Zepbound pricing | $299–$699 (vials); $1,086 (list) | Zepbound.lilly.com |
| Head-to-head trial | TRIUMPH-5 ongoing | ClinicalTrials.gov NCT06662383 |
| Compounding status | Tirzepatide compounding restricted | FDA guidance |
| Dysesthesia signal | 20.9% at 12 mg in TRIUMPH-4 | Eli Lilly press release, Zepbound FDA label |
This content is for informational purposes only. It is not medical advice. Always consult a licensed healthcare provider before starting, stopping, or changing any medication. Weight loss medications should be used under medical supervision as part of a comprehensive plan that includes diet and physical activity.
Related Articles
What Is GLP-1?
A complete beginner's guide to GLP-1 medications, how they work, and what to expect.
GLP-1s for Weight Loss
How GLP-1 receptor agonists promote weight loss through appetite reduction and metabolic changes.
Learn About GLP-1
Educational hub covering GLP-1 science, medications, eligibility, and treatment options.
History of GLP-1 Drugs
The complete timeline of GLP-1 drug development from discovery to modern weight loss treatments.
Semaglutide vs Tirzepatide
Head-to-head comparison of semaglutide and tirzepatide for weight loss effectiveness and cost.
Willow vs Ozempic
Willow telehealth service vs FDA-approved Ozempic: real pricing, safety, eligibility, and regulatory differences compared.