GLP-1 Telehealth Safety Checklist (2026): 15 Checks Before You Start
By WPG Editorial Team · Last updated: · Last regulatory verification:
Last verified: May 6, 2026

This GLP-1 telehealth safety checklist gives you the answer up front. A GLP-1 telehealth provider passes the safety check when all four of these are true:
- 1A licensed clinician — in the state where you're located for care — actually evaluates you.
- 2A verifiable U.S. pharmacy fills your prescription.
- 3The medication is clearly identified: brand-name FDA-approved, or compounded with full disclosure.
- 4You get real follow-up care, not just a shipment.
If a provider can't confirm all four, walk away.
The FDA says that in the six months leading up to March 3, 2026, it sent thousands of letters telling pharmaceutical and telehealth firms to remove misleading ads — more than it had sent in the entire previous decade. That same day, it announced 30 formal warning letters to telehealth firms making false or misleading claims about compounded GLP-1 products. That's the climate you're shopping in.We built this page to be a working tool. By the end, you'll be able to vet any GLP-1 telehealth company in about ten minutes, know what labs to expect, know which red flags should stop you cold, know what to do if your medication arrives warm, and know exactly when to call your prescriber versus 911.
What we verified for this page:
FDA enforcement and regulatory documents, FDA prescribing information for Wegovy, Zepbound, Ozempic, Mounjaro, and Foundayo, FDA BeSafeRx and NABP pharmacy verification resources, FSMB and CCHP telehealth policy sources, and clinical references listed at the bottom. Last regulatory verification: May 6, 2026.The 60-Second GLP-1 Telehealth Safety Check
A safe GLP-1 telehealth provider passes 15 checks: a state-licensed clinician reviews you, intake screens for thyroid cancer history and pancreatitis, a named U.S. pharmacy fills the prescription, the medication identity is disclosed, dosing is in clear milligrams, follow-up is scheduled, side-effect support is real, and the platform handles your data, your records, and your money transparently.
| Safety check | ✅ Pass | ⚠️ Caution | 🛑 Stop |
|---|---|---|---|
| Prescription required | Yes, after clinician review | Questionnaire, then later review | "No prescription needed" |
| Clinician licensed in your state | Name and license shown | Credentials hidden until visit | No clinician named |
| Medical evaluation depth | Full history, meds, contraindications | Short intake, limited follow-up | Instant approval, no risk questions |
| Pharmacy named | Name and license available | Named after checkout only | Not disclosed |
| Product identity | Brand name or compounded clearly stated | Vague "GLP-1 blend" | "Generic Ozempic" or "generic Wegovy" |
| FDA status disclosure | "Compounded, not FDA-approved" if compounded | Ambiguous wording | "FDA-approved compounded GLP-1" |
| Dosing | Clear mg, titration plan, supplies, training | Confusing "units only" | No mg, no concentration |
| Lab work | Required before or shortly after starting | Recommended but not required | Never required, never reviewed |
| Follow-up | Scheduled check-ins, dose review | Chat or ticket only | Nothing after shipment |
| Shipping | Cold-chain, replacement plan | Unclear timing | Arrives warm, told to use anyway |
| Side-effect plan | Urgent vs non-urgent guidance with response window | Generic FAQ only | No medical support |
| Privacy/data policy | HIPAA notice, health-data sharing explained | Generic privacy page only | No privacy policy or unclear data use |
| Cancellation/refund terms | Subscription, refills, cancellation explained before payment | Terms visible only at checkout | Multi-month prepay with no cancellation path |
| Medical records access | Records and prescription details available on request | Records require a support ticket | Refuses records or hides prescription details |
| Primary-care coordination | Encourages PCP sharing, provides a visit summary | Patient must request summary | Discourages PCP involvement |
Why This Checklist Matters Right Now (2026)
GLP-1 telehealth is under heavy FDA scrutiny in 2026. The FDA announced 30 warning letters to telehealth firms on March 3, 2026 — the second formal wave following an earlier round tied to its September 2025 enforcement crackdown. The National Association of Boards of Pharmacy (NABP) — the body that represents state pharmacy regulators — estimates only about 5% of online pharmacies meet patient safety standards. The shortage that opened the door to widespread compounded GLP-1 prescribing has ended, which means the rules changed.
Three things shifted in the last 18 months:
1. The shortage was officially resolved.
The FDA determined the tirzepatide injection shortage was resolved on December 19, 2024, and the semaglutide injection shortage on February 21, 2025. The shortage-based enforcement-discretion windows tied to those determinations have since closed. Lawful individualized compounding still has to meet the conditions for 503A or 503B compounding — and the bar got higher.
2. Enforcement got real.
Between September 2025 and March 2026, the FDA sent letters to dozens of telehealth firms — Bliv Wellness, Belle Health, FitRX, BluefitMD, Viv Health, 24HrDoc, GoodGirlRx, and others. The cited violations: claims that compounded products are "the same as" FDA-approved drugs, and brand names that hide who actually compounds the medication.
3. The FDA proposed shutting down 503B GLP-1 compounding.
On April 30, 2026, the FDA proposed excluding semaglutide, tirzepatide, and liraglutide from the 503B bulks list, after finding no clinical need for outsourcing facilities to compound them from bulk substances. The agency is taking comments through June 29, 2026 before making a final determination. If finalized, this further narrows the legal lane for compounded GLP-1.
Regulatory timeline at a glance
| Date | What changed | Why it matters to patients |
|---|---|---|
| Dec. 19, 2024 | FDA determined tirzepatide injection shortage resolved | Shortage exception for compounded tirzepatide started winding down |
| Feb. 21, 2025 | FDA determined semaglutide injection shortage resolved | Same — for compounded semaglutide |
| Sep. 2025 | FDA's first telehealth crackdown wave | Misleading direct-to-consumer GLP-1 ads put on notice |
| Feb. 6, 2026 | FDA announced restrictions on bulk GLP-1 APIs used in non-approved compounding | Hims & Hers named specifically; pulled compounded oral semaglutide product days later |
| Mar. 3, 2026 | FDA announced 30 warning letters to telehealth firms | Specific firms cited for "sameness" claims and brand-name obfuscation |
| Apr. 1, 2026 | FDA clarified 503B status for semaglutide and tirzepatide | Both confirmed off the 503B bulks list and shortage list |
| Apr. 30, 2026 | FDA proposed excluding semaglutide, tirzepatide, liraglutide from 503B bulks list | Comment period through June 29, 2026 — could further restrict 503B compounding |
The 4-Layer Safety Model
Safe GLP-1 telehealth has four required layers — a real clinician, a verifiable pharmacy, a clearly identified medication, and ongoing follow-up. If one layer fails, the entire system fails. Most patient harm we've seen in 2024–2026 traced back to a single missing layer the patient didn't know to check.
Think of it as a chain. A great prescriber paired with an unknown pharmacy is unsafe. A reputable pharmacy with no follow-up is unsafe. We've watched providers excel at one or two layers and quietly skip the others.
Layer 1: Clinician
Licensed where you're located for care, performs a real evaluation
Layer 2: Pharmacy
Named, U.S.-licensed, verifiable
Layer 3: Medication
Clearly identified, properly sourced, lawfully compounded if compounded
Layer 4: Follow-up
Scheduled, with a real path to a clinician for side effects and dose decisions
Layer 1: Verify the Clinician
A safe GLP-1 telehealth clinician is named, credentialed (MD, DO, NP, or PA), and licensed or otherwise authorized in the state where you're located when you receive care. You can verify them in about two minutes using state medical board databases and the National Provider Identifier (NPI) Registry. If the platform won't tell you who is signing your prescription, treat that as disqualifying.
The clinician must be licensed where you receive care
Telehealth medicine is generally treated as practiced where the patient is located, not where the clinician is sitting. The Federation of State Medical Boards (FSMB) — the organization that represents the U.S. medical and osteopathic boards — has a model telemedicine policy stating that physicians prescribing through online services need a license in the patient's jurisdiction. State rules vary; the Center for Connected Health Policy (CCHP) tracks them.
This is one of the quietest failures. A provider may be licensed in 15 states but not yours, and the platform sometimes won't tell you up front.
A questionnaire is not a medical evaluation
CCHP reports that most state medical boards do not consider an online questionnaire alone enough to establish the patient-clinician relationship needed to prescribe. A real evaluation should cover:
- Height, weight, BMI, weight history
- Current prescriptions, OTC drugs, and supplements
- Allergies and prior drug reactions
- Diabetes history and current diabetes medications
- Personal or family thyroid cancer or MEN2 history
- Pancreatitis or gallbladder history
- Kidney disease and dehydration risk
- Severe GI disease, gastroparesis, or reflux history
- Pregnancy, breastfeeding, or pregnancy plans
- Mental health and eating disorder history
- Prior weight-loss medication use
- A defined follow-up plan
If your intake skipped half of these, your prescription was written without information the FDA prescribing label assumes the prescriber has.
How to verify a clinician in three steps
If the clinician can't be matched on the state board, ask the platform for clarification in writing before paying.
Five questions to ask before paying
- Who reviews my intake?
- Are they licensed where I'm located?
- Is my visit live video, phone, or asynchronous review only?
- Who handles side effects after I start?
- Who decides when my dose changes?
A provider that can't answer all five in writing isn't ready to be your prescriber.
Layer 2: Verify the Pharmacy
The pharmacy filling your prescription should be named, U.S.-licensed, and verifiable through your state board of pharmacy. The FDA's BeSafeRx tool links to every state's pharmacy database. Foreign-shipping pharmacies and pharmacies that won't disclose their name are red flags.
The pharmacy name should be known before you pay
This is non-negotiable. A safe provider tells you who fills the prescription. Some platforms only reveal it at checkout. Others never reveal it. Both are warning signs.
How to verify a pharmacy in three steps
Cross-check with NABP
The NABP Safe Site Search at safe.pharmacy lets you check whether a pharmacy website is accredited or "not recommended." NABP has identified more than 40,000 websites that fail to meet patient safety or pharmacy practice standards. Your prescription should not be coming from one of them.
Pharmacy red flags
- No pharmacy name disclosed
- Foreign pharmacy shipping prescription drugs to U.S. patients
- No way to contact a pharmacist
- No state license
- Label has spelling errors or no expiration date
- Medication arrives warm and the pharmacy says use it anyway
- Pharmacy address is a P.O. box only
503A vs 503B compounding pharmacies
If your medication is compounded — meaning a pharmacy mixed it for you instead of receiving it pre-made from a drug manufacturer — the pharmacy will be one of two types:
| Type | What it is | Regulated by |
|---|---|---|
| 503A pharmacy | Traditional compounder — makes medications for individual patients with a specific prescription | State-licensed, state-regulated |
| 503B pharmacy | FDA-registered "outsourcing facility" — operates under a different federal framework including CGMP standards | FDA-registered + state oversight |
Layer 3: Verify the Medication (FDA-Approved vs Compounded)
FDA-approved GLP-1 or GIP/GLP-1 products that show up in weight-loss telehealth include Wegovy injection and tablets, Zepbound, Saxenda, and Foundayo for chronic weight management, plus Ozempic, Mounjaro, and Rybelsus for type 2 diabetes (sometimes prescribed off-label for weight). Anything else marketed as "generic Ozempic," "generic Wegovy," or "FDA-approved compounded semaglutide" is misleading. Compounded GLP-1 medications are not FDA-approved finished products and are not generics — even when the active ingredient name matches.
Medication status matrix
| Product | FDA-approved for weight management | FDA-approved for diabetes | Compounded? | Generic available? |
|---|---|---|---|---|
| Wegovy injection | Yes (BMI ≥30, or ≥27 with comorbidity; pediatric 12+) | No | No | No |
| Wegovy tablets | Yes (adults) | No | No | No |
| Zepbound | Yes (adults) | No | No | No |
| Saxenda | Yes | No | No | No |
| Foundayo (orforglipron) | Yes (adults) | No | No | No |
| Ozempic | No | Yes | No | No |
| Mounjaro | No | Yes | No | No |
| Rybelsus | No | Yes | No | No |
| Compounded semaglutide | No (not FDA-approved) | No (not FDA-approved) | Yes | No FDA-approved generic exists |
| Compounded tirzepatide | No (not FDA-approved) | No (not FDA-approved) | Yes | No FDA-approved generic exists |
| Semaglutide sodium / acetate | Not lawful for compounding per FDA | Not lawful for compounding per FDA | Should not be used | N/A |
| Retatrutide / cagrilintide | Cannot lawfully be compounded per FDA | Cannot lawfully be compounded per FDA | Should not be used | N/A |
| "Research use only" GLP-1 | Not for human use | Not for human use | Diverted product | N/A |
Know exactly what is in your vial or pen
Brand-name FDA-approved products arrive in original manufacturer packaging. Wegovy and Ozempic and Rybelsus come from Novo Nordisk. Zepbound, Mounjaro, and Foundayo come from Eli Lilly. If your medication does not arrive in original branded packaging, it's compounded. That is not automatically dangerous. It's automatically a different regulatory category.
What "compounded" actually means
A compounded drug is mixed by a pharmacy from raw ingredients (called active pharmaceutical ingredients, or APIs). The FDA does not review compounded drugs for safety, effectiveness, or quality before they reach patients. The FDA's March 2026 announcement is direct: compounded drugs are not FDA-approved, and they are not the same as generic drugs.
No generic exists.
A "generic Ozempic" does not legally exist in the United States. There is no FDA-approved generic semaglutide. If a telehealth company calls their compounded product "generic Ozempic," they are using a phrase the FDA has cited as misleading.When compounded GLP-1 is still legitimate
Compounding can be appropriate when the prescriber documents that the compounded product offers a clinically meaningful difference for that specific patient that an FDA-approved product can't provide — for example:
- Documented allergy to an inactive ingredient in the FDA-approved product
- A specific dose or formulation that's not commercially available and is clinically needed
- A customized formulation a manufacturer doesn't make
It is generally not appropriate to compound a copy of a commercially available FDA-approved drug just because it's cheaper. With the GLP-1 shortage resolved, the lane for routine compounded GLP-1 narrowed substantially.
Compounded GLP-1 red flags that should stop you
- Semaglutide sodium or semaglutide acetate. These are salt forms. The FDA has stated it is unaware of any lawful basis for using semaglutide salt forms in compounding. The active ingredient should be semaglutide base.
- Retatrutide or cagrilintide offered as compounded weight-loss treatment. The FDA has stated these substances cannot lawfully be used in compounding and have not been found safe and effective for any condition.
- "Research use only" or "not for human consumption" labels. These are products diverted from research supply chains and were never intended to be injected into people.
- Vague "GLP-1 blend" with no mg or concentration disclosed.
- No certificate of analysis available for the batch.
- Telehealth firm's brand name on the drug instead of the actual molecule. The FDA cited this in the March 2026 letters as misleading.
Questions to ask before accepting a compounded prescription
- Is this compounded semaglutide or compounded tirzepatide?
- Why am I being prescribed a compounded version instead of an FDA-approved one?
- Is the pharmacy a 503A or a 503B?
- What is the active pharmaceutical ingredient — semaglutide base, sodium, or acetate?
- Does the pharmacy provide a certificate of analysis?
- Has the pharmacy had recent state board inspection issues or disciplinary actions?
- What is the concentration of my vial?
- How is my dose measured (mg, mL, units)?
- What is the beyond-use date?
- What happens if my vial arrives warm?
Layer 4: Verify the Follow-Up Plan
Safe GLP-1 telehealth includes scheduled follow-up at month 1, month 3, and month 6 minimum, with a defined path to a clinician for side effects and dose decisions. "Ship and disappear" — where you get a prescription and never hear from a clinician again — is unsafe and out of step with current obesity medicine practice.
What ongoing care should look like
Per the StatPearls obesity medication management guidance and the AMA's GLP-1 monitoring framework, ongoing care should include:
- Weight and BMI trend
- Blood pressure and heart rate
- A1C or glucose if relevant
- Kidney function if relevant
- Side-effect tracking
- Nutrition adequacy and protein intake
- Muscle mass concerns
- Dose tolerance and adjustments
- Mental health and eating-behavior screening
- Adjustment of any diabetes medications
A program that doesn't address most of these is not following clinical best practice.
Five questions to ask about follow-up
- How often do I see a clinician after starting?
- Who decides when I move up in dose?
- What symptoms make you pause my titration?
- How do I reach you outside of business hours?
- What's the plan if I want to stop?
12 Red Flags That Should Stop You Cold
A GLP-1 telehealth provider should be rejected if they advertise "no prescription needed," sell a compounded product as "the same as" Wegovy or Ozempic, hide their pharmacy, sell semaglutide sodium or acetate, ship without cold packs, or guarantee specific weight-loss numbers. Several of these track directly to FDA enforcement language from 2025–2026 warning letters; the rest are operational checks that should make you investigate before paying.
- 1
"No prescription needed." GLP-1 medications are prescription-only in the U.S. There are no exceptions.
- 2
Very low advertised pricing with no breakdown of what's included. Real GLP-1 programs include clinician time, medication, pharmacy dispensing, supplies, shipping, dose changes, and follow-up — and the math has to add up somewhere. Vague or unitemized pricing is the red flag, not the dollar amount itself.
- 3
Questionnaire only, no clinician interaction at any point.
- 4
Intake doesn't ask about MTC, MEN2, pancreatitis, or pregnancy. These are FDA-labeled contraindications and warnings. Skipping them violates the prescribing label.
- 5
Pharmacy source is unnamed or "our partner pharmacy."
- 6
Drug sold under the telehealth firm's own brand name instead of the actual molecule. The FDA cited this in the March 2026 letters as misleading.
- 7
Site claims compounded version is "the same as" Wegovy, Ozempic, or Zepbound. This is the exact language the FDA cited.
- 8
Specific weight-loss guarantees ("lose 30 pounds in 30 days").
- 9
No physical business address listed.
- 10
No clinician credentials or NPI numbers visible.
- 11
No defined response window for severe side effects.
- 12
Multi-month prepayment required with no cancellation option.
Are You Personally a Candidate? The Patient Self-Screen
GLP-1 medications are contraindicated for people with personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN2), and for prior serious hypersensitivity to that medication's active ingredient or excipients. Strong precautions apply for prior pancreatitis, severe gastroparesis, gallbladder disease, severe kidney disease, and pregnancy. A safe telehealth provider screens for all of these in intake — not after prescribing.
Vetting the provider is half the work. The other half is checking yourself.
FDA-labeled contraindications and pregnancy stop points
These are pulled from the current FDA prescribing labels for Wegovy, Zepbound, Ozempic, Mounjaro, and Foundayo:
- Personal or family history of medullary thyroid carcinoma (MTC) — a rare type of thyroid cancer
- Multiple Endocrine Neoplasia syndrome type 2 (MEN2) — a rare genetic condition linked to MTC
- Prior serious hypersensitivity reaction to the medication's active ingredient or excipients
- Pregnancy (GLP-1 medications may cause fetal harm; Wegovy label says discontinue at least 2 months before a planned pregnancy)
FDA-labeled precautions (not automatic disqualifiers — discuss with your prescriber)
- Personal history of pancreatitis
- Active or history of gallbladder disease
- Severe gastroparesis or severe GI disease
- Severe kidney disease
- Use of insulin or a sulfonylurea (raises hypoglycemia risk)
- Use of oral contraceptives (tirzepatide and Foundayo require backup contraception)
- Planned surgery or procedures requiring anesthesia
- Eating disorder history
- Adolescent patients 12–17 (Wegovy injection only has established pediatric evidence)
Side Effect Triage: Common, Call, or 911?
Common GLP-1 side effects include nausea, vomiting, diarrhea, constipation, and reduced appetite, and they're most common when starting and after a dose increase. Call your prescriber for persistent vomiting, severe constipation, or symptoms that worsen after a dose change. Seek emergency care for severe abdominal pain (especially radiating to the back), allergic reaction symptoms, severe dehydration, or fainting.
Side effects are most common when starting and after a dose increase, and typically improve as your body adjusts. Knowing what's normal versus what isn't is the difference between toughing it out and ending up in an ER.
Common (manage at home)
- · Mild to moderate nausea
- · Occasional vomiting
- · Diarrhea or constipation
- · Reduced appetite
- · Mild headache
- · Fatigue
- · Mild injection-site irritation
Call your prescriber
- · Vomiting that won't stop or prevents keeping fluids down
- · Severe constipation lasting more than 3 days
- · Worsening reflux or upper abdominal discomfort
- · Significant dizziness or weakness
- · Low blood sugar symptoms (if on insulin or sulfonylurea)
- · Side effects that worsen after a dose increase
- · Mood or appetite changes that concern you
911 / Emergency room
- · Severe persistent abdominal pain, especially radiating to the back (possible pancreatitis)
- · Severe right-upper-quadrant pain, fever, yellowing skin (possible gallbladder)
- · Trouble breathing, swelling of face/throat, hives (possible allergic reaction)
- · Repeated vomiting with signs of severe dehydration
- · Confusion, fainting, or severe low blood sugar
- · Chest pain
- · Sudden vision changes
Surgery and anesthesia note:
Before any scheduled surgery or procedure with anesthesia or deep sedation, tell the surgical and anesthesia team you take a GLP-1. The Wegovy and Zepbound labels include pulmonary aspiration warnings because the medication slows stomach emptying. This needs to be on the surgical team's radar in advance.Special Situations That Need Extra Caution
Pregnancy planning, surgery, oral birth control with tirzepatide or Foundayo, type 2 diabetes with insulin, prior bariatric surgery, and adolescents 12–17 each require modified protocols.
Planning pregnancy
The Wegovy label says to discontinue at least 2 months before a planned pregnancy because of semaglutide's long half-life. The current U.S. Zepbound label says the medication may cause fetal harm and should be discontinued when pregnancy is recognized — it does not use Wegovy's 2-month planned-pregnancy washout language. If you're trying to conceive, ask your prescriber for the specific timing for the medication you're on.
Tirzepatide and oral birth control
The Zepbound label specifically advises patients on oral hormonal contraceptives to switch to non-oral contraception or add a barrier method for 4 weeks after starting and for 4 weeks after every dose increase. This is one of the most commonly missed safety steps in fast telehealth visits. If your prescriber doesn't bring it up, you should.
Foundayo and oral birth control
The Foundayo label advises patients using oral contraceptives to switch to a non-oral method or add a barrier method for 30 days after starting and for 30 days after each dose escalation. This is a longer window than tirzepatide. Don't assume your prescriber automatically caught it.
Surgery or anesthesia
Tell your surgeon and anesthesia team you're on a GLP-1 well in advance of any procedure. Both the Wegovy and Zepbound labels reference aspiration risk because the medication slows stomach emptying. Many surgical teams now have specific pre-op protocols for GLP-1 patients.
Type 2 diabetes on insulin or a sulfonylurea
These combinations raise hypoglycemia risk. Doses of insulin or sulfonylurea often need to come down when starting a GLP-1. A telehealth provider who isn't actively coordinating those changes — or coordinating with your primary care diabetes clinician — is missing a critical piece.
Adolescents 12–17
The American Academy of Pediatrics' 2023 clinical practice guideline says clinicians should offer adolescents 12 and older with obesity weight-loss pharmacotherapy as an adjunct to lifestyle treatment. Wegovy injection has established safety and effectiveness for pediatric patients 12 and older with obesity. Wegovy tablets, Zepbound, and Foundayo do not have pediatric safety/effectiveness established in their current labels. Adolescent care should also include eating disorder screening, depression screening, and clinicians experienced with pediatric care.
Older adults
Watch for nutrition adequacy, protein intake, fall risk from rapid weight loss, and muscle mass concerns. Resistance exercise and adequate protein intake matter even more in this population.
Eating disorder history
This isn't disqualifying, but it requires honest disclosure and a clinician who is comfortable managing the overlap. A provider who doesn't ask is not the right provider for this situation.
Prior bariatric surgery
Coordination with your bariatric team matters. Don't add a GLP-1 to your post-bariatric care without that loop closed.
How to Verify a GLP-1 Telehealth Provider in 10 Minutes
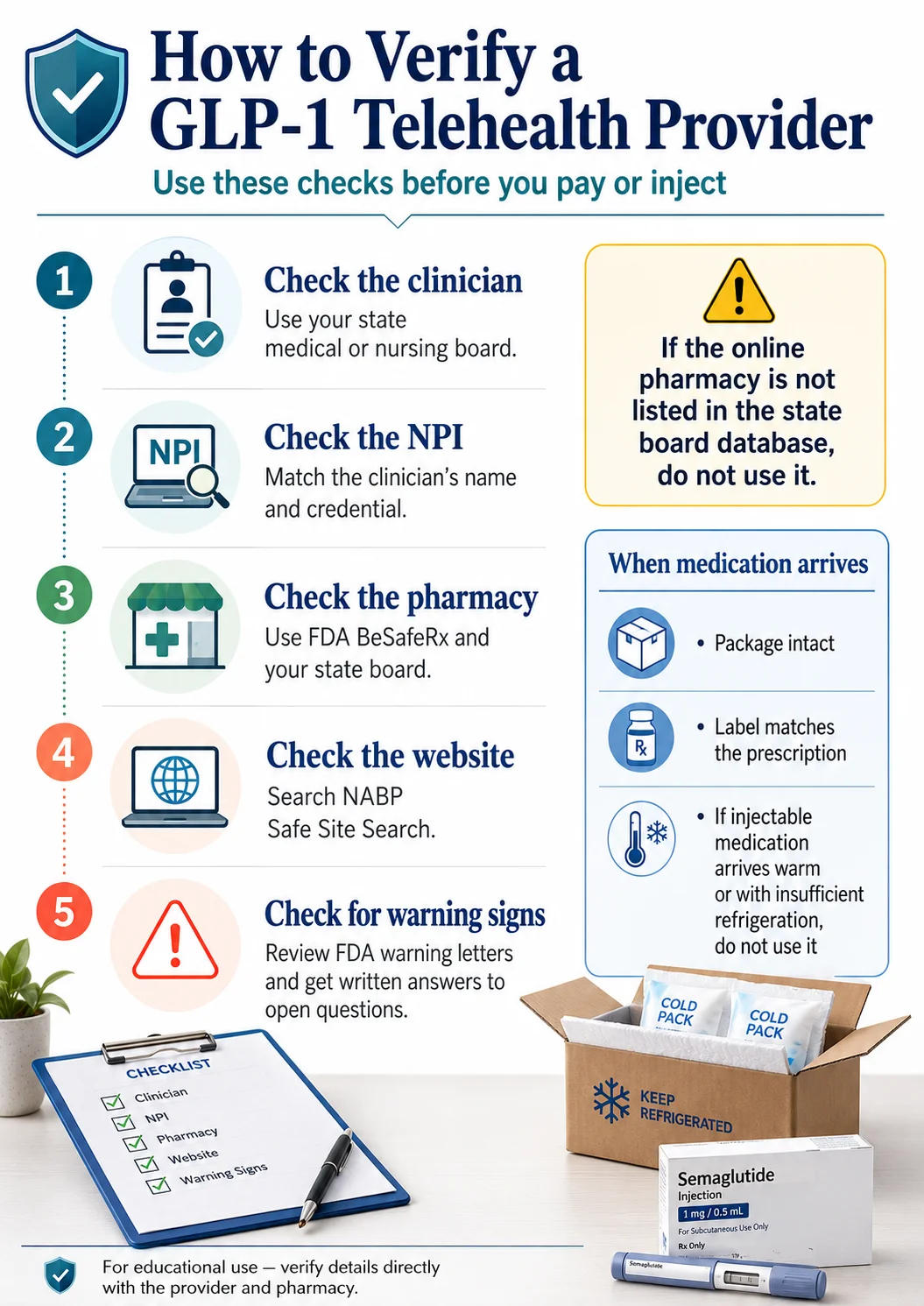
Vetting a GLP-1 telehealth provider takes about 10 minutes using six public checks — your state medical or nursing board, the NPI Registry, FDA BeSafeRx and your state pharmacy board, NABP Safe Site Search, the FDA warning letter database, and any state Attorney General or consumer-protection complaint records available in your state.
Look up the clinician on your state's medical board
Every state has a free public lookup. Search "[your state] medical board license lookup." For nurse practitioners, use the state nursing board. You're looking for: license active, no disciplinary actions, license matches the state where you're located for care.
Check the NPI Registry
Go to npiregistry.cms.hhs.gov. Search by clinician name. Most prescribing clinicians in the U.S. have an NPI. The result should match the clinician's name, state, and credential. If you can't find a match, ask the platform for clarification in writing.
Check the platform's certification status
Go to legitscript.com and use the search tool. Many GLP-1 telehealth platforms display a LegitScript seal — verify it through LegitScript's website rather than trusting the displayed graphic. Note the exact status that comes back.
Check the pharmacy on FDA BeSafeRx
Go to fda.gov/besaferx. Click "Locate a State-Licensed Online Pharmacy" and find your state's pharmacy database. Search the pharmacy name. You're looking for: an active license matching the pharmacy's stated name and address.
Cross-check with NABP Safe Site Search
Go to safe.pharmacy. Search the pharmacy or telehealth website. NABP categorizes online pharmacies and flags ones that fail patient safety standards.
Search the FDA warning letter database
Go to fda.gov, search "warning letters," and use the database search. Search by company name. You're looking for: no recent warning letters. If there are any, read them in full before deciding.
(Optional) Check your state AG consumer complaints
Search "[your state] attorney general consumer complaints." A pattern of unresolved complaints is a signal worth taking seriously.
What "passing" looks like
A safe provider passes the license, pharmacy, and prescription-requirement checks cleanly. If the clinician license, pharmacy license, or "is a prescription required" check fails, that's a hard stop. Other checks raising questions means get answers in writing before you pay.
What to Do If a Provider Fails the Checklist
If a provider fails the checklist before you've started, don't enroll. If you've already started, do not inject any medication you suspect is counterfeit, contaminated, mislabeled, improperly shipped, or incorrectly dosed.
If you haven't started yet
Walk away. Don't engage further. There are providers that pass the full checklist; spend your time finding one of those.
If you've already started
This is more careful. Don't inject anything you suspect is counterfeit, contaminated, mislabeled, improperly shipped, or incorrectly dosed. Then:
- 1
Request your medical records and prescription history in writing. They're legally yours.
- 2
Get transition guidance from a licensed clinician or pharmacist — your PCP, an obesity medicine specialist, or a new vetted telehealth provider — before you stop the medication. If you're using GLP-1 for diabetes, this is non-negotiable. Glucose control can change quickly when GLP-1 therapy stops.
- 3
Share the medication label, dose, pharmacy name, and any shipment issues with the replacement clinician.
- 4
Cancel only after you understand the clinical and billing implications of leaving.
Where to report
| Problem | Where to report | What to save |
|---|---|---|
| Suspected counterfeit medication | FDA MedWatch (fda.gov/safety/medwatch) | Photos, packaging, label, pharmacy name |
| Adverse event from medication | FDA MedWatch | Symptom log, dose, timing, prescriber name |
| Warm or damaged shipment | Pharmacy first; FDA MedWatch if unresolved | Photos of package and contents on arrival |
| False or misleading advertising | FDA Office of Prescription Drug Promotion (OPDP) | Screenshots of ads, URLs, dates |
| Deceptive marketing or billing | FTC (reportfraud.ftc.gov) | Order confirmations, ad copy, billing statements |
| Clinician misconduct | State medical or nursing board | Clinician name, NPI, dates, case details |
| Pharmacy license concern | State board of pharmacy | Pharmacy name, license number, prescription details |
| Subscription or refund dispute | State Attorney General + BBB | Terms of service, billing records, communication |
The FDA actively encourages patients and professionals to report adverse events or quality problems with GLP-1 medications to MedWatch. Your report contributes to the agency's enforcement decisions.
The Printable GLP-1 Telehealth Safety Checklist Worksheet
Use this worksheet during the provider vetting process. Fill it out before you pay. Keep it for your records. It also serves as documentation if you later need to file a complaint.
Provider information
- Company name: ___________________________
- Website URL: ___________________________
- Legal business name: ___________________________
- Clinician name: ___________________________
- Clinician credential (MD, DO, NP, PA): ___________________________
- State license verified? Yes / No: ___________________________
- NPI verified? Yes / No: ___________________________
- Date checked: ___________________________
Pharmacy information
- Pharmacy name: ___________________________
- Pharmacy city/state: ___________________________
- 503A or 503B (if compounding): ___________________________
- State license verified through FDA BeSafeRx? Yes / No: ___________________________
- NABP Safe Site verified? Yes / No: ___________________________
- LegitScript status: ___________________________
Medication information
- Medication name: ___________________________
- Brand-name FDA-approved or compounded: ___________________________
- Active ingredient: ___________________________
- If semaglutide: base, sodium, or acetate?: ___________________________
- Dose in mg: ___________________________
- Concentration (mg/mL): ___________________________
- Starting dose: ___________________________
- Titration plan: ___________________________
- Beyond-use date: ___________________________
Medical review checklist
- BMI/eligibility reviewed: Yes / No
- Current medications reviewed: Yes / No
- Diabetes medications reviewed: Yes / No
- Pregnancy plans reviewed: Yes / No
- Thyroid/MEN2 history reviewed: Yes / No
- Pancreatitis history reviewed: Yes / No
- Gallbladder history reviewed: Yes / No
- GI disease reviewed: Yes / No
- Kidney/liver disease reviewed: Yes / No
- Allergies reviewed: Yes / No
- Lab work ordered or recent results reviewed: Yes / No
- Follow-up scheduled: Yes / No
Shipment inspection
- Arrived cold (if required): Yes / No
- Label matches prescription: Yes / No
- Expiration/BUD present: Yes / No
- Packaging intact: Yes / No
- Instructions clear: Yes / No
- Pharmacy contact info on label: Yes / No
Frequently Asked Questions
Is GLP-1 telehealth safe in 2026?
GLP-1 telehealth can be safe when a licensed clinician performs a real evaluation, the pharmacy is verifiable, the medication is clearly identified, and follow-up care is included. It is unsafe when those layers are missing. The FDA announced 30 telehealth warning letters on March 3, 2026, after an earlier round tied to its September 2025 enforcement crackdown, and says it sent thousands of letters warning pharmaceutical and telehealth firms to remove misleading ads in the six months before March 3, 2026. Vetting matters more now than it did a year ago.
Can I get GLP-1 medication online without a prescription?
No. GLP-1 medications are prescription-only in the U.S. Any service offering GLP-1 without a prescription is operating illegally and should be avoided.
How do I know if my GLP-1 telehealth provider is legit?
Verify three things in 10 minutes: the clinician's state license through your state medical board, the pharmacy's state license through FDA BeSafeRx, and any platform certification through LegitScript or NABP Safe Site Search. Then cross-check the FDA warning letter database. If those four come back clean, the provider is likely legitimate.
Are compounded GLP-1 medications FDA-approved?
No. Compounded medications are not FDA-approved finished products. The FDA does not review them for safety, effectiveness, or quality before they reach patients. Compounded medications are also not the same as FDA-approved generic drugs.
Is "generic Ozempic" or "generic Wegovy" a real thing?
No. There is no FDA-approved generic semaglutide or generic tirzepatide in the United States. A telehealth company that markets a compounded product as "generic Ozempic" or "generic Wegovy" is using language the FDA has cited as misleading.
What's the difference between semaglutide base and semaglutide sodium?
Semaglutide base is the active ingredient used in FDA-approved semaglutide products. Semaglutide sodium and semaglutide acetate are salt forms. The FDA has stated it is unaware of any lawful basis for using semaglutide salt forms in compounding. If your compounded prescription lists semaglutide sodium or acetate, ask the pharmacy directly why.
What should I do if my GLP-1 medication arrives warm?
Do not inject it. Take photos of the package and contents. Contact the pharmacy and prescriber the same day. Ask them in writing whether the shipment is usable under the labeled storage rules or whether they'll replace it under their shipping policy. The FDA has stated that injectable GLP-1 medications that arrive warm or with insufficient refrigeration should not be used because quality can be affected.
What labs should be checked before starting a GLP-1?
There is no single universal lab list. Clinicians commonly consider A1C or fasting glucose, kidney function (creatinine, eGFR), liver function, lipids, and pregnancy testing when relevant. Other labs may be ordered based on individual history. A provider who orders no labs and reviews no recent records is cutting a corner the prescribing label assumes will be filled.
How often should I see my prescriber after starting?
Standard practice is monthly during the titration phase (the first 3–4 months while you're moving up in dose) and every 3 months after reaching maintenance dose. Yale Medicine notes that ongoing follow-up is critical because about half of patients stop within a year, often due to side effects, cost, or lack of support.
Can my regular doctor prescribe GLP-1 medications?
Yes. Any licensed clinician — primary care physician, nurse practitioner, physician assistant, endocrinologist, or obesity medicine specialist — can prescribe GLP-1 medications. Telehealth is one option among several. Your primary care doctor may already prescribe these medications.
What's the difference between a 503A and 503B pharmacy?
A 503A pharmacy is a traditional compounding pharmacy that prepares medications for individual patients with a specific prescription. A 503B pharmacy is an FDA-registered "outsourcing facility" that operates under a different federal framework, including current good manufacturing practice (CGMP) standards. Neither type is the same as an FDA-approved drug manufacturer.
Should I tell my primary care doctor I'm using a telehealth GLP-1 service?
Yes. Care coordination matters. GLP-1 therapy can affect diabetes medications, surgery planning, GI symptoms, kidney function, and long-term weight management. A 2025 Omada Health survey of more than 2,000 U.S. primary care physicians found that 67% agreed or strongly agreed that third-party telehealth GLP-1 prescribing for weight loss may put patient health at risk, and 50% named continuity of care — follow-up visits, titration, and tapering — as a top concern. That gap is a known patient safety issue.
Where do I report a sketchy GLP-1 telehealth company?
Multiple paths: your state medical board for clinician issues, FDA MedWatch for medication quality issues, FDA OPDP for false advertising, FTC for deceptive marketing, your state Attorney General for consumer protection, and the BBB for documented complaint patterns.
What questions should I ask before paying?
Ask who prescribes, whether they're licensed where you're located, what pharmacy fills the prescription, whether the medication is FDA-approved or compounded, how dosing is written, what follow-up is included, what happens if you have side effects, and what the cancellation policy is. Get answers in writing before you pay.
Sources
This page was built using:
- ·FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss (current as of February 2026)
- ·FDA Press Announcement: Warning Letters to 30 Telehealth Companies (March 3, 2026)
- ·FDA Press Announcement: Proposal to Exclude Semaglutide, Tirzepatide, and Liraglutide from the 503B Bulks List (April 30, 2026)
- ·FDA's clarification on policies for compounders as national GLP-1 supply stabilizes (April 1, 2026)
- ·FDA BeSafeRx — Locate a State-Licensed Online Pharmacy
- ·Wegovy prescribing information (current label)
- ·Zepbound prescribing information (current label)
- ·Foundayo (orforglipron) prescribing information (current label)
- ·Ozempic, Mounjaro, and Rybelsus prescribing information
- ·FSMB Model Telemedicine Policy
- ·CCHP State Telehealth Policies — Cross-State Licensing and Online Prescribing
- ·NABP Safe Site Search Tool
- ·StatPearls Obesity Medications: Evidence-Based Management (NCBI)
- ·AMA Ed Hub — Weight Management Implications Applying GLP-1s
- ·Cleveland Clinic — GLP-1 Agonists overview
- ·MDedge — Compounded Semaglutide/Tirzepatide Checklist
- ·Yale Medicine — GLP-1 Medications: How to Get Started
- ·American Academy of Pediatrics 2023 Clinical Practice Guideline on Childhood Obesity
- ·Omada Health 2025 PCP survey on telehealth GLP-1 prescribing
- ·Novo Nordisk published position on illicit compounding
Update log
- May 6, 2026: Initial publication. Verified the full FDA enforcement timeline through April 30, 2026, current shortage status for semaglutide and tirzepatide, current Wegovy, Zepbound, and Foundayo label language for contraception, pregnancy guidance, and pediatric use, and current NABP and FDA BeSafeRx verification workflows.
- Next scheduled review: August 2026 — earlier if FDA finalizes its 503B bulks list proposal, issues new warning letters, or updates GLP-1 prescribing labels.
Editorial independence
We may earn a commission if a reader chooses a partnered provider through our comparison pages. No provider paid to be included in or excluded from this safety checklist. This page is a patient resource, not a provider ranking.