Willow vs Ozempic: They're Not the Same Thing — Here's Which Fits You
By WPG Research Team | Pricing Last Verified: March 14, 2026 | Disclosure: We earn commissions from some links below. This does not affect our rankings. Full editorial policy
If you're comparing Willow vs Ozempic, you're not actually comparing two medications. You're comparing a telehealth weight-loss service to an FDA-approved prescription drug — and that difference changes everything about cost, safety, who qualifies, and which one you should actually choose.
We verified pricing from official sources, reviewed FDA records and regulatory filings, and analyzed real patient reviews across multiple platforms to build this comparison. Here's where we landed:
Willow is a telehealth platform that prescribes compounded GLP-1 medications (including compounded semaglutide) for weight loss. Plans start at $299/month, no insurance needed, and they'll prescribe to people who don't meet traditional BMI requirements. Their compounded medications are not FDA-approved.
Ozempic is an FDA-approved semaglutide injection made by Novo Nordisk, indicated for adults with type 2 diabetes. It's backed by large-scale clinical trials. Self-pay pricing through NovoCare recently dropped to $349/month — not the $1,000+ figure most articles still quote.
The Bottom Line
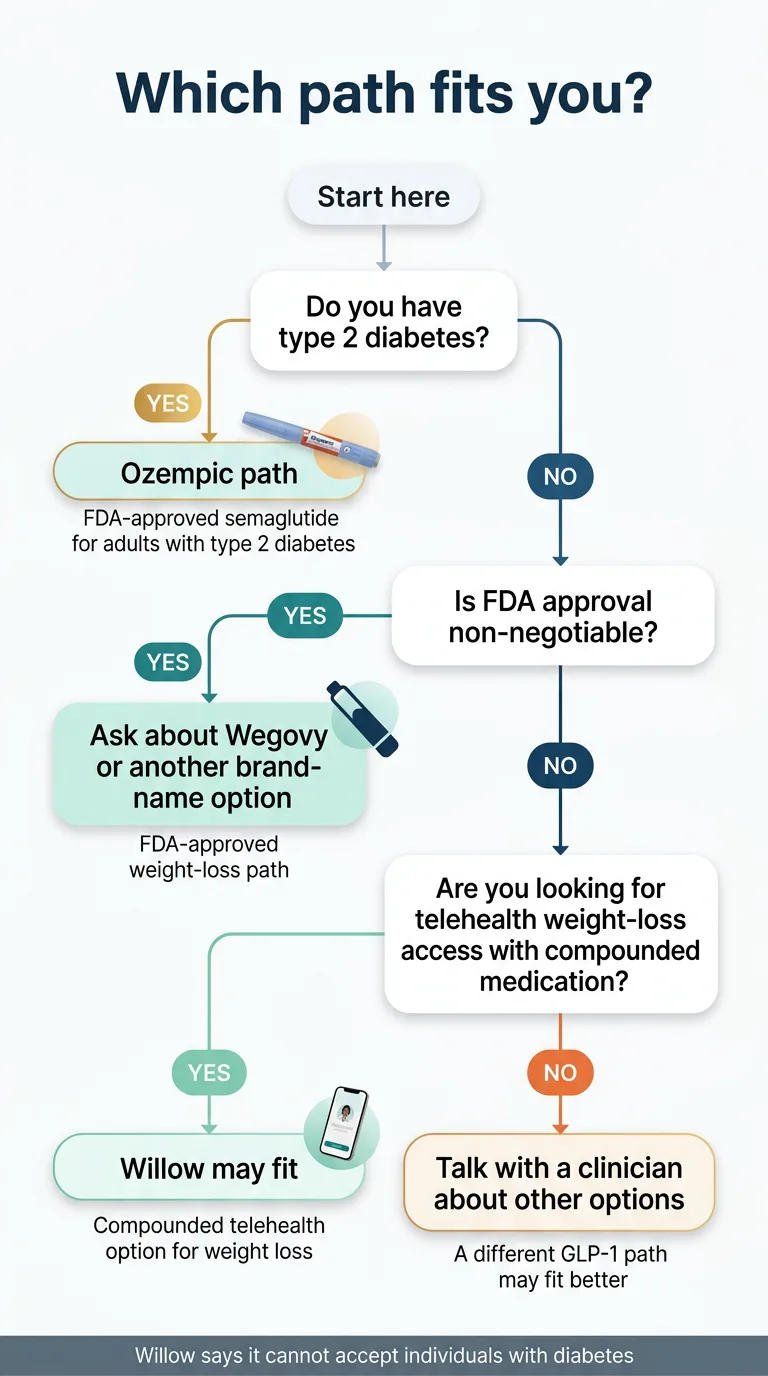
- Have type 2 diabetes? Ozempic is the medically aligned path. Willow explicitly cannot accept patients with diabetes.
- Want weight-loss access without insurance, fast, at a lower monthly cost? Willow may fit — if you understand you're getting a compounded product, not an FDA-approved one.
- Want FDA-approved semaglutide specifically for weight loss? You probably want Wegovy, not Ozempic. (We explain why most people confuse this below.)
- Needle-averse? Willow offers oral semaglutide tablets. But read our section on how those differ from FDA-approved oral options before deciding.
There's more to unpack here than any single comparison table can capture — regulatory changes, the oral semaglutide distinction most sites get wrong, what happened when the FDA issued warning letters to 30 telehealth companies (including Willow) in March 2026, and real pricing breakdowns that account for savings cards and intro offers.
Keep reading. We built this page so you wouldn't need another tab.

Willow vs Ozempic at a Glance: The Fast Answer
They are not the same kind of product. One is a service. The other is a specific medication. Here's how they actually stack up:
| Feature | Willow | Ozempic |
|---|---|---|
| What it is | Telehealth service prescribing compounded GLP-1 medications | FDA-approved semaglutide injection (Novo Nordisk) |
| FDA-approved? | No — compounded by licensed pharmacies, not FDA-reviewed as a finished product | Yes — for type 2 diabetes, cardiovascular risk reduction, and kidney disease risk in diabetic adults |
| Best for | Weight-loss access without insurance or BMI gatekeeping | Adults with type 2 diabetes; off-label weight loss with a prescription |
| Type 2 diabetes? | Cannot accept patients with diabetes | FDA-approved specifically for type 2 diabetes |
| Delivery formats | Injectable semaglutide, oral semaglutide (sublingual tablet), injectable tirzepatide | Injectable pen (pre-filled, pre-measured doses). FDA-approved Ozempic tablets exist as of Jan 2026 via Rx |
| Self-pay cost | Semaglutide: $299/mo; Tirzepatide: from $399/mo (all-inclusive) | $349/mo (NovoCare self-pay for 0.25–1 mg); $499/mo for 2 mg. Intro: $199/mo first 2 months |
| With insurance | Not covered by insurance (HSA/FSA accepted) | As low as $25/mo with commercial insurance + savings card |
| Speed to start | Same-day doctor review, ships in ~5–7 days total | Requires separate Rx from a doctor, then pharmacy fill |
| Cancellation | Cancel anytime (2 business days before next shipment) | No subscription — fill your Rx or don't |
| Available in | 35 states | All 50 states |
| Clinical trial data | None for Willow's specific compounded formulations | Extensive — SUSTAIN (diabetes); STEP and SELECT (Wegovy-dose semaglutide) |
| Regulatory flags | FDA warning letter sweep (March 2026, per industry reporting); NAD referral to regulators (Dec 2025) | None for the medication itself |
| What's included | Medication, doctor consult, unlimited messaging, free shipping, support | Medication only — doctor visits, support are separate |
Prices verified March 14, 2026 from startwillow.com, novocare.com, and ozempic.com. Prices change — we check monthly.
If you have type 2 diabetes, stop here.
Willow's FAQ explicitly states they cannot accept individuals with diabetes. Ozempic is FDA-approved for your condition and may be covered by insurance. Talk to your doctor about Ozempic, Mounjaro, or Wegovy. Read our GLP-1 guide for diabetes
Willow
$299/mo — All-Inclusive, No Insurance Needed
Are Willow and Ozempic Even the Same Thing?
No. And understanding why they're different is the single most important thing on this page.
Ozempic is a specific medication. Novo Nordisk manufactures it in FDA-inspected facilities. It went through years of clinical trials involving tens of thousands of patients. The FDA reviewed the safety data, the manufacturing process, the labeling, and the dosing. Every pre-filled pen contains an exact, verified dose of semaglutide. When your doctor prescribes Ozempic, you know exactly what you're getting because the FDA verified it.
Willow is a telehealth service — a platform that connects you with licensed physicians who can prescribe compounded GLP-1 medications. The medications are prepared by licensed compounding pharmacies, not by Novo Nordisk. Willow says this plainly on their site: their products "are not substitutes for FDA-approved medications such as... Ozempic."
Think of it this way: Ozempic is a specific car built in a specific factory with a specific safety rating. Willow is a car dealership that sells custom-built vehicles using similar parts — but those vehicles haven't been crash-tested by the same agency.
That doesn't automatically make one "good" and the other "bad." Compounding pharmacies are licensed, regulated by state boards, and serve a legitimate medical role. But the tradeoffs are real, and you deserve to understand them before you spend $299+ a month.
Here's the key distinction in one sentence: Ozempic is a product. Willow is a service that provides a different type of product.
The rest of this page breaks down exactly what that means for your wallet, your safety, and your results.
Who Should Choose Willow? Who Should Choose Ozempic?
Not everyone reading this page should end up in the same place. Here's how to find your lane:
You have type 2 diabetes → Ozempic (or talk to your doctor about alternatives)
This one isn't close. Ozempic is FDA-approved for type 2 diabetes. Willow explicitly says they cannot accept patients with diabetes. If you have diabetes, your path goes through your doctor, not through a telehealth weight-loss platform.
You want weight loss, no insurance, and the fastest online option → Willow may fit
If you're paying cash, don't have insurance that covers GLP-1 medications, and want a streamlined telehealth experience — Willow is designed for exactly this. Starting at $299/month all-inclusive (medication, doctor, shipping, support), it's one of the more affordable compounded options available. Just understand that you're getting a compounded product, not an FDA-approved one.
FDA approval is non-negotiable for you → Brand-name path (Ozempic, Wegovy, or Zepbound)
If the idea of using a non-FDA-approved medication doesn't sit right with you, that's a completely valid position. Ozempic (for diabetes), Wegovy (for weight loss), or Zepbound (tirzepatide for weight loss) are your FDA-approved options. They cost more without insurance, but they come with clinical trial data and FDA manufacturing oversight.
You want a pill instead of injections → Read our oral semaglutide section carefully
Willow offers a compounded sublingual semaglutide tablet. Ozempic now has FDA-approved tablets too (as of January 2026). These are not the same product, and the distinction matters more than most sites will tell you. We break this down in detail below.
You want the most regulatory certainty → Brand-name path
The regulatory landscape for compounded GLP-1s is shifting fast. The semaglutide shortage was officially resolved in February 2025, which limits the legal basis for compounding copies of FDA-approved drugs. Willow is still operating as of March 2026, but the future is uncertain. If you want a path that won't be affected by regulatory changes, FDA-approved medications are the safer bet.
Your insurance covers GLP-1s → Skip Willow, use your insurance
If your insurance covers Ozempic, Wegovy, or Zepbound, you could pay as little as $25/month with manufacturer savings cards. That's significantly less than Willow's $299/month — and you'd be getting an FDA-approved product. Always check your coverage first.
Wait — Do You Actually Mean Wegovy Instead of Ozempic?
This is a question most comparison pages never think to ask. But we've seen enough search data and reader questions to know: a lot of people who type "Ozempic" are really looking for semaglutide for weight loss. And if that's you, Ozempic might not actually be the right comparator.
Here's the distinction:
- Ozempic = semaglutide, FDA-approved for type 2 diabetes
- Wegovy = semaglutide, FDA-approved for weight management (obesity or overweight with weight-related conditions)
Same class of medication. Different approved uses. Different dosing. And in 2026, Wegovy now includes tablets — which makes the oral semaglutide conversation even more relevant.
If your primary goal is weight loss and you don't have type 2 diabetes, the most apples-to-apples FDA-approved comparison to Willow is actually Wegovy, not Ozempic. Many doctors prescribe Ozempic off-label for weight loss, which is common and legal, but it's worth knowing that Wegovy was specifically designed and tested for that purpose.
Why this matters for your decision: If your insurance covers Wegovy for weight management, that's likely a better (and possibly cheaper) path than either Willow or off-label Ozempic. Check with your insurer before assuming you need to go the compounded route.
What Is Willow, Exactly?
Willow (formally Willow Health Services, Inc.) is an online telehealth platform that specializes in prescribing compounded GLP-1 medications for weight loss. They've positioned themselves as the accessible, affordable alternative to brand-name medications — particularly for people who don't have insurance or don't meet traditional BMI requirements.
How Willow Works
- Take a 2-minute online quiz to check basic eligibility
- Share your health history and choose a medication plan
- Get reviewed by a licensed physician — often same-day
- Receive your medication by mail — pharmacy processing takes 2–4 business days, then 2–3 day delivery via FedEx or UPS
- Ongoing support — unlimited same-day messaging with your Willow doctor, follow-up appointments included
What Willow Prescribes
Willow offers three main medication options, all compounded by licensed U.S. pharmacies:
- Compounded semaglutide injection — weekly injection, $299/month
- Compounded semaglutide tablets — daily sublingual (under-the-tongue) dissolving tablet, $299/month
- Compounded tirzepatide injection — weekly injection, starts at $399/month
Formulations may include additional ingredients like Vitamin B12 or L-Carnitine, customized to the patient's treatment plan.
What's Included in the Price
The monthly fee covers everything: medication, physician consultation, treatment plan, unlimited doctor messaging, follow-up appointments, and free 2-day shipping. No hidden fees. HSA/FSA accepted. No insurance required.
Where Willow Is Available
As of March 2026, Willow operates in 35 states. They're not available nationwide. Check their site for the current list, as this changes periodically.
What Makes Willow Different
One thing that sets Willow apart from many compounded GLP-1 providers: oral semaglutide option. Most telehealth GLP-1 providers only offer injections. Willow's sublingual tablet option appeals to needle-averse patients. However, as we cover below, the user reviews on the oral form are notably more mixed than the injectable.
The Regulatory Picture
We believe in transparency, even when it's uncomfortable:
- March 2026: The FDA announced a sweep of 30 warning letters to telehealth companies over compounded GLP-1 marketing claims. Industry reporting identified Willow Health Services as one of the companies included. The FDA flagged language suggesting equivalence with FDA-approved drugs.
- December 2025: The National Advertising Division (NAD) found that Willow did not provide evidence to substantiate its express claims about health benefits, weight loss, or efficacy. Willow declined to commit to compliance, so NAD referred the matter to state attorneys general and federal regulators.
- Semaglutide shortage resolved: The FDA declared the semaglutide injection shortage over in February 2025, which limits the legal basis for compounding "essentially copies" of FDA-approved drugs. Enforcement discretion windows have largely expired.
What this means for you: Willow is still operating. Many patients report positive experiences, particularly with injectable semaglutide. But you should know the regulatory context before signing up — and have a backup plan in case the compounding landscape changes further.
What Is Ozempic, Exactly?
Ozempic is an FDA-approved semaglutide injection manufactured by Novo Nordisk. It's one of the most prescribed GLP-1 receptor agonist medications in the world.
What Ozempic Is Approved For
Ozempic is FDA-approved for three specific uses in adults with type 2 diabetes:
- Blood sugar management — in combination with diet and exercise
- Cardiovascular risk reduction — reducing the risk of major cardiac events like heart attack and stroke
- Kidney disease risk reduction — lowering the risk of worsening kidney disease and heart-related death
It is not FDA-approved for weight loss. However, doctors commonly prescribe it off-label for weight management, and its effectiveness for weight loss is well-documented in clinical trials. (The FDA-approved semaglutide for weight loss is Wegovy, not Ozempic.)
The 2026 Ozempic Tablets Update
In January 2026, the FDA approved a labeling change that brought tablets under the Ozempic brand name. Previously, oral semaglutide was only available as Rybelsus (a diabetes medication). This means Ozempic now includes both injection and tablet formulations — though both require a prescription and are primarily indicated for type 2 diabetes.
This matters because it's easy to confuse Willow's compounded sublingual tablets with FDA-approved Ozempic tablets. They are not the same thing. We break down the differences in detail below.
The Clinical Evidence Behind Ozempic
Ozempic's safety and effectiveness for type 2 diabetes have been evaluated in the SUSTAIN trial program (1–9), which demonstrated blood sugar reduction and weight loss in adults with type 2 diabetes.
Separately, the STEP trial program (1–5) used the higher-dose semaglutide formulation marketed as Wegovy — not Ozempic — and showed significant weight loss (average ~15% of body weight over 68 weeks) in adults with obesity or overweight. The SELECT trial demonstrated cardiovascular risk reduction in adults with obesity using that same Wegovy-dose formulation.
The distinction matters: the ~15% weight loss figure that gets cited everywhere comes from Wegovy's trial program, not Ozempic's. Ozempic's clinical data is primarily about diabetes management, though off-label weight loss is well-documented in practice.
No compounded semaglutide product has this kind of clinical trial data. It simply doesn't exist for pharmacy-prepared formulations.
How to Get Ozempic
Unlike Willow's all-in-one platform, getting Ozempic requires a few more steps:
- See a doctor (in-person or telehealth) and get a prescription
- Check your insurance coverage — many plans cover Ozempic for diabetes
- Fill your prescription at a pharmacy or through NovoCare's online pharmacy
- Apply the savings card if commercially insured (as low as $25/month)
It takes more effort. But you're getting an FDA-reviewed product with established clinical evidence.
Can I Use Willow If I Have Type 2 Diabetes?
No. Willow's FAQ is clear: they cannot accept individuals with diabetes. They do accept patients with pre-diabetes and hypertension.
This is a dealbreaker worth its own section because a surprising number of people searching "Willow vs Ozempic" have diabetes and are looking for a cheaper alternative to their current medication. If that's you, Willow is not an option.
Your best paths:
- Ozempic through insurance — if your plan covers it, copays can be as low as $25/month with the manufacturer savings card
- NovoCare self-pay — $349/month if you don't have coverage (or $199/month for the first two months on lower doses through March 31, 2026)
- Mounjaro — another GLP-1 option for type 2 diabetes, ask your doctor
- Patient assistance programs — Novo Nordisk offers a Patient Assistance Program (PAP) for eligible patients who meet income criteria
Don't settle for a provider that can't legally serve your condition. Your diabetes management is too important.
Which One Is FDA-Approved — and Why Does That Matter?
This might be the most important section on this page. Take two minutes here. It could save you real money and real worry.
What "FDA-Approved" Actually Means
When a drug is FDA-approved, it means the manufacturer submitted years of clinical trial data — safety, efficacy, manufacturing quality, labeling accuracy — and the FDA independently reviewed all of it before allowing the product to be sold. The FDA also inspects manufacturing facilities and monitors ongoing safety after approval.
Ozempic went through this entire process. The data is public. You can read the prescribing information, the clinical trial results, and the FDA's review documents yourself.
What "Compounded" Actually Means
Compounded medications are custom-prepared by licensed pharmacies, often to meet a specific patient need — like adjusting a dose, removing an allergen, or changing a delivery format. Compounding is legal, regulated by state pharmacy boards, and serves an important medical role.
However, compounded drugs do not go through FDA review. The FDA does not verify the safety, efficacy, or manufacturing quality of the finished compounded product. Willow acknowledges this directly on their website: their formulations "have not undergone clinical trials to evaluate their safety, efficacy, or therapeutic equivalence to any FDA-approved medications."

Why the Shortage Context Matters
From 2022 through early 2025, semaglutide injection products (Ozempic and Wegovy) were on the FDA's drug shortage list. During a shortage, compounding pharmacies are legally allowed to prepare copies of FDA-approved drugs to meet patient demand.
That shortage was officially resolved on February 21, 2025. Once resolved, the legal exception that permitted broad compounding of semaglutide copies began to close. The FDA set enforcement discretion windows — for 503A pharmacies through April 22, 2025, and for 503B outsourcing facilities through May 22, 2025.
Those windows have passed. Some compounders have pivoted to formulations they argue are "clinically different" (different doses, added ingredients like B12 or L-Carnitine), which may fall outside the "essentially a copy" restriction. This is the legal theory some providers, including Willow, appear to be operating under. It's an area of active legal and regulatory uncertainty.
What the FDA Has Flagged
The FDA has raised several specific concerns about compounded GLP-1 products:
- Dosing errors: Patients measuring and self-administering incorrect doses — a risk that doesn't exist with Ozempic's pre-filled, pre-measured pens
- Salt form concerns: Some compounders have used semaglutide sodium or semaglutide acetate rather than semaglutide base. The FDA says these are different active ingredients with "no known lawful basis for compounding"
- Adverse events: As of mid-2025, the FDA reported receiving 605 adverse event reports associated with compounded semaglutide and 545 associated with compounded tirzepatide (as of July 31, 2025)
Additionally, in March 2026, the FDA announced a sweep of 30 warning letters to telehealth companies over compounded GLP-1 marketing. Industry reporting identified Willow Health Services as one of the companies included.
The NAD Decision
Separately, in December 2025, the Better Business Bureau's National Advertising Division (NAD) reviewed Willow's advertising claims after a challenge from Novo Nordisk (the maker of Ozempic). NAD found that Willow did not provide sufficient evidence to substantiate its claims about health benefits, weight loss, or efficacy for its compounded semaglutide products. The studies Willow referenced were for FDA-approved products, which NAD said were "insufficient to validate claims for compounded alternatives."
When Willow declined to commit to complying with NAD's recommendations, NAD referred the matter to state attorneys general and other regulators.
How to Think About This
We're not here to scare you away from Willow. Thousands of people use compounded semaglutide and report genuine results, particularly with the injectable form.
But we are here to make sure you understand the tradeoff: with compounded medications, you're trading regulatory certainty and clinical trial validation for lower cost and easier access. For some people, that tradeoff makes sense. For others, it doesn't. Neither answer is wrong — as long as you're making the choice with your eyes open.
If FDA approval and clinical evidence are important to you, the brand-name path (Ozempic, Wegovy, Zepbound) is the right move, even if it costs more.
If access and affordability are your top priorities and you're comfortable with the regulatory context we've outlined, Willow offers a legitimate telehealth pathway with real physician oversight.
How Much Does Willow Cost vs Ozempic in 2026?
Let's kill the biggest myth right now: Ozempic does not cost $1,000/month for most people. That $997 list price is what manufacturers set before discounts — and almost nobody pays it. Most comparison sites still use this number, which makes Willow look like a dramatically better deal than it actually is.
Here's what you'll really pay:
Willow Pricing (Verified March 2026)
| Medication | Monthly Cost |
|---|---|
| Compounded semaglutide injection | $299/mo |
| Compounded semaglutide tablets (oral) | $299/mo |
| Compounded tirzepatide injection | Starts at $399/mo |
All Willow prices include medication, doctor consultation, treatment plan, unlimited messaging, follow-ups, and free 2-day shipping. Willow's FAQ notes that costs may vary by dosage and prescription. Source: startwillow.com product pages, verified March 14, 2026.
Ozempic Pricing (Verified March 2026)
| Scenario | Monthly Cost |
|---|---|
| List price (no insurance, no discounts) | ~$997/mo |
| NovoCare self-pay program (0.25 mg – 1 mg) | $349/mo |
| NovoCare self-pay (2 mg dose) | $499/mo |
| NovoCare intro offer (new self-pay patients, 0.25–0.5 mg) | $199/mo for first 2 months |
| Commercial insurance + savings card | As low as $25/mo |
Sources: novocare.com, ozempic.com. Savings card terms: max $100/mo savings, up to 48 months, for commercially insured patients. Patient Assistance Program available for qualifying individuals. Intro offer available through March 31, 2026.
The 6-Month Real-Cost Comparison
| Scenario | 6-Month Total | Includes |
|---|---|---|
| Willow (semaglutide injection) | $1,794 | Medication, doctor, shipping, support |
| Ozempic (NovoCare self-pay, starting dose) | $1,794 – $2,094 | Medication only — doctor visits separate |
| Ozempic (insurance + savings card) | $150 – $600 | Medication + copay reduction |
The real cost gap isn't what most sites claim.
When you compare actual prices (not list prices), Willow and Ozempic self-pay are surprisingly close over 6 months. The gap widens at higher doses. But if insurance covers Ozempic, it's substantially cheaper. The tradeoff between the two is about more than just price — it's about what you're getting for that price: compounded vs FDA-approved, all-in-one vs separate components, speed vs regulatory certainty. Which matters more depends entirely on your priorities.
Willow
$299/mo — Medication + Doctor + Shipping Included
Is Willow's Oral Semaglutide the Same as Ozempic Tablets?
No. And this distinction is one of the biggest things other comparison pages get wrong.
Three Different "Oral Semaglutide" Products Exist
This is confusing, so let's lay it out clearly:
- Willow's compounded sublingual semaglutide tablet — A daily dissolving tablet placed under the tongue. Prepared by a compounding pharmacy. Not FDA-approved. Not clinically tested as a specific product.
- Rybelsus (now also labeled as Ozempic tablets) — An FDA-approved oral semaglutide tablet. Swallowed whole on an empty stomach with no more than 4 ounces of water. Approved for type 2 diabetes. Made by Novo Nordisk.
- Wegovy tablets — FDA-approved oral semaglutide for weight management. Launched in 2025/2026.
These are three different products with different formulations, different administration methods, different approval statuses, and different evidence bases.

Why the Route of Administration Matters
Willow's sublingual tablet dissolves under the tongue and is absorbed through the oral mucosa (the lining of your mouth). FDA-approved Ozempic/Rybelsus tablets are swallowed whole and absorbed through the GI tract using a specific absorption-enhancing technology (SNAC).
This is not a trivial difference. The way a medication enters your body affects how much of it reaches your bloodstream (bioavailability), how consistently it works, and how predictable the dosing is. FDA labeling explicitly states that Rybelsus and Ozempic tablets are not substitutable on a milligram-to-milligram basis — even between FDA-approved oral formulations, dosing is not interchangeable.
Willow's own website states their compounded formulations have not undergone clinical trials to evaluate "therapeutic equivalence to any FDA-approved medications." In other words: we simply don't have the data to confirm that Willow's sublingual tablets deliver semaglutide as effectively as the FDA-approved versions.
What Patient Reviews Say About Willow's Oral Option
This is where the conversation gets real. We analyzed reviews across Trustpilot, ConsumerAffairs, and Reddit, and a clear pattern emerged:
- Willow's injectable semaglutide → Majority of reviews are positive. Users report appetite suppression, weight loss of 15–30+ pounds, and reduced food noise. Consistent with what you'd expect from semaglutide treatment.
- Willow's oral semaglutide tablets → Significantly more mixed. Multiple reviewers across platforms report zero weight loss after 2–5 months. Common complaints include: "didn't lose a single pound," "no appetite suppression," "felt like a mint candy with no effect." Some users were eventually told to switch to the injectable.
We're not saying the oral form can't work for anyone. But the pattern in user reviews is clear enough to warrant caution. If you choose Willow and weight loss is your primary goal, the injectable form appears to have substantially better user-reported outcomes.
Our Recommendation
If you're needle-averse and considering Willow's oral semaglutide specifically, set clear expectations:
- Understand it's a compounded sublingual tablet — not the same as FDA-approved oral semaglutide
- Give it a fair trial (most providers recommend at least 3 months), but track your results
- If you see no progress after 2–3 months, discuss switching to the injectable with your Willow doctor
- Consider whether FDA-approved Wegovy tablets might be a better fit, especially if you have insurance
Which One Is More Likely to Help With Weight Loss?
We're going to be very careful here — and very honest.
The Evidence for Ozempic (and Semaglutide Generally)
Semaglutide has strong clinical trial evidence for weight loss. The STEP trial program, using the higher-dose Wegovy formulation (semaglutide 2.4 mg weekly), showed average weight loss of approximately 15% of body weight over 68 weeks.
Ozempic uses lower doses (up to 2 mg weekly) and is indicated for diabetes, not weight loss. But off-label weight loss results are well-documented — typically 5–10% body weight loss, sometimes more at higher doses.
This evidence applies to FDA-approved semaglutide products — manufactured by Novo Nordisk, with verified dosing and quality.
The Evidence for Willow
Here's where we have to be straightforward: there is no clinical trial data for Willow's specific compounded formulations. None. Willow's own disclaimers confirm this.
Willow's compounded semaglutide is prepared by third-party pharmacies. The ingredients, formulation, manufacturing process, and quality controls are different from FDA-approved products. We cannot assume that what's been proven for Ozempic or Wegovy automatically applies to Willow's compounded versions.
What we do have is anecdotal evidence — patient reviews. And those paint a mixed picture:
- Injectable semaglutide from Willow: Most reviewers report real weight loss results. Many describe meaningful appetite reduction and weight loss consistent with what GLP-1 medications are known for.
- Oral semaglutide from Willow: Results are significantly more inconsistent. A notable portion of reviewers report no measurable weight loss.
The Evidence Hierarchy
When evaluating weight-loss claims, here's how we rank the evidence:
| Evidence Level | What It Means | Applies To |
|---|---|---|
| 1. FDA-approved labeling | Rigorously tested, independently verified | Ozempic, Wegovy, Zepbound |
| 2. Published clinical trials | Large-scale data on specific formulations | Ozempic (SUSTAIN), Wegovy (STEP/SELECT) |
| 3. Real-world prescription data | Doctors and patients reporting outcomes | Both FDA-approved and some compounded |
| 4. Patient reviews | Self-reported, unverified, variable quality | Willow, other compounded providers |
| 5. Marketing claims | What the company says on their website | Both — always verify independently |
Willow's evidence sits at levels 4 and 5. Ozempic's sits at levels 1 through 3. That's a real difference.
What Are the Side Effects and Safety Differences?
Both Willow's compounded semaglutide and Ozempic come with GLP-1-class side effects. But there are important differences in what we know and how the risks are managed.
Side Effects Common to GLP-1 Treatment Generally
These are reported across all semaglutide products, both FDA-approved and compounded:
- Nausea (most common, especially during dose escalation)
- Vomiting
- Diarrhea
- Constipation
- Abdominal pain
- Decreased appetite (this is also the mechanism of action for weight loss)
- Headache
- Indigestion
Most of these improve over time as your body adjusts. Both Willow and Ozempic prescribers typically start at a low dose and increase gradually to minimize side effects.
Ozempic-Specific Warnings (From FDA-Approved Labeling)
Because Ozempic went through FDA review, it carries specific labeled warnings:
- Boxed warning: Thyroid C-cell tumors observed in animal studies. Not recommended for patients with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2
- Pancreatitis risk
- Vision changes (diabetic retinopathy complications)
- Hypoglycemia risk when combined with certain diabetes medications
- Kidney injury related to dehydration from GI side effects
- Gallbladder problems
- Gastroparesis-related concerns
These warnings exist because the data was collected, analyzed, and published as part of the FDA approval process. It's not that Ozempic is necessarily riskier — it's that we actually have the data to identify and quantify these risks.
Compounded Semaglutide-Specific Concerns
For compounded products like Willow's, the risk profile is less well-defined because the products haven't gone through FDA safety review. However, the FDA has flagged several concerns specific to compounded GLP-1 medications:
- Dosing errors: Patients self-measuring and administering incorrect doses. Ozempic's pre-filled pens eliminate this risk entirely.
- Salt form issues: Some compounders have used semaglutide sodium or semaglutide acetate instead of semaglutide base. The FDA has stated these are different active ingredients with unknown safety profiles.
- Manufacturing variability: Without FDA manufacturing oversight, batch-to-batch consistency may vary.
- Adverse event reports: The FDA has received hundreds of adverse event reports related to compounded semaglutide, though the exact breakdown by severity isn't fully public.
Willow states they use the base form of semaglutide (not salt forms) and work with licensed compounding pharmacies. However, independent verification of these claims is limited compared to FDA-approved products.
The Safety Bottom Line
If safety certainty is your top priority, FDA-approved semaglutide (Ozempic, Wegovy) provides a known, documented risk profile backed by clinical data and ongoing FDA monitoring.
If you choose a compounded option like Willow, you're accepting some additional uncertainty — which may be reasonable for many people, but it's a tradeoff you should make consciously.
Regardless of which path you choose: Start at a low dose, escalate slowly, stay hydrated, eat smaller meals, and keep your prescribing doctor informed about any side effects. Both Willow and Ozempic prescribers can adjust your treatment if side effects are problematic.
How Fast Can I Start — and How Hard Is It to Cancel?
Willow: Speed Is a Major Selling Point
- Day 1: Complete the online quiz and health history (2–5 minutes)
- Day 1: Physician reviews your case (often same-day)
- Days 2–5: Pharmacy compounds and processes your medication (2–4 business days)
- Days 4–8: Medication delivered via FedEx or UPS (2–3 day shipping)
Total time from sign-up to medication in hand: roughly 5–8 days in most cases.
Willow's program auto-renews monthly. To cancel, you need to submit a cancellation request at least 2 business days before your next processing date to avoid being charged for the next month. There's no long-term contract — cancel anytime.
Ozempic: More Steps, More Gatekeeping
- Step 1: Schedule an appointment with a doctor (could be same-day with telehealth, or weeks with traditional care)
- Step 2: Get a prescription (if the doctor agrees it's appropriate for your condition)
- Step 3: Check insurance coverage / apply for savings card
- Step 4: Fill the prescription at a pharmacy
- Step 5: Pick up or receive by mail
Total time: varies wildly — from a few days (telehealth + fast pharmacy) to several weeks (traditional doctor + insurance prior authorization).
There's no subscription to cancel with Ozempic. You simply fill or don't fill your prescription. If you stop filling it, you stop getting it.
The Friction Comparison
Willow wins on speed and simplicity. One platform, one payment, medication at your door in about a week.
Ozempic wins on flexibility. No subscription, no auto-renewal, no cancellation process to navigate.
Will Insurance Cover Willow or Ozempic?
Willow
Insurance does not cover Willow's compounded medications. This is standard across the compounded GLP-1 space — insurance companies do not pay for non-FDA-approved compounded drugs. Willow's Terms of Service state that neither Willow nor its pharmacy partners submit claims to third parties, and users agree not to submit reimbursement claims for the services.
HSA and FSA funds are accepted. No insurance is required to use Willow.
Ozempic
Insurance coverage for Ozempic varies significantly:
- Type 2 diabetes: Most commercial insurance plans and Medicare Part D cover Ozempic for diabetes. Most commercial health plans cover GLP-1s for diabetes.
- Weight loss (off-label): Coverage is much spottier. Coverage for weight management is less common but expanding.
- Medicare: Covers for diabetes. Does not cover for weight loss under current rules.
- Medicaid: Varies by state.
With commercial insurance + manufacturer savings card: As low as $25/month (max savings of $100/month for up to 48 months).
Self-pay through NovoCare: $349/month for most doses (or $199/month for the first two months on lower doses through March 31, 2026).
Patient Assistance Program (PAP): Novo Nordisk offers free or reduced-cost Ozempic for eligible patients who meet income requirements and are prescribed for type 2 diabetes.
The Insurance Decision Tree
- Does your insurance cover GLP-1s? → If yes, get Ozempic or Wegovy. Much cheaper than Willow.
- No insurance or no GLP-1 coverage? → Compare NovoCare self-pay ($349/mo) vs Willow ($299/mo) and decide based on your comfort with compounded vs FDA-approved.
- Have HSA/FSA funds? → Both options accept them.
Where Is Willow Available Right Now?
As of March 2026, Willow operates in 35 states: Arizona, California, Colorado, Connecticut, Florida, Georgia, Hawaii, Idaho, Illinois, Indiana, Iowa, Kentucky, Maine, Maryland, Massachusetts, Michigan, Minnesota, Missouri, Montana, Nebraska, Nevada, New Hampshire, New Jersey, New York, North Carolina, Ohio, Oklahoma, Oregon, Pennsylvania, South Carolina, Tennessee, Texas, Virginia, Washington, and Wisconsin.
This list was verified on March 14, 2026 from Willow's Terms and Conditions. Willow adds and removes states periodically — check their website for the most current availability.
Ozempic, by contrast, is available through any licensed pharmacy in all 50 states. If you're in a state where Willow doesn't operate and you want a telehealth GLP-1 option, other providers may serve your area. See our full telehealth provider comparison
What Happens After Promo Pricing Ends — or If I Stop?
Ozempic Intro Pricing Has an Expiration Date
Novo Nordisk's $199/month introductory offer for new self-pay patients applies only to the first two monthly fills of 0.25 mg and 0.5 mg doses, and it ends March 31, 2026. After that, self-pay reverts to $349/month (or $499 for the 2 mg dose).
If you're considering Ozempic, be aware that your costs will increase after the intro period and as your dose escalates.
Willow's Prices Increase With Dose
Willow keeps it simple at $299/month for most dose levels. But if you need a higher dose — 7 mg injectable semaglutide or above 12 mg oral — the price jumps to $399/month. Tirzepatide at higher doses reaches $549/month.
The Harder Question: What Happens When You Stop?
This applies to both Willow and Ozempic equally, and it's something most comparison pages gloss over. But it might be the most important financial and health consideration you face.
Research on FDA-approved semaglutide shows that most patients regain approximately two-thirds of their lost weight within a year of stopping the medication. Appetite typically returns. Food noise comes back. The biological signals that drove weight gain don't disappear just because you lost the weight.
The STEP 1 extension trial is the clearest data point: participants who stopped semaglutide after 68 weeks regained approximately two-thirds of their prior weight loss by week 120 (Wilding et al., Diabetes, Obesity and Metabolism, 2022; PMID: 35441470). This isn't a failure of the medication — it's how our biology works. Obesity is increasingly understood as a chronic condition, not a one-time problem to fix.
This doesn't mean the medication was a waste — many patients maintain improved metabolic health, reduced A1C levels, and better cardiovascular markers even after partial weight regain. But it does mean you should think of GLP-1 treatment as potentially an ongoing commitment, not a 3-month quick fix.
Both Willow and Ozempic prescribers should help you with a tapering plan if you decide to stop. Willow specifically recommends working with their physicians to gradually decrease your dose rather than stopping abruptly, noting that abrupt discontinuation can lead to increased appetite and weight regain.
Plan for the long game. When you're comparing Willow's $299/month to Ozempic's $349/month, don't just think about month one. Think about months 6, 12, and 24. Factor in dose escalations that may increase your monthly cost. And have a realistic conversation with your prescribing doctor about what a sustainable long-term plan looks like — whether that means ongoing medication, a transition to maintenance dosing, or a gradual taper with lifestyle changes to support your new weight.
The best time to think about your exit strategy is before you start.
So Which One Wins for Your Situation?
Neither one "wins" universally. The right choice depends on who you are. Here's our verdict after verifying pricing, reading FDA records, analyzing hundreds of reviews, and building this comparison from the ground up:
Best for Type 2 Diabetes: Ozempic
Willow can't serve you — their platform explicitly excludes patients with diabetes. Ozempic is FDA-approved for exactly your condition, with clinical trial data backing its effectiveness for blood sugar management and cardiovascular risk reduction. Check your insurance first — with a savings card, you may pay as little as $25/month.
Best for Self-Pay Weight-Loss Access: Willow
If you don't have insurance that covers GLP-1s, want a streamlined online experience, and are comfortable with compounded medication, Willow offers a genuinely convenient, lower-cost path at $299/month all-in. The process is fast (medication at your door in about a week), the support is included (unlimited doctor messaging). Just go in with your eyes open about FDA status and pick the injectable — the oral tablets have too many mixed reviews to recommend confidently.
Best for FDA-Approved Weight Loss: Wegovy
Neither Willow nor Ozempic is the perfect answer here. Wegovy is semaglutide FDA-approved specifically for weight management, available in both injection and tablet form, and backed by the STEP clinical trial program showing ~15% body weight loss. Ask your doctor — and check if your insurance covers it.
Best for Maximum Regulatory Certainty: Brand-Name Path
If FDA approval, clinical trial data, and manufacturing oversight are important to you, the brand-name route (Ozempic, Wegovy, or Zepbound) is the clear choice — even if it costs more. You're paying for verified dosing, ongoing FDA safety monitoring, and a product that has been tested in tens of thousands of patients. That peace of mind has real value.
Your move. If Willow sounds like the right fit after everything you've read, their eligibility quiz takes 2 minutes. You'll know immediately if you qualify, and a doctor reviews your case the same day.
Willow
$299/mo — Same-Day Doctor Review, Free 2-Day Shipping
Same-day doctor review if appropriate. No insurance required. Free 2-day shipping. Cancel anytime.
If Ozempic is the better path for you, check your insurance coverage and visit novocare.com for current savings options.
How We Researched This Comparison
Here's exactly how this comparison was built:
Pricing: Checked directly on startwillow.com, novocare.com, and ozempic.com on March 14, 2026. Compared list prices, self-pay programs, savings card terms, and intro pricing.
FDA records: Drug shortage database, warning letter archives, compounding policy guidance, the February 2025 shortage resolution declaratory order, and the January 2026 Ozempic tablet labeling approval (supplement s030).
Regulatory filings: BBB National Programs / NAD decision regarding Willow Health Services (Case #7488, November 2025, published December 2025); Novo Nordisk challenge filing; Outsourcing Facilities Association v. FDA litigation docket.
Clinical evidence: Published FDA prescribing information for Ozempic (NDA 209637) and peer-reviewed publications of the STEP, SUSTAIN, and SELECT clinical trial programs. Trial data is cited only for the specific formulations that were tested — not as evidence for compounded products.
Patient reviews: Reviewed across Trustpilot, ConsumerAffairs, and Reddit (r/semaglutide, r/Ozempic, r/GLP1Agonists). The injectable-vs-oral review pattern we describe reflects what multiple reviewers report across platforms.
What this page does not do:
- Present trial data for FDA-approved products as evidence for compounded formulations
- Make health claims not substantiated by the relevant product's own evidence base
- Provide medical advice — always consult a qualified healthcare provider
Affiliate relationship: We may earn a commission when you sign up through our Willow links. This does not influence our analysis. We do not have an affiliate relationship with Novo Nordisk (Ozempic). Full editorial policy
Willow vs Ozempic: Frequently Asked Questions
Still Not Sure Which GLP-1 Program Is Right for You?
Answer 5 quick questions about your health goals, insurance status, and preferences. We'll match you with the GLP-1 provider and program that fits your situation — whether that's Willow, a brand-name path, or something else entirely. No email required.
This article is for informational purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider before starting any medication, including GLP-1 receptor agonists. Individual results vary. GLP-1 medications carry risks and side effects that should be discussed with your doctor.
Willow's compounded medications are prepared by licensed compounding pharmacies. They are not FDA-approved and have not undergone clinical trials to evaluate their safety, efficacy, or therapeutic equivalence to FDA-approved medications. They are not substitutes for Ozempic, Wegovy, or other FDA-approved semaglutide products.
Medical DisclaimerAdvertising DisclosureEditorial StandardsHow We Rank
Related Articles
What Is GLP-1?
A complete beginner's guide to GLP-1 medications, how they work, and what to expect.
GLP-1s for Weight Loss
How GLP-1 receptor agonists promote weight loss through appetite reduction and metabolic changes.
Learn About GLP-1
Educational hub covering GLP-1 science, medications, eligibility, and treatment options.
History of GLP-1 Drugs
The complete timeline of GLP-1 drug development from discovery to modern weight loss treatments.
Semaglutide vs Tirzepatide
Head-to-head comparison of semaglutide and tirzepatide for weight loss effectiveness and cost.
Retatrutide vs Tirzepatide
Triple agonist vs dual agonist: trial data, side effects, availability, and what you can actually do today.