Best GLP-1 for Sleep Apnea: Which Medication Actually Works?
Disclosure: Some links on this site are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you. Thank you for supporting our site.·For informational purposes only—not medical advice.
Sources: FDA Prescribing Information (Zepbound), SURMOUNT-OSA Trial (NEJM 2024), SCALE Sleep Apnea Trial, CMS Medicare Part D Guidelines · Jump to Sources ↓
If you landed here, you're probably exhausted — literally — and wondering whether a GLP-1 can do something about your sleep apnea that your CPAP hasn't fully solved (or that you'd rather not strap to your face every night). We get it.
Here's the short answer: The best GLP-1 for sleep apnea is Zepbound (tirzepatide). It's the first and only medication the FDA has ever approved specifically for obstructive sleep apnea. In two 52-week clinical trials, it reduced breathing interruptions by 25.3 and 29.3 events per hour (versus 5.3 and 5.5 on placebo), and 42–50% of participants improved to remission or mild, non-symptomatic OSA. Any decision to reduce or stop PAP therapy should be based on follow-up testing with your clinician.
Wegovy and Ozempic may help indirectly through weight loss, but neither carries an FDA approval for OSA. And no — a GLP-1 is not automatic permission to toss your CPAP machine. For some people it could eventually mean less dependence on it. For others, CPAP will still be part of the picture. The honest answer depends on your specific situation.
What makes this guide different from everything else out there: We're not going to give you a 2,000-word pharmacology lesson before telling you what to do. Below, you'll find a clear comparison table for GLP-1s and sleep apnea, a breakdown by your actual situation, the real insurance numbers, and a clear next step — whether that's talking to your doctor, checking insurance, or exploring telehealth options.

Not sure which GLP-1 path fits your situation? Our free eligibility quiz matches you with the right program based on your diagnosis, insurance, and budget.
Take the 60-Second QuizIn This Guide
How Do GLP-1 Medications Compare for Sleep Apnea?
Not all GLP-1s are equal when it comes to sleep apnea. Some have large-scale clinical evidence for OSA specifically. Others may help through weight loss alone. And one — only one — has an FDA approval for this condition.
We built this comparison table so you can see the differences at a glance. Every cell is based on FDA labeling, published clinical trial data, or verified pricing. No guesswork.

| Zepbound (tirzepatide) | Wegovy (semaglutide) | Ozempic (semaglutide) | Mounjaro (tirzepatide) | Saxenda (liraglutide) | |
|---|---|---|---|---|---|
| FDA Approved for OSA? | ✅ Yes — Dec 2024 | ❌ No | ❌ No | ❌ No (diabetes label) | ❌ No |
| OSA-Specific Clinical Trial | SURMOUNT-OSA: 469 patients, 52 weeks, published in NEJM | None | None | Same molecule as Zepbound, different label | SCALE Sleep Apnea: 359 patients, 32 weeks |
| AHI Reduction | −25.3 and −29.3 events/hr (placebo: −5.3 and −5.5) | No direct OSA data | No direct OSA data | Same molecule as Zepbound | ~6 more events/hr vs placebo |
| Disease Resolution Rate | 42.2% (Study 1) and 50.2% (Study 2) | Not studied for OSA | Not studied for OSA | Same molecule as Zepbound | Not reported |
| Avg. Weight Loss | 18–20% body weight | ~15% body weight | Labeled for diabetes/CV/kidney — not obesity | 18–20% body weight | ~8% body weight |
| Mechanism | Dual GIP + GLP-1 agonist | Single GLP-1 agonist | Single GLP-1 agonist | Dual GIP + GLP-1 agonist | Single GLP-1 agonist |
| Insurance for OSA | Growing coverage; Medicare Part D eligible for OSA | Not covered for OSA | Covered for diabetes, CV, kidney — not OSA | Covered for diabetes only | Rarely covered |
| List Price | ~$1,060/mo | ~$1,350/mo | ~$935/mo | ~$1,060/mo | ~$1,350/mo |
| Best For | The strongest OSA evidence available | Broader weight/CV goals; not OSA-specific | Already prescribed for type 2 diabetes | Already prescribed for type 2 diabetes | Outdated; weaker data |
| Our Take | The clear #1 for sleep apnea | Good medication, wrong indication for OSA | Talk to your doctor about OSA evidence | Same drug, different label — ask your doctor | Not recommended when better options exist |
Table based on FDA-approved prescribing information, SURMOUNT-OSA trial (Malhotra et al., NEJM 2024), SCALE Sleep Apnea trial (Blackman et al., 2016), and manufacturer pricing as of March 2026. Last verified: March 2026.
The takeaway: Zepbound stands alone with both an FDA approval and large-scale phase 3 evidence for obstructive sleep apnea. If sleep apnea treatment is your primary goal, it's the most supported answer right now.
Which GLP-1 Is Best for YOUR Sleep Apnea Situation?
This is where most guides fall short. They tell you what the "best" drug is, but not what the best drug is for you. Your insurance status, current medications, and diagnosis all change the answer.
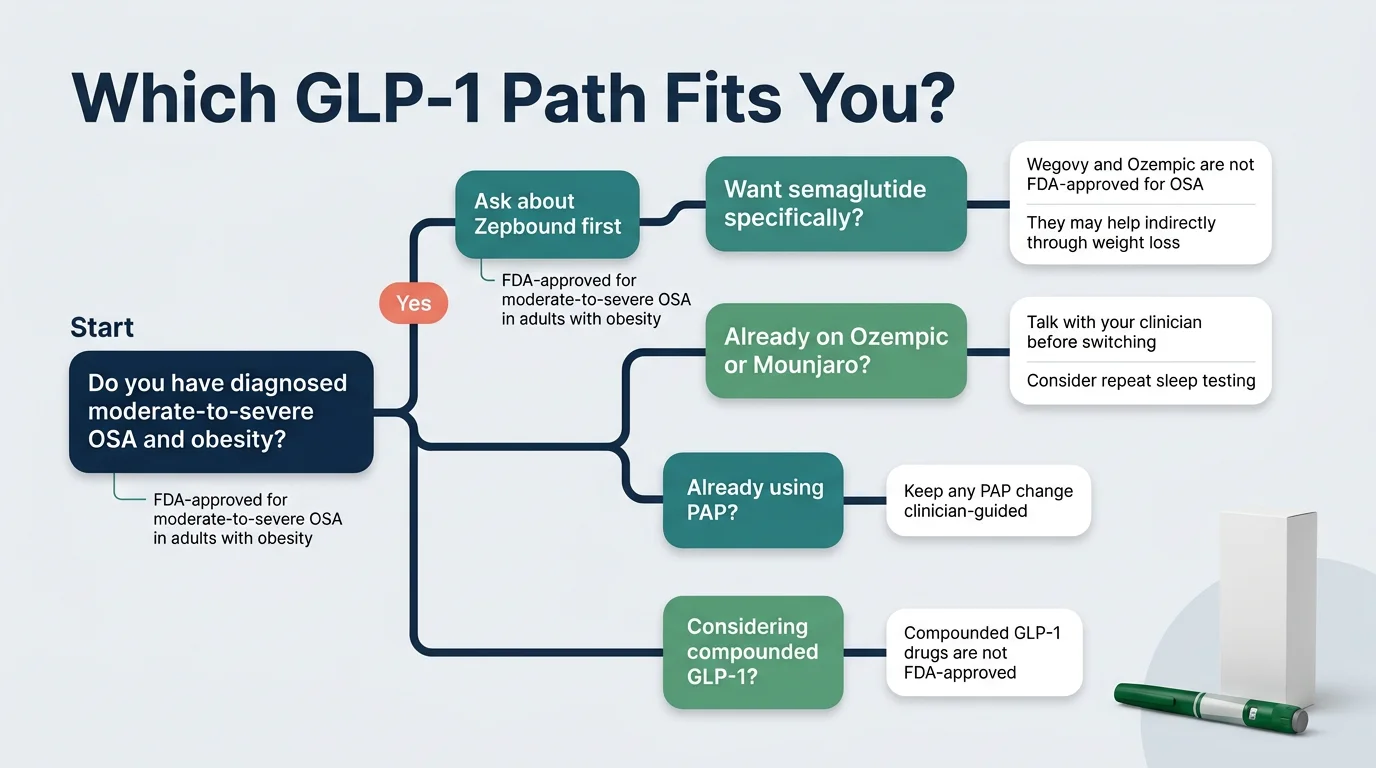
If you want the strongest evidence for sleep apnea specifically
Start with Zepbound. It's the only GLP-1 backed by a dedicated phase 3 OSA trial and an FDA approval for this use. Ask your doctor to prescribe it specifically for obstructive sleep apnea (ICD-10 code G47.33) — this matters more than you think for insurance purposes, which we'll cover below.
If you specifically want semaglutide
Wegovy is the weight-management brand; Ozempic is the diabetes brand. Neither is FDA-approved for OSA. Semaglutide may improve your sleep apnea through weight loss — roughly 15% average body weight reduction — but there's no dedicated OSA trial behind it. If semaglutide is your preference, know that you're relying on indirect benefit, not the OSA-specific data that Zepbound has. As of March 2026, Hims & Hers now offers FDA-approved branded Wegovy and Ozempic through their telehealth platform following a deal with Novo Nordisk.
If you already take Ozempic or Mounjaro for another condition
Don't switch without talking to your doctor. If you're on Ozempic for diabetes and also have OSA, the weight loss may already be helping. Your doctor can order an updated sleep study to see if your AHI has improved. If you're on Mounjaro (same active ingredient as Zepbound, different label), your doctor may be able to help you navigate the coverage angle — since Zepbound specifically carries the OSA approval.
If insurance is your biggest concern
Your sleep apnea diagnosis may actually unlock coverage. Medicare Part D can cover Zepbound when prescribed for moderate-to-severe OSA — through its regular formulary and prior-authorization process. Many commercial plans are more likely to approve Zepbound for an OSA diagnosis than for weight loss alone. A separate Medicare GLP-1 Bridge program launching in July 2026 will offer a $50/month copay for weight-loss uses, but Zepbound prescribed specifically for OSA goes through the standard Part D pathway. More details in our insurance section below.
If cost is the main barrier and you have no insurance
Eli Lilly's LillyDirect program offers Zepbound at cash-pay pricing starting around $299–$449/month. Telehealth platforms like MEDVi also offer doctor-guided GLP-1 programs starting at $179/month for compounded semaglutide or $279/month for compounded tirzepatide — with no contracts, month-to-month billing, and provider consultations included. If budget is the deciding factor between starting treatment now or waiting indefinitely, an affordable program that gets you on medication under medical supervision beats doing nothing. We break down the brand-name vs compounded landscape in detail below.
If you're on CPAP and wondering whether you can reduce dependence
This is a real possibility — but not a guarantee. In the SURMOUNT-OSA trials, 42.2% of patients not using PAP therapy and 50.2% of those on PAP therapy reached remission or mild, non-symptomatic OSA. That's encouraging. But don't stop PAP therapy on your own — let your sleep doctor make that call based on a follow-up sleep study.
Our eligibility quiz factors in your diagnosis, insurance, and preferences to recommend the safest path forward.
Find Your Best GLP-1 Path — Free, 60 SecondsWhy Zepbound Is the Best GLP-1 for Most People With Obesity-Related Sleep Apnea
Let's talk about why Zepbound rose to the top — and what the actual data looks like when you strip away the marketing language.
The first (and only) FDA-approved medication for OSA
On December 20, 2024, the FDA approved Zepbound for adults with moderate-to-severe obstructive sleep apnea and obesity. This was a landmark moment: before Zepbound, there had never been a pharmaceutical treatment approved specifically for OSA. The only options were mechanical — CPAP machines, oral appliances, surgery, and nerve stimulators.
Zepbound is a dual GIP and GLP-1 receptor agonist. In plain terms, it activates two gut hormone pathways (not just one, like semaglutide), which reduces appetite, slows stomach emptying, and promotes significant weight loss. For sleep apnea, that weight loss translates directly to reduced fat around the neck and throat — the tissue that collapses and blocks your airway while you sleep.
What the SURMOUNT-OSA trials actually showed
The evidence behind the approval came from two phase 3 randomized controlled trials involving 469 adults with moderate-to-severe OSA and obesity (BMI ≥30). The trials were published in The New England Journal of Medicine in 2024.
Study 1 enrolled people who were NOT using PAP (positive airway pressure) therapy.
Study 2 enrolled people who WERE using PAP therapy alongside the medication.
Both groups received either tirzepatide (10 or 15 mg once weekly) or placebo for 52 weeks. Here's what happened:
−25.3 to −29.3
events/hr AHI reduction (vs −5.3 to −5.5 placebo)
42–50%
reached remission or mild, non-symptomatic OSA
18–20%
body weight lost (avg. 45–50 lbs)
−9.5 mmHg
systolic blood pressure drop + CRP reduction
These are not small, ambiguous improvements. This is a medication that moved about half of study participants from moderate-to-severe sleep apnea to a level where mechanical treatment may no longer be necessary.
What Zepbound does NOT prove
We believe in being straight with you, because that's how you earn trust — and because you deserve the full picture before making a medical decision.
- Zepbound is not a cure for every type of sleep apnea. It's approved for obesity-related OSA specifically. If your sleep apnea is driven by jaw structure, enlarged tonsils, or neurological factors, weight loss alone may not resolve it.
- Long-term durability is still being studied. The SURMOUNT-OSA trial ran 52 weeks. We don't yet have 5- or 10-year data. In other GLP-1 studies, patients who stopped the medication regained weight — and there's reason to believe sleep apnea could return with the weight.
- It does not work overnight. Zepbound starts at 2.5 mg weekly and increases every 4 weeks. If titration proceeds on schedule, 10 mg can be reached by about week 13 and 15 mg by about week 21. Meaningful AHI improvements follow significant weight loss, which takes months.
None of these limitations change the verdict: Zepbound has the strongest evidence for sleep apnea of any medication that exists. But you should go in with realistic expectations.
Zepbound vs Wegovy for Sleep Apnea: Which One Should You Choose?
This is one of the most common questions we see, and it deserves a clean answer.
Zepbound wins the sleep apnea question. It has a dedicated FDA approval for OSA, a dedicated phase 3 trial measuring AHI reduction, and substantially larger weight loss (18–20% vs ~15%).
Wegovy can still win a broader health question. Its label covers weight management, cardiovascular risk reduction, and more recently, MASH (metabolic dysfunction-associated steatohepatitis). If your doctor has you on Wegovy for heart health and you also have OSA, you may already be getting some benefit for your sleep apnea through weight loss — even though it isn't specifically approved for it.
The mechanism matters. Zepbound activates two hormone receptors (GIP and GLP-1). Wegovy activates one (GLP-1 only). Research suggests this dual mechanism leads to greater weight loss, and for OSA, more weight loss generally translates to more airway improvement.
The insurance angle matters too. Zepbound's OSA approval creates insurance pathways that Wegovy cannot access. If your insurer declines Wegovy for "weight management," the OSA-specific approval on Zepbound gives your doctor a different avenue to pursue coverage — especially through Medicare Part D.
The quick verdict
| Scenario | Choose This |
|---|---|
| You want the strongest sleep apnea evidence | Zepbound |
| Your doctor recommends semaglutide for CV risk + you also have OSA | Wegovy (discuss OSA monitoring with your doctor) |
| You're already on Wegovy and sleeping better | Stay on it, get a follow-up sleep study |
| Insurance covers one but not the other | Work with your doctor on the covered option |
| You want FDA-approved GLP-1 access via telehealth | Hims & Hers now carries branded Wegovy (as of March 2026) |
| Sleep apnea is your #1 treatment priority | Zepbound |
How Do GLP-1 Medications Actually Help Sleep Apnea?
Understanding the "why" helps you set better expectations — and helps you have a smarter conversation with your doctor.
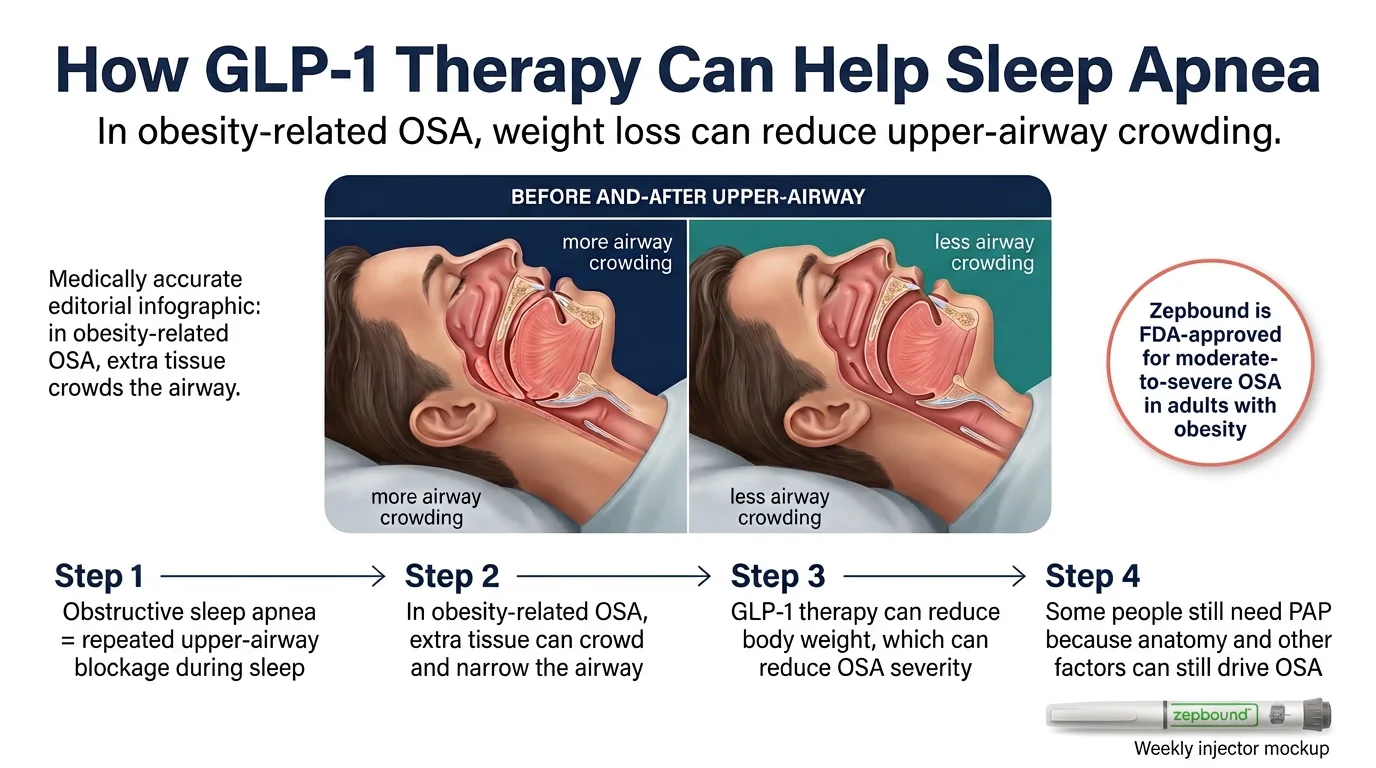
The weight-airway connection
Obstructive sleep apnea happens when the soft tissue in your throat collapses during sleep, blocking airflow. In people with obesity, excess fat deposits around the neck, tongue, and pharynx physically narrow the airway. More tissue means a narrower passage. A narrower passage means it's more likely to collapse when your muscles relax during sleep.
When Zepbound participants lost an average of 45–50 pounds, that represented a substantial reduction in pharyngeal fat. Less tissue pressing on the airway means fewer collapses per hour. Fewer collapses means a lower AHI. It's straightforward physics.
Beyond weight loss: what else GLP-1s may be doing
Reduced systemic inflammation. OSA and obesity both drive chronic inflammation. In the SURMOUNT-OSA trial, tirzepatide significantly reduced high-sensitivity CRP — a key inflammation marker. Since inflammation can contribute to airway swelling and cardiovascular risk, this reduction may have independent benefits for sleep apnea patients.
Improved cardiovascular markers. The trial showed blood pressure reductions of up to 9.5 mmHg — meaningful because OSA and hypertension frequently co-occur and amplify each other's health risks.
Potential effects on sleep regulation. Some researchers are exploring whether GLP-1 receptors in the brain may directly influence sleep-wake cycles. This research is still early, but it may partly explain why some patients report improved sleep quality before significant weight loss occurs.
Why the dual-agonist mechanism may matter for OSA
Zepbound activates two hormone receptors: GLP-1 and GIP. Wegovy and Ozempic activate only GLP-1. Research consistently shows that this dual mechanism produces more weight loss — around 18–20% of body weight with tirzepatide versus ~15% with semaglutide. For sleep apnea specifically, more weight loss generally means more airway improvement. This pharmacological difference is one reason tirzepatide produced the impressive AHI reductions seen in SURMOUNT-OSA.
What About Ozempic and Mounjaro for Sleep Apnea?
People ask about these two constantly, so let's be clear.
Ozempic (semaglutide) is FDA-approved for type 2 diabetes, cardiovascular risk reduction, and slowing chronic kidney disease progression. It is NOT approved for weight loss or sleep apnea. Some doctors prescribe it off-label, but for OSA specifically, there is no clinical trial supporting this use.
Mounjaro (tirzepatide) is FDA-approved for type 2 diabetes. It contains the same active ingredient as Zepbound. In Europe, regulators have noted that a separate sleep apnea approval isn't needed since the molecule is the same. In the U.S., only Zepbound carries the official OSA indication.
If you're already on Mounjaro for diabetes and also have sleep apnea, talk to your doctor. You're already on the same drug that proved effective for OSA. Your doctor can discuss whether switching to or adding the Zepbound label makes sense for your insurance coverage.
If you're already on Ozempic, the weight loss you're experiencing may be helping your sleep apnea, but the evidence base is weaker than tirzepatide's. Ask your doctor about monitoring your AHI and whether a switch could benefit you.
Can a GLP-1 Replace CPAP?
We're going to be direct with you here, because this is the question that matters most — and the one most websites dance around.
For some people, eventually, yes. For everyone? No.

The SURMOUNT-OSA data is genuinely encouraging. On the FDA-reviewed label, 42.2% and 50.2% of tirzepatide-treated participants reached remission or mild, non-symptomatic OSA. But PAP therapy remains the single most effective intervention for reducing AHI directly. CPAP and similar devices mechanically splint your airway open every night, while Zepbound addresses the root cause — the excess tissue compressing your airway — over months.
Why the answer depends on YOUR type of sleep apnea
Obstructive sleep apnea isn't one-size-fits-all. In many people, obesity is the dominant driver. GLP-1s can dramatically improve this type. But some people have OSA driven by:
- Jaw structure or craniofacial anatomy
- Enlarged tonsils or adenoids
- Nasal obstruction
- Neurological factors
If your sleep apnea has significant anatomical contributions beyond weight, a GLP-1 alone may not be sufficient. This is why the AASM (American Academy of Sleep Medicine) has said Zepbound is a "positive development" but should be seen as part of a comprehensive treatment plan, not a standalone solution for everyone.
The "do not stop CPAP" checklist
Before reducing or stopping CPAP therapy, make sure:
- You've been on GLP-1 treatment for at least several months with significant weight loss
- Your doctor has ordered a follow-up sleep study (polysomnography or home sleep test)
- Your AHI on the new study is below 15 events/hour (or your doctor determines CPAP is no longer needed)
- You understand that if weight returns, OSA can return with it
- You have a monitoring plan in place with your sleep medicine provider
Never stop CPAP on your own because you "feel better." Subjective improvement doesn't always match objective AHI numbers. Get tested.
How Much Weight Loss Usually Improves Sleep Apnea?
You don't need to hit your ideal body weight to start seeing improvement. Research consistently shows that even modest weight loss can meaningfully reduce sleep apnea severity — and GLP-1 medications can produce weight loss well beyond "modest."
The numbers that matter
A single-point change in BMI has been shown to significantly impact AHI. The American Academy of Sleep Medicine considers a 50% improvement in AHI as clinically relevant. In the SURMOUNT-OSA trial, tirzepatide achieved average AHI reductions of 55–63%.
- 18% body weight loss (Study 1, no CPAP): AHI dropped from ~51 to ~26 events/hour
- 20% body weight loss (Study 2, with CPAP): AHI dropped from ~50 to ~20 events/hour
- Placebo groups (lost ~2% body weight): AHI barely budged
Why some people still have OSA after losing weight
Not everyone who loses 50+ pounds resolves their sleep apnea. Anatomy varies — some people have naturally narrow airways, recessed jaws, or enlarged tongue bases. Severity matters — if you start with severe OSA (AHI of 60+), even a dramatic reduction might leave you at moderate levels. Neck circumference is a factor — people who carry more weight around their neck tend to see more improvement from weight loss.
When improvements start
Some patients report sleeping better within the first 2–3 months as the initial pounds come off. But formal AHI reduction — the kind your doctor measures with a sleep study — typically takes longer to become significant. The SURMOUNT-OSA trial measured primary outcomes at 52 weeks for good reason. Go in with optimism and patience.

“Lost 16 lbs in 10 weeks — no side effects. Down two sizes. I wish I’d started sooner.”
— Verified MEDVi patient on ConsumerAffairs ★★★★★
Who Is a Good Candidate for a GLP-1 if They Have Sleep Apnea?
Based on the FDA approval and clinical trial criteria, the strongest candidates are:
Adults with all three of these:
- Diagnosed moderate-to-severe obstructive sleep apnea (AHI ≥15 events/hour, confirmed by sleep study)
- Obesity (BMI ≥30)
- No type 1 diabetes or history of medullary thyroid carcinoma / MEN2 syndrome
You may also be a candidate if:
- You're exploring GLP-1 therapy for weight management alongside OSA
- You're on CPAP but want to potentially reduce reliance over time
- You've been unable or unwilling to use CPAP consistently
- You're already on a GLP-1 for another condition and want to know if it's helping your OSA
You are probably NOT a good candidate if:
- Your BMI is under 27
- Your sleep apnea is primarily caused by jaw structure, tonsils, or other non-weight factors
- You're pregnant, planning pregnancy within 2 months, or nursing
- You have central sleep apnea (CSA) — a different condition caused by the brain not signaling properly
Haven't been formally diagnosed yet? Start there. A sleep study (either in-lab polysomnography or an at-home sleep test) is typically required before any GLP-1 prescription for OSA. This also gives you the AHI number you'll need for insurance prior authorization.
What Dose of Zepbound Is Used for Sleep Apnea?
The dosing for sleep apnea follows the same titration schedule as for weight management:
Starting dose: 2.5 mg once weekly (subcutaneous injection) for the first 4 weeks. This is a titration dose — not a treatment dose. Its purpose is to let your body adjust and minimize GI side effects.
Dose increases: Every 4 weeks, the dose increases by 2.5 mg until you reach your maximum tolerated dose.
Maintenance dose: 10 mg or 15 mg once weekly. In the SURMOUNT-OSA trials, participants were titrated to the highest dose they could tolerate (10 or 15 mg).
Timeline to full dose: If titration proceeds without delays, 10 mg can be reached by about week 13 and 15 mg by about week 21. Your provider will adjust based on tolerability.
If side effects slow you down: Your provider can keep you at a lower dose for longer before increasing. Some patients stay at 10 mg rather than pushing to 15 mg, and still see meaningful benefit. The goal is the maximum dose you can tolerate, not necessarily the maximum dose available.
Related: Tirzepatide Dosing Schedule · Tirzepatide Side Effects Guide
GLP-1 Side Effects for Sleep Apnea Patients: What to Expect
Let's talk about the part nobody loves — but that you need to know before starting treatment.
The common stuff (and why it usually gets better)
The most frequently reported side effects in the SURMOUNT-OSA trials were gastrointestinal:
21–25%
Nausea
21–26%
Diarrhea
~17%
Vomiting
~15%
Constipation
Here's the important context: most of these were mild to moderate and improved over time. The slow titration schedule exists specifically to minimize these effects. For the vast majority of patients in the trial, the side effects were a temporary adjustment period. The improvement in sleep, energy, and breathing was lasting.
The serious warnings (know these before starting)
Zepbound carries a boxed warning about thyroid C-cell tumors observed in rodent studies. It's contraindicated in people with a personal or family history of medullary thyroid carcinoma or MEN2. Other serious but less common risks include:
- Pancreatitis
- Gallbladder problems
- Hypoglycemia (especially if combined with insulin or sulfonylureas)
- Kidney injury (usually related to dehydration from GI effects)
- Suicidal ideation (rare, but FDA monitors this across the GLP-1 class)
- Allergic reactions
When GLP-1s may not be the right fit
If you have a history of pancreatitis, severe GI disease, or the thyroid conditions mentioned above, GLP-1s may not be appropriate. Pregnancy is a hard contraindication — stop the medication at least 2 months before attempting conception.
A note for surgery and anesthesia
If you have sleep apnea and are planning any procedure requiring sedation, tell your surgeon and anesthesiologist you're on a GLP-1. These medications slow gastric emptying, which can increase aspiration risk during anesthesia. Current multi-society guidance (2024) says most patients on stable, long-term GLP-1 therapy can continue before elective procedures — with individualized planning for higher-risk patients.
For a deep dive: Tirzepatide Side Effects: What's Normal, What's Serious · Cons of GLP-1 Medications
Will Insurance Cover a GLP-1 for Sleep Apnea?
This section could save you hundreds — or even thousands — of dollars. Pay attention, because your OSA diagnosis may be the key to unlocking coverage that weight-loss-only prescriptions cannot access.
Why your sleep apnea diagnosis changes everything
Many insurance plans that deny GLP-1 coverage for "weight management" will approve it when prescribed for a different FDA-approved indication — like obstructive sleep apnea. The December 2024 FDA approval of Zepbound for OSA opened a completely separate coverage pathway.
Medicare Part D
Medicare Part D can cover Zepbound when prescribed for moderate-to-severe obstructive sleep apnea. This matters enormously because federal law prohibits Medicare from covering weight-loss drugs — but the OSA indication bypasses that exclusion.
Requirements typically include:
- Confirmed OSA diagnosis (AHI ≥15 events/hour via sleep study)
- BMI ≥30 (the FDA-approved OSA indication specifies obesity)
- Enrollment in a Medicare Part D plan
- Prior authorization with sleep study documentation
When your doctor prescribes Zepbound for moderate-to-severe OSA in adults with obesity, the claim goes through your Part D plan's regular formulary and prior-authorization process. Under the Inflation Reduction Act, annual out-of-pocket costs for Part D drugs are capped at $2,000 (2025) and $2,100 (2026).
The separate Medicare GLP-1 Bridge program (launching July 2026) offers a $50/month copay — but that program is specifically for weight-loss uses, not OSA. CMS has explicitly stated that Zepbound prescribed for moderate-to-severe OSA goes through the regular Part D route, not the bridge.
Commercial insurance (employer-sponsored plans)
- About 64% of employers with 20,000+ employees now cover GLP-1 medications
- About 43% of employers with 5,000+ employees cover them
- Many plans are more likely to approve the OSA indication than weight management alone
- Prior authorization is almost always required
The Lilly Savings Card: If you have commercial insurance that covers Zepbound, the manufacturer's savings card can bring your copay down to as low as $25/month (up to $100 max savings per 1-month fill, $1,300/year cap).
What to do if you're denied
This is critical: the vast majority of prior authorization appeals succeed. According to the AMA, over 80% of appealed prior-auth denials in Medicare Advantage were partially or fully overturned — yet only about 1 in 10 denials were ever appealed. If your first request is denied, don't give up. Have your doctor submit an appeal with:
- Your sleep study results showing moderate-to-severe OSA
- Documentation of your BMI
- A letter of medical necessity referencing the FDA-approved OSA indication
- Your ICD-10 code (G47.33 for obstructive sleep apnea)
No insurance? Your options
- LillyDirect: Brand-name Zepbound at cash-pay pricing starting around $299–$449/month
- Lilly Savings Card (cash-pay version): May bring costs down further for uninsured patients
- MEDVi: Compounded semaglutide programs start at $179/month, compounded tirzepatide at $279/month. Month-to-month billing, no long-term contracts, includes provider consultation and medication delivery. Over 100,000 patients have used the platform. HSA/FSA accepted.
- Hims & Hers: Now carries FDA-approved branded Wegovy and Ozempic following a deal with Novo Nordisk announced March 2026
- HSA/FSA: GLP-1 medications prescribed for a medical condition like OSA are typically eligible for HSA/FSA funds
Related: Can You Use HSA for GLP-1? · Cheapest Tirzepatide Online
Navigating insurance for GLP-1s can be confusing. Our quiz helps you figure out the most affordable path based on your specific coverage.
Check Your Eligibility — Free, 60 SecondsHow to Get a GLP-1 for Sleep Apnea: Step by Step
Path 1: Through your doctor (best for brand-name Zepbound with insurance)
- Get a sleep study if you haven't already. This confirms your OSA diagnosis, measures your AHI, and gives your doctor the documentation needed for insurance.
- Confirm your BMI meets the threshold (≥30 for the FDA-approved OSA indication).
- Ask your doctor to prescribe Zepbound specifically for obstructive sleep apnea — not for weight management. This distinction matters for insurance coding.
- Your doctor submits prior authorization with your sleep study results, BMI, and a letter of medical necessity.
- If approved: Use the Lilly savings card to reduce your copay.
- If denied: File an appeal. Success rates are high, and the FDA-approved OSA indication gives you strong clinical grounds.
Path 2: Through telehealth (best for faster access or cash-pay)
Several telehealth platforms connect you with licensed providers who can evaluate whether a GLP-1 is appropriate for your situation. This route is typically faster than scheduling multiple in-person visits.
MEDVi is one of the more widely used platforms in this space. The process is straightforward: you complete a health questionnaire, a licensed physician reviews your information (usually within 24–48 hours), and if approved, your medication ships directly to your door. Programs start at $179/month for compounded semaglutide or $279/month for compounded tirzepatide, with no contracts — you can cancel anytime. They also offer 24/7 provider messaging for questions about dosing or side effects.

“Lost 16 lbs in 10 weeks — no side effects. Down two sizes. I wish I’d started sooner.”
— Verified MEDVi patient on ConsumerAffairs ★★★★★
For brand-name semaglutide via telehealth: Hims & Hers now carries FDA-approved Wegovy and Ozempic following their March 2026 agreement with Novo Nordisk.
Path 3: Already on a GLP-1 for weight or diabetes
- Tell your sleep medicine doctor about your GLP-1 treatment
- Request an updated sleep study to measure whether your AHI has improved
- Let your doctor assess whether CPAP settings can be adjusted or whether you may eventually reduce reliance
- Do not stop CPAP on your own based on how you feel — get tested
Brand-Name vs Compounded GLP-1s for Sleep Apnea: What You Need to Know
If you're exploring GLP-1 options for sleep apnea, you'll encounter two categories: FDA-approved brand-name medications and compounded versions available through telehealth. Here's what the difference actually means in practical terms.

What "compounded" actually means
Compounded medications are prepared by U.S.-licensed compounding pharmacies using bulk active pharmaceutical ingredients. They are not FDA-approved as finished products — meaning the FDA has not individually evaluated the final compounded medication for safety, effectiveness, or quality. Compounding has been part of medicine for decades. In the GLP-1 space, compounding has also made these medications accessible to hundreds of thousands of people who could not afford brand-name pricing.
The landscape in 2026
Brand-name supply has stabilized. Both tirzepatide and semaglutide shortages have been resolved, which changes the regulatory context around compounding.
The FDA is paying closer attention to marketing claims. The agency has issued guidance reminding telehealth companies that compounded products should not be marketed as equivalent to brand-name medications.
Some platforms are adding brand-name options. Hims & Hers recently announced a deal with Novo Nordisk to offer branded Wegovy and Ozempic alongside their existing services.
How to choose
If you have insurance that covers Zepbound for OSA: Brand-name is the straightforward choice. You get the exact medication used in the clinical trials, with the FDA's full regulatory oversight.
If you're paying cash and brand-name pricing is out of reach: A reputable telehealth program with licensed physicians and licensed U.S. compounding pharmacies — like MEDVi — can be a practical path to medically supervised GLP-1 treatment.
What matters most, regardless of which path you take: That you're under the care of a licensed medical provider who is monitoring your progress, adjusting your dose appropriately, and coordinating with your sleep medicine team.
Related: Best Compounded Tirzepatide Programs · Is Compounded Semaglutide Safe?
What the Sleep Apnea Community Is Talking About
We spend a lot of time reading forums, patient groups, and clinical discussions to understand what real people dealing with sleep apnea are actually asking and experiencing. Here's what we're hearing consistently:
The "CPAP escape" hope is real — and sometimes warranted. Many people in sleep apnea forums express deep frustration with CPAP therapy. Mask leaks, dry mouth, claustrophobia, travel inconvenience, and partner disruption come up constantly. When the Zepbound FDA approval happened, the conversation shifted from "I hate my CPAP" to "Wait, there's actually a medication that might help?" The enthusiasm is understandable. Just make sure it's paired with realistic expectations and follow-up testing.
Insurance confusion is the #1 barrier. More than anything, people want to know: "Will my insurance cover this?" The OSA coverage pathway is genuinely new and many patients — and even some doctors — don't fully understand it yet. If you read nothing else on this page, read the insurance section above.
People on existing GLP-1s are noticing improvement. In patient discussions, we see a recurring pattern: someone starts Ozempic or Wegovy for weight loss or diabetes, then notices their CPAP machine shows fewer events, or their partner mentions the snoring has stopped. These anecdotal reports align with the mechanism — weight loss reduces airway obstruction — but they're not a substitute for formal evaluation.
The compounded vs brand-name conversation is evolving. The most important factor, based on what we see consistently, is the quality of the medical supervision behind the medication. A good telehealth program with responsive providers and clear communication makes a bigger difference than the brand-vs-compounded label.
What Should You Do Next?
You've read the evidence. You understand the options. Now here's how to move forward based on where you are today.
If you have diagnosed moderate-to-severe OSA and obesity:
Talk to your doctor about Zepbound. Bring a copy of your sleep study. Ask about insurance coverage using the OSA indication. This is the most evidence-supported path available right now.
If you're on CPAP and want to explore reducing dependence:
Start the conversation with your sleep medicine doctor. The SURMOUNT-OSA data showing 42–50% of participants reaching remission gives you a legitimate, evidence-based reason to have this conversation.
If you haven't been formally diagnosed yet:
Step one is a sleep study. Many can now be done at home. You need the AHI number — both for treatment decisions and for insurance.
If insurance is the main barrier:
Use the OSA coverage pathway. Ask your doctor to code the prescription for obstructive sleep apnea, not weight loss. Appeal any denial — success rates are high.
If you want FDA-approved access without traditional insurance:
LillyDirect offers Zepbound at reduced cash-pay rates. Hims & Hers now carries branded Wegovy and Ozempic.
Still not sure which GLP-1 program is right for you?
Take our free 60-second matching quiz. We'll recommend the safest path based on your diagnosis, insurance status, budget, and whether you want FDA-approved only.
Take the Quiz NowHow We Evaluated the Best GLP-1 for Sleep Apnea
We believe in showing our work. Here's how we ranked the medications and built this guide:
- FDA indication status was our #1 criterion. A medication with an FDA-approved indication for OSA outranks one without it, regardless of price or popularity.
- Clinical evidence quality mattered next. We prioritized randomized, placebo-controlled trials published in peer-reviewed journals (specifically the SURMOUNT-OSA trial in NEJM) over retrospective analyses or anecdotal reports.
- OSA-specific outcome measures — particularly AHI reduction and disease resolution rates — carried more weight than general weight-loss percentages.
- Insurance and access reality was evaluated based on current Medicare/Medicaid policies, employer coverage surveys (Mercer 2026), and manufacturer savings programs.
- Safety and regulatory standing of providers and medications was checked against FDA warning letters, prescribing information, and recent enforcement actions.
- All pricing and insurance information was verified against official manufacturer sites, CMS data, and plan-specific formulary documents as of March 2026.
Questions to Bring to Your Doctor About GLP-1s and Sleep Apnea
Walking into your appointment with the right questions can save you weeks of back-and-forth and help your doctor advocate more effectively with your insurance.
Before your first appointment
Write down or bring:
- Your most recent sleep study results — specifically your AHI number and severity classification
- Your current BMI
- A list of current medications including supplements
- Your insurance card and pharmacy benefit information
- Whether you've tried CPAP — how consistently you use it and whether you tolerate it well
The conversation-starter questions
"Is Zepbound appropriate for my specific type of sleep apnea?" This opens the door for your doctor to explain whether your OSA is primarily obesity-driven or has significant anatomical components.
"Can we code the prescription for obstructive sleep apnea rather than weight management?" This isn't gaming the system — Zepbound genuinely has an FDA-approved OSA indication. But the coding matters enormously for insurance, especially Medicare.
"What's your experience prescribing GLP-1s for sleep apnea patients?" Not all physicians are up to speed on the OSA indication. If your PCP isn't comfortable, ask for a referral to an obesity medicine specialist or sleep medicine physician.
"When should we retest?" Knowing the follow-up plan upfront helps you stay accountable. Most clinicians will want a repeat sleep study after 6–12 months of treatment.
"What should I do about CPAP while on this medication?" Your doctor will almost certainly tell you to keep using it — at least initially. But they can explain what benchmarks you'd need to hit before reconsidering.
Frequently Asked Questions
Which GLP-1 is FDA-approved for sleep apnea?
Zepbound (tirzepatide) is the only GLP-1 medication with an FDA-approved indication for obstructive sleep apnea. The approval, granted in December 2024, specifically covers adults with moderate-to-severe OSA and obesity. No other GLP-1 — including Wegovy, Ozempic, Mounjaro, or Saxenda — carries this approval.
Is Zepbound the same as Mounjaro?
They contain the same active ingredient (tirzepatide) but are different brands with different FDA-approved uses. Mounjaro is approved for type 2 diabetes. Zepbound is approved for weight management and obstructive sleep apnea. If you're on Mounjaro for diabetes and also have OSA, talk to your doctor about whether the Zepbound label makes sense for your situation.
Can Ozempic or Wegovy help sleep apnea?
Possibly — but indirectly. Both contain semaglutide, which promotes weight loss that can reduce airway obstruction. However, neither has an FDA approval for OSA, and there are no dedicated large-scale OSA trials for semaglutide. Tirzepatide (Zepbound) has stronger weight loss data and a dedicated OSA trial, making it the more supported choice.
Can a GLP-1 replace CPAP?
For some people, eventually. In the SURMOUNT-OSA trials, 42.2% and 50.2% of participants reached remission or mild, non-symptomatic OSA. But PAP therapy remains the most effective direct treatment for reducing breathing interruptions. Never stop PAP therapy without a follow-up sleep study and your doctor's guidance.
How long before sleep apnea improves on a GLP-1?
Meaningful improvement typically occurs over several months as weight loss accumulates. The SURMOUNT-OSA trial measured primary outcomes at 52 weeks. Some patients report better sleep within 2–3 months, but formal AHI reduction takes longer. Patience is part of the process.
Does Medicare cover Zepbound for sleep apnea?
Yes — Medicare Part D can cover Zepbound when prescribed for moderate-to-severe obstructive sleep apnea (not for weight loss). Coverage goes through your plan's regular formulary and prior-authorization process, so your actual copay depends on your specific Part D plan. A separate Medicare GLP-1 Bridge program launching July 2026 offers a $50/month copay for weight-loss uses, but Zepbound for OSA goes through the standard Part D route.
Does commercial insurance cover Zepbound for sleep apnea?
Many commercial plans do, especially for the OSA indication. About 64% of large employers (20,000+ employees) now cover GLP-1 medications. Prior authorization is almost always required. If denied, file an appeal — the AMA reports that over 80% of appealed denials are overturned.
What dose of Zepbound is used for sleep apnea?
Treatment starts at 2.5 mg once weekly and increases every 4 weeks. The maintenance dose used in the SURMOUNT-OSA trials was 10 or 15 mg once weekly, depending on tolerability. If titration proceeds on schedule, 10 mg can be reached by about week 13 and 15 mg by about week 21.
Do I need a sleep study before starting?
In most cases, yes. Insurance typically requires a sleep study confirming moderate-to-severe OSA (AHI ≥15) for Zepbound coverage. Even without insurance requirements, a sleep study gives you a baseline AHI to measure improvement against. Many can now be done at home.
What if my sleep apnea is mild?
Zepbound is specifically approved for moderate-to-severe OSA (AHI ≥15). For mild sleep apnea (AHI 5–15), lifestyle changes, positional therapy, oral appliances, and general weight loss through any method may be more appropriate first steps.
What happens if I stop taking the medication?
Weight regain is common after stopping GLP-1 medications, and sleep apnea can return with the weight. This is consistent across all GLP-1 studies. If you stop treatment, work with your doctor on a monitoring plan that includes repeat sleep studies.
Are compounded GLP-1s the same as brand-name?
They are different regulatory categories. Compounded versions are prepared by U.S.-licensed pharmacies but are not FDA-approved as finished products. Brand-name medications (like Zepbound and Wegovy) go through FDA's full approval process. If you have insurance coverage or can afford brand-name pricing, that path offers the most regulatory assurance. If cost is a barrier, a reputable telehealth program with licensed physicians and U.S.-based pharmacies can provide medically supervised access at a fraction of the cost.
Is there a pill form of GLP-1 for sleep apnea?
No GLP-1 pill is FDA-approved for sleep apnea. However, Novo Nordisk recently launched an oral Wegovy pill for weight management. It's not approved for OSA. The only FDA-approved sleep apnea treatment in this class is injectable Zepbound.
What if I already take Wegovy or Ozempic?
Don't stop your current medication without talking to your doctor. The weight loss you're experiencing may already be improving your OSA. Ask your doctor about getting an updated sleep study to check your AHI, and discuss whether switching to or adding Zepbound makes sense for your specific goals and insurance situation.
What questions should I ask my doctor?
Key questions include: "Is Zepbound appropriate for my specific type of sleep apnea?" "Can we code the prescription for obstructive sleep apnea rather than weight management?" "What's your experience prescribing GLP-1s for sleep apnea patients?" "When should we schedule a follow-up sleep study to measure improvement?" and "At what point might we discuss reducing CPAP reliance?"
Sources
- U.S. Food and Drug Administration. FDA Approves First Medication for Obstructive Sleep Apnea. Press release. December 20, 2024.
- Malhotra A, Grunstein RR, Fietze I, et al. Tirzepatide for the Treatment of Obstructive Sleep Apnea and Obesity. N Engl J Med. 2024;391(13):1193-1205. doi:10.1056/NEJMoa2404881
- Eli Lilly and Company. FDA approves Zepbound® (tirzepatide) as the first and only prescription medicine for moderate-to-severe obstructive sleep apnea in adults with obesity. Press release. December 20, 2024.
- Eli Lilly and Company. Lilly's tirzepatide reduced obstructive sleep apnea (OSA) severity, with up to 51.5% of participants meeting the criteria for disease resolution. Press release. June 21, 2024.
- American Academy of Sleep Medicine (AASM). Statement on Zepbound approval for sleep apnea. December 23, 2024.
- DailyMed. Zepbound (tirzepatide) — Prescribing Information. National Library of Medicine.
- DailyMed. Wegovy (semaglutide) — Prescribing Information. National Library of Medicine.
- Blackman A, et al. Effect of liraglutide 3.0 mg in individuals with obesity and moderate or severe obstructive sleep apnea: the SCALE Sleep Apnea randomized clinical trial. Int J Obes. 2016;40(8):1310-1319.
- Reuters. Eli Lilly's obesity drug gets Medicare coverage for sleep apnea. January 8, 2025.
- Mercer. GLP-1 considerations for 2026: Your questions answered. 2026.
- U.S. Food and Drug Administration. FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss.
- Centers for Medicare and Medicaid Services. Medicare GLP-1 Bridge FAQ. Updated March 2026.
- Hims & Hers Health. Strategic Shift for US Weight Loss Business — Novo Nordisk Agreement. March 9, 2026.
- Bing Webmaster Blog. Introducing AI Performance in Bing Webmaster Tools. February 2026.
- American Medical Association. Over 80% of prior auth appeals succeed. Why aren't there more?
- Multi-society Clinical Practice Guidance for the Safe Use of GLP-1 Receptor Agonists in the Perioperative Period. Clin Gastroenterol Hepatol. 2025;23(12):2083-2085.