Zepbound Sleep Apnea Prior Authorization: How to Get Approved in 2026
By Weight Loss Provider Guide Team · Published April 7, 2026 · Last verified: April 7, 2026
Zepbound (tirzepatide) is the only FDA-approved medication for obstructive sleep apnea — approved December 20, 2024. That distinction matters enormously for prior authorization: when your plan covers OSA treatment, Zepbound has no formulary alternative your insurer can steer you toward. That's a stronger position than any other GLP-1 drug can claim.
But there's a catch. There is no universal prior authorization checklist. What UnitedHealthcare requires is different from CVS Caremark, Highmark, or Medicare Part D. Most denials aren't about whether you qualify — they're about whether the paperwork matched the plan's rules. This guide shows you exactly what each major insurer requires, what to put in front of your doctor, and how to fix a denial when it happens.
Bottom Line
Zepbound sleep apnea PA gets approved when the first submission matches your plan's written criteria and documents three things: moderate-to-severe obstructive sleep apnea on a sleep study (AHI ≥15), obesity (BMI ≥30), and the exact paperwork your specific plan requires. If denied, the next step is a corrected resubmission or formal appeal — and Zepbound has the strongest appeal argument of any GLP-1 because it is the only medication FDA-approved for OSA.

What Does Zepbound Sleep Apnea Prior Authorization Actually Require?
Most public policies ask for the same core proof: documented moderate-to-severe obstructive sleep apnea, documented obesity, and a complete submission that hits every checkbox your specific plan requires. In the public criteria we reviewed, the basics center on AHI/REI/RDI of at least 15 and BMI of at least 30 — but the extras range from simple to surprisingly strict.
The 7 Documents to Gather Before Your Doctor Submits Anything
- 1
Sleep study results
Polysomnography (PSG) or home sleep apnea test (HSAT) showing AHI, REI, or RDI of 15 or higher. Some plans require the study within 12 months. Attach the full report — not just the diagnosis.
- 2
BMI documentation
Current height and weight with calculated BMI of 30+. Dating requirements vary (some plans specify within 30 days, others within 90 days). Must be from a clinical visit, not self-reported.
- 3
Diagnosis codes
G47.33 (obstructive sleep apnea) as primary diagnosis, with applicable obesity codes as secondary. The framing matters — submitting under a weight-loss diagnosis instead of OSA can trigger a different (often worse) coverage evaluation.
- 4
Treatment history
Any prior or current CPAP/PAP therapy, other OSA treatments, and response to each. Include compliance data if available. Some plans require documented failure or intolerance.
- 5
Comorbidity documentation
Hypertension, type 2 diabetes, cardiovascular disease, or other obesity-related conditions strengthen the medical necessity argument. List each with ICD-10 codes.
- 6
Prior weight-management efforts
Many plans require evidence of previous diet, exercise, or medication-based weight-loss attempts — with dates and outcomes.
- 7
Letter of Medical Necessity
Not always required, but helps in borderline cases or appeals. Should cite: specific AHI score, BMI, prior treatment history, and the fact that Zepbound is the only FDA-approved medication for OSA.
What "moderate-to-severe OSA" means in payer language
What counts as "obesity" in public criteria
The most common reason for PA denial: incomplete documentation
Sources: UHC commercial Zepbound OSA PA criteria; CVS Caremark Zepbound criteria; Eli Lilly PA Resource Guide

What Are the Actual Insurer Criteria Right Now?
There is no single national checklist — that's the root of most denials. Public criteria range from relatively straightforward AHI-plus-BMI rules to forms that ask for CPAP compliance logs, specialist involvement, and annual renewal documentation. We reviewed publicly available PA criteria from major insurers and government programs.
| Plan / PBM | Covers OSA? | Key Public Criteria | Notable Extra Requirements | Renewal Rule |
|---|---|---|---|---|
| UnitedHealthcare (Commercial) | Yes — OSA-specific | Age ≥18, BMI ≥30, AHI/REI/RDI >15, documented OSA, prior dietary effort | Sleep specialist involvement; PAP symptoms/intolerance path; excludes predominant central/mixed apnea | 50% AHI/RDI/REI decrease + ≥10% weight loss after 52 weeks |
| CVS Caremark / Aetna (Commercial) | Via formulary exception (removed 2025) | Moderate-to-severe OSA, AHI ≥15 on PSG or HSAT, BMI ≥30 | Exception required. Strong argument: Zepbound is the only GLP-1 FDA-approved for OSA | Positive response in OSA symptoms for continuation |
| Highmark | Yes (strict criteria) | Baseline AHI/RDI, optimized-care data, BMI documentation | PAP compliance or failure documentation, oral appliance assessment, sleep hygiene records, current-vs-baseline metrics | Extensive renewal documentation required |
| Medicare Part D | Yes — OSA with obesity | Moderate-to-severe OSA, BMI ≥30, documented on sleep study | NOT the GLP-1 Bridge (that covers weight loss only, starting Jul 2026). Goes through Part D plan's normal utilization management. | Plan-specific |
| WV Medicaid | Yes (specific form exists) | Documented OSA, BMI, measurements within last 90 days | State-specific form with detailed fields | State-specific |
Last verified April 2026. Requirements change frequently — always call the number on your insurance card and ask: "Is Zepbound covered for obstructive sleep apnea under my specific plan? What are the prior authorization criteria, and where is the PA form?"
UnitedHealthcare Commercial
UHC has one of the more detailed public PA criteria for Zepbound OSA: age 18+, treatment for obstructive sleep apnea, BMI ≥30, sleep study with AHI/REI/RDI >15, prior dietary effort documentation, and a pathway addressing PAP symptoms or intolerance. They also exclude patients with predominant central or mixed sleep apnea. For reauthorization after the initial 52-week period, UHC requires a 50% decrease in AHI/RDI/REI plus at least 10% weight loss — one of the stricter renewal standards we've seen.
UHC's path exists — but make sure every document on their checklist is attached to the first submission.
CVS Caremark / Aetna Commercial
CVS Caremark removed Zepbound from its standard formulary in mid-2025. Under current rules, Zepbound coverage for OSA requires a formulary exception. You have a particularly strong argument here: Wegovy is not FDA-approved for sleep apnea. Zepbound is the only GLP-1 with that indication — meaning there's no true formulary alternative for OSA. Your doctor submits the exception request via fax or electronic PA with full clinical documentation.
The formulary-exception argument for OSA is stronger than for weight loss because no other GLP-1 carries the OSA indication.
Highmark
Highmark is one of the strictest examples of how demanding a PA form can get. Their public form asks for baseline AHI/RDI data, documentation of "optimized care," PAP compliance records or documented PAP failure, oral appliance assessment, sleep hygiene documentation, and current-vs-baseline metrics. If your plan is through Highmark, expect to gather significantly more documentation than average.
Source: Highmark Zepbound PA form (PDF)
Skip the paperwork maze — work with a provider who handles PA for you
Some GLP-1 telehealth programs manage the prior authorization process on your behalf, including for the OSA indication.
The Complete PA Packet Checklist
The first submission should be built like a complete packet, not a casual prescription request. Eli Lilly's own PA resource guide emphasizes providing complete and correct information to avoid denied claims. Print this, bring it to your next appointment, and go through it with your doctor before anything is submitted.
Correct diagnosis framing
The PA should explicitly state the prescription is for obstructive sleep apnea — not weight loss, not obesity management. ICD-10 code G47.33 (OSA) should be the primary diagnosis. This framing determines which coverage pathway the insurer uses.
Sleep study report (full report, not just a summary)
Attach the actual PSG or HSAT report showing the AHI, REI, or RDI score. Highlight the severity measurement. Some plans want to see the raw number, not just a physician's interpretation.
Current BMI documentation
Height, weight, and calculated BMI per your plan's recency requirement. Must be from a clinical visit, not self-reported.
Treatment history
Current and prior OSA treatments: CPAP/PAP therapy (dates, compliance data if available), oral appliances, positional therapy. Include response to each treatment.
Comorbidity documentation
List obesity-related comorbidities with ICD-10 codes: hypertension, type 2 diabetes, cardiovascular disease, hyperlipidemia. These strengthen medical necessity.
Prior weight-management efforts
Diet, exercise, or medication-based attempts with dates, programs, and outcomes.
Letter of Medical Necessity
Should cite: your specific AHI score, BMI, weight-related nature of the OSA, prior treatment history, and the fact that Zepbound is the only FDA-approved medication for obstructive sleep apnea.
Correct PA form for your specific insurer
Don't use a generic form. Some insurers — like Highmark — have Zepbound-specific OSA forms with fields that generic forms miss. Call your plan or check the provider portal.
Message to Send Your Doctor Right Now
Copy, paste, and customize:
"Hi [Doctor's Name], I'd like to request prior authorization for Zepbound (tirzepatide) for my obstructive sleep apnea. My understanding is that the PA submission should include: my sleep study results showing my AHI score, my current BMI documentation, my treatment history including CPAP/PAP therapy, my comorbidity diagnoses, and ideally a Letter of Medical Necessity. Zepbound is the only FDA-approved medication for OSA, which may help with the insurer's review. Can we review these documents at my next visit and submit the PA together? I'll call my insurance beforehand to get the exact PA form and criteria."
Sending this message before your appointment can shave weeks off the process. Most patients show up hoping the doctor "handles it" — and that's exactly how incomplete submissions happen.
The Honest Truth About Getting Zepbound Covered
Zepbound does not have one universal prior-authorization checklist. Public criteria already range from simple AHI-plus-BMI policies to forms that require CPAP compliance logs, specialist consult notes, optimized-care documentation, and baseline-vs-current metrics. And private employer group plans may have criteria that differ from anything we can publicly verify.
What we can tell you is this: the strongest submissions match the plan's written criteria exactly. The weakest submissions assume the doctor knows what to include. That gap — between assuming and verifying — is where most denials happen.
Your fastest path: call your insurer before submission, get the exact criteria, and hand your doctor a complete packet. That's not complicated, and it's dramatically more effective than the alternative.
What If Insurance Denied Zepbound as a Weight-Loss Drug?
This is one of the most underused pathways in GLP-1 coverage. Some insurers that categorically deny Zepbound for weight management do cover it when prescribed for obstructive sleep apnea — because the OSA indication is a different FDA-approved use with different coverage criteria.
If you were denied Zepbound for weight loss and you have sleep symptoms — loud snoring, daytime sleepiness, your partner notices you stop breathing at night, morning headaches — talk to your doctor about a sleep study. If your AHI comes back at 15 or higher, you may qualify through the OSA pathway entirely.
Important: this is a new PA submission, not an appeal of the weight-loss denial
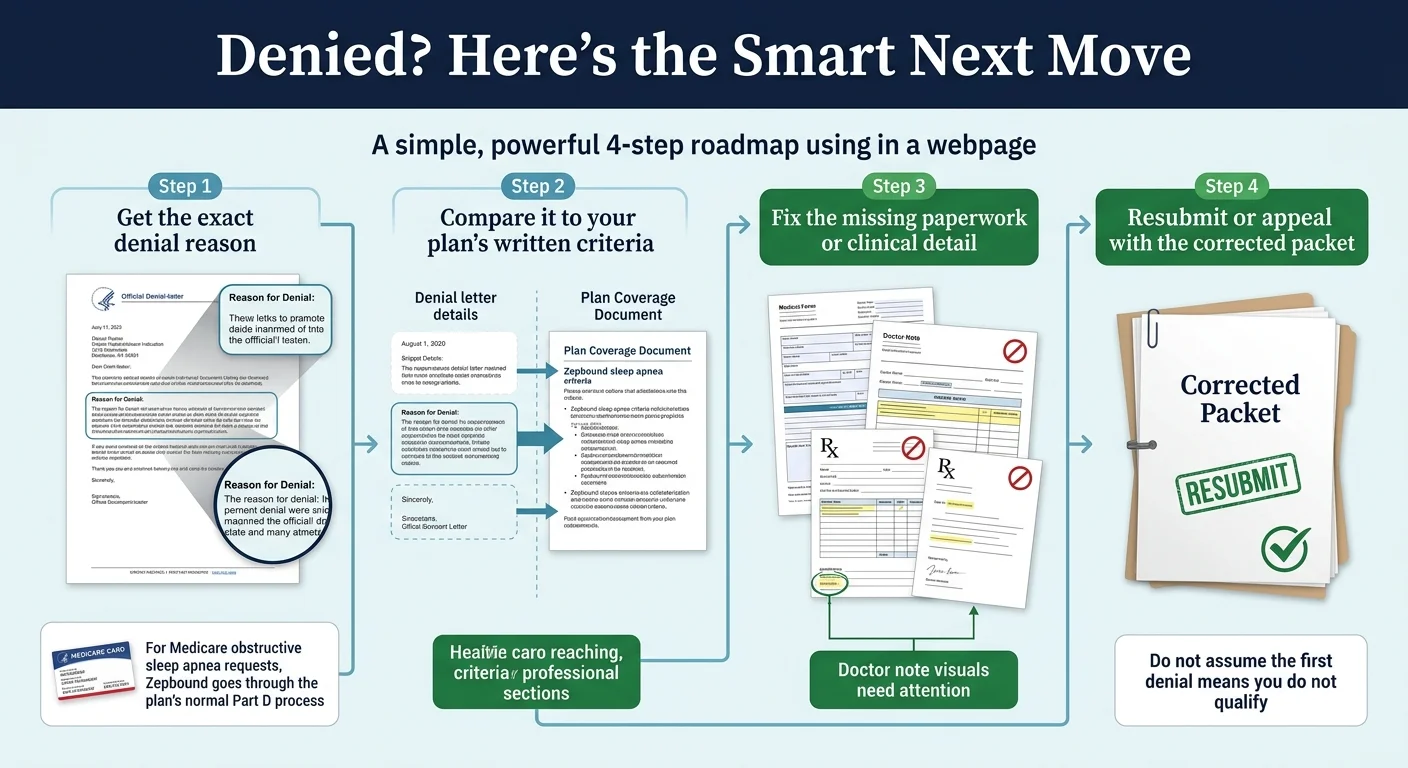
Get the exact denial reason
Pull the official denial letter — look for the specific wording, not just "denied".
Compare to written criteria
Request your plan's PA criteria document (you have the right to see it).
Fix the missing detail
Address the exact gap: wrong diagnosis framing, missing sleep study, BMI outdated, etc.
Resubmit or appeal
Corrected resubmission for paperwork issues. Formal appeal for medical necessity denials.
Check if the Zepbound OSA pathway applies to you
If you've already been denied for weight loss and suspect you have sleep apnea, a consultation with a licensed provider can clarify whether the OSA coverage route is worth pursuing.
How to Appeal a Denied Zepbound Sleep Apnea Prior Authorization
Don't give up after the first denial. Appeals work — and Zepbound's appeal argument for OSA is stronger than for any other GLP-1 because no other medication carries that FDA indication. That's not a talking point — it's a fact your appeal letter should state directly.
Get the denial letter and identify the exact reason
Look for language like "insufficient documentation," "not medically necessary," "not on formulary," or "weight loss — plan exclusion." The specific wording tells you exactly what the appeal needs to address.
Decide: new PA or formal appeal?
If the denial is due to missing documentation or incorrect diagnosis framing, a corrected PA resubmission is often faster than a formal appeal. If the denial is a medical necessity or formulary decision, file an appeal.
Build the appeal packet
- Copy of the denial letter
- Complete sleep study report with AHI/REI/RDI highlighted
- Current BMI documentation
- Updated provider notes
- Letter of Medical Necessity (cite Zepbound's unique FDA approval for OSA)
- Any additional documentation addressing the specific denial reason
Your doctor requests a peer-to-peer review (optional but powerful)
Your prescriber can request a phone conversation with the insurer's medical reviewer. This is often where borderline denials get overturned — a physician explaining clinical necessity directly to another physician.
File the internal appeal
Check your denial letter for the appeal deadline and required format. Don't wait — submit within 2 weeks while the documentation is fresh.
If internal appeal fails — external review
You have the right to an independent external review by a third party not employed by your insurer. This is separate from the internal appeal and carries significant weight.
Medicare Part D appeal path
When to request expedited review
Need help navigating a denial?
Licensed providers who specialize in GLP-1 prescriptions can manage the PA process — and the appeal — on your behalf.
Does Medicare Cover Zepbound for Sleep Apnea?
Yes. Medicare Part D can cover Zepbound when prescribed for moderate-to-severe obstructive sleep apnea in adults with obesity. This is a real, established pathway — not a loophole or workaround.
To qualify under Medicare Part D, you need:
- Enrollment in a Medicare Part D plan that lists Zepbound on its formulary (or willingness to request a formulary exception)
- Confirmed diagnosis of moderate-to-severe OSA from a sleep study
- BMI of 30 or higher
- Prior authorization from your Part D plan
Question to ask your Part D plan, word for word:
"Is Zepbound covered under my Part D plan for the treatment of moderate-to-severe obstructive sleep apnea? What are the utilization-management criteria, and where can I find the PA form?"
Use Medicare's online plan comparison tool to check whether your specific plan covers Zepbound and estimate your cost. The 2026 maximum Part D deductible is $615, and the total annual out-of-pocket cap is $2,100.
Medicare GLP-1 Bridge — why it doesn't apply to OSA
Lilly savings cards don't work with Medicare
How Much Does Zepbound Cost for Sleep Apnea?
The cost depends entirely on your coverage situation. Here's what we can verify from current official sources:
| Coverage Scenario | What You'd Pay | Source / Notes |
|---|---|---|
| Commercial insurance, PA approved, eligible for Lilly savings card | As low as $25/month (single-dose pen) | Max savings $100/fill, $1,300/year cap. Expires 12/31/2026 |
| Commercial insurance, NOT covered, eligible for savings card | As low as $499/month (single-dose pen) | Lilly savings program, uncovered tier |
| LillyDirect self-pay (KwikPen) | $299–$449/month (by dose) | LillyDirect.lilly.com. No insurance needed. 2.5 mg starts at $299 |
| Medicare Part D, PA approved for OSA | Plan-specific | $615 max 2026 deductible; $2,100 annual OOP cap applies |
| Government-insured (Medicare, Medicaid, Tricare) | Savings card NOT available | Use Part D/Medicaid coverage pathway instead |
Sources: Zepbound savings page; LillyDirect. Pricing verified April 2026. Subject to change.
What If Insurance Won't Cover It at All?
If your plan categorically excludes Zepbound, your appeal fails, and you need treatment now, LillyDirect offers brand-name Zepbound starting at $299/month with no insurance needed. Many patients use this to start treatment while their appeal processes, then switch to insurance once approved.
Note on compounded tirzepatide: Some telehealth platforms offer compounded tirzepatide at lower price points. Compounded tirzepatide is not FDA-approved Zepbound and is not specifically indicated for sleep apnea. If you're pursuing the PA route for OSA, you need the brand-name product.
Real patient experience: One user described getting their PA approved after their doctor submitted sleep study results showing moderate OSA. Coverage started the following month at $25 per fill with the savings card. The key was making sure the full sleep study report — not just the diagnosis code — was attached to the submission. — Anecdotal patient report (r/Zepbound). Not typical or guaranteed.
Start treatment while your PA processes
Telehealth GLP-1 programs can get you started on a physician-supervised weight-loss program — and some specialize in navigating the insurance process for you.
Can Zepbound Replace Your CPAP?
For some people, eventually — but not necessarily right away, and not for everyone. In the two 52-week SURMOUNT-OSA clinical trials, 42.2% and 50.2% of patients on Zepbound achieved disease remission or only mild, non-symptomatic OSA. Those are meaningful numbers.

Zepbound + CPAP (combination)
- ✓ Both approaches studied in SURMOUNT-OSA trials
- ✓ Some plans view continued CPAP favorably in PA evaluation
- ✓ Continuing CPAP while on Zepbound is a reasonable path
- ✓ Follow-up sleep study assesses whether CPAP can be reduced
Zepbound without PAP
- ✓ FDA approval doesn't require concurrent CPAP use
- ✓ Some patients unable or unwilling to use PAP therapy
- ⚠ For severe OSA, Zepbound alone may not be sufficient
- ⚠ Do not stop CPAP on your own — get a follow-up sleep study first
Do not stop CPAP without a follow-up sleep study
What If Your Sleep Study Shows RDI but Not AHI?
This is a real edge case that catches more people than you'd expect. Some sleep studies report RDI (respiratory disturbance index) or REI (respiratory event index) prominently, while the AHI number is lower or calculated differently. If your RDI is 15+ but your AHI is under 15, you may run into trouble — because some payer criteria name AHI specifically.
The good news: some public criteria explicitly accept AHI, REI, or RDI at the ≥15 threshold. UnitedHealthcare's public policy, for example, references AHI/REI/RDI. Others are narrower.
What to do if you're in this situation:
- 1.Check your plan's written PA criteria to see which metrics they accept.
- 2.If they only list AHI and yours is borderline, ask your sleep specialist whether the study can be re-scored or whether a new study with different scoring criteria would produce a qualifying AHI.
- 3.If the metric issue caused a denial, your appeal should explicitly cite which measurement your plan accepts, provide the qualifying score, and reference any plan language that supports alternative metrics.
"Their insurer only looked at my AHI and denied the PA despite years of CPAP use and a strong RDI score. The appeal was also denied." — Anecdotal patient report (r/Zepbound). Not typical. This edge case is addressable with the right documentation and metric emphasis.
How Long Does Zepbound Prior Authorization Take?
The fastest path to approval is a complete first submission. Every missing document adds days or weeks to the process.
Medicare Part D (federally mandated)
- Standard coverage determination72 hours
- Expedited determination24 hours
- Redetermination (appeal)7 calendar days
- Expedited appeal72 hours
Source: Medicare appeals process (CMS)
Commercial plans
Commercial plans set their own timelines. Check your plan's PA instructions and denial letter for specific deadlines.
- • Complete submissions are processed faster
- • Every missing document adds days or weeks
- • Expedited available for urgent medical need
How Quickly Can Zepbound Improve Sleep Apnea?
The SURMOUNT-OSA trials measured outcomes at 52 weeks. Participants who received Zepbound (10 or 15 mg weekly) experienced statistically significant and clinically meaningful reductions in breathing interruptions compared to placebo. Individual timelines vary.
This matters for PA renewals — plans have different continuation criteria:
- • CVS Caremark: Positive response evidenced by decreased OSA symptoms
- • Virginia Medicaid: Documentation of improvement in OSA symptoms
- • UHC: 50% decrease in AHI/RDI/REI plus at least 10% weight loss (after 52 weeks)
Start documenting your weight and any changes in sleep quality from day one. This data becomes your renewal evidence.
How We Verified This Page
This guide was built from publicly available payer criteria documents, CMS guidance, Eli Lilly's official PA resource materials, FDA approval documents, clinical trial data, and patient reports. What "public criteria" means: the insurer requirements on this page come from publicly available policy documents and PA forms. Your exact employer-group plan terms may differ — always verify with your specific plan before submitting.
Sources reviewed:
- UHC commercial Zepbound OSA PA criteria (PDF)
- CVS Caremark Zepbound criteria (PDF)
- Highmark Zepbound PA form (PDF)
- WV Medicaid Zepbound OSA form
- CMS Medicare GLP-1 Bridge guidance
- FDA OSA approval announcement (Dec. 20, 2024)
- Eli Lilly PA Resource Guide
- Zepbound prescribing information (FDA)
- SURMOUNT-OSA trial (NEJM)
Not a guarantee: We don't guarantee any PA outcome. Individual plan criteria, coverage decisions, and eligibility determinations are unique to each patient and plan.
How we make money: This page includes links to providers we may earn a commission from if you sign up. This doesn't affect our editorial process.
Not medical advice: This page is for informational purposes only. Consult your healthcare provider and insurer for decisions about your specific situation.
Last verified: April 7, 2026. We review and update this guide monthly as insurer requirements change.
Frequently Asked Questions
Ready to start your path to treatment?
Whether you're navigating prior authorization or looking for a physician-supervised GLP-1 program without the insurance delays, licensed providers are available to help you start today.