Sterile Compounding Red Flags: How to Evaluate a 503A Pharmacy Before You Order
By Weight Loss Provider Guide Research Team · Last verified: · Next check: July 2026
Advertising disclosure: We earn affiliate commissions from some links on this page. Our rankings reflect verified safety signals and editorial judgment — not commission rates. Full disclosure →
The short answer, before you scroll
How to evaluate a 503A pharmacy before ordering compounded GLP-1: get the pharmacy's legal name before you pay, verify it holds an active license through your state's board of pharmacy, confirm it has no open FDA warning letter tied to sterile compounding or GLP-1s, and ask — in writing — how the pharmacy verifies sterility and potency. If the seller refuses to name the pharmacy, uses phrases like "generic Wegovy" or "same active ingredient as Ozempic," delivers a vial that arrives warm, or ships a product labeled semaglutide sodium or acetate (the FDA has said these salt forms are different active ingredients from semaglutide and have not been evaluated by the agency), walk away. No partial credit on any of those. [FDA, Feb. 6, 2026] [FDA GLP-1 safety page]
| Verdict | What it means |
|---|---|
| ✅ Proceed | Named pharmacy, active state license, clean public record, full answers in writing, score of 80+ on the audit below, and zero automatic-fail triggers |
| ⚠️ Pause | Basics check out, score of 60–79, no automatic-fail triggers — resolve the gaps in writing before re-ordering |
| 🚫 Walk away | Score below 60, or any single automatic-fail trigger from the section below |
Primary next step: run the audit yourself. It's free, sits on this page, and takes about ten minutes.
Jump to the 11-check pharmacy audit ↓
Or if you'd rather skip the DIY and get matched with a GLP-1 provider that fits your situation:
Take the free 60-second matching quiz →What we actually verified for this page
We built this guide on April 23, 2026 by reviewing:
- FDA's BeSafeRx state-licensed online pharmacy guidance
- FDA's April 1, 2026 clarification on compounding policy after the national GLP-1 supply stabilized
- FDA's February 6, 2026 press announcement about enforcement against non-FDA-approved GLP-1 drugs
- FDA's March 3, 2026 action warning 30 telehealth companies about illegal marketing of compounded GLP-1s
- FDA's ongoing safety communications on fraudulent labels, warm shipments, dosing errors, and semaglutide salt forms
- USP chapters <795>, <797>, and <800> — the compounding standards 503A pharmacies are typically held to under state law
- Pharmacy Compounding Accreditation Board (PCAB) standards, a service of ACHC
- NABP Verified Pharmacy Program and Compounding Pharmacy Accreditation
- Eli Lilly's March 12, 2026 open letter on a novel impurity in tirzepatide compounded with vitamin B12 (company statement)
- Outsourcing Facilities Association v. FDA, 4:25-cv-00174 (N.D. Tex. 2025) court rulings
- FDA's Boothwyn Pharmacy LLC warning letter, issued January 16, 2026
What we did not independently verify: the sterility, potency, or impurity profile of any single pharmacy's product. No consumer-side audit can substitute for end-product lab testing. We tell you what to ask for so the pharmacy does that job on the record.
What people type when this fear hits
Public-forum shopper language, shown for intent context — not as evidence of safety or of any particular pharmacy:
- “How do you know it's legit?”
- “This doesn't feel safe to inject??”
- “Delivered warm(ish)?”
- “Which is better 503a or 503b?”
- “My understanding is that it's safer to find a PCAB-certified 503A, or ideally a 503B.”
If any of those sound like what you almost typed, you're in the right place.
Is a 503A pharmacy automatically unsafe for compounded GLP-1?
Answer: No. A 503A pharmacy (a state-licensed compounding pharmacy that fills patient-specific prescriptions under Section 503A of the Federal Food, Drug, and Cosmetic Act) isn't automatically unsafe — but it's a weaker baseline trust signal than an FDA-approved medication dispensed from a standard retail pharmacy, and a weaker signal than a clean 503B outsourcing facility (an FDA-registered compounder that operates under current Good Manufacturing Practice, or cGMP). The real question is never the acronym. It's whether the specific pharmacy is named, licensed in your state, operating on a patient-specific prescription basis, transparent about sourcing, and clean on public records.
- •503A pharmacies are regulated day-to-day by state boards of pharmacy, not the FDA. [FDA Q&A]
- •They must comply — where applicable under state law — with USP <795> (non-sterile), USP <797> (sterile), and USP <800> (hazardous-drug handling). State adoption and enforcement vary.
- •They do not have to meet cGMP — the stricter, pharma-manufacturer standard that requires validated processes, lab controls, release testing, and full documentation traceability.
- •Inspections vary widely. A 2018 Pew Charitable Trusts and NABP review found that about half of U.S. states did not conduct routine inspections of 503A sterile compounders, and inspection intervals varied widely among those that did. State practices have shifted since 2018, but the patchwork structure is the same.
One more thing worth saying out loud. Compounded drugs are not FDA-approved — full stop. That's true whether the pharmacy is 503A or 503B. "Not FDA-approved" doesn't mean unsafe, but it does mean the normal safety net of FDA premarket review isn't there. Everything below is how you build your own version of it.
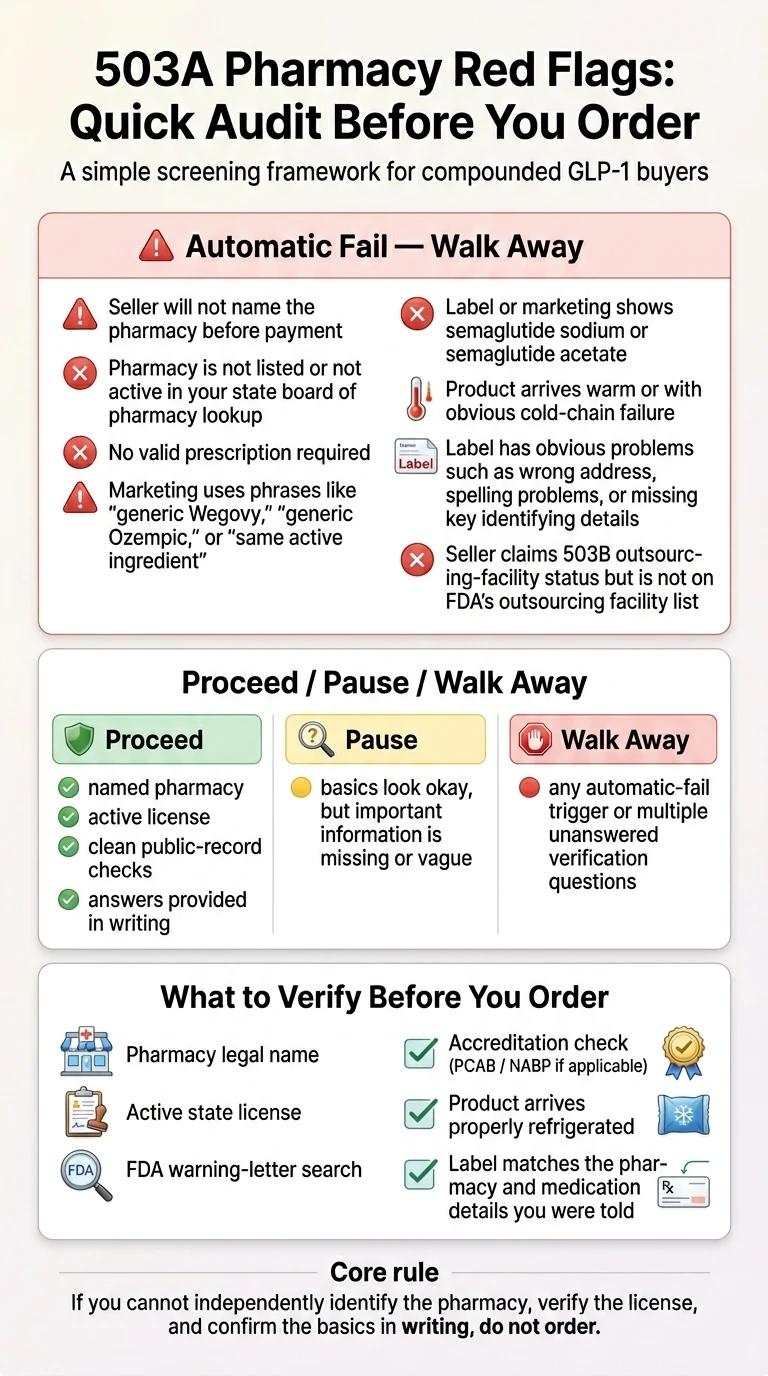
Sterile compounding red flags that are automatic walk-aways
Answer: Eight conditions override every other positive signal. If any single one of these is true, cancel the order, don't ship the vial, don't give the benefit of the doubt. Most of these map directly to FDA safety communications from 2024–2026.
Hard-fails first. Weighted audit next. Hard-fails aren't negotiable.
🚫 Automatic fail #1 — The seller won't name the pharmacy before you pay
FDA has publicly documented fraudulent compounded GLP-1s circulating with labels citing pharmacies that didn't exist, or naming real pharmacies that hadn't actually compounded the product. If you can't independently identify the legal pharmacy name before your card is charged, the core verification chain is broken from the start. Every real 503A pharmacy has a legal name and an address. If the provider's customer service gives you "we use a network of licensed pharmacies" and nothing more — that's the answer.
[FDA GLP-1 safety page]🚫 Automatic fail #2 — The pharmacy isn't in your state's board-of-pharmacy database
FDA's BeSafeRx guidance says plainly: if an online pharmacy isn't listed in the state pharmacy board database, you should not use that pharmacy. For out-of-state pharmacies shipping to you, the relevant license is often a non-resident pharmacy license in your home state.
[FDA BeSafeRx]🚫 Automatic fail #3 — No valid prescription required
A 503A pharmacy's legal basis for compounding is a patient-specific prescription from a licensed prescriber for an identified individual patient. If a seller ships you a compounded injectable without a prescription — or labels it "research use only" or "not for human consumption" — that isn't a compounding pharmacy at all.
[FDA 503A guidance]🚫 Automatic fail #4 — The label or marketing uses prohibited comparison language
On February 6, 2026, the FDA announced its intention to take action against non-FDA-approved GLP-1 drugs and made clear that compounded GLP-1 marketing cannot claim the products are "generic versions" of Wegovy, Ozempic, Zepbound, or Mounjaro, or that they contain the "same active ingredient" as FDA-approved products. If the seller is still using that language in 2026, they're either ignoring the FDA or betting you won't notice.
[FDA, Feb. 6, 2026]🚫 Automatic fail #5 — Semaglutide sodium, semaglutide acetate, retatrutide, or cagrilintide on the label
The FDA has said semaglutide sodium and semaglutide acetate are salt forms that are different active ingredients from semaglutide and have not been evaluated by the agency. The FDA has also said retatrutide and cagrilintide cannot be used as active ingredients for compounding under federal law. Any of these on your label or in the seller's marketing is a hard stop.
[FDA GLP-1 safety page]🚫 Automatic fail #6 — The vial arrives warm or the cold chain obviously failed
FDA has told patients, in writing, not to use injectable GLP-1 medication that arrives warm or without adequate refrigeration. Semaglutide and tirzepatide are peptide drugs. Heat exposure can degrade peptide potency and creates opportunities for microbial growth. If the insulated box is warm, the ice packs are fully liquid and room temperature, or the packaging is insufficient for your climate and transit time, don't inject it.
[FDA GLP-1 safety page]🚫 Automatic fail #7 — Obvious label problems
FDA has documented compounded GLP-1 labels with misspellings, wrong addresses, nonexistent pharmacies, and pharmacy names that didn't match the actual compounder. If the label is missing a lot number, a beyond-use date (BUD), or the pharmacy address, that's a serious red flag. If the pharmacy name on the label doesn't match the pharmacy your provider told you about, treat it as a hard stop until the pharmacy itself explains the discrepancy.
[FDA GLP-1 safety page]🚫 Automatic fail #8 — The seller claims 503B outsourcing-facility status but isn't on FDA's list
FDA maintains a public list of registered outsourcing facilities. Registration is annual. If a seller markets itself as a 503B and the facility isn't on FDA's list, the registration claim is false. That's fraud, not carelessness.
[FDA registered outsourcing facilities]Decision checkpoint:
Any one of those eight triggers and you stop. Full stop. If your situation clears all eight, keep going. That's the bar for the rest of the framework to matter.
How to evaluate a 503A pharmacy before ordering: the 11-check audit
Answer: After the hard-fails, there are eleven weighted checks that separate a pharmacy you can actually trust from one that's skating on baseline licensing alone. The highest-weight checks are state licensing (12 points), FDA enforcement record (12 points), PCAB or NABP accreditation (10 points), sterile-compounding capability (10 points), and testing and quality program (10 points). Pass is 80+. Pause is 60–79. Fail is below 60. Any automatic-fail trigger overrides the weighted score.
This scorecard is our original framework. The individual facts are public; the assembled scoring model is ours. You're welcome to copy it — just cite us.
| # | Check | Points | Why it matters | How to verify |
|---|---|---|---|---|
| 1 | Pharmacy's legal name is disclosed before payment | 10 | FDA has documented compounded GLP-1 labels citing pharmacies that didn't exist or didn't actually compound the product [FDA] | Ask in writing; confirm on the eventual label |
| 2 | Active, unrestricted state license in your state (resident or non-resident) | 12 | Federal law requires state licensure; non-resident license needed for ship-to-your-state [FDA BeSafeRx] | Your state board of pharmacy lookup (see Step 1 below) |
| 3 | Patient-specific prescription basis for the compound | 8 | Section 503A is anchored to patient-specific prescribing; office-stock shipments to patients are not 503A-eligible [FDA] | Confirm with provider; the prescription number appears on your label |
| 4 | No open FDA warning letter, Form 483, or consent decree related to compounding or GLP-1s | 12 | FDA warned 30 telehealth companies on March 3, 2026; warning letters continue on a rolling basis [FDA March 2026] | FDA Warning Letter database (walkthrough below) |
| 5 | Performs sterile compounding under USP <797> with environmental monitoring at least every six months | 10 | Top sterile-compounding violations cluster around facility/environmental controls, cleaning/disinfection, and personnel gowning [ScienceDirect, 2023] | Ask pharmacy; cross-check with state inspection reports |
| 6 | PCAB/ACHC accreditation or NABP Compounding Pharmacy Accreditation | 10 | Voluntary third-party programs built on USP compounding standards; a meaningful independent quality signal [ACHC/NABP] | ACHC and NABP directories |
| 7 | Pharmacy has a documented testing and quality program covering sterility, potency, and impurities | 10 | Eli Lilly's March 12, 2026 open letter reported impurity concerns in tirzepatide compounded with B12; asking about testing surfaces whether real quality controls exist [Eli Lilly open letter] | Ask the pharmacy in writing; request documentation |
| 8 | Active pharmaceutical ingredient (API) sourced from an FDA-registered manufacturer | 8 | FDA's Import Alert 66-80 "Green List" identifies acceptable API sources; imported API from unregistered facilities is a recurring warning-letter issue [FDA] | Ask which API supplier and for its FDA registration |
| 9 | Documented clinical-differentiation rationale for the compound, post-April 2025 | 8 | After FDA resolved the shortage and ended shortage-based enforcement discretion, ongoing compounding generally requires documented clinical difference [FDA April 1, 2026] | Provider must document a qualifying reason in your chart |
| 10 | Product ships ready-to-inject (not powder-plus-diluent for you to mix), with visible lot number and BUD | 6 | User-reconstitution kits are a documented sterility and dosing-error vector; FDA has received adverse-event reports tied to measurement-unit confusion [FDA] | Visual inspection on arrival |
| 11 | Physical address and direct phone published; a licensed pharmacist is reachable during business hours | 6 | FDA's BeSafeRx program flags hidden addresses and web-form-only contact as core counterfeit indicators [FDA BeSafeRx] | Website check and a test call |
One honest note: no weighted model is a guarantee. A pharmacy that scored 95 last quarter can be the subject of a new warning letter by next Tuesday. That's why we recommend re-running this audit at renewal, not just at first order.

How to verify any compounding pharmacy in under 10 minutes (with direct links)
Answer: Five lookups cover ninety percent of what matters: your state pharmacy board's license database, the FDA warning-letter database, the PCAB/ACHC accredited-pharmacy directory, the NABP Verified Pharmacy Program or NABP Safe Site Search Tool, and the Alliance for Pharmacy Compounding "Is It Legit?" tool. Each one is free, takes two minutes or less, and gives you an objective yes-or-no answer.
1Verify the state-board license
Federal law requires every U.S. pharmacy to hold an active pharmacy license from its home state's Board of Pharmacy. If it ships to patients in other states, it also needs non-resident licensure in each state it ships to. Your ship-to address determines which state's rules apply.
- NABP's member directory — links to every U.S. state and territory board of pharmacy
- FDA's BeSafeRx state map — same starting point with consumer-friendly framing
What you're looking for: active status, no current disciplinary action, an expiration date in the future, and — if relevant — a non-resident license in your state. A pharmacy that's active in its home state but has no non-resident license for your state is not legally authorized to ship to you. Clean walk-away.
2Search the FDA warning-letter database
Go to FDA's Warning Letter database and search by the pharmacy's legal name. Also try searching "semaglutide" or "tirzepatide" to see the full cohort of recent GLP-1-related letters. As of spring 2026, FDA is continuing to publish new letters on a rolling basis.
3Check PCAB accreditation
PCAB (operated as a service of ACHC) is the most widely recognized independent accreditation for compounding pharmacies. Its standards are built on USP <795>, <797>, and <800>. PCAB is voluntary, resource-intensive, and re-surveyed on a three-year cycle. Check directly at achc.org/pcab. A compounded injectable GLP-1 requires sterile accreditation — non-sterile alone isn't enough.
4Check NABP
The National Association of Boards of Pharmacy runs two relevant programs:
- NABP Compounding Pharmacy Accreditation — a parallel track to PCAB
- NABP Verified Pharmacy Program (VPP) — a uniform inspection program used by many state boards for non-resident licensure
Neither program is mandatory for a 503A. The presence of either is a positive signal. The absence of both is information, not necessarily a fail.
5APC "Is It Legit?" and Safe Pharmacy resources
The Alliance for Pharmacy Compounding (APC — note: an industry trade association, not a government regulator) maintains a4pc.org/isitlegit for confirming whether a compounder is legitimately state-licensed. NABP's Safe Pharmacy resources flag known illegitimate sites. Use both as one more cross-check, not the sole answer.
Decision checkpoint
If you've cleared all five lookups and haven't tripped an automatic-fail trigger, you're in Proceed or Pause territory. If you're in Pause territory — basics check out but something's missing — it's time to put your questions to the provider in writing.
503A vs. 503B: what the distinction actually tells you (and what it doesn't)
Answer: 503A pharmacies compound patient-specific prescriptions under state board of pharmacy oversight. 503B outsourcing facilities are FDA-registered sterile compounders that operate under cGMP, including lab controls and release testing. 503B is a stronger baseline trust signal because of FDA registration and cGMP compliance, but FDA has been explicit that registration alone doesn't guarantee compliance or product safety — a 503B facility with a recent warning letter is not safer than a well-run 503A.
| Feature | 503A pharmacy | 503B outsourcing facility |
|---|---|---|
| Primary regulator | State board of pharmacy | FDA |
| FDA registration | Not required | Required (annual) |
| Patient-specific prescription | Required | Not required (can compound office stock) |
| USP <795>/<797>/<800> compliance | Required where applicable under state law | Required |
| cGMP (validated manufacturing, lab controls, release testing) | Not required | Required |
| Inspection cadence | Varies widely by state | FDA inspects on a risk-based schedule |
| Common use case for GLP-1 in 2026 | Patient-specific differentiated formulations with documented clinical rationale | Now limited for GLP-1 — see legal section below |
"503B is always safer." It's usually a stronger baseline, but the track record of a specific facility matters more than the letter. The FDA has issued warning letters to 503B outsourcing facilities that included cGMP violations, sterile-compounding deficiencies, and insanitary conditions. A 503B warning letter is as much a walk-away as a 503A one.
"503A means they'll customize for me." In theory yes, in practice the compound you receive from most telehealth-affiliated 503A pharmacies is a standard formulation the prescriber selected from the pharmacy's menu. The "patient-specific" language is the legal basis for dispensing; it doesn't mean the pharmacist is engineering a bespoke blend just for you.
Can a 503A pharmacy legally compound semaglutide or tirzepatide right now?
Answer, current as of April 23, 2026: Only in narrow circumstances. FDA resolved the semaglutide shortage on February 21, 2025, with shortage-based enforcement discretion for 503A semaglutide ending on April 24, 2025, and enforcement discretion for 503B facilities ending on May 22, 2025. FDA reaffirmed the tirzepatide shortage resolved in December 2024. FDA's April 1, 2026 update confirmed neither drug appears on the 503B bulks list or the current shortage list. Continued 503A compounding generally requires documented clinical differentiation. The agency also stated it does not currently intend to take action if a 503A compounder fills four or fewer prescriptions per month of an essentially-copy product.
2026 legal status timeline at a glance
| Date | Event |
|---|---|
| Dec. 2024 | FDA reaffirms tirzepatide shortage resolved |
| Feb. 21, 2025 | FDA announces semaglutide shortage resolved |
| Mar. 5, 2025 | Federal district court denies preliminary injunction to compounders |
| Apr. 24, 2025 | Semaglutide 503A shortage-based enforcement discretion ends (window closes on later of Apr. 22 or court ruling) |
| May 22, 2025 | Semaglutide 503B enforcement discretion ends |
| Sept. 2025 | FDA opens first wave of warning letters to GLP-1 compounders and telehealth marketers |
| Jan. 16, 2026 | FDA issues warning letter to Boothwyn Pharmacy citing sterile deficiencies plus subpotent semaglutide and tirzepatide |
| Feb. 6, 2026 | FDA press announcement announcing enforcement against non-FDA-approved GLP-1 drugs |
| Mar. 3, 2026 | FDA warns 30 telehealth companies over illegal compounded-GLP-1 marketing |
| Mar. 12, 2026 | Eli Lilly open letter flags novel impurity in tirzepatide compounded with B12 based on company testing |
| Apr. 1, 2026 | FDA policy clarification: 503A essentially-copy restrictions apply; FDA does not currently intend to act on four-or-fewer prescriptions per month for essentially-copy products |
"Essentially a copy" test. FDA's guidance prohibits 503A pharmacies from compounding a drug that is essentially a copy of a commercially available FDA-approved product, unless there's a documented clinical difference on file. A pharmacy simply labeling a product "semaglutide with B12" doesn't automatically satisfy it.
The semaglutide + B12 question. Some pharmacies argue that adding vitamin B12 (cyanocobalamin) creates clinical differentiation. FDA has not formally endorsed that position. Eli Lilly's March 12, 2026 open letter flagged a novel impurity in tirzepatide compounded with vitamin B12 whose short- and long-term effects in humans are unknown.
Adverse-event data. As of July 31, 2025, the FDA had received 605 adverse-event reports for compounded semaglutide and 545 adverse-event reports for compounded tirzepatide. [FDA] Federal law doesn't require 503A pharmacies to report adverse events to FDA — so actual numbers are likely higher.
How to check warning letters, recalls, and inspection problems (case study: Boothwyn)
Answer: A clean public-record search is one of the few high-leverage checks you can run without trusting marketing copy. FDA publishes warning letters, Form 483 observations, and product recalls in free searchable databases. The highest-value searches: the pharmacy's legal name in the warning-letter database and on FDA's Enforcement Report for recalls.
How to run the search
- Warning letters: search by company name or product/topic, filter by date
- Recalls: cross-reference FDA's Enforcement Report for recall classifications
- Form 483 observations (public via FOIA and sometimes on FDA's site): what an FDA inspector documents at a facility during an inspection. A 483 isn't a warning letter, but a pattern of repeat 483s on the same issues is meaningful.
How to read what you find
| Phrase in warning letter | What it actually means |
|---|---|
| "Failure to follow established procedures for sterile-drug-product production" | The facility didn't follow its own cleanroom or gowning procedures — the most common class of sterile-compounding violation |
| "Lack of assurance of sterility" | The pharmacy can't prove what it sent you was sterile; products made under those conditions are often recalled |
| "Subpotent" or "out-of-specification" batch | Lab testing found less active ingredient than the label claimed; a subpotent GLP-1 won't work as expected |
| "Bulk drug substance from a source that is not an FDA-registered establishment" | The API itself was sourced from a facility FDA doesn't know — an upstream quality problem |
One thing that matters: a warning letter is not a conviction. The pharmacy has an opportunity to respond, remediate, and get back into good standing. What separates a concerning warning letter from a disqualifying one is whether the pharmacy publicly addressed the findings, committed to remediation, and has operated clean since. Silence is the bad sign.
Boothwyn as a teaching case
We use Boothwyn as the concrete example because the warning letter is recent, public, and covers three of the failure modes this guide exists to help you avoid:
- Sterile-compounding deficiencies (what USP <797> is designed to prevent)
- Tirzepatide sterility failure (end-product testing found the product was not sterile)
- Subpotent semaglutide and tirzepatide lots (end-product testing found less active ingredient than labeled)
Decision checkpoint: clean search = keep going. Recent unresolved warning letter = stop here.
What to check when the vial arrives
Answer: An order isn't safe just because it shipped. FDA has explicitly warned patients not to use injectable GLP-1 products that arrive warm or with insufficient refrigeration, and has documented compounded GLP-1 labels with misspellings, wrong addresses, and pharmacy identities that didn't match the actual compounder. At minimum, check: package temperature and ice-pack state on arrival, label completeness, and product appearance in the vial. If anything looks off, don't inject — photograph everything and call the pharmacy before the product goes in your body.
❄️ Cold-chain checks
- Box exterior feels cold to the touch
- Ice packs at minimum still partially frozen
- Vial itself is cool, not at ambient temperature
- Transit time accounted for (hot porch risk)
🏷️ Label checks
- Legal pharmacy name (matches what you verified)
- Your name spelled correctly
- Medication name and strength (not "sodium" or "acetate")
- Lot number and beyond-use date (BUD)
- Dose in clear units (mg or mL, not "units")
- Prescriber's name
🧪 Product checks
- No particulate matter floating in vial
- No cloudiness where product should be clear
- No crystals or unexpected color
- If anything looks off: photograph and call pharmacy first
If something is wrong with the product you've received, you can report the adverse event to FDA through MedWatch. Your report helps FDA build the safety signal for compounded GLP-1s that the agency itself has said is likely underreported.
The 6 questions to send your telehealth provider before you pay
Answer: Legitimate providers answer these six questions in plain language, in writing, before you're charged. Evasion, "our pharmacy network is confidential," or "we can't share that" are not acceptable answers in a context where you're paying to inject a non-FDA-approved peptide drug into your body.
Copy the script below. Send it via chat, message, or email. Save the response.
How to read the answers:
- ✅ All six answered in writing, in plain language, with no hedging → Proceed.
- ⚠️ Four or five answered; one or two dodged → Pause. Get the outstanding ones in writing before you re-ask.
- 🚫 Three or fewer answered, or any hard-fail trigger → Walk away.
Who should NOT use a compounded 503A GLP-1 (the honest version)
We try not to talk anyone out of a legitimate path. But this page doesn't earn its trust unless we're straight about when compounded is the wrong fit.
The damaging admission:
No audit framework — ours or anyone else's — fully replaces the regulatory certainty of an FDA-approved medication. Compounded GLP-1s are not FDA-approved. They don't go through FDA's premarket evaluation for safety, efficacy, or quality. An 11-point scorecard reduces risk materially. It cannot eliminate it.
If any of these describe you, compounded is the wrong path:
- ✗You want maximum manufacturing oversight — every vial produced under validated cGMP processes, from a drug that went through FDA's full premarket review. That's only available with FDA-approved medications (Wegovy, Zepbound, Ozempic, Mounjaro, Foundayo, Rybelsus).
- ✗You want to use insurance or pay through HSA/FSA for a branded medication — compounded GLP-1s are generally not covered by insurance; FDA-approved ones often are, and the paperwork pathway is predictable.
- ✗You cannot tolerate regulatory ambiguity on a sensitive medication — the legal status of compounded semaglutide and tirzepatide has been contested in federal court throughout 2025–2026.
- ✗You've had a significant adverse event on any GLP-1 before — compounded formulations add variability on top of the underlying medication profile.
- ✗You'd rather not do any of the verification in this guide — that's a legitimate choice. It means compounded isn't your fit, not that you're lazy.
The pivot: a clean path if compounded isn't for you
If you want the FDA-approved route, Ro is currently the most direct bridge for cash-pay patients. Ro carries FDA-approved GLP-1 medications including Zepbound® (tirzepatide) and Foundayo™ (orforglipron) and includes an insurance concierge that handles prior-authorization paperwork plus a free GLP-1 Insurance Coverage Checker.
If compounded is right for your situation — you're cost-sensitive, you have a documented clinical reason, and you're willing to do the verification — we point you at providers that publicly name their pharmacy partner. The quiz gets you there in about a minute.
Match with a GLP-1 provider that fits →How we built this guide and what we actually verified
Answer: This guide is a research product, not a personal medical recommendation. On April 23, 2026, we reviewed FDA online-pharmacy guidance, FDA's compounding rules and the April 1, 2026 GLP-1 policy update, FDA warning letters and safety communications from 2024–2026, USP <795>/<797>/<800> standards, NABP accreditation and inspection materials, PCAB/ACHC accreditation criteria, Eli Lilly and Novo Nordisk public statements on compounded versions, and the current public docket in Outsourcing Facilities Association v. FDA.
What we did
- ✅ Read the primary sources — not summaries or blog posts
- ✅ Tested every public-record lookup link on April 23, 2026
- ✅ Kept editorial judgment separate from fact
- ✅ Separated authorities: FDA, USP, NABP, court rulings vs. manufacturer statements vs. trade groups
What we didn't do
- ✗ We did not test any pharmacy's product
- ✗ We did not rank or score named pharmacies on this page
- ✗ We did not include patient testimonials
- ✗ No fabricated "medically reviewed by" credentials
If you spot an error, a stale citation, or a broken public-record link, tell us. This page is only as useful as it is accurate, and accurate doesn't stay accurate without maintenance.
Frequently asked questions
Still not sure which GLP-1 program is right for you?
Take our free 60-second matching quiz. We'll ask you about your state, your goals, your budget, and whether you prefer the FDA-approved route or a compounded path — and match you with providers that fit.
Start the free 60-second matching quiz →Author and disclosure
Written by the Weight Loss Provider Guide Research Team. Weight Loss Provider Guide is an independent comparison resource for GLP-1 telehealth providers. We may earn a commission when readers sign up with providers we cover — see our editorial policy and how we make money. This guide is for consumer education; it is not medical advice, and it is not a substitute for a conversation with your prescribing clinician.
Changelog
- April 23, 2026: Initial publication. FDA warning-letter database links, state-board directory links, legal-status timeline, and adverse-event counts verified against primary sources on this date.
Sources
Primary sources cited on this page (verified April 23, 2026):
Federal regulators and statute
- U.S. Food and Drug Administration. "FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss."
- U.S. Food and Drug Administration. "FDA Clarifies Policies for Compounders as National GLP-1 Supply Begins to Stabilize." April 1, 2026.
- U.S. Food and Drug Administration. "FDA Intends to Take Action Against Non-FDA-Approved GLP-1 Drugs." February 6, 2026.
- U.S. Food and Drug Administration. "FDA Warns 30 Telehealth Companies Against Illegal Marketing of Compounded GLP-1s." March 3, 2026.
- U.S. Food and Drug Administration. "BeSafeRx: Your Source for Online Pharmacy Information."
- U.S. Food and Drug Administration. Warning Letter to Boothwyn Pharmacy LLC. January 16, 2026.
- U.S. Food and Drug Administration. "Registered Outsourcing Facilities" list.
- Outsourcing Facilities Association v. FDA, 4:25-cv-00174 (N.D. Tex. 2025).
- Federal Trade Commission. Final order against NextMed. December 2025.
Standards and accreditation
- U.S. Pharmacopeia. Chapters <795>, <797>, and <800>.
- ACHC. Pharmacy Compounding Accreditation Board (PCAB) standards and directory.
- National Association of Boards of Pharmacy. Compounding Pharmacy Accreditation and Verified Pharmacy Program.
- The Pew Charitable Trusts and NABP. State Oversight of Drug Compounding, 2018.
Published literature
Manufacturer communications (company statements, not independent research)
All statistics and dates on this page are linked to their source at point of use. All pricing is verified as of the publication date and should be re-checked at checkout.