Incretin: What It Is, How It Works & Drugs Explained (2026)
Disclosure: Some links on this site are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you. Thank you for supporting our site. For informational purposes only—not medical advice.

On this page
Incretin in 60 Seconds
Incretins are gut hormones that help control your blood sugar after you eat. When food enters your digestive system, your intestines release two main incretins—GLP-1 and GIP—which signal your pancreas to release insulin. In healthy people, the incretin effect is often estimated to account for roughly 50–70% of insulin secretion after oral nutrient intake (estimates vary by study and measurement method).[1]
In people with type 2 diabetes, this incretin system doesn't work properly. That discovery led to the development of medications like Ozempic, Wegovy, Mounjaro, and Zepbound that either mimic these hormones or help them last longer in your body.
| What Incretins Are | What They Do | Why They Matter |
|---|---|---|
| Gut hormones (GLP-1 and GIP) | Stimulate insulin release after eating | A large portion of meal-related insulin depends on them |
| Released by intestinal cells | Slow stomach emptying, reduce appetite | Foundation for diabetes and weight loss medications |
| Broken down within minutes by DPP-4 enzyme | Work only when blood sugar is elevated | Explains why newer drugs were designed to resist breakdown |
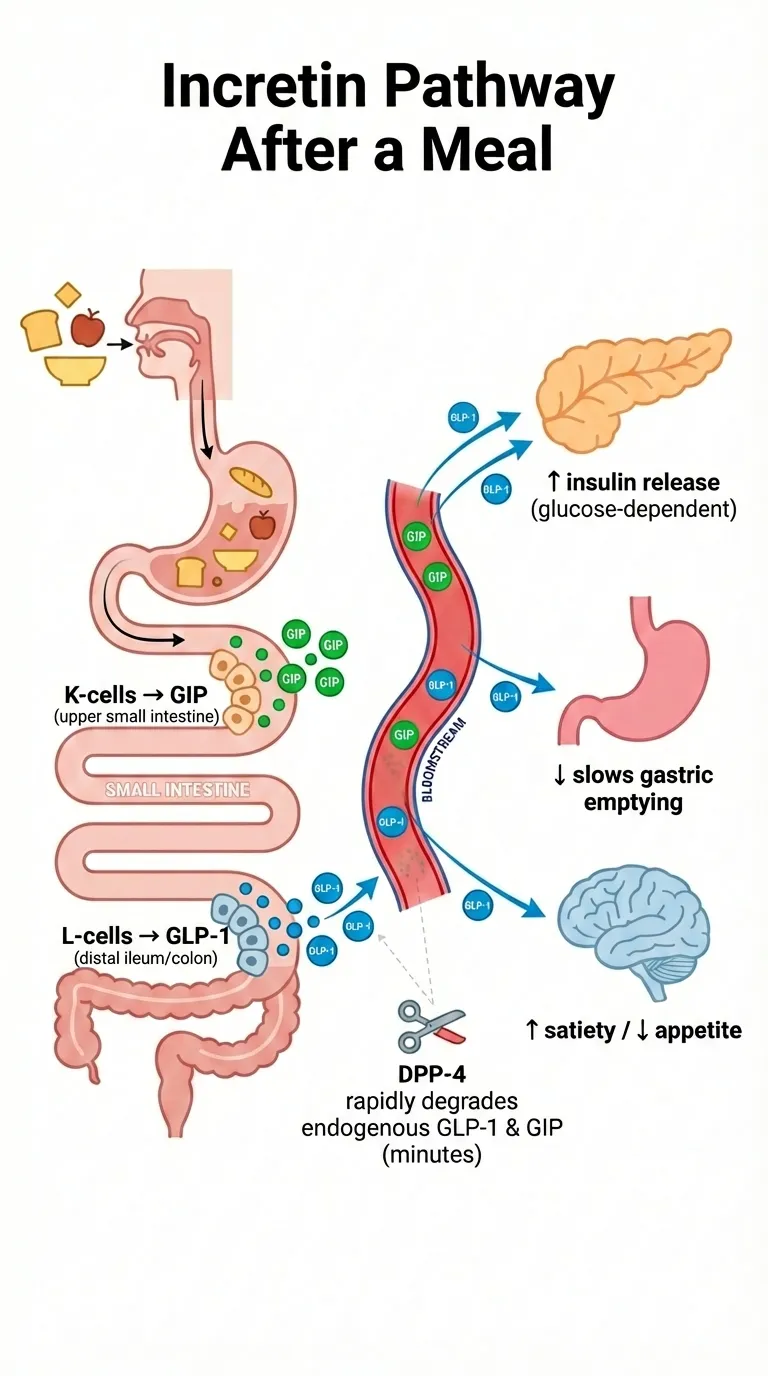
How incretins work after you eat: K-cells and L-cells in your intestines release GIP and GLP-1, which trigger multiple effects including insulin release and appetite reduction.
What Is an Incretin?
An incretin is a type of hormone released by your gut after you eat. The name comes from "INtestine seCRETion INsulin"—coined by Belgian scientist Jean La Barre in 1932—because these hormones cause your intestines to trigger insulin release.
Here's the simple version: When you eat, specialized cells in your intestines detect the incoming nutrients and release incretin hormones into your bloodstream. These hormones travel to your pancreas and tell it to prepare for the incoming sugar by releasing insulin. They also signal your brain that food is coming, which helps regulate your appetite.
The two main incretin hormones in humans are:
- GLP-1 (glucagon-like peptide-1)
- GIP (glucose-dependent insulinotropic polypeptide)
Together, these hormones are responsible for what scientists call the "incretin effect"—and understanding this effect has revolutionized how we treat type 2 diabetes and obesity.
Why You're Hearing About Incretins Everywhere
If "incretin" sounds familiar, it's probably because of medications like Ozempic, Wegovy, Mounjaro, and Zepbound. These drugs work by targeting the incretin system:
- GLP-1 receptor agonists (like semaglutide in Ozempic/Wegovy) mimic the GLP-1 hormone
- Dual GIP/GLP-1 agonists (like tirzepatide in Mounjaro/Zepbound) mimic both incretin hormones
- DPP-4 inhibitors (like Januvia) prevent the breakdown of your body's natural incretins
The explosive popularity of these medications—for both diabetes management and weight loss—has put the word "incretin" into everyday conversation.
How to Pronounce "Incretin"
Incretin is pronounced: IN-kreh-tin (stress on the first syllable).
Quick Glossary of Related Terms
Before we go deeper, here are the key terms you'll encounter:
| Term | What It Means |
|---|---|
| Incretin | A gut hormone that stimulates insulin release after eating |
| GLP-1 | Glucagon-like peptide-1; one of the two main incretins |
| GIP | Glucose-dependent insulinotropic polypeptide; the other main incretin |
| Incretin effect | The phenomenon where eating triggers more insulin than IV glucose would |
| Incretin mimetic | A drug that mimics incretin hormones (like GLP-1 agonists) |
| DPP-4 | The enzyme that breaks down natural incretins in your body |
| DPP-4 inhibitor | A drug that blocks DPP-4, helping your natural incretins last longer |
| GLP-1 receptor agonist | A medication that activates GLP-1 receptors (Ozempic, Wegovy, etc.) |
The Incretin Effect
The incretin effect is one of the most important discoveries in diabetes research. It explains why your body handles sugar from food differently than sugar injected directly into your bloodstream.
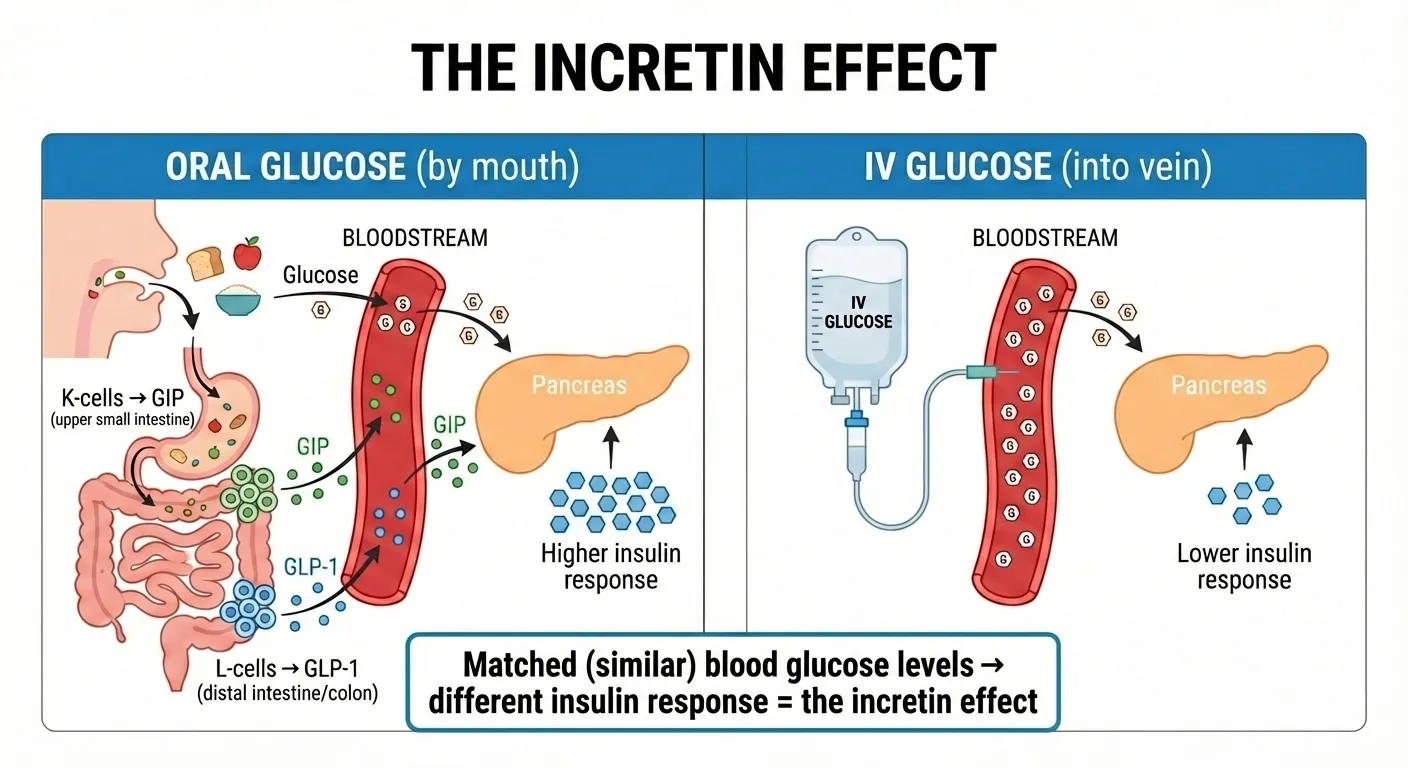
The incretin effect: When you eat glucose by mouth, your gut releases GLP-1 and GIP, producing a much higher insulin response than IV glucose at matched blood sugar levels.
The Discovery That Changed Everything
In the 1960s, researchers made a surprising observation. When they gave people glucose by mouth (drinking a sugary solution), their bodies released significantly more insulin than when they gave the exact same amount of glucose through an IV directly into the bloodstream.
This didn't make sense at first. If blood sugar levels were the same, why would the route of delivery matter?
The answer: the gut was sending signals to the pancreas.
When glucose travels through your digestive system, it triggers the release of incretin hormones from intestinal cells. These hormones amplify insulin release beyond what glucose alone would cause. Scientists named this the "incretin effect."
The Numbers Behind the Incretin Effect
The incretin effect isn't a small contribution—it's massive:
- Oral glucose triggers 2-3 times more insulin than the same amount of IV glucose (when blood sugar levels are matched)[2]
- Across studies, the incretin effect contributes a large portion of the post-meal insulin response in healthy people—often estimated at 50-70% depending on the study and measurement method[1]
In practical terms, this means your gut hormones are doing much of the heavy lifting when it comes to managing blood sugar after you eat.
How the Incretin Effect Works Step-by-Step
Here's what happens in your body when you eat:
Step 1: Food enters your gut As nutrients move through your stomach and into your small intestine, specialized cells detect their presence.
Step 2: K-cells release GIP In the upper part of your small intestine (duodenum and jejunum), K-cells release GIP into your bloodstream. This happens within minutes of eating.
Step 3: L-cells release GLP-1 Further down in your intestine (ileum and colon), L-cells release GLP-1. This continues as food moves through your digestive tract.
Step 4: Incretins reach the pancreas GLP-1 and GIP travel through your bloodstream to the pancreas, where they bind to specific receptors on beta cells.
Step 5: Insulin release is amplified The combination of rising blood glucose PLUS incretin signaling causes beta cells to release significantly more insulin than glucose alone would trigger.
Step 6: Additional effects kick in GLP-1 also slows stomach emptying, signals your brain to reduce appetite, and suppresses glucagon (a hormone that raises blood sugar).
Step 7: DPP-4 breaks down the incretins Within 1-2 minutes for GLP-1 and 5-7 minutes for GIP, the enzyme DPP-4 degrades these hormones, ending their effects.
Why the Incretin Effect Fails in Type 2 Diabetes
In people with type 2 diabetes, the incretin effect is significantly reduced—sometimes almost completely absent.
A landmark 1986 study by Nauck and colleagues measured the incretin effect in people with and without diabetes.[3] The results were striking:
- Healthy individuals (controls): insulin-based incretin effect ~73%
- Type 2 diabetes: insulin-based incretin effect ~30% (markedly reduced)
Important note: The exact percentage varies depending on whether insulin or C-peptide is used for measurement (because insulin clearance affects insulin-based calculations).[3]
This means people with type 2 diabetes are missing a major driver of insulin release after meals—which helps explain why their blood sugar spikes after eating.
What goes wrong:
- GIP resistance: The pancreas stops responding normally to GIP, even though GIP secretion is often normal
- Reduced GLP-1 secretion: Some people with diabetes produce less GLP-1 after meals
- Preserved GLP-1 response: Importantly, the pancreas still responds to GLP-1 in diabetes—just not as strongly as in healthy individuals
That last point is crucial. The fact that GLP-1 still works in type 2 diabetes is exactly why GLP-1-based medications are so effective.
Incretin Hormones: GLP-1 and GIP
While researchers have identified other gut hormones with potential incretin-like effects, only two meet the strict scientific definition of an incretin in humans: GLP-1 and GIP.
GLP-1 (Glucagon-Like Peptide-1)
GLP-1 is the incretin hormone that has received the most attention—and for good reason. It's the basis for some of the most effective diabetes and weight loss medications available today.
Basic Facts:
- Size: 30-31 amino acids
- Produced by: L-cells in the lower small intestine and colon
- Half-life: Only 1-2 minutes (quickly broken down by DPP-4)
What GLP-1 Does:
- Stimulates insulin release (only when blood sugar is elevated—this is key)
- Suppresses glucagon (reduces the hormone that raises blood sugar)
- Slows gastric emptying (food stays in your stomach longer)
- Reduces appetite (signals the brain to decrease hunger)
- May protect beta cells (the insulin-producing cells in the pancreas)
The combination of these effects explains why GLP-1-based medications are so powerful for both blood sugar control and weight loss.
GIP (Glucose-Dependent Insulinotropic Polypeptide)
GIP was actually discovered before GLP-1, but it received less attention for decades because researchers found it didn't work well in people with type 2 diabetes.
Basic Facts:
- Size: 42 amino acids
- Produced by: K-cells in the upper small intestine (duodenum and jejunum)
- Half-life: 5-7 minutes
What GIP Does:
- Stimulates insulin release (accounts for a significant portion of the incretin effect in healthy people)
- Affects glucagon (increases it, but in a glucose-dependent way)
- Influences fat tissue (effects are complex and context-dependent)
- Supports bone health (promotes bone formation)
- May reduce nausea (emerging research suggests GIP could counteract GLP-1 side effects)
For years, GIP was considered a poor drug target because people with type 2 diabetes showed "GIP resistance"—their pancreas didn't respond to GIP properly. However, the success of tirzepatide (Mounjaro/Zepbound), which activates both GIP and GLP-1 receptors, has renewed interest in GIP's therapeutic potential.
GLP-1 vs GIP: Head-to-Head Comparison
| Feature | GLP-1 | GIP |
|---|---|---|
| Full name | Glucagon-like peptide-1 | Glucose-dependent insulinotropic polypeptide |
| Where produced | L-cells (lower intestine) | K-cells (upper intestine) |
| Number of amino acids | 30-31 | 42 |
| Half-life | 1-2 minutes | 5-7 minutes |
| Effect on insulin | Increases (glucose-dependent) | Increases (glucose-dependent) |
| Effect on glucagon | Decreases | Increases (but glucose-dependent)[4] |
| Effect on appetite | Strongly reduces | Minimal direct effect |
| Effect on gastric emptying | Slows significantly | Minimal effect |
| Effect on fat tissue | Complex; context-dependent | Complex; context-dependent |
| Effect on bone | Under investigation | May promote bone formation |
| Response in T2 diabetes | Largely preserved | Significantly reduced |
| FDA-approved drug target | Yes (multiple drugs) | Yes (as part of tirzepatide) |
Note: Effects on fat tissue and bone are areas of active research. The therapeutic story with tirzepatide (which produces substantial weight loss despite GIP activation) shows the physiology is more nuanced than early assumptions suggested.
Why GLP-1 Became the Primary Drug Target
Given that both GLP-1 and GIP contribute to the incretin effect, why did drug companies focus mainly on GLP-1?
The answer lies in what happens in type 2 diabetes:
- GIP stops working: The pancreatic beta cells become resistant to GIP. Even though GIP is still released normally, it doesn't stimulate much insulin release.
- GLP-1 keeps working: While GLP-1's effects are somewhat reduced in diabetes, the beta cells still respond to it. More importantly, giving people extra GLP-1 (through medications) can still lower blood sugar effectively.
- GLP-1 has extra benefits: Unlike GIP, GLP-1 strongly reduces appetite and slows gastric emptying—effects that help with weight loss.
This made GLP-1 the obvious choice for drug development. The first GLP-1 receptor agonist (exenatide/Byetta) was approved in 2005, and the field has exploded since then.
The Resurgence of GIP (Through Dual Agonists)
The story took an interesting turn with tirzepatide (Mounjaro/Zepbound), which activates both GIP and GLP-1 receptors.
Clinical trials showed that tirzepatide produced better blood sugar control AND more weight loss than GLP-1-only medications like semaglutide. This surprised many researchers who had written off GIP as a therapeutic target.
Why might dual agonism work better? Several theories:
- GIP may reduce GLP-1 side effects: GIP might counteract the nausea that makes GLP-1 drugs hard to tolerate
- Synergistic effects: The two hormones may work better together than either alone
- Different tissue effects: GIP affects fat and bone in ways that complement GLP-1's actions
Research is ongoing to understand exactly why targeting both incretins seems to be more effective than targeting just one.
Incretin Drugs: The Complete FDA-Sourced Index
Understanding incretin hormones is interesting, but what most people really want to know is: what medications target this system, and what are they used for?
There are three main classes of incretin-based medications:
- GLP-1 receptor agonists (GLP-1 RAs) — mimic GLP-1
- Dual GIP/GLP-1 receptor agonists — mimic both incretin hormones
- DPP-4 inhibitors — prevent the breakdown of your natural incretins
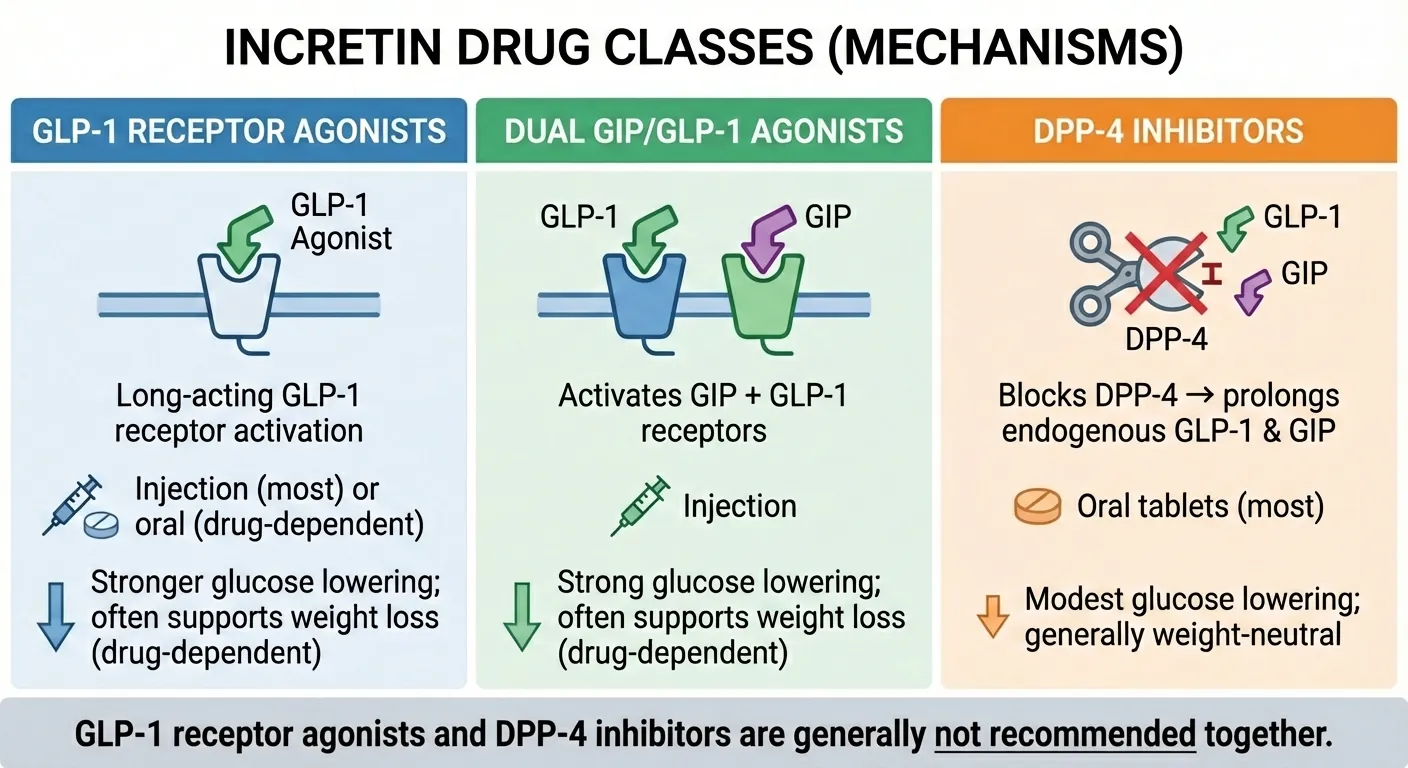
The three classes of incretin drugs work through different mechanisms: GLP-1 agonists directly activate GLP-1 receptors, dual agonists activate both GIP and GLP-1 receptors, and DPP-4 inhibitors help your natural hormones last longer.
Understanding the Terminology
Before we look at the drug lists, let's clarify some confusing terminology:
Terminology note: In clinical writing, "incretin-based therapies" typically includes GLP-1 receptor agonists and DPP-4 inhibitors. The FDA has also used the phrase "incretin mimetic drugs" in some safety information as an umbrella for these incretin-pathway drug classes—even though mechanistically DPP-4 inhibitors don't mimic an incretin hormone; they prolong endogenous GLP-1 and GIP by inhibiting DPP-4.[5]
Some sources use these terms interchangeably, which can be confusing. The tables below clarify exactly what each drug does.
How to read these lists: Some incretin drugs remain FDA-approved but are no longer marketed in the U.S. We flag these so readers don't waste time trying to fill a prescription that's not available.
GLP-1 Receptor Agonists: Currently Marketed in the U.S.
These medications mimic the GLP-1 hormone. Because they're designed to resist breakdown by DPP-4, they last much longer than natural GLP-1 (hours to days instead of minutes).
| Generic Name | Brand Name(s) | FDA Approved | How Given | Frequency | Approved For | U.S. Market Status |
|---|---|---|---|---|---|---|
| Liraglutide | Victoza | 2010 | Injection | Daily | Type 2 diabetes | Available |
| Liraglutide | Saxenda | 2014 | Injection | Daily | Weight management | Available |
| Dulaglutide | Trulicity | 2014 | Injection | Weekly | Type 2 diabetes | Available |
| Semaglutide | Ozempic | 2017 | Injection | Weekly | Type 2 diabetes | Available |
| Semaglutide | Rybelsus | 2019 | Oral tablet | Daily | Type 2 diabetes | Available |
| Semaglutide | Wegovy (injection) | 2021 | Injection | Weekly | Weight management | Available |
| Semaglutide | Wegovy (tablets) | 2025 | Oral tablet | Daily | Chronic weight management AND to reduce risk of major adverse cardiovascular events in certain adults with established CV disease and overweight/obesity | Available |
Sources: FDA drug labels accessed January 2026
Note: Wegovy exists as both weekly injection and daily tablets; approval dates and labeled indications differ by formulation—always verify against the current FDA label.[6]
GLP-1 Receptor Agonists: FDA-Approved but Discontinued in the U.S.
| Generic Name | Brand Name(s) | FDA Approved | How Given | Frequency | Original Indication | Status |
|---|---|---|---|---|---|---|
| Exenatide | Byetta | 2005 | Injection | Twice daily | Type 2 diabetes | Discontinued (2024)[7] |
| Exenatide ER | Bydureon BCise | 2012 | Injection | Weekly | Type 2 diabetes | Discontinued (2024)[7] |
| Albiglutide | Tanzeum | 2014 | Injection | Weekly | Type 2 diabetes | Discontinued[8] |
| Lixisenatide | Adlyxin | 2016 | Injection | Daily | Type 2 diabetes | Discontinued[9] |
Note: Discontinuation does not mean safety concerns—manufacturers may discontinue products for business reasons.
Dual GIP/GLP-1 Receptor Agonists
These newer medications activate receptors for both incretin hormones.
| Generic Name | Brand Name | FDA Approved | How Given | Frequency | Approved For | U.S. Market Status |
|---|---|---|---|---|---|---|
| Tirzepatide | Mounjaro | 2022 | Injection | Weekly | Type 2 diabetes | Available |
| Tirzepatide | Zepbound | 2023 | Injection | Weekly | Weight management | Available |
Sources: FDA drug labels accessed January 2026
Why tirzepatide is significant: Clinical trials showed tirzepatide produced greater A1C reductions and more weight loss than semaglutide (Ozempic), the previous leading GLP-1 agonist. This is why Mounjaro/Zepbound generated so much excitement—it appears to be the most effective incretin-based medication to date.
DPP-4 Inhibitors (Incretin Enhancers)
Rather than mimicking incretins, these oral medications block the enzyme (DPP-4) that breaks down your body's natural GLP-1 and GIP. This allows your own incretin hormones to last longer and have stronger effects.
| Generic Name | Brand Name | FDA Approved | Dosing | Notes |
|---|---|---|---|---|
| Sitagliptin | Januvia | 2006 | 100mg once daily | Dose adjust for kidney function |
| Saxagliptin | Onglyza | 2009 | 2.5-5mg once daily | Dose adjust for kidney function; heart failure warning |
| Linagliptin | Tradjenta | 2011 | 5mg once daily | No dose adjustment needed for kidney/liver |
| Alogliptin | Nesina | 2013 | 25mg once daily | Dose adjust for kidney function; heart failure warning |
Note: Vildagliptin (Galvus) is approved in Europe but not in the United States.
Sources: FDA drug labels accessed January 2026
DPP-4 inhibitors vs GLP-1 agonists: DPP-4 inhibitors are less powerful than GLP-1 agonists. They raise GLP-1 levels by about 2-3 times normal, while GLP-1 agonists raise levels to 5-10 times normal. This is why DPP-4 inhibitors:
- Cause less nausea
- Don't significantly affect weight (weight-neutral)
- Produce smaller A1C reductions
- Come as convenient oral tablets
- Don't require injection
Incretin Combination Products
These products combine an incretin-based medication with insulin:
| Generic Name | Brand Name | Components | FDA Approved | How Given | Approved For |
|---|---|---|---|---|---|
| Insulin glargine + lixisenatide | Soliqua 100/33 | Basal insulin + GLP-1 RA | 2016 | Injection | Type 2 diabetes |
| Insulin degludec + liraglutide | Xultophy 100/3.6 | Basal insulin + GLP-1 RA | 2016 | Injection | Type 2 diabetes |
Sources: FDA drug labels[10][11]
How We Maintain This Drug Index
We verify this information by:
- Reviewing current FDA drug labels at FDA.gov
- Checking FDA drug approval announcements
- Monitoring manufacturer discontinuation notices
- Updating when new drugs are approved or label changes occur
- Noting the "last verified" date at the top of this page
If you notice an error or a recent approval we've missed, please contact us.
How Incretin Medications Work
Now that you know what incretin drugs are available, let's understand how they actually work in your body.
GLP-1 Receptor Agonists: Mechanism of Action
GLP-1 receptor agonists (like semaglutide in Ozempic/Wegovy) are synthetic versions of the GLP-1 hormone that have been modified to last much longer in your body.
How they're different from natural GLP-1:
- Natural GLP-1 half-life: 1-2 minutes
- Semaglutide half-life: About 1 week
This longer duration is achieved through chemical modifications that prevent DPP-4 from breaking down the medication.
What happens when you take a GLP-1 agonist:
- Pancreas effects:
- Beta cells release more insulin (but only when blood sugar is elevated)
- Alpha cells release less glucagon (reducing liver glucose output)
- May help preserve beta cell function over time
- Stomach effects:
- Gastric emptying slows significantly
- Food stays in your stomach longer
- This contributes to feeling full and reduces blood sugar spikes after meals
- Brain effects:
- Appetite signals are suppressed
- Feelings of fullness increase
- Many patients report reduced "food noise" (constant thoughts about food)
- May affect reward pathways related to eating
- Other effects:
- Some evidence of cardiovascular benefits (reduced heart attacks, strokes)
- Potential kidney protective effects
- Effects on liver fat (being studied)
Dual GIP/GLP-1 Agonists: How Tirzepatide Works
Tirzepatide (Mounjaro/Zepbound) is a single molecule that activates both GIP receptors and GLP-1 receptors.
The dual mechanism:
- GLP-1 receptor activation: Provides all the benefits described above
- GIP receptor activation: Adds effects on fat tissue, potentially improves tolerability
Why two targets might be better than one:
The combination appears to produce:
- Better blood sugar control than GLP-1-only drugs
- More weight loss than GLP-1-only drugs
- Possibly better tolerability (GIP may reduce nausea)
Researchers are still working to understand exactly why the combination is so effective.
DPP-4 Inhibitors: How They Enhance Natural Incretins
DPP-4 inhibitors take a different approach. Instead of adding synthetic hormones, they help your body's natural incretins work better.
The mechanism:
- You eat a meal
- Your gut releases GLP-1 and GIP (as normal)
- Normally, DPP-4 would quickly break these down
- The DPP-4 inhibitor blocks this breakdown
- Your natural GLP-1 and GIP last longer and reach higher levels
- More insulin is released in response to the meal
Why DPP-4 inhibitors are less powerful:
Even with DPP-4 blocked, your natural incretin levels only rise to about 2-3 times normal. Compare this to GLP-1 agonists, which raise levels to 5-10 times normal. This is why:
- DPP-4 inhibitors cause less nausea (levels aren't as high)
- They don't significantly affect appetite or weight
- A1C reductions are more modest (typically 0.5-1% vs 1-2%)
Why You Can't Combine GLP-1 Agonists and DPP-4 Inhibitors
A common question: "Can I take both a GLP-1 agonist and a DPP-4 inhibitor for extra benefit?"
The answer is no—and here's why:
DPP-4 inhibitors work by protecting your body's natural GLP-1 from breakdown. But GLP-1 agonists (like semaglutide) are already designed to resist DPP-4 breakdown. They don't need protection.
Since the DPP-4 inhibitor can't enhance the synthetic GLP-1 from the agonist, adding it provides no additional benefit. The American Diabetes Association does not recommend this combination.
Incretin Drugs Compared: Which Class Is Used for What?
With so many options, how do doctors choose which incretin medication to prescribe? Here's a practical comparison.
Quick Comparison Table
| Feature | GLP-1 Agonists | Dual GIP/GLP-1 | DPP-4 Inhibitors |
|---|---|---|---|
| How given | Injection (most) or oral | Injection | Oral tablet |
| GLP-1 increase | 5-10x normal | 5-10x normal | 2-3x normal |
| Typical A1C reduction | 1-2% | 1.5-2.5% | 0.5-1% |
| Weight effect | Loss (significant) | Loss (most significant) | Neutral |
| Hypoglycemia risk alone | Low | Low | Low |
| Common side effects | Nausea, vomiting, diarrhea | Nausea, vomiting, diarrhea | Generally well-tolerated |
| Cardiovascular benefit proven | Yes (some agents) | Under study | No |
| FDA-approved for weight loss | Yes (Wegovy, Saxenda) | Yes (Zepbound) | No |
| Cost | Higher | Highest | Moderate |
When Each Class Is Typically Used
GLP-1 Receptor Agonists are often chosen when:
- Significant weight loss is a goal
- Cardiovascular protection is important (established heart disease)
- A1C needs substantial reduction
- Patient can tolerate injections (or can use oral Rybelsus)
Dual GIP/GLP-1 Agonists may be preferred when:
- Maximum weight loss is the priority
- Previous GLP-1 agonists weren't effective enough
- Patient tolerates injections
DPP-4 Inhibitors are often chosen when:
- Patient strongly prefers oral medication
- Modest A1C reduction is needed
- Weight neutrality is acceptable or preferred
- GI side effects from GLP-1 agonists were intolerable
- Cost is a significant factor
Benefits and Outcomes: What Incretin Therapy Can Achieve
Understanding the potential benefits helps set realistic expectations. Here's what the research shows.
Blood Sugar Control
All incretin-based medications improve blood sugar control, but the magnitude varies:
A1C reductions (approximate averages):
- DPP-4 inhibitors: 0.5-1.0%
- GLP-1 agonists: 1.0-1.8%
- Dual GIP/GLP-1 (tirzepatide): 1.5-2.4%
For reference, a "normal" A1C is below 5.7%, prediabetes is 5.7-6.4%, and diabetes is diagnosed at 6.5% or above. Reducing A1C from, say, 8.5% to 7% significantly reduces the risk of diabetes complications.
Weight Loss
Weight effects vary dramatically by drug class:
DPP-4 inhibitors: Generally weight-neutral (no significant gain or loss)
GLP-1 agonists:
- Semaglutide 2.4mg (Wegovy): Mean −14.9% body weight at 68 weeks in the STEP-1 trial[12]
- Liraglutide (Saxenda): Average 5-8% body weight loss
- Other GLP-1 agonists: Generally 3-6% body weight loss
Dual GIP/GLP-1:
- Tirzepatide: Mean −15% to −20.9% body weight depending on dose at 72 weeks in the SURMOUNT-1 trial[13]
These are averages—individual results vary significantly. Some people lose more, others less.
Cardiovascular Outcomes
Several GLP-1 agonists have demonstrated cardiovascular benefits in large trials (these are relative risk reductions in specific trial populations):
- Liraglutide (LEADER trial): 13% relative reduction in major cardiovascular events (heart attack, stroke, CV death)[14]
- Semaglutide injection (SUSTAIN-6 trial): 26% relative reduction in major cardiovascular events[15]
- Dulaglutide (REWIND trial): 12% relative reduction in major cardiovascular events[16]
These benefits have led to some GLP-1 agonists being specifically approved or recommended for people with type 2 diabetes and established cardiovascular disease.
DPP-4 inhibitors have NOT shown cardiovascular benefit—they appear to be neutral (neither helpful nor harmful for heart outcomes).
Tirzepatide cardiovascular outcome trials are ongoing.
Other Potential Benefits Under Study
Researchers are investigating incretin-based therapies for:
- Non-alcoholic fatty liver disease (NAFLD/NASH)
- Chronic kidney disease
- Parkinson's disease and other neurological conditions
- Sleep apnea
- Polycystic ovary syndrome (PCOS)
Some of these show promise, but none are FDA-approved indications yet.
Side Effects and Safety: What You Need to Know
No medication is without risks. Understanding potential side effects helps you make informed decisions and know when to contact your healthcare provider.
Common Side Effects of GLP-1 Agonists and Dual Agonists
The most frequent side effects are gastrointestinal:
| Side Effect | Frequency | Notes |
|---|---|---|
| Nausea | Very common (20-40%) | Usually improves over time; worse at dose increases |
| Vomiting | Common (5-20%) | More common early in treatment |
| Diarrhea | Common (10-20%) | Usually mild |
| Constipation | Common (5-15%) | Some people experience this instead of diarrhea |
| Abdominal pain | Common (5-15%) | Usually mild to moderate |
| Decreased appetite | Common | This is often desired for weight management |
| Injection site reactions | Occasional | Redness, itching, or swelling at injection site |
Managing GI side effects:
- Start at the lowest dose and increase slowly (titration)
- Eat smaller meals
- Avoid high-fat and fried foods
- Stay hydrated
- Most people find symptoms improve after 4-8 weeks
Common Side Effects of DPP-4 Inhibitors
DPP-4 inhibitors are generally well-tolerated:
- Nasopharyngitis (cold-like symptoms)
- Headache
- Upper respiratory infection
- Joint pain (FDA issued a warning about severe joint pain in 2015)
Serious Risks and Warnings
Pancreatitis: Both GLP-1 agonists and DPP-4 inhibitors have been associated with cases of pancreatitis (inflammation of the pancreas). The FDA continues to monitor this.
What to watch for: Severe, persistent abdominal pain (sometimes radiating to the back) with or without vomiting. Seek medical attention immediately if this occurs.
Gallbladder problems: GLP-1 agonists may increase the risk of gallstones and gallbladder inflammation.
What to watch for: Pain in the upper right abdomen, especially after eating fatty foods, with or without nausea/vomiting.
Thyroid tumors (boxed warning for GLP-1 agonists): In rodent studies, GLP-1 agonists caused thyroid C-cell tumors. It's unknown if this applies to humans, but as a precaution:
- GLP-1 agonists are contraindicated in people with a personal or family history of medullary thyroid carcinoma (MTC)
- They're also contraindicated in people with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
Heart failure (DPP-4 inhibitors): Saxagliptin (Onglyza) and alogliptin (Nesina) have shown a small increased risk of hospitalization for heart failure. The FDA added warnings to these labels in 2016.
Kidney effects: Both drug classes can affect the kidneys. Some GLP-1 agonists have shown kidney protective effects, but there have also been reports of acute kidney injury, particularly with dehydration from GI side effects.
Mental health: In January 2024, FDA reported its preliminary review did not find evidence that GLP-1 receptor agonists cause suicidal thoughts or actions, while noting evaluation would continue. In January 2026, FDA said it completed a comprehensive review and asked manufacturers to remove warnings about suicidal thoughts/actions from GLP-1 labels.[17] If you notice mood changes while on any medication, discuss them promptly with a clinician.
Red Flag Symptoms: When to Seek Medical Attention
Contact your healthcare provider or seek emergency care if you experience:
- Severe, persistent abdominal pain
- Persistent vomiting you can't control
- Signs of allergic reaction (rash, itching, swelling, difficulty breathing)
- Symptoms of low blood sugar (if taking with insulin or sulfonylureas)
- Severe constipation lasting more than a few days
- Yellowing of skin or eyes (jaundice)
- Unusual mood changes, thoughts of self-harm
Before Surgery/Anesthesia: Important Considerations
Because GLP-1 medicines can delay gastric emptying, FDA labeling for some products includes warnings about pulmonary aspiration (food/liquid entering the lungs) during general anesthesia or deep sedation.
Professional guidance has evolved:
- ASA's 2023 consensus suggested holding GLP-1 drugs before elective procedures
- 2024 multi-society guidance said most patients can continue and emphasized individualized risk assessment (considering factors like dose-escalation phase, significant GI symptoms, and high aspiration risk)
Follow your surgical/anesthesia team's specific instructions. Inform them that you take a GLP-1 agonist or dual agonist before any procedure requiring sedation or anesthesia.
Questions to Ask Your Healthcare Provider
Before starting an incretin-based medication, consider asking:
- Why is this medication the best choice for me?
- What side effects should I watch for?
- How should I take this medication (timing, with or without food)?
- How will we monitor whether it's working?
- What should I do if I miss a dose?
- Will this interact with my other medications?
- What happens if I need surgery while taking this?
- How long will I need to take this medication?
- What are the costs, and are there assistance programs?
- Are there lifestyle changes that could make this more effective?
Special Situations
Certain circumstances require extra consideration when using incretin-based medications.
Pregnancy and Breastfeeding
Current guidance:
- Incretin-based medications are generally avoided in pregnancy due to limited human safety data
- For semaglutide products (Ozempic/Wegovy/Rybelsus), labeling recommends discontinuing at least 2 months before a planned pregnancy because semaglutide can remain in the body for weeks[18]
- For other incretin drugs, follow the specific product label and clinician guidance
- Limited data on breastfeeding; discuss with your healthcare provider
Why this matters: Animal studies have shown potential risks, and there isn't enough human data to confirm safety. If you become pregnant while taking these medications, contact your healthcare provider promptly.
Kidney Disease
GLP-1 agonists: Some have shown kidney-protective effects, but dose adjustments or monitoring may be needed depending on the specific drug and severity of kidney disease.
DPP-4 inhibitors: Most require dose adjustment in kidney disease, EXCEPT linagliptin (Tradjenta), which can be used at the same dose regardless of kidney function.
Liver Disease
Generally, incretin-based medications are not extensively studied in severe liver disease. Linagliptin is eliminated through the gut rather than kidneys or liver, which may make it preferable in some cases.
If You Take Insulin or Sulfonylureas
Adding incretin-based medications to insulin or sulfonylureas increases the risk of hypoglycemia (low blood sugar). Your healthcare provider will likely:
- Reduce your insulin or sulfonylurea dose when starting the incretin medication
- Provide instructions for monitoring blood sugar more frequently
- Explain symptoms of low blood sugar and how to treat them
Oral Medication Absorption
Because GLP-1 agonists slow stomach emptying, they can affect how quickly other oral medications are absorbed. This is particularly important for:
- Medications that need to be absorbed quickly
- Medications where timing matters (like certain thyroid medications)
- Oral contraceptives (birth control pills)
Regarding birth control pills: Some labels suggest using backup contraception or alternative methods, especially during dose escalation when GI side effects may cause vomiting. Discuss with your healthcare provider if this applies to you.
Tirzepatide (Mounjaro/Zepbound) specific note: Labeling advises switching to a non-oral contraceptive method or adding a barrier method for 4 weeks after starting tirzepatide and for 4 weeks after each dose increase, due to delayed gastric emptying potentially reducing oral contraceptive absorption.[19]
Natural Incretins and Supplements: What Actually Works
Many people wonder if they can boost their incretin levels naturally, without prescription medications. Let's look at what the evidence actually shows.
The Honest Truth About Natural Approaches
First, the reality check:
- Natural GLP-1 lasts only 1-2 minutes in your body
- Foods and lifestyle can temporarily increase GLP-1 release
- These temporary increases are MUCH smaller than what medications produce
- No natural approach can match the weight loss or blood sugar effects of GLP-1 medications
That said, certain foods and habits may support your body's incretin system as part of an overall healthy lifestyle.
Foods That May Support Incretin Function
| Food/Nutrient | Evidence Level | How It May Help |
|---|---|---|
| Soluble fiber (oats, legumes, flaxseed) | Moderate | Fermentation produces short-chain fatty acids that may stimulate L-cells |
| Resistant starch (green bananas, cooled potatoes) | Moderate | Similar fermentation mechanism |
| Healthy fats (olive oil, avocados, nuts) | Moderate | May activate receptors that enhance GLP-1 release |
| Lean protein | Moderate | Amino acids can stimulate incretin release |
| Fermented foods | Low-Moderate | May support gut microbiome health |
What the research shows: One study found that a plant-based meal stimulated more incretin and insulin release than an energy-matched meat-based meal in people with type 2 diabetes. The researchers suggested that fiber, polyphenols, and other plant compounds may be responsible.
Lifestyle Factors
Exercise: Research suggests that both single exercise sessions and regular training may enhance GLP-1 levels, particularly in people with type 2 diabetes. The mechanisms are still being studied, but this adds to the many reasons to stay physically active.
Sleep: Your circadian rhythm affects incretin secretion. Studies show that people with disrupted sleep patterns (like shift workers) tend to have reduced GLP-1 responses and higher risk of insulin resistance. Getting 7-9 hours of quality sleep may help maintain normal incretin function.
Gut microbiome: The bacteria in your gut influence incretin secretion. Diets high in fiber and fermented foods support a healthy microbiome, which may indirectly support incretin function.
What About Supplements?
Several supplements are marketed as "natural GLP-1 boosters" or even "nature's Ozempic." Here's what the evidence actually shows:
| Supplement | Claim | Reality |
|---|---|---|
| Berberine | "Nature's Ozempic" | Animal studies show increased GLP-1; human data limited. Meta-analyses show ~4 lbs weight loss on average—far less than actual GLP-1 medications. May cause similar GI side effects. |
| Psyllium fiber | Boosts GLP-1 | May modestly support GLP-1 through fiber mechanisms. Safe and beneficial for other reasons. |
| Curcumin | Inhibits DPP-4 | Test tube studies show DPP-4 inhibition; human evidence for GLP-1 effects is minimal. |
| Probiotics | Improve incretin response | Some strains may have modest effects; research is early. |
| Cinnamon | Blood sugar support | May have small effects on blood sugar; no strong evidence for incretin effects. |
The "Don't Get Scammed" Checklist
Be skeptical of any product that claims to:
- Work "just like Ozempic" without a prescription
- Cause rapid weight loss naturally
- Boost GLP-1 levels to "therapeutic" ranges
- Replace diabetes medications
- Have no side effects while matching drug effectiveness
Red flags:
- Claims sound too good to be true
- No citations to peer-reviewed research
- Before/after photos with dramatic results
- Pressure to buy immediately
- "Doctors don't want you to know" language
The Bottom Line on Natural Approaches
Natural strategies can support overall metabolic health, but they cannot:
- Replace diabetes medications for people who need them
- Produce weight loss comparable to GLP-1 medications
- Restore incretin function in people with type 2 diabetes
Think of diet, exercise, and sleep as the foundation—medications as tools that can be added when lifestyle alone isn't enough.
Frequently Asked Questions
Basic Questions
What is an incretin? An incretin is a hormone released by your gut after you eat that helps control blood sugar. The two main incretins are GLP-1 and GIP. They signal your pancreas to release insulin and have other effects on appetite and digestion.
What does incretin mean? The term "incretin" comes from "INtestine seCRETion INsulin"—referring to hormones from the intestine that promote insulin release. The term was coined by Belgian scientist Jean La Barre in 1932.
How do you pronounce incretin? Incretin is pronounced IN-kreh-tin, with the stress on the first syllable.
What is the incretin effect? The incretin effect is the observation that eating food triggers much more insulin release (2-3 times more) than if you received the same amount of glucose directly into your bloodstream. This happens because gut hormones (incretins) amplify the pancreas's insulin response.
What cells secrete incretin hormones? GLP-1 is secreted by L-cells, which are found mainly in the lower small intestine and colon. GIP is secreted by K-cells, which are found mainly in the upper small intestine (duodenum and jejunum).
About the Hormones
What's the difference between GLP-1 and GIP? Both are incretin hormones that stimulate insulin release, but they differ in where they're made, their effects beyond insulin, and how well they work in diabetes. GLP-1 strongly reduces appetite and slows stomach emptying; GIP has minimal effects on appetite. In type 2 diabetes, GIP's insulin-stimulating ability is greatly reduced, while GLP-1's ability is largely preserved.
What are examples of incretins? The two established incretin hormones in humans are GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide). Other gut hormones like oxyntomodulin and peptide YY have some incretin-like effects but don't meet the strict scientific definition.
About Medications
What are incretin drugs? Incretin drugs are medications that work through the incretin system. They include GLP-1 receptor agonists (like Ozempic), dual GIP/GLP-1 agonists (like Mounjaro), and DPP-4 inhibitors (like Januvia). They're used mainly to treat type 2 diabetes, and some are approved for weight management.
What are incretin mimetics? Incretin mimetics are drugs that mimic incretin hormones. This term typically refers to GLP-1 receptor agonists and dual GIP/GLP-1 agonists. The FDA has used "incretin mimetic" in some communications to describe these drug classes.
Is Ozempic an incretin drug? Yes. Ozempic contains semaglutide, which is a GLP-1 receptor agonist. It mimics the incretin hormone GLP-1 and is approved for type 2 diabetes. Wegovy contains the same active ingredient at a higher dose and is approved for weight management.
Is Mounjaro an incretin drug? Yes. Mounjaro contains tirzepatide, which is a dual GIP/GLP-1 receptor agonist. It activates receptors for both major incretin hormones and is approved for type 2 diabetes. Zepbound is the same medication approved for weight management.
What's the difference between GLP-1 agonists and DPP-4 inhibitors? GLP-1 agonists are synthetic hormones that directly activate GLP-1 receptors (most are injections). DPP-4 inhibitors are oral pills that block the enzyme that breaks down your natural incretins. GLP-1 agonists are more powerful, cause more weight loss, but have more GI side effects. DPP-4 inhibitors are gentler, weight-neutral, and better tolerated.
Can I take a GLP-1 agonist and a DPP-4 inhibitor together? No, this combination is not recommended. GLP-1 agonists are designed to resist DPP-4 breakdown, so adding a DPP-4 inhibitor provides no additional benefit. Guidelines from the American Diabetes Association do not support this combination.
About Weight Loss
Are incretin drugs used for weight loss? Yes, some are. Wegovy (semaglutide), Saxenda (liraglutide), and Zepbound (tirzepatide) are FDA-approved specifically for chronic weight management in adults with obesity or overweight with weight-related conditions. Other incretin drugs cause weight loss but are only approved for diabetes.
How much weight can you lose on incretin medications? Results vary widely between individuals. In clinical trials:
- Semaglutide 2.4mg (Wegovy): Mean −14.9% body weight at 68 weeks (STEP-1 trial)[12]
- Tirzepatide (Zepbound): Mean −15% to −20.9% body weight depending on dose at 72 weeks (SURMOUNT-1 trial)[13]
- Liraglutide (Saxenda): Average 5-8% body weight loss
About Safety
Do incretin drugs cause pancreatitis? Cases of pancreatitis have been reported with both GLP-1 agonists and DPP-4 inhibitors. The FDA monitors this risk. If you experience severe, persistent abdominal pain, stop the medication and seek medical attention. People with a history of pancreatitis should use these medications with caution.
Do incretin drugs increase thyroid cancer risk? In rodent studies, GLP-1 agonists caused thyroid C-cell tumors. It's unknown if this applies to humans. As a precaution, these medications carry a boxed warning and are contraindicated in people with medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Should I stop my GLP-1 medication before surgery? Discuss this with your surgical team. Because GLP-1 agonists slow stomach emptying, there may be increased risk of aspiration during procedures involving anesthesia. Professional guidance has evolved (see the Safety section above), so follow your specific medical team's recommendations.
About Natural Alternatives
What are incretin supplements? "Incretin supplements" typically refers to products marketed as natural ways to boost GLP-1 levels. Common ingredients include berberine, fiber supplements, and various herbal extracts. However, no supplement can match the effects of prescription incretin medications.
Is berberine "nature's Ozempic"? This is a marketing claim that oversells berberine's effects. While berberine may modestly affect blood sugar and cause small amounts of weight loss (~4 lbs on average), it is nowhere near as effective as semaglutide (Ozempic), which produces 15%+ body weight loss in clinical trials.
Can you increase incretins naturally? You can temporarily increase incretin release through eating (especially high-fiber foods), exercise, and maintaining good sleep habits. However, natural GLP-1 only lasts 1-2 minutes, so these effects are brief and much weaker than what medications provide.
Complete Glossary of Incretin Terms
| Term | Definition |
|---|---|
| Beta cell | Insulin-producing cell in the pancreas |
| DPP-4 | Dipeptidyl peptidase-4; the enzyme that breaks down incretins |
| DPP-4 inhibitor | A drug that blocks DPP-4, prolonging natural incretin action |
| Dual agonist | A drug that activates two different receptors (e.g., GIP and GLP-1) |
| GIP | Glucose-dependent insulinotropic polypeptide; one of two main incretins |
| GLP-1 | Glucagon-like peptide-1; one of two main incretins |
| GLP-1 receptor agonist | A drug that mimics GLP-1 by activating its receptor |
| Glucagon | A hormone that raises blood sugar (opposite of insulin) |
| Glucose-dependent | Working only when blood sugar is elevated |
| Incretin | A gut hormone that enhances insulin release after eating |
| Incretin effect | Enhanced insulin release from oral vs IV glucose |
| Incretin enhancer | Another term for DPP-4 inhibitors |
| Incretin mimetic | A drug that mimics incretin hormones |
| Incretin-based therapy | Any treatment targeting the incretin system |
| Insulin | A hormone that lowers blood sugar |
| Insulinotropic | Stimulating insulin release |
| K-cells | Intestinal cells that produce GIP |
| L-cells | Intestinal cells that produce GLP-1 |
| Receptor agonist | A drug that binds to and activates a receptor |
| Semaglutide | A GLP-1 receptor agonist (Ozempic, Wegovy, Rybelsus) |
| Tirzepatide | A dual GIP/GLP-1 agonist (Mounjaro, Zepbound) |
| Type 2 diabetes | A condition where blood sugar is too high due to insulin resistance |
Sources and Editorial Standards
How We Created This Guide
This guide was developed by reviewing:
- FDA drug labels and safety communications
- Peer-reviewed medical literature
- Clinical guidelines from the American Diabetes Association
- Physiology textbooks and review articles
We aim to present accurate, balanced information that helps readers understand incretin biology and medications without promoting or discouraging any specific treatment.
Our Editorial Standards
What we do:
- Cite primary sources (FDA labels, peer-reviewed research) with inline references
- Clearly distinguish between established facts and emerging research
- Note when evidence is limited or conflicting
- Update content when new information becomes available
- Disclose financial relationships
- Flag discontinued medications so readers don't waste time
What we don't do:
- Make promises about specific health outcomes
- Recommend one treatment over another
- Provide individual medical advice
- Hide uncertainties or limitations
Primary Sources We Rely On
- FDA Drug Labels — accessed via FDA.gov for current prescribing information
- FDA Safety Communications — for warnings, updates, and regulatory actions
- American Diabetes Association Standards of Medical Care — for clinical practice guidelines
- PubMed/NCBI — for peer-reviewed research articles
- Manufacturer prescribing information — for drug-specific details
Key References
[1] Gasbjerg LS, et al. The incretin system in healthy humans: The role of GIP and GLP-1. Metabolism. 2019;96:46-55.
[2] Nauck MA, Meier JJ. The incretin effect in healthy individuals and those with type 2 diabetes: physiology, pathophysiology, and response to therapeutic interventions. Lancet Diabetes Endocrinol. 2016;4(6):525-536.
[3] Nauck MA, et al. Reduced incretin effect in type 2 (non-insulin-dependent) diabetes. Diabetologia. 1986;29:46-52.
[4] Gasbjerg LS, et al. GIP and GLP-1 receptor antagonism during a meal in healthy individuals. JCI Insight. 2024.
[5] PMC. The Origin and Understanding of the Incretin Concept. PMCID: PMC6054964.
[6] FDA Label: Wegovy tablets. FDA.gov, accessed January 2026.
[7] UHC Provider. Discontinuation of Bydureon BCise and Byetta. 2024.
[8] FDA. Tanzeum (albiglutide) information. FDA.gov.
[9] TruLaw. Adlyxin discontinuation information.
[10] FDA Label: Soliqua 100/33. FDA.gov.
[11] FDA Label: Xultophy 100/3.6. FDA.gov.
[12] Wilding JPH, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP-1). N Engl J Med. 2021;384:989-1002.
[13] Jastreboff AM, et al. Tirzepatide Once Weekly for the Treatment of Obesity (SURMOUNT-1). N Engl J Med. 2022;387:205-216.
[14] Marso SP, et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes (LEADER). N Engl J Med. 2016;375:311-322.
[15] Marso SP, et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes (SUSTAIN-6). N Engl J Med. 2016;375:1834-1844.
[16] Gerstein HC, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND). Lancet. 2019;394(10193):121-130.
[17] FDA. FDA Requests Removal of Suicidal Behavior and Ideation Warning from Glucagon-Like Peptide-1 Receptor Agonist (GLP-1 RA) Labels. January 2026.
[18] FDA Label: Ozempic (semaglutide). FDA.gov, accessed January 2026.
[19] FDA Label: Mounjaro (tirzepatide). FDA.gov, accessed January 2026.
Update Log
| Date | Update |
|---|---|
| January 2026 | Initial publication; includes oral Wegovy approval, updated mental health guidance per FDA Jan 2026 communication, discontinued drug flags |
Fact-Checking Methodology
This content was fact-checked by our editorial team using the following process:
- All drug approval dates verified against FDA records
- Clinical trial data verified against published peer-reviewed studies
- Safety warnings verified against current FDA communications
- Market availability verified against manufacturer announcements
- All claims requiring citations have inline references
We do not have a medical doctor review each article. For medical advice, please consult your healthcare provider.
What to Read Next
If you found this guide helpful, you may also be interested in:
- How GLP-1 Works for Weight Loss — A detailed mechanism guide with clinical trial data
- What Is GIP? — Deep dive into the other incretin hormone and dual agonist medications
- GLP-1 Cost Guide 2026 — Real pricing for Wegovy, Zepbound, and other options
- Understanding GLP-1 Side Effects — Practical tips for managing common issues
- GLP-1 Contraindications — Who should and shouldn't take these medications
This page was last updated January 2026. Incretin drug information was last verified against FDA sources January 2026.
How we rank + verify
Last verified: March 3, 2026
What we verified: FDA drug labels, prescribing information, and safety communications for all listed medications. Peer-reviewed research from NEJM, Lancet, Diabetologia, and NCBI databases.
Sources: FDA labels accessed January 2026 [6][7][10][11][18][19], peer-reviewed studies [1][2][3][4][12][13][14][15][16], ADA clinical guidelines
Related Articles
What is GIP? GIP (glucose-dependent insulinotropic polypeptide) is the gut hormone behind Mounjaro and Zepbound. Learn how dual GIP/GLP-1 agonists work.
How GLP-1 medications like Ozempic and Wegovy work for weight loss. Mechanism map, clinical trial results, side effects, and decision framework.
GLP-1 cost breakdown for 2026. Real prices for Wegovy, Zepbound, Ozempic with insurance, cash-pay, and Medicare options. Updated January 2026.
A comprehensive guide to GLP-1 medication side effects, how to manage them, and when to contact your doctor. Based on FDA prescribing information.
This content is educational only and does not replace professional medical advice. Always consult a qualified healthcare provider before starting any medication.