Disclosure: Some links on this site are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you. Thank you for supporting our site.·For informational purposes only—not medical advice.
Microdosing Tirzepatide: Chart, Dosage & Complete Guide (2026)
Microdosing tirzepatide means taking doses below the standard 2.5 mg starting point — typically 0.5 mg to 2 mg per week — with the goal of reducing side effects, cutting costs, or maintaining weight loss at a lower dose. It's off-label, unproven in clinical trials, and increasingly popular.
Here's what you need to know before you do anything: no large outcomes trial has ever evaluated tirzepatide below 2.5 mg as a weight-loss strategy. Early-phase studies tested lower doses for pharmacokinetics and tolerability, but the pivotal trials that proved tirzepatide works started at 2.5 mg. We do know from the landmark SURMOUNT-1 trial that even the lowest therapeutic dose (5 mg) produced roughly 15% body weight loss over 72 weeks — which tells you you don't need to max out at 15 mg to see real results. And real-world physicians are prescribing microdoses. Dr. Craig Koniver, profiled in Medscape in early 2025, starts women at 1.5 mg and men at 1.8 mg, with a goal of 2 pounds per week or less.
So microdosing isn't made up. But it isn't proven either. And the difference between “my doctor prescribed a careful low dose” and “I'm eyeballing doses from a TikTok chart” is the difference between reasonable medicine and a real safety risk.
This guide gives you everything: the actual dosing charts, what trial data exists at each dose level, the honest risks, the 2026 legal reality around compounded tirzepatide, real cost numbers, and a clear decision framework — so you can have an informed conversation with a provider instead of guessing.

Illustration only. Not medical advice. Actual dosing must be determined by a licensed provider.
Microdosing vs. Standard Dosing at a Glance
| Factor | Microdosing Tirzepatide | Standard Tirzepatide Dosing |
|---|---|---|
| Typical Starting Dose | 0.5–2 mg/week | 2.5 mg/week (FDA-approved) |
| Maintenance Range | 1–5 mg/week | 5–15 mg/week |
| Studied in Clinical Trials? | Early-phase PK/tolerability only; no large outcomes trials validating microdosing | Yes — SURMOUNT and SURPASS trials |
| Primary Goal | Maintenance, side effect reduction, metabolic support | Significant weight loss, blood sugar control |
| Average Weight Loss (trial data) | Unknown | 15–21% body weight over 72 weeks |
| Side Effects | Expected milder (anecdotal only) | Nausea, diarrhea, constipation (dose-dependent) |
| Requires Compounded Version? | Usually (brand pens start at 2.5 mg) | No — FDA-approved pens and vials available |
| Monthly Cost Range | $150–$350 (telehealth/compounded) | $299–$1,100+ (brand, cash price) |
| Best For | Weight maintenance, GI-sensitive patients, metabolic support | Obesity, Type 2 diabetes, significant weight loss goals |
What Is Microdosing Tirzepatide?
Let's get the definition straight, because social media has muddied it.
Tirzepatide is the active ingredient in two FDA-approved brand-name medications: Mounjaro (approved for Type 2 diabetes) and Zepbound (approved for weight management and obstructive sleep apnea). Both are made by Eli Lilly and administered as once-weekly injections.
The FDA-approved dosing schedule starts at 2.5 mg per week for 4 weeks — and that 2.5 mg dose is specifically labeled as an initiation dose, not a maintenance dose. After 4 weeks, patients move to 5 mg, and from there can increase in 2.5 mg increments every 4+ weeks up to a maximum of 15 mg per week (Source: Zepbound prescribing information, FDA 2025).
Microdosing means going below that starting line. Taking 0.25 mg, 0.5 mg, 1 mg, or 1.5 mg per week — doses the FDA has never reviewed, tested, or approved.
Some people also use “microdosing” to mean staying at 2.5 mg or 5 mg long-term instead of escalating higher. That's technically not microdosing — it's lower-dose maintenance — but the term gets used loosely. For this guide, we'll cover both: true sub-2.5 mg microdosing and lower-dose maintenance strategies. For a deeper dive into how GLP-1 medications work, see our beginner's guide.

FDA-labeled doses vs. off-label microdosing. Illustration only.
Three things that matter right away:
- You can't microdose with standard Mounjaro or Zepbound pens. They're single-dose, pre-filled devices delivering fixed amounts (2.5 mg through 15 mg). There's no way to reliably extract a partial dose. Microdosing below 2.5 mg requires either compounded tirzepatide (legally restricted in 2026 — more on that below) or Zepbound single-dose vials, which can be drawn in partial amounts with a syringe under provider guidance.
- “Microdosing” in the GLP-1 world has nothing to do with psychedelic microdosing. Different concept entirely. Here it simply means sub-standard dosing of a prescription weight-loss medication.
- This is an off-label practice. Off-label prescribing is legal and common in medicine — your doctor does it all the time. But it means you're in territory without the safety net of large clinical trials. That makes provider supervision non-negotiable, not optional.
Why Are People Microdosing Right Now?
People don't search “microdosing tirzepatide” out of idle curiosity. Something specific brought them here. Understanding the real reasons helps you figure out if you're making a decision for the right reasons — or reacting to hype.
The Side-Effect Escape
This is the biggest driver. Standard tirzepatide titration causes nausea in roughly 25–29% of patients, diarrhea in 19–23%, and constipation in 11–17%, according to Zepbound weight-loss trial data. Those numbers are dose-dependent — they get worse as doses climb. Some people hit a wall at 5 mg or 7.5 mg and can't push higher without feeling terrible. Microdosing promises a gentler on-ramp. Whether it delivers is another question. For strategies on managing GI symptoms, see our GLP-1 nausea guide and Zepbound nausea relief tips.
The Cost Crunch
Brand-name Zepbound lists at roughly $1,000+ per month without insurance. Even with Eli Lilly's LillyDirect vial pricing ($299 for 2.5 mg, $399–$449 for higher doses as of December 2025), it's expensive. Compounded tirzepatide ran $150–$600 per month when it was widely available. Stretching a lower dose = stretching a budget. The math is appealing — but only if the lower dose actually works. See our GLP-1 cost breakdown for the full pricing picture.
The Maintenance Question
You hit your goal weight on 10 mg or 15 mg. Now what? The SURMOUNT-4 trial showed that people who stopped tirzepatide regained substantially — an average of 14 percentage points of body weight within a year, meaning most participants regained the majority of the weight they had originally lost. The medication works best as an ongoing treatment, not a short course. But do you need the same dose that got you there? Many patients and physicians think not — and that's where stepping down to a micro or low dose enters the picture.
The Menopause and Metabolic Health Angle
A growing number of physicians, including OB-GYN Dr. Shamsah Amersi, advocate microdosing specifically for perimenopausal and menopausal women. The rationale: hormonal transitions can meaningfully reduce metabolic rate — though the magnitude varies widely from person to person — and these patients often need metabolic support more than aggressive weight loss. Microdosing may offer insulin sensitization and appetite regulation without overwhelming a body already in flux.
The Social Media Effect
Andy Cohen told TODAY he lost weight microdosing a GLP-1 drug. Rebel Wilson described using microdoses for weight maintenance. Reddit's r/GLP1microdosing community has over 1,700 members sharing protocols. The conversation has gone mainstream — and the information quality ranges from genuinely helpful to dangerously wrong.
Not every reason here is a bad reason. But every single one requires medical supervision to execute safely.
How Does Tirzepatide Work — and Why Does the Dose Matter?
Understanding the mechanism isn't just academic. It explains why microdosing might work, why it might not, and why guessing your dose is risky.
Tirzepatide is the only FDA-approved dual GIP/GLP-1 receptor agonist. Your body naturally produces two hormones after you eat — GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide). Both signal your brain that food is arriving. GLP-1 slows your stomach emptying, makes you feel full, and helps regulate blood sugar. GIP works alongside it to fine-tune insulin response and may help reduce fat storage. Learn more about this mechanism in our What Is GLP-1? guide.
Most GLP-1 drugs (like semaglutide/Ozempic/Wegovy) only target the GLP-1 receptor. Tirzepatide hits both. That dual action is why it consistently outperforms semaglutide in head-to-head trials — the SURPASS-2 trial showed tirzepatide produced significantly greater weight loss and HbA1c reductions than semaglutide at comparable doses (Frías et al., New England Journal of Medicine, 2021). For a full comparison, see our semaglutide vs tirzepatide guide.
Here's why dose matters for microdosing: Tirzepatide's effects are dose-dependent. Both receptors need sufficient activation to produce meaningful appetite suppression, insulin sensitization, and metabolic changes. At ultra-low doses — say, 0.25 mg — there's a real question of whether you're reaching the threshold needed to activate either receptor in a clinically meaningful way.
The honest answer: we don't know the minimum effective dose. No trial has tested it. What we do know is that the 5 mg dose (the lowest therapeutic dose) works powerfully. The 2.5 mg initiation dose shows some early-phase effects in trial data. And below 2.5 mg? It's educated guessing based on pharmacological principles, not evidence.
Tirzepatide has a half-life of approximately 5 days (Source: PMC pharmacokinetics review). That means it takes about 4 weeks of consistent weekly dosing to reach steady-state levels in your blood. This has a practical implication for microdosing: you need to stay at a dose long enough to actually evaluate whether it's working. Changing doses every week or two — which some online charts suggest — doesn't give the drug time to stabilize.
What Does the Research Actually Say?
We're going to be ruthlessly honest here, because your health decisions deserve better than cherry-picked data. We'll use a simple evidence rating:
- Level A — Supported by randomized clinical trial data
- Level B — Supported by real-world observational data or published medical commentary
- Level C — Based on pharmacological reasoning and mechanism, not direct testing
- Level D — Anecdotal (patient reports, social media, physician interviews)
What Clinical Trials Show (Level A)
The SURMOUNT-1 trial is the gold standard. Here's what tirzepatide achieved at each dose over 72 weeks:
| Weekly Dose | Avg. Body Weight Loss | Study | Evidence Level |
|---|---|---|---|
| Placebo | -3.1% | SURMOUNT-1 (Jastreboff et al., NEJM 2022) | A |
| 5 mg | -15.0% | SURMOUNT-1 | A |
| 10 mg | -19.5% | SURMOUNT-1 | A |
| 15 mg | -20.9% | SURMOUNT-1 | A |
Two things jump out. First, even at 5 mg — the lowest therapeutic dose — patients lost an average of 15% body weight. That's substantial. You don't need to push to 15 mg to get meaningful results.
Second, look at the gap between 5 mg and 15 mg: only about 6 percentage points. The biggest jump is from placebo to 5 mg. That matters for the microdosing conversation — it suggests a floor effect exists, but the precise floor hasn't been mapped.
What Real-World Data Shows (Level B)
A large real-world study analyzed Optum insurance claims for 20,998 tirzepatide users. By their sixth fill, 74.2% of patients were on doses below 10 mg, and the average weight loss was 11.9% at 6 months (Source: PMC). This tells us something the trials don't: in the real world, most people don't escalate to the max dose, and they still lose meaningful weight.
In 2025, Diabetes Care published the first academic commentary specifically discussing microdosing of semaglutide and tirzepatide. The authors described it as “potentially useful” for certain populations but noted it “lacks clinical guideline support” (Source: PMC12178608).

Risk ladder: from safest (FDA-labeled) to riskiest (unsupervised). Illustration only.
What Physicians Report (Level D)
Dr. Craig Koniver, a South Carolina family medicine physician, told Medscape in February 2025 that he has all 200 of his GLP-1 patients on microdoses. He starts women at 1.5 mg of tirzepatide, men at 1.8–2 mg, and targets weight loss of 2 pounds per week or less. Some patients stay at these lower doses; others eventually move higher. He describes significant variation between patients.
Dr. Shamsah Amersi, an OB-GYN, has published protocols starting semaglutide patients as low as 0.05–0.1 mg and applies similar principles to tirzepatide for perimenopausal and menopausal women.
What We Genuinely Don't Know
- Whether doses below 2.5 mg produce meaningful receptor activation in most people
- Long-term outcomes (weight trajectory, metabolic markers) for microdosing specifically
- Whether microdosing would affect cardiovascular outcomes (CV outcomes data for tirzepatide at therapeutic doses are emerging and context-dependent)
- How microdosing affects different populations differently (age, sex, metabolic status)
- Optimal microdosing schedules (weekly vs. twice weekly vs. extended intervals)
Bottom line: There's enough signal — from lower-dose trial arms, real-world prescribing patterns, and physician experience — to make microdosing a reasonable conversation with your doctor. But calling it “proven” would be dishonest.
Microdosing Tirzepatide Chart, Dosage & Schedule
This is probably why you're here. So let's give you what you came for — with important context.
Disclaimer: These charts are based on published physician protocols and patient reports. They are not FDA-approved dosing guidelines and no large outcomes trials have validated these specific schedules. Any microdosing protocol should be supervised by a licensed healthcare provider who monitors your individual response. Do not self-prescribe.
The Standard FDA Titration Schedule (Your Baseline Reference)
| Week | Weekly Dose | Purpose |
|---|---|---|
| Weeks 1–4 | 2.5 mg | Initiation only — not approved for maintenance |
| Weeks 5–8 | 5 mg | First therapeutic dose |
| Weeks 9–12 | 7.5 mg | If needed based on response and tolerability |
| Weeks 13–16 | 10 mg | Further escalation if needed |
| Weeks 17–20 | 12.5 mg | Further escalation if needed |
| Weeks 21+ | 15 mg | Maximum approved dose |
Source: Zepbound prescribing information, FDA 2025. Dose increases occur after minimum 4 weeks at each level.
Physician-Reported Microdosing Starting Points
Rather than a rigid month-by-month schedule, here's what physicians who prescribe microdoses have reported:
- Dr. Craig Koniver (Medscape, Feb 2025): Starts women at ~1.5 mg/week, men at ~1.8–2 mg/week. Targets no more than 2 lbs/week weight loss. Titrates based on individual response. (Reported in physician interview; not a clinical guideline.)
- Dr. Shamsah Amersi (OB-GYN): Uses similar low-dose principles for perimenopausal and menopausal women, starting well below standard doses and adjusting based on symptoms and metabolic markers.
The starting dose, pace of increase, and maintenance level should be determined by your prescribing provider based on your individual situation — not copied from a chart online.
Maintenance Step-Down Protocol (After Reaching Goal Weight)
For people who've already lost weight on standard doses (5–15 mg) and want to maintain at a lower level:
| Phase | Weekly Dose | Duration | Goal |
|---|---|---|---|
| Active loss | 5–15 mg | Until target weight reached | Weight reduction |
| Taper | Reduce by 2.5 mg every 4–6 weeks | 8–16+ weeks | Gradual step-down |
| Low-dose maintenance | 2.5–5 mg | Ongoing | Sustain weight loss, manage appetite |
| Micro-maintenance | 1–2.5 mg (some patients) | Ongoing — provider monitored | Lowest effective dose |
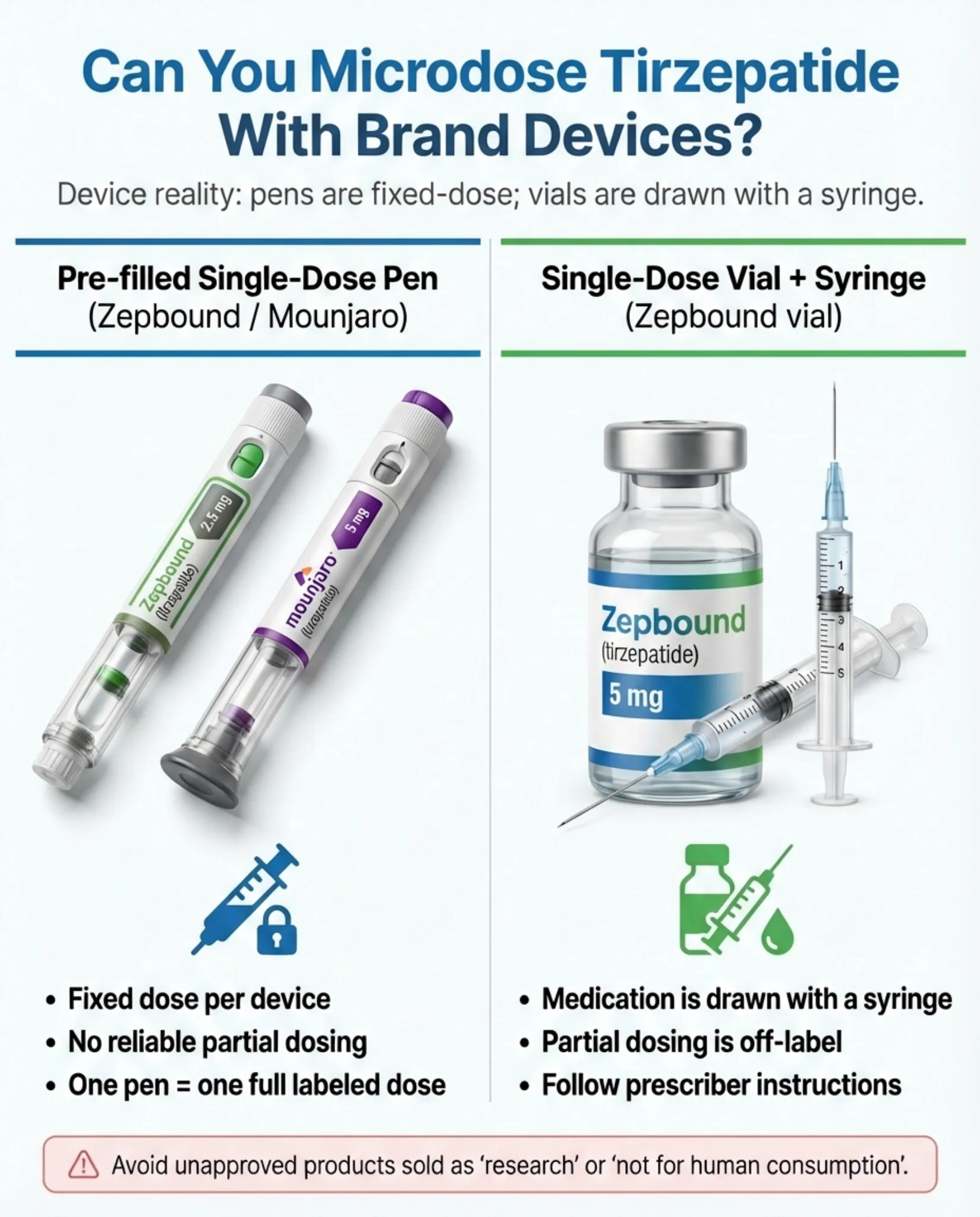
Device comparison: pre-filled pens vs. vials. Illustration only.
Critical Safety Notes on These Charts
Units matter — and confusion kills. The FDA has flagged dosing errors with compounded GLP-1 medications as a serious safety issue. Different compounding pharmacies use different concentrations, which means the same mg dose can require different mL volumes depending on the product. Don't convert mg → mL → units yourself — ask your prescriber and pharmacist for the exact draw instructions for your specific concentration. Getting this wrong is how people end up hospitalized.
Brand-name pens can't be microdosed. Mounjaro and Zepbound pens are single-use, fixed-dose devices. You cannot split them. Zepbound single-dose vials (2.5 mg and 5 mg) can be drawn in partial amounts with a syringe — but they don't contain preservatives and are designed for single use, so dose-splitting introduces contamination risk. This should only be done under direct provider instruction.
Stick with your dose long enough to evaluate it. Tirzepatide takes about 4 weeks to reach steady state. Changing your dose every week based on how you feel day-to-day isn't microdosing — it's unpredictable dosing. Stay at each level for at least 4 weeks before assessing.
Should You Microdose? A Decision Framework
Not everyone searching this term has the same goal. The right approach depends on where you are and what you're trying to accomplish.
Microdosing May Make Sense If You...
- Have reached your weight loss goal and want to maintain. This is the strongest use case. Stepping down to a lower dose under medical supervision is reasonable and aligns with how many physicians manage long-term GLP-1 therapy. You're not trying to get results from a micro dose — you're trying to keep results at a lower dose.
- Are extremely GI-sensitive. If you've tried standard tirzepatide and the nausea, diarrhea, or vomiting was unbearable even at 2.5 mg, a supervised ultra-slow start may help your body adapt.
- Want metabolic support, not dramatic weight loss. If you're in perimenopause, have mild insulin resistance, or want appetite regulation without losing 40 pounds, a lower dose may be appropriate under provider guidance.
- Are cost-conscious and your provider agrees. Using Zepbound vials at a lower dose with provider oversight can stretch your medication budget. This is fundamentally different from buying unregulated compounded products to save money.
Standard Dosing Is Better If You...
- Need to lose significant weight (20+ pounds). Microdosing has no evidence for major weight loss. Every clinical trial showing 15–21% body weight reduction used 5–15 mg doses.
- Have Type 2 diabetes requiring blood sugar control. The ADA's 2025 Standards of Care recommend tirzepatide at therapeutic doses for glycemic management. Microdosing for diabetes is not supported by any guideline.
- Are tempted to self-dose without medical supervision. This is where microdosing gets genuinely dangerous. Without a provider monitoring your response, adjusting your dose, and catching problems early, you're experimenting on yourself with a powerful medication.
Don't Take Tirzepatide at Any Dose If You...
- Have a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2) — this is a boxed warning contraindication
- Are pregnant or planning to become pregnant — animal studies showed fetal harm
- Have severe gastroparesis or history of pancreatitis — discuss risks with your doctor first
Not sure where you fall? Check your GLP-1 eligibility or use the decision tool below.
Microdosing Tirzepatide Decision Tool
Answer 3 questions to get a personalized next step and specific questions to bring to your clinician. No dosing instructions provided.
Should You Microdose Tirzepatide?
Answer 3 quick questions. Get a personalized recommendation and specific questions to bring to your clinician.
Takes about 30 seconds. Educational only — not medical advice. No dosing instructions provided.
Microdosing Tirzepatide Side Effects
Side effects are the #1 reason people search for microdosing. Let's break down what the data shows and what lower-dose users actually report. For comprehensive side effect management, see our GLP-1 SOS guide.
What Clinical Trials Tell Us (Standard Doses)
From Zepbound weight-loss trials, the most common side effects at therapeutic doses (5–15 mg) include:
| Side Effect | Frequency (approx.) | Dose-Dependent? |
|---|---|---|
| Nausea | ~25–29% | Yes — worse at higher doses and during escalation |
| Diarrhea | ~19–23% | Yes |
| Constipation | ~11–17% | Yes |
| Vomiting | ~6–12% | Yes |
| Decreased appetite | ~15–20% | Yes |
| Injection site reactions | ~3–7% | Mild in most cases |
Source: Zepbound prescribing information (weight management trials)
Most GI side effects peak during dose escalation and tend to diminish within 2–4 weeks at a stable dose. This is actually the clinical rationale behind the standard titration schedule — it's already designed to minimize side effects by starting low and going slow.
What Microdosing Users Report (Anecdotal)
Community forums, physician interviews, and patient reports consistently describe:
- Significantly less nausea at doses under 2 mg — some report none at all
- Appetite changes that feel more like “the food noise turned down” rather than complete appetite loss
- More stable energy levels compared to higher doses
- Fewer instances of the extreme fullness and aversion to food that some higher-dose users describe
- Some users at ultra-low doses (under 1 mg) report feeling essentially nothing
Serious Risks That Apply at Any Dose
These are from the Zepbound prescribing information and apply regardless of how much you take:
- Thyroid C-cell tumors: Tirzepatide caused thyroid tumors in rats at clinically relevant doses. It's unknown whether this occurs in humans. This is the FDA's boxed warning — the most serious warning category.
- Pancreatitis: Rare but serious. Seek immediate medical attention for severe, persistent abdominal pain.
- Gallbladder disease: Increased risk observed in clinical trials.
- Acute kidney injury: Usually secondary to severe dehydration from GI side effects.
- Serious allergic reactions: Including anaphylaxis (rare).
Risks Specific to Microdosing
- Dosing errors. This is the biggest practical danger. Compounded products come in varying concentrations, and converting between mg, mL, and “units” on a syringe has led to serious adverse events. The FDA has received multiple reports of adverse events, some requiring hospitalization, related to dosing errors with compounded injectable GLP-1 products. As of July 31, 2025, the FDA had received 545 adverse event reports associated with compounded tirzepatide and 605 associated with compounded semaglutide — and these are likely underreported since most compounding pharmacies aren't required to submit them (Source: FDA).
- Contamination. Compounded products lack FDA manufacturing oversight. Sterility and potency can vary.
- Inconsistent results. Sub-therapeutic dosing may produce unpredictable effects — some weeks you feel something, some weeks nothing.
- False reassurance. A lower dose isn't a zero-risk dose. All of the serious warnings above still apply.
When to Stop and Call Your Doctor
If you experience any of the following, stop your injection and contact your provider immediately:
- Severe abdominal pain that doesn't resolve
- Signs of allergic reaction (swelling of face/throat, difficulty breathing, rapid heartbeat)
- Persistent vomiting you can't keep fluids down through
- Signs of dehydration (dark urine, dizziness, rapid heart rate)
- A lump or swelling in your neck, hoarseness, or difficulty swallowing
Microdosing Tirzepatide Cost in 2026
Cost drives a lot of microdosing interest, so let's do the real math. For the full picture across all GLP-1 options, see our GLP-1 cost guide and cheapest tirzepatide options.
Current Pricing Landscape
| Access Path | Monthly Cost (Approx.) | What You Get | Microdosing-Friendly? |
|---|---|---|---|
| Zepbound vials (brand, LillyDirect self-pay) | $299 (2.5 mg) / $399 (5 mg) / $449 (7.5–15 mg with Journey Program*) | FDA-approved medication; Eli Lilly direct pricing (reduced Dec 2025) | Partial — vials allow syringe draws at smaller amounts |
| Zepbound pens (brand, cash) | $550–$1,100 | FDA-approved, fixed-dose pens | No — can't split pen doses |
| Zepbound (with insurance + savings card) | As low as $25/mo (varies) | Depends on formulary, prior auth, copay tier | Same as cash path for dose flexibility |
| MEDVi (telehealth) | Varies — check current pricing | Licensed physician, personalized tirzepatide dosing | Yes — flexible protocols |
| iThriveMD microdosing program | $195/mo | 3-month program, monthly provider visits, up to 3 mg/week | Yes — dedicated microdosing |
| WeightCare micro program | $349/mo | Board-certified physician, medication included, max 5 mg/week | Yes — specific micro protocol |
| Compounded tirzepatide | $150–$600/mo (where still legally available) | Non-FDA-approved, variable quality | Yes — but legally restricted in 2026 |
*Journey Program pricing ($449 for 7.5–15 mg) requires refilling within 45 days of previous delivery. Regular self-pay prices without the Journey Program range from $599 (7.5 mg) to $1,049 (15 mg).
Last verified: February 2026. Pricing changes frequently — always confirm directly with the provider.
The Cost Math Most People Miss
Microdosing can save money, but not always as much as it seems:
Brand-name math: As of December 2025, a Zepbound 2.5 mg vial costs $299/month and 5 mg costs $399/month through LillyDirect's self-pay program. That's already competitive with many compounded options — and it's FDA-approved medication. However, taking less than the full vial amount is off-label and requires your prescriber's specific instructions. Don't assume you can split a single-dose container for later use — ask your clinician and pharmacist what's appropriate for your specific product and format.
The hidden cost of going cheap: Unregulated compounded products might cost $150/month, but if the potency is inconsistent, you may be paying for medication that isn't doing anything. Or worse — a dosing error from incorrect concentration could land you in an ER. The FDA's documented adverse events from compounded GLP-1s are a real financial (and health) risk.
Insurance tip: If your insurance covers Zepbound, even a high copay might be competitive with out-of-pocket microdosing programs. Eli Lilly's savings card can bring copays on Zepbound pens to as low as $25/month for eligible patients whose commercial insurance covers Zepbound. Check Zepbound.com for current savings programs before assuming you need an alternative path.
Can You Still Get Compounded Tirzepatide in 2026?
This section matters because microdosing below 2.5 mg almost always requires compounded medication — and the legal landscape has changed dramatically. For more on this topic, see our best compounded tirzepatide providers guide.
The Timeline
- December 2022: FDA adds tirzepatide to the drug shortage list. Compounding pharmacies can legally produce copies.
- October 2024: FDA removes tirzepatide from the shortage list, declaring the shortage resolved.
- December 2024: FDA confirms resolution, sets grace periods for compounders to transition.
- February 18, 2025: Grace period ends for 503A state-licensed pharmacies.
- March 19, 2025: Grace period ends for 503B outsourcing facilities.
- May 7, 2025: Federal court upholds FDA's decision (Outsourcing Facilities Association v. FDA, Northern District of Texas).
- 2025–2026: Eli Lilly files lawsuits against individual compounding pharmacies.
Where Things Stand Now
As of 2026, the FDA states that compounded GLP-1 drugs “should only be used” when a patient's medical needs cannot be met by an FDA-approved drug, or when the FDA-approved drug is not commercially available. Compounding for cost savings, dose flexibility, or convenience alone does not meet this standard.
The practical reality: Some compounding pharmacies continue to operate under various legal theories — court challenges, “combination” products that aren't exact copies, or other formulation differences. Additionally, the FDA has warned that some compounders use semaglutide salt forms (semaglutide sodium or acetate), which are different active ingredients than approved semaglutide, and the FDA says it is not aware of a lawful basis for their use in compounding (Source: FDA). The legal ground for compounded GLP-1 products is less stable than it was during the shortage era.
Red Flags to Watch For
- “For research use only” or “not for human consumption” — The FDA has specifically warned about unapproved GLP-1 products marketed with these labels while including dosing instructions for human use.
- No licensed prescriber involved — A legitimate pharmacy requires a prescription from a licensed provider.
- Unclear ingredients or additives — Some compounded versions include B vitamins, NAD+, or other additives. These combinations haven't been studied for safety.
- Pricing that seems impossibly low — Under $100/month should raise serious questions about what you're actually getting.
- No verifiable pharmacy license — Check your state board of pharmacy website. For 503B facilities, verify on the FDA's registered outsourcing facility list.

Discussion checklist for your doctor appointment. Illustration only — actual product packaging may vary.
Microdosing Tirzepatide for Maintenance
If there's one scenario where microdosing makes the most clinical sense, it's this: you've already lost weight on standard-dose tirzepatide and want to maintain your results at a lower, more sustainable dose.
Why Maintenance Matters
The SURMOUNT-4 trial answered a critical question: what happens when you stop tirzepatide? Participants who switched from tirzepatide to placebo after 36 weeks gained approximately 14 percentage points of body weight over the following year (relative to their Week 36 weight), meaning most regained the majority of their initial weight loss. Those who continued treatment maintained their results.
The takeaway is clear: tirzepatide works best as an ongoing treatment, not a short course. But here's the nuance — the trial didn't test whether maintenance requires the same dose that caused the loss.
What the Data Suggests About Lower Maintenance Doses
A 2025 mathematical modeling study published in a peer-reviewed journal suggested that extending dosing intervals to every two weeks at maintenance could sustain weight loss for many patients. While this isn't the same as weekly microdosing, it supports the concept that full-dose escalation isn't necessarily required long-term.
The real-world Optum data is also telling: 74.2% of tirzepatide patients were on doses below 10 mg by their sixth refill, suggesting that in clinical practice, many providers and patients naturally settle at moderate doses rather than pushing to the maximum.
How to Step Down Safely
If you're currently on standard-dose tirzepatide and want to explore maintenance at a lower dose, here's what a responsible approach looks like:
- Don't change your dose without your provider's involvement. This isn't optional.
- Taper gradually. Reduce by 2.5 mg every 4–6 weeks while monitoring your weight, appetite, and energy.
- Set a floor. Have a conversation with your provider about what the lowest acceptable maintenance dose might be for your situation.
- Track your data. Weigh yourself weekly (same day, same conditions). If you see consistent upward trend over 4+ weeks, your maintenance dose may be too low.
- Be honest about food noise. If cravings and “food noise” return significantly, your dose may need to come back up. That's not failure — it's information.
- Don't neglect the lifestyle foundation. Tirzepatide — at any dose — works best alongside protein-adequate nutrition (aim for 0.7–1 g per pound of lean body mass), resistance training to preserve muscle, adequate sleep (7+ hours), and stress management. These aren't optional add-ons. At a maintenance microdose, they become even more important because the pharmaceutical support is lighter.
- Have a “floor plan.” Before you taper, agree with your provider on what signals should trigger a dose increase. This might be: regaining more than 5 pounds over 4 weeks, return of significant food cravings, or lab values moving in the wrong direction. Having pre-set triggers removes the emotional guesswork.
The Maintenance Mindset Shift
Here's something that trips people up: maintenance isn't just a lower dose of what you were doing before. It's a fundamentally different phase. During active weight loss, the medication is doing heavy metabolic lifting. During maintenance, you're looking for the minimum dose that keeps your appetite regulated, your metabolic markers stable, and your weight roughly where you want it — plus or minus a few pounds.
Some patients maintain beautifully at 2.5 mg. Others need 5 mg. A small number find they need to go back to their active-loss dose to prevent regain. There's no failure in that — everyone's biology responds differently. The goal is finding your floor, not someone else's.
Microdosing Tirzepatide Results: What People Actually Report
People search “microdosing tirzepatide reviews” and “microdosing tirzepatide results” because they want to know what it's actually like. Clinical trials don't answer that — they measure averages. Here's what individual patients and online communities consistently describe.
Important context: These are anecdotal reports from online communities (Reddit r/GLP1microdosing, patient forums), physician interviews, and telehealth program feedback. They are not clinical evidence. Individual results vary significantly based on dose, body composition, metabolic health, diet, and activity level.
What People Report at Sub-2.5 mg Doses
- “The food noise turned down, not off.” The most consistent report. At doses under 2 mg, people describe thinking less about food between meals, having an easier time stopping when full, and fewer cravings — but still having a normal appetite.
- Minimal to no nausea. The most commonly cited benefit. Many people who couldn't tolerate standard titration describe smooth experiences at lower doses.
- Slow, gradual weight changes. People report 1–4 pounds per month at micro doses — not the rapid early losses seen at therapeutic doses. Some report no weight change at all but improved metabolic markers (lower fasting glucose, better energy).
- Maintenance success. The strongest positive reports come from people who lost weight at higher doses and stepped down. Many describe holding their weight stable at 2.5–5 mg after active loss at 10–15 mg.
- Some people feel nothing. At doses below 1 mg, a meaningful number of people report no discernible effects. This aligns with the pharmacological uncertainty about minimum effective dose.
- Energy and mood. Some users report improved energy and more stable mood, though this is hard to separate from the effects of weight loss and better eating habits.
What Doesn't Get Reported as Often
- Stalls. People who start at micro doses for weight loss (not maintenance) frequently report frustrating plateaus. This is expected — the evidence for significant weight loss at sub-therapeutic doses is weak.
- Eventual need to increase. Many microdosing starters eventually move up to standard doses when results plateau. This isn't failure — it may simply mean a higher dose is needed for their goals.
Bottom line: Anecdotal reports are encouraging for maintenance and side-effect management, less convincing for primary weight loss. Take online reviews seriously as signal, not as evidence.
Microdosing Tirzepatide for Menopause, Inflammation, and Other Emerging Uses
These are the searches that come up in autocomplete: “microdosing tirzepatide for menopause,” “for inflammation,” and “for PCOS.” Here's what we actually know.
Menopause and Perimenopause
Hormonal transitions during perimenopause and menopause can meaningfully reduce basal metabolic rate, though the magnitude varies widely from person to person. Add shifting hormone levels, increased insulin resistance, and redistribution of body fat, and you have a population genuinely struggling with metabolic changes.
Some physicians — notably Dr. Amersi — specifically advocate lower-dose GLP-1 protocols for this group, arguing that menopausal women need metabolic stabilization more than aggressive weight loss, and that lower doses minimize the GI distress that can compound hormonal symptoms.
The evidence grade: Level D (physician protocols, not clinical trials). No study has tested tirzepatide microdosing specifically in menopausal women. The reasoning is sound, but the data is absent.
Inflammation
GLP-1 receptor agonists have demonstrated anti-inflammatory effects in clinical studies — in Zepbound's obstructive sleep apnea studies, high-sensitivity C-reactive protein (hs-CRP) decreased versus placebo. The mechanism likely involves improvements in insulin sensitivity, reduced visceral fat, and direct anti-inflammatory signaling through GLP-1 receptors.
Whether these effects persist at micro doses is genuinely unknown. The inflammatory benefits observed in trials used therapeutic doses at 10–15 mg. The evidence grade: Level C (mechanism-based reasoning, no direct microdosing data).
PCOS and Insulin Resistance
Tirzepatide's insulin-sensitizing effects may benefit women with PCOS, where insulin resistance drives many symptoms. Some providers use lower doses to address metabolic dysfunction without the dramatic weight changes that PCOS patients sometimes find destabilizing.
The evidence grade: Level C (plausible mechanism, no microdosing-specific data).
The Longevity and “Metabolic Optimization” Trend
Some telehealth companies market microdosing for “healthy aging,” “longevity,” and “metabolic optimization” in people who don't have obesity or diabetes. This is the most speculative end of the spectrum. The clinical trials that established tirzepatide's benefits enrolled people with obesity or Type 2 diabetes. Extrapolating those benefits to metabolically healthy people at micro doses is a leap without a bridge.
The evidence grade: Level D (marketing claims, minimal supporting data).
If you're exploring any of these use cases, work with a provider who understands the speculative nature and is ordering appropriate lab work — metabolic panel, HbA1c, lipid panel, CRP, thyroid function — to objectively measure whether the medication is doing what you hope. For more on the latest tirzepatide developments including oral formulations, see our dedicated guide.
Microdosing Tirzepatide vs. Semaglutide: Which Is Better for Low Doses?
If you're considering microdosing, you might be weighing tirzepatide against semaglutide (Ozempic/Wegovy). Here's how they compare specifically for low-dose use. For the full comparison at standard doses, see our semaglutide vs tirzepatide guide.
| Factor | Tirzepatide | Semaglutide |
|---|---|---|
| Mechanism | Dual GIP + GLP-1 | GLP-1 only |
| Lowest Brand-Name Dose | 2.5 mg/week (Zepbound/Mounjaro) | 0.25 mg/week (Ozempic) |
| Easier to Microdose? | Harder — pens start at 2.5 mg; need vials or compounded | Easier — Ozempic multi-dose pens allow “click counting” for smaller doses |
| Weight Loss at Standard Doses | ~15–21% (SURMOUNT-1) | ~12–15% (STEP trials) |
| Cost (brand, cash) | $299–$449+/mo (LillyDirect vials) | Varies; Ozempic/Wegovy shortage resolved Feb 2025 |
| Compounding Availability (2026) | Severely restricted | Similar enforcement timelines to tirzepatide |
The key tradeoff: Semaglutide is technically easier to microdose because Ozempic comes in multi-dose pens that patients can dial to smaller increments. Tirzepatide's single-dose pens don't offer that flexibility. But tirzepatide's dual mechanism may deliver more metabolic benefit per milligram — it consistently outperforms semaglutide in head-to-head trials.
If microdosing convenience is your primary concern, semaglutide has a practical edge. If metabolic potency matters more, tirzepatide may be worth the extra logistics.
How to Talk to Your Doctor About Lower Doses
A lot of people feel awkward bringing this up. You don't want to sound like you're prescribing to yourself or second-guessing your doctor. Here's a practical approach.
What to Say
You don't need a script — but if it helps, something like this works:
“I've been reading about lower-dose and maintenance-dose approaches to tirzepatide. My goal is [reduce side effects / maintain my weight loss / manage costs]. I'd like to discuss whether adjusting my dose might make sense for my situation, and I want to do it safely with your guidance.”
That's it. You're not demanding a microdose. You're opening a conversation and putting the medical judgment where it belongs — with your provider.
What to Bring to the Appointment
- Your weight trend over the last 4–8 weeks
- A list of side effects you're experiencing and their severity
- Your current dose and how long you've been on it
- Your goal (maintaining weight? reducing side effects? reducing cost?)
- Any lab work you've had recently
What a Good Response Looks Like from Your Provider
A provider who's worth working with will:
- Take your concerns seriously instead of dismissing them
- Explain the tradeoffs of dose adjustment for your specific situation
- Set up a monitoring plan (weight checks, labs, follow-up timing)
- Be transparent about what's evidence-based versus experimental
- Adjust based on your response over time, not a fixed one-size-fits-all protocol
If your current provider won't discuss dose adjustments at all, it may be worth seeking a second opinion from a physician experienced with GLP-1 medications.
Where to Get Supervised Tirzepatide Safely in 2026
You've read the guide. You've decided you want to explore tirzepatide — whether at standard or lower doses. Here's how to actually do it. For a broader comparison of all programs, see our best tirzepatide online and best GLP-1 programs guides.
Option 1: Your Own Doctor
This is the best starting point for most people. Any licensed physician can prescribe tirzepatide. If you have an existing relationship with a PCP, endocrinologist, or obesity medicine specialist, start there. They know your medical history, can order labs, and can adjust dosing based on your response over time.
Best for: People with an established provider relationship and insurance that covers tirzepatide.
Option 2: Telehealth Provider with Flexible Dosing
If you don't have a local provider experienced with GLP-1s, or you want the convenience of virtual care, telehealth platforms offer licensed physician consultations and shipped medication.
MEDVi offers personalized tirzepatide dosing with licensed physicians who tailor your protocol to your goals — whether that's standard titration or a more conservative approach. No forced escalation schedules. See our MEDVi tirzepatide reviews for a detailed breakdown.
Best for: People who want medical supervision + dosing flexibility + the convenience of telehealth.
Option 3: Brand-Name Zepbound Vials Through Your Provider
Eli Lilly's Zepbound vials (starting at $299 for 2.5 mg through LillyDirect) can be drawn in partial doses with a syringe under provider guidance. This may be the most accessible path to lower-dose tirzepatide in 2026, since compounding access is restricted.
Best for: Cost-conscious patients who want FDA-approved medication at a lower dose point.
What to Look for in Any Provider
- A licensed physician (MD or DO) making dosing decisions — not just a health coach or NP without physician oversight
- Transparent pricing with no hidden fees or forced upsells
- The ability to adjust your dose based on your response, not a rigid protocol
- Access to messaging your provider between scheduled visits
- Clear disclosure about whether medication is FDA-approved brand-name or compounded
Red Flags
- No physician involved in prescribing decisions
- Medication sourced from unverifiable suppliers
- Monthly cost under $100 (raises questions about product legitimacy)
- Guarantees of specific weight-loss outcomes
- Pressure to commit to long-term contracts before starting
How We Researched This Guide
Transparency matters, especially for health content. Here's exactly what we reviewed and how. For more about our editorial standards, see our Learn hub.
- Primary sources: FDA prescribing information for Zepbound (2025 label) and Mounjaro (2025 label), accessed via FDA.gov. These are the authoritative documents on approved dosing, safety, and contraindications.
- Clinical trial data: SURMOUNT-1 (Jastreboff et al., NEJM, 2022), SURMOUNT-4 (Aronne et al., JAMA, 2024), and SURPASS-2 (Frías et al., NEJM, 2021). These are the landmark trials establishing tirzepatide's efficacy and safety at therapeutic doses.
- Published medical commentary: Diabetes Care 2025 commentary on microdosing semaglutide/tirzepatide (PMC12178608). This is the first peer-reviewed academic discussion of the microdosing concept.
- Real-world data: Optum insurance claims study of 20,998 tirzepatide users (PMC11964993).
- Physician interviews and protocols: Medscape coverage of Dr. Craig Koniver's microdosing approach (February 2025), Dr. Shamsah Amersi's published GLP-1 microdosing protocols.
- Regulatory sources: FDA drug shortage database, FDA safety alerts on compounded GLP-1 medications, NCPA statements on compounding enforcement, McDermott Will & Emery legal analysis of OFA v. FDA (June 2025).
- Pricing data: Verified directly from provider websites and Eli Lilly's published vial pricing. Verification date noted on all pricing claims.
What we didn't do: We did not test these medications. We are not physicians. This guide is informational and does not constitute medical advice. We earn affiliate commissions from some providers mentioned — this is disclosed at the top of the page. We include providers we don't earn from, and we will always tell you when your own doctor or a non-affiliate option is the better path.
Frequently Asked Questions
Sources
- Zepbound (tirzepatide) prescribing information, FDA 2025. FDA.gov
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387(3):205-216. (SURMOUNT-1)
- Aronne LJ, Sattar N, Horn DB, et al. Continued Treatment With Tirzepatide for Maintenance of Weight Reduction in Adults With Obesity. JAMA. 2024;331(1):38-48. (SURMOUNT-4)
- Frías JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N Engl J Med. 2021;385(6):503-515. (SURPASS-2)
- Tirzepatide pharmacokinetics review. PMC10962491
- Real-world tirzepatide dosing patterns (Optum claims, 20,998 patients). PMC11964993
- Microdosing semaglutide/tirzepatide commentary, Diabetes Care 2025. PMC12178608
- FDA's Concerns About Unapproved GLP-1 Drugs Used for Weight Loss. FDA.gov
- FDA Registered Outsourcing Facilities list. FDA.gov
- Medscape: Dr. Craig Koniver microdosing protocols (February 2025).
- Outsourcing Facilities Association v. FDA, N.D. Tex., May 2025.
Disclosure: Some links on this site are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you. Thank you for supporting our site.
This guide is reviewed and updated as new clinical data, pricing changes, and regulatory developments emerge. Last comprehensive review: February 2026.