By the Weight Loss Provider Guide Editorial Team · Last verified: April 20, 2026
Last updated: April 20, 2026. Next scheduled verification: May 20, 2026.
Sourced from peer-reviewed studies, systematic reviews, official FDA prescribing information, and authoritative reporting on newly published research. Weight Loss Provider Guide is an independent comparison resource. We earn commissions on some provider links — that does not affect this page's editorial judgment. Disclosure →
Does GLP-1 affect men and women differently? Yes — but probably less than the headlines suggest, and not in a way that means men and women need different drugs. Within a 2026 Johns Hopkins meta-analysis, in the six trials with sex-stratified data (19,906 patients), women lost about 10.88% of body weight vs. 6.78% for men on GLP-1 receptor agonists (Mehta et al., JAMA Internal Medicine, March 2026). Women also tend to get more nausea. The glucose, heart, and kidney benefits look broadly similar across sexes.
That's the answer. The rest of this page explains what "different" actually means in the clinic, which differences matter for your decision, and which headlines are overblown — including one surprising 2026 finding that flips a common assumption about muscle loss on its head.

Not sure what this means for your situation?
Take our free 60-second matching quiz for a personalized action plan based on your sex, life stage, medications, and goals.
What Actually Changes for Men and Women on GLP-1s
First-scroll summary — full evidence in each section below.
| Area | Women | Men | Does It Matter for Your Choice? |
|---|---|---|---|
| Average weight loss (GLP-1 RAs) | ~10.88% of body weight | ~6.78% of body weight | Informs expectations, not drug choice |
| Blood sugar (HbA1c) | Similar reduction | Similar reduction | No |
| Heart / kidney benefit | Similar | Similar | No |
| Nausea and vomiting | More common | Less common | Affects titration pace, not drug choice |
| Pregnancy planning (semaglutide) | Stop 2 months before a planned pregnancy (Wegovy label) | N/A | Yes — plan ahead |
| Pregnancy planning (tirzepatide, Foundayo) | Discontinue when pregnancy recognized; discuss timing with prescriber | N/A | Yes — plan ahead |
| Oral birth control interaction | Tirzepatide: 4-week backup. Foundayo: 30-day backup. Semaglutide: no warning | N/A | Yes — may change drug or method |
| PCOS / menopause context | Often a major reason to start | N/A | Yes — influences fit |
| Lean mass preservation | Slightly worse on average | Slightly better on average | Both should lift and eat protein |
| Testosterone (obesity-related low T) | Modest drop in PCOS (often wanted) | Modest rise (often welcomed) | Informs expectations |
Sources: Mehta et al., JAMA Internal Medicine (March 2026); current FDA-approved prescribing information for Wegovy, Zepbound, Mounjaro, and Foundayo (verified April 20, 2026); Stokes et al., JAMA Network Open (2026); peer-reviewed systematic reviews cited throughout.
What People Are Actually Asking
Before we get into the evidence, here are the real questions behind this query — pulled from public forums and search data:
- ›"My husband and I are both on semaglutide. He's lost 25 pounds. I've lost 8. What am I doing wrong?"
- ›"Nobody told me tirzepatide could mess with my birth control. Is that actually true?"
- ›"I'm a guy worried about losing muscle. Should I even start?"
- ›"I heard women lose more weight — does that mean it won't work as well for me?"
- ›"Can my husband keep taking Mounjaro while we're trying to have a baby?"
Forum snippets above reflect real user questions, not medical evidence. Every clinical claim on this page is sourced below.
Does GLP-1 Affect Men and Women Differently? The Short Answer
Yes — mostly in three places: average weight-loss response, side-effect tolerability, and a short list of sex-specific practical issues (pregnancy, contraception, and reproductive hormones). The glucose, cardiovascular, and kidney benefits look broadly similar across sexes. There is no "male GLP-1" and no "female GLP-1."
The shortest honest answer
Women tend to lose somewhat more weight and tend to have more GI side effects. Men tend to protect muscle a little better and, if they had obesity-related low testosterone, tend to see it rebound. Everything else is largely a wash.
What changes in practice
Four things actually change the conversation:
- Average weight-loss expectations. A man losing 7% and a woman losing 11% on the same drug is the statistical norm, not a sign either is doing it wrong.
- Dose titration. Women often need a slower ramp to manage nausea.
- Pregnancy and contraception planning. This is the single most actionable sex difference — and the details are molecule-specific.
- Body-composition strategy. Both sexes should resistance-train and prioritize protein — but women may need to push harder on this, which reverses a long-standing assumption.
What does not change
Men and women appear to get similar HbA1c reductions, similar cardiovascular event reductions, and similar kidney benefits across the GLP-1 class. That's the part most pages skip — and it matters most if the reason you're on a GLP-1 is diabetes, heart disease, or kidney protection rather than weight loss.
What the Best Current Evidence Actually Says
Three independent research layers point to the same conclusion: women lose more weight on GLP-1s than men on average, while non-weight outcomes track similarly across sexes.
| Study | Scope | Key Finding |
|---|---|---|
| 2026 Johns Hopkins / JAMA Internal Medicine (Mehta et al.) | 64 trials total; 6 sex-stratified trials, 19,906 patients | Women: 10.88% weight loss. Men: 6.78%. HbA1c and CV outcomes similar across sexes. |
| 2025 sex-difference meta-analysis (Yang et al., Journal of Diabetes) | 14 studies, 5 molecules (dulaglutide, exenatide, liraglutide, semaglutide, retatrutide) | Women lost 1.04 kg more and 1.69 percentage points more. Gap widened at higher doses and longer durations. |
| 2025 GLIMPLES 4-year cohort (Marassi et al., Pharmacological Research) | Type 2 diabetes patients; median 4-year follow-up | 66.5% of women vs. 58.0% of men lost ≥5%. 40.0% vs. 30.7% lost ≥10%. HbA1c difference: not significant (p = 0.21). |
The 2026 Johns Hopkins meta-analysis (the headline study)
Published in JAMA Internal Medicine (March 2026), this is the largest look to date at subgroup responses. The sex-stratified subset: women lost 10.88% of baseline body weight; men lost 6.78%. For every other demographic studied — race, age, starting BMI, baseline HbA1c — response was essentially similar.
What this evidence can and cannot prove
It proves the pattern. It does not prove the mechanism. It also doesn't prove every woman will lose more than every man — individual variation inside each sex is much larger than the average gap between them.
Do Women Lose More Weight on GLP-1s Than Men?
Yes, on average. The 2026 Johns Hopkins sex-stratified data pegged the gap at roughly 10.88% vs. 6.78% of body weight across pure GLP-1 receptor agonists. But "on average" is the most important phrase in that sentence.
What the gap really means
If you and your partner are on the same drug at the same dose and your numbers are on opposite sides of this range, you're not doing something wrong. You're at the statistical norm.
- If you're a woman, you have a higher chance of being a high responder. The gap widens at higher doses and longer durations.
- If you're a man, a 6–7% loss is clinically meaningful — that's the threshold where cardiovascular risk markers improve.
- If you're a man losing much less than that at a therapeutic dose, audit dose, titration, protein, and activity with your clinician before assuming the drug isn't working.
Does the sex gap hold for tirzepatide?
For semaglutide and dulaglutide, yes — statistically significant sex differences in weight reduction are established. Exenatide did not show a clear sex difference. Tirzepatide-specific sex-stratified data is still being accumulated — early signals are consistent with the class pattern, but the 10.88% vs. 6.78% figure is not a tirzepatide number.
Why couples on the same GLP-1 see very different results
- Biology. Women average ~4 percentage points more weight loss on these drugs.
- Dose-to-body-size ratio. A 2 mg dose in a 200-lb man delivers a different exposure than the same dose in a 140-lb woman.
- Adherence timing. Men are slightly more adherent at 12 months in older diabetes cohorts; women sometimes pause or titrate down due to nausea.
- Diet and activity context. Women in weight-loss trials tend to engage more with lifestyle modification.
Are GLP-1 Side Effects Worse for Women?
Probably, yes — especially for nausea, vomiting, and GI symptoms. Multiple sources show women experience more tolerability issues and discontinue treatment more often than men. We'll be clear about which claims are established and which are emerging.
What's established
- Women report persistent GI side effects at higher rates than men (KFF Health Tracking Poll, 2024–2025; RAND, 2025).
- In older adherence studies, women's 12-month adherence was lower (~48.4% vs. ~54.2% for men), with tolerability cited as a common reason for discontinuation.
- Nausea is the most common GLP-1 side effect overall, reaching about 44% in semaglutide weight-management trials.
What's emerging (labeled carefully)
Why the nausea gap matters
Nausea predicts whether you stay on the medication long enough to benefit. The playbook for managing it:
- Slower dose titration. Ask your clinician about holding at a lower dose longer before stepping up.
- Protein-forward, lower-fat meals. Delayed gastric emptying punishes heavy, greasy food.
- Hydration between meals, not during.
- Cycle tracking. Some women find side effects track with hormonal phases — useful data for your provider.
- Consider a different molecule. Dulaglutide (Trulicity) trends toward less nausea than semaglutide in head-to-head comparisons.
The menstrual-cycle signal
A 2026 University of Pennsylvania analysis of roughly 410,000 Reddit posts from approximately 67,000 GLP-1 users (Sehgal et al., arXiv preprint) flagged two symptom clusters not prominent in clinical trial reporting: menstrual irregularities (including unexpected bleeding) and temperature-related symptoms like hot flashes and chills. These are hypothesis-generating — Reddit users skew younger, more male, and more U.S.-based than the broader GLP-1 population — but consistent with GLP-1 acting on the hypothalamus, and worth tracking if you notice them.
Do Men and Women Get Different Blood Sugar, Heart, or Kidney Benefits?
Not meaningfully, based on current evidence. The sex gap lives in weight loss and tolerability — not in the outcomes that matter most for diabetes, heart disease, and kidney disease.
| Outcome | Women | Men | Source |
|---|---|---|---|
| HbA1c reduction | Significant improvement | Significant improvement — no sex difference (p = 0.21) | GLIMPLES cohort, Marassi et al., 2025 |
| Cardiovascular outcomes (MACE) | Consistent reduction | Consistent reduction | 2025 CV outcomes meta-analysis |
| Kidney / CKD protection | Extends to both sexes | Extends to both sexes | Current GLP-1 renal data |
| Weight loss | ~10.88% average | ~6.78% average | Mehta et al., JAMA IM, 2026 |
Don't assume men get less heart benefit just because women may lose more weight — the outcomes data says otherwise.
What Women Need to Know Before Starting a GLP-1
Four sex-specific issues matter more than the weight-loss gap: pregnancy timing (which varies by molecule), oral contraceptive interactions with tirzepatide and Foundayo, PCOS context, and menopause context.
The molecule-by-molecule label matrix
Most articles say "stop GLP-1s 2 months before pregnancy" and "GLP-1s affect birth control." Both statements are oversimplified in ways that matter. Here is what the current U.S. labels actually say:
| Medication | Molecule | Pregnancy / Preconception Label | Oral Contraceptive Warning | Lactation Label |
|---|---|---|---|---|
| Wegovy | Semaglutide | Stop at least 2 months before a planned pregnancy | No formal interaction warning | Weighs maternal need vs. potential infant risk |
| Ozempic | Semaglutide | Discontinue when pregnancy is recognized | No formal interaction warning | Weighs maternal need vs. potential infant risk |
| Zepbound / Mounjaro | Tirzepatide | Discontinue when pregnancy is recognized; discuss preconception timing with prescriber | Switch to non-oral or add barrier for 4 weeks after start and after each dose escalation | Weighs maternal need vs. potential infant risk |
| Foundayo | Orforglipron | Discontinue when pregnancy is recognized; discuss preconception timing with prescriber | Switch to non-oral or add barrier for 30 days after start and after each dose escalation | Not recommended for nursing women per label |
Source: current U.S. FDA-approved prescribing information for each product, verified April 20, 2026. Always confirm against the current product insert before starting or making decisions.
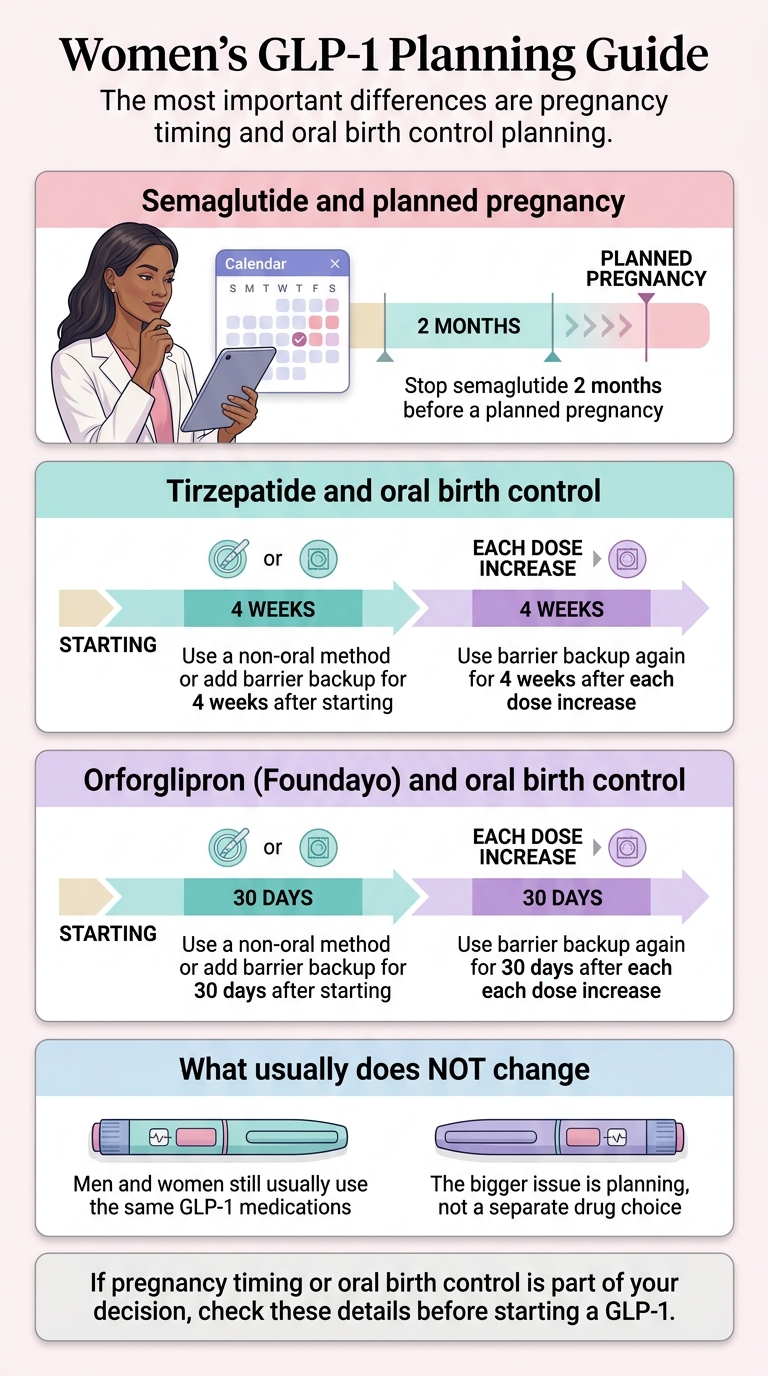
Why tirzepatide carries a contraceptive warning but semaglutide doesn't
Tirzepatide slows gastric emptying more than other GLP-1s, especially after the first dose and after dose increases, which can reduce absorption of ethinyl estradiol and progestins. One pharmacokinetic study showed oral contraceptive hormone levels dropped by about 20% after a single tirzepatide dose. Semaglutide's pharmacokinetic studies have not shown the same effect. Unaffected contraceptive options: IUD, hormonal implant, injection, patch, vaginal ring.
Foundayo (orforglipron) is an oral GLP-1, so it competes directly with oral contraceptives for gut absorption — not just indirectly through gastric emptying. That's why its backup-contraception window is 30 days instead of 4 weeks.
PCOS and perimenopause
PCOS affects an estimated 7–10% of women of reproductive age. A 2023 meta-analysis in BMC Endocrine Disorders (Zhou et al.) found that women with PCOS on GLP-1 receptor agonists had improved spontaneous pregnancy rates — largely through weight loss, insulin-sensitivity improvement, and restored ovulation. If you have PCOS and have not been using contraception because you were told you couldn't get pregnant, reconsider that assumption on a GLP-1.
Perimenopause and menopause change the body-composition context: women gain more visceral fat, lose more muscle, and experience appetite and sleep shifts that make weight management harder. GLP-1s appear to work across life stages, but the data specifically in post-menopausal women is thinner. Expect your clinician to weigh bone and muscle concerns more heavily if you're over 55.
What to ask your clinician
- "Am I planning pregnancy in the next 12 months, and do we need a timing plan for my specific molecule?"
- "If I'm on tirzepatide or Foundayo, what's my contraceptive plan during start and titration?"
- "Do I have PCOS or insulin resistance that makes a GLP-1 especially appropriate?"
- "If I'm post-menopausal, what's our plan for protecting muscle and bone?"
What Men Need to Know Before Starting a GLP-1
Three issues matter more for men than for women: lean mass preservation, testosterone and sexual function, and fertility. The news is mostly good — but the evidence on ED and fertility is mixed rather than one-directional, and we'll say so clearly.
| Outcome | Evidence Direction | How Settled | What to Do |
|---|---|---|---|
| Testosterone (obesity-related low T) | Positive — modest rise in bioavailable T | Moderately settled in target population | Baseline labs if symptomatic; don't expect a boost if T is normal |
| Erectile function | Mixed — most data leans positive; newest study is a counter-signal | Not settled | Discuss with clinician; baseline IIEF-5 if you have ED concerns; flag any change |
| Fertility / sperm parameters | Neutral-to-positive in obese men | Limited — long-term data sparse | Flag if actively trying to conceive; get a semen analysis if concerned |
| Lean mass preservation | Men preserve better than women (reverses the common assumption) | Emerging but consistent | Resistance train + eat 1.2–1.6 g/kg protein; DEXA scan if possible |
1. Lean mass — the assumption that gets flipped
Across GLP-1 therapies, roughly 25% of total weight lost is lean mass. But a 2026 JAMA Network Open body-composition study found men preserved fat-free mass better than women on GLP-1 receptor agonists — not worse. This reverses the common assumption that men are at greater risk of muscle loss, and suggests post-menopausal women may need the more aggressive protein-and-resistance-training protocol.
2. Testosterone: promising for men with obesity-related low T
If you have obesity-related functional hypogonadism (low testosterone driven by excess body fat), GLP-1 therapy is broadly associated with a rise in bioavailable testosterone — largely through weight loss and insulin-sensitivity correction.
- A 2025 systematic review and meta-analysis found significant rises in bioavailable testosterone in men with obesity or type 2 diabetes.
- A November 2025 Journal of Sexual Medicine systematic review (Mahmood et al.) reported 7 of 9 human studies showed statistically significant rises in testosterone after GLP-1 treatment.
- 2 studies showed no change in metabolically healthy, eugonadal men — if your baseline testosterone is normal, don't expect a boost.
3. Erectile function: the evidence is mixed
The positive signal:
- A 2024 Mendelian-randomization study found a genetic GLP-1 profile was associated with approximately 50% lower odds of erectile dysfunction, mediated largely through reductions in obesity, diabetes, and blood pressure.
- A 1-year retrospective cohort study (ECE 2024) showed significant improvements on the IIEF-5 compared to metformin alone.
The counter-signal (2026 EClinicalMedicine):
4. Fertility and sperm parameters
Early evidence in obese men shows improvements in sperm concentration, motility, and morphology alongside weight loss. Current human evidence does not show a clear direct fertility-harm signal, but long-term male reproductive data remain sparse. If you're actively trying to conceive, have the conversation with your clinician.
Why Might GLP-1s Affect Men and Women Differently? (The Mechanism)
Researchers have three plausible explanations: estrogen-GLP-1 interaction, body-size and drug-exposure differences, and sex-specific receptor density in key brain regions. The clinical pattern is well-established; the exact mechanism is still being worked out.
1. Estrogen synergizes with GLP-1 signaling (clinical + translational evidence)
Estrogen appears to amplify the satiety and metabolic effects of GLP-1. A 2025 Endocrinology review (Börchers & Skibicka) concluded that quantitative sex differences are real but qualitative differences (i.e., the drug fundamentally working differently) are not. This is consistent with observations that women's GLP-1 response can vary across the menstrual cycle and differ pre- vs. post-menopause.
2. Body size and drug exposure (clinical evidence)
Women on average weigh less than men, so a fixed dose delivers a higher dose-per-kilogram exposure. This partially explains why the same 2.4 mg semaglutide or 15 mg tirzepatide produces a bigger relative weight-loss result in women. It also partially explains the side-effect asymmetry — higher exposure per kilogram drives more GI symptoms.
3. Sex-specific brain receptor density (preclinical evidence)
In early 2026, a team at Mount Sinai (Ryu, Zaidi et al.) published the first comprehensive sex-specific atlas of GLP-1 expression in the mammalian brain, mapping GLP-1 at single-transcript resolution across 25 brain regions. GLP-1 density was significantly higher in certain female hindbrain regions linked to both appetite suppression and nausea. The work is in rodent models; translation to humans is still pending. This is one of the cleanest biological explanations yet for why the same drug might produce more weight loss and more side effects in women.
Note: This is fascinating preclinical evidence — not a clinical tool. Nobody should change their dose based on it.
Does Sex Change Which GLP-1 Medication You Should Choose?
Usually not by itself. The primary factors for drug choice remain your goal, medical contraindications, insurance and cost, and medication form. Sex matters at the margins — specifically around pregnancy planning, oral contraceptives, and certain reproductive considerations.
When sex matters a lot
| Scenario | What Sex Adds to the Decision |
|---|---|
| Woman planning pregnancy within 12 months | Semaglutide: stop at least 2 months pre-conception. Tirzepatide/Foundayo: discuss timing with your prescriber. Factor into start timing. |
| Woman on oral hormonal contraceptives | Tirzepatide requires 4-week backup after start and each dose increase. Foundayo requires 30-day backup. Semaglutide does not carry this warning. Non-oral contraceptives (IUD, implant, injection, patch, ring) are not affected. |
| Man with obesity-related low testosterone or ED | GLP-1 weight loss may produce welcome reproductive-hormone improvements; the 2026 ED counter-signal means baseline labs and follow-up matter. |
| Adults over 60 or with low baseline muscle (either sex) | Consider resistance training and protein intake as non-negotiable before starting, and a baseline DEXA scan if accessible. |
A simple decision framework
- Woman, no pregnancy plans, IUD or implant in place:Sex is largely a non-factor for drug choice. Pick based on goal, molecule, and cost.
- Woman on oral birth control pills:Prefer semaglutide over tirzepatide or Foundayo, or plan backup contraception aligned to the start/titration schedule.
- Woman with PCOS and irregular cycles:GLP-1s may restore ovulation and fertility — use contraception if you're trying to prevent pregnancy.
- Post-menopausal woman:Push harder on muscle and bone protection than the default protocol.
- Man with obesity-related metabolic issues:The testosterone and ED picture is promising but mixed — establish a baseline before starting.
- Man focused on athletic performance / muscle preservation:Plan protein and resistance training from day one; consider a slower dose ramp.
This is exactly the kind of situational question our quiz was built for.
Two minutes, a handful of questions, and you get a personalized plan that factors in your sex, life stage, medications, and goals.
What's Still Unknown
There's enough evidence to answer "does GLP-1 affect men and women differently?" — but not enough to pretend every detail is settled. Here's what we're still waiting on.
- Specific-drug side-effect gaps by sex. We know women have more GI side effects overall. Clean, molecule-by-molecule, peer-reviewed sex-stratified data isn't yet available for every approved GLP-1. Claims of precise multipliers (like 2.5× nausea) come from emerging real-world data that needs replication.
- Dose-response differences by sex. Researchers have proposed cycle-phase-based or body-weight-adjusted dosing for women, but none of that is standard of care yet.
- Long-term reproductive outcomes. The Lilly and Novo Nordisk pregnancy registries are still accumulating data. Large-sample answers on pregnancy outcomes after GLP-1 exposure in either parent are not yet available.
- Male-specific endpoints at scale. Testosterone, ED, sperm, and long-term sexual function studies in men exist — but sample sizes and follow-up durations are smaller than the corresponding female data. The 2026 EClinicalMedicine ED counter-signal shows the male picture is still forming.
- GLP-1 effects across the menstrual cycle. The hypothesis that side effects track hormonal phases is mechanistically plausible and supported by real-world signals. It hasn't been tested in a clean randomized trial.
We'll update this page when the answers move.
How We Built This Page
We are Weight Loss Provider Guide — an independent comparison resource for GLP-1 telehealth providers. We write these evidence explainers because the existing top-ranked results either go too academic or too narrow. This page pulls the full picture together in one place.
What we actually verified for this page (April 20, 2026):
- Reviewed the 2026 Johns Hopkins / JAMA Internal Medicine meta-analysis (Mehta et al.; 64 trials total; 6 sex-stratified trials, 19,906 patients) and its coverage in Medscape, Hub JHU, and the Bloomberg School of Public Health press release.
- Reviewed the 2025 sex-difference meta-analysis (Yang et al., Journal of Diabetes) covering 5 molecules.
- Reviewed the 2025 GLIMPLES multi-year real-world cohort (Marassi et al., Pharmacological Research).
- Reviewed the 2026 JAMA Network Open body-composition study on sex differences in fat-free mass preservation.
- Checked current FDA-approved prescribing information for Wegovy, Ozempic, Zepbound, Mounjaro, and Foundayo on April 20, 2026.
- Reviewed the 2025 Endocrinology review on GLP-1 and sex (Börchers & Skibicka).
- Reviewed the 2025 systematic review and meta-analysis on GLP-1 and testosterone.
- Reviewed the Journal of Sexual Medicine November 2025 systematic review on GLP-1 agonists and testosterone deficiency (Mahmood et al.).
- Reviewed the 2023 BMC Endocrine Disorders meta-analysis on GLP-1 and PCOS-related pregnancy rates (Zhou et al.).
- Reviewed Truveta / Olio Labs real-world EHR research on GLP-1 side-effect asymmetry (labeled as emerging).
- Reviewed the 2026 University of Pennsylvania Reddit analysis on menstrual and temperature-related side effects (Sehgal et al., arXiv preprint; hypothesis-generating).
- Reviewed the 2026 EClinicalMedicine target trial emulation on GLP-1 receptor agonists and ED risk in men with type 2 diabetes.
Proven means multiple high-quality studies or a direct regulatory label. Emerging means supported by real-world or preliminary data — directionally consistent but not yet replicated at scale.
Frequently Asked Questions
Where to Go Next
When you're ready for the next step, pick the door that matches where you are:
Not sure which GLP-1 fits your situation?
Take the Free 60-Second Quiz →Woman comparing provider options?
Best GLP-1 for Women →Man comparing provider options?
Best GLP-1 for Men →Have PCOS?
Best GLP-1 for PCOS →Perimenopausal?
Best GLP-1 for Perimenopause →Want to compare pricing?
GLP-1 Provider Comparison →