🚨 If this is a medical emergency, call 911.
If you took too much GLP-1, took the wrong dose, or used a product you suspect is counterfeit, call Poison Help at 1-800-222-1222 right now. It’s free, confidential, and available 24/7. Reporting to the FDA comes after you’re safe, not instead of it.
How to Report a Bad GLP-1 Experience to FDA MedWatch
By Weight Loss Provider Guide editorial team · Last verified: April 21, 2026 · 14 min read

5-step GLP-1 adverse event reporting guide. Verified against FDA sources, April 21, 2026.
The short answer
If you’ve had a bad reaction to a GLP-1, here’s exactly how to report it: file FDA Form 3500B, the consumer version of the federal adverse-event report. This covers brand-name products like Ozempic®, Wegovy®, Zepbound®, Mounjaro®, Foundayo™, Rybelsus®, and Saxenda®, and it covers compounded semaglutide and tirzepatide from telehealth providers. You can submit at fda.gov/medwatch, by mail, or by fax. The FDA estimates the form takes about 30 minutes on average when your details are handy. It’s free. It’s voluntary. You can ask FDA not to share your identity with the manufacturer.
Your reporting path in one table
| Your situation | File this first |
|---|---|
| Bad reaction to an FDA-approved GLP-1 (Wegovy, Ozempic, Zepbound, Mounjaro, Foundayo, Rybelsus, Saxenda) | MedWatch Form 3500B (online, mail, or fax) |
| Bad reaction to a compounded GLP-1 (compounded semaglutide, compounded tirzepatide, sublingual, microdose, oral drops, custom vials) | MedWatch Form 3500B |
| Suspected counterfeit GLP-1 (misspellings, mystery seller, suspiciously cheap, "research use only") | MedWatch Form 3500B — mark it as a product problem |
Below we’ll walk you through every field of the current Form 3500B in plain English, including the exact language to use if your product was compounded. First, the question most readers actually arrived with: is what happened to me even something the FDA wants to hear about?
When to call 911 or Poison Help first
MedWatch is a reporting system, not a medical helpline. If your situation is urgent, reporting can wait a few hours or a few days. Your safety can’t.
Call 911 now if you have any of these
Call Poison Help (1-800-222-1222) if
- You or someone else took more GLP-1 than prescribed
- You injected or took a product you now suspect is counterfeit or tampered with
- A child accidentally got into the medication
- You're not sure whether what you took is safe
This matters especially for compounded injectables. The FDA has publicly said it has received multiple reports of hospitalizations tied to dose-measurement errors with compounded semaglutide — people measuring in the wrong units, using the wrong syringe, or misreading instructions that asked for “units” on a vial labeled in mg/mL. If that’s you or a loved one right now, Poison Help comes before MedWatch.
Call your prescriber
If you’re medically stable but something’s clearly not right — symptoms that aren’t in your medication’s patient leaflet, symptoms that keep getting worse, or an injection site that’s becoming infected — contact your prescriber. Then file your report yourself. Your doctor is not required to file MedWatch on your behalf.
Not sure if your symptoms are normal? Our guide to understanding GLP-1 side effects breaks down what’s common, what’s concerning, and what earns a same-day call.
What actually counts as “a bad GLP-1 experience” for MedWatch?
If any of the below describes what happened to you or someone you care for, the FDA wants to know.
Your body reacted badly
- ER visit or hospitalization after a dose
- Pancreatitis, gallbladder attack, or severe abdominal pain
- Persistent vomiting or dehydration requiring IV fluids
- Injection-site infection, abscess, or unusual lump
- Sudden vision changes
- New burning, pins and needles, or "sunburned" skin sensations
- Menstrual changes, hair loss, or severe fatigue
- A reaction that required you to stop the medication
Something was wrong with the product
- Pen or vial arrived warm, or shipping cold packs were melted
- Pen doesn't work, leaks, won't click through doses
- Liquid looks cloudy, discolored, or has particles
- Packaging looks different from prior shipments
- Pharmacy name on label doesn't match the one that confirmed your order
- Label has misspellings, wrong addresses, or non-English text
- Missing lot number, expiration date, or NDC
Someone made a dosing mistake
- You were told to measure "10 units" from a vial labeled in mg/mL and the math didn't match
- The dose in the vial doesn't match what was prescribed
- Telehealth clinician changed your dose without clear instructions
- You realized you'd been giving yourself 5× or 10× the intended dose
Symptoms started after a switch
- You moved from brand-name to compounded, or between compounded providers, and something changed
- Efficacy changed unexpectedly after a new shipment
How to file Form 3500B: the three ways
| Route | Speed | How |
|---|---|---|
| Online | Fastest — ~30 min | fda.gov/medwatch → "Report a Problem" → Consumer/Patient (Form 3500B) |
| Several days | Download the PDF, complete it, mail to: MedWatch Program, White Oak Campus, Building 22, G0207, 10903 New Hampshire Ave., Silver Spring, MD 20993 | |
| Fax | Same-day | Complete the PDF and fax to 1-800-FDA-0178 (toll-free) |
| Phone assistance | Depends | 1-800-FDA-1088 (1-800-332-1088), Mon–Fri, 8:00 a.m.–4:30 p.m. ET |
For issues connected to illegal online sales: FDA’s Office of Criminal Investigations runs 1-866-300-4374 or 301-796-8240.
Before you open the form: what to gather
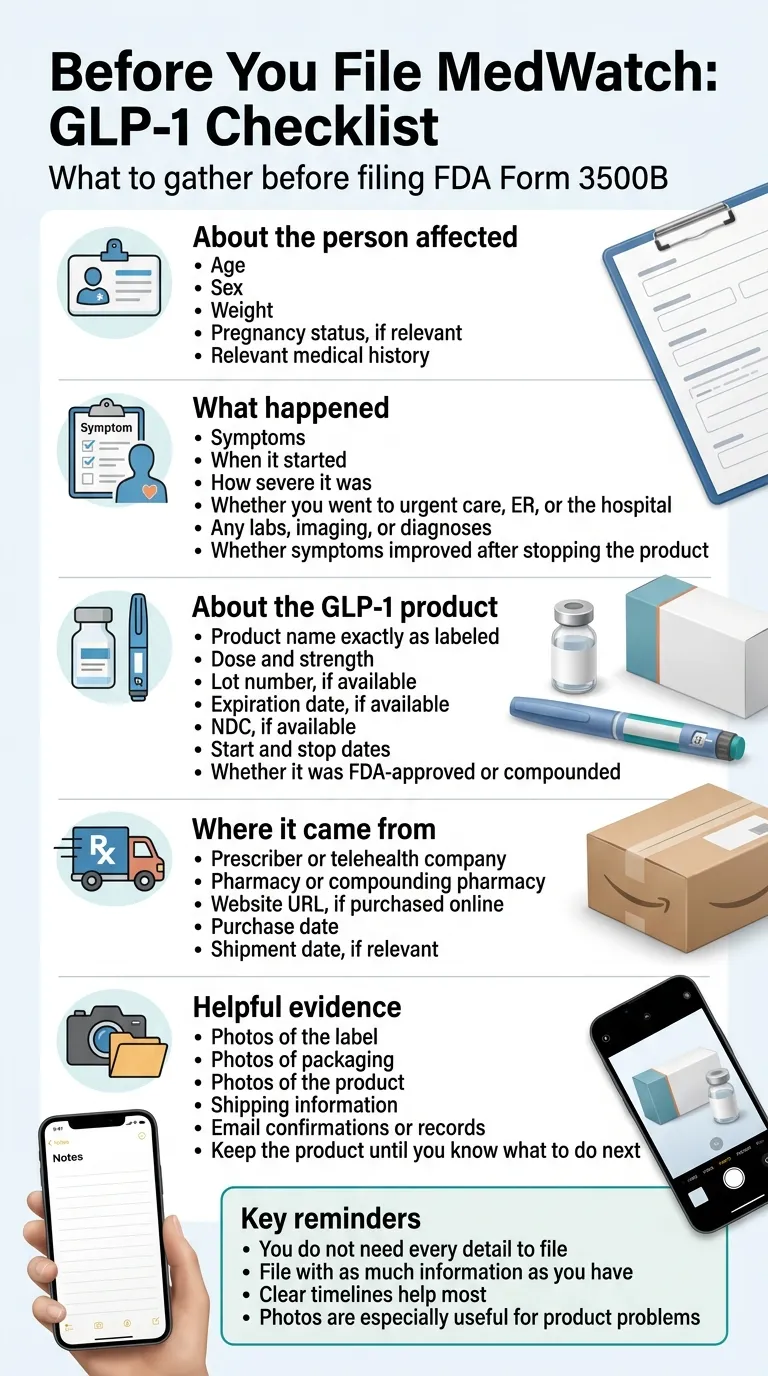
Before you file MedWatch Form 3500B: what to gather. Key reminder: you don’t need every field — file with as much as you have.
About the person affected
- Age, sex, weight, ethnicity (FDA uses demographic patterns to spot signals)
- Pregnancy status
- Relevant medical history (diabetes, thyroid, pancreatitis, etc.)
About what happened
- A timeline — when it started, what changed, what made it better or worse
- What medical care was received (urgent care, ER, hospitalization)
- Test results you have (lipase, amylase, liver enzymes, imaging)
- Whether symptoms improved after stopping the medication
About the product
- Exact name as it appears on the label
- Strength and concentration
- Lot number (critical for product-quality reports)
- Expiration date
- NDC number (for FDA-approved brands)
- Your dose and how often you took it
- Dates you started and stopped
- Who prescribed it and who dispensed it
About where it came from
- The telehealth company or clinic name
- The compounding pharmacy name printed on the label
- The website URL if you bought online
- The date of purchase and shipment date
You do not need every field to file. The FDA says to submit as much as possible even if you don’t have everything. A report from someone who’s lost the packaging and doesn’t remember the lot number is still useful. Don’t let missing details stop you.
Field-by-field walkthrough of the current Form 3500B
Section A — About the problem
This is the narrative — the most important thing you’ll write. Tell the story in order. You’ll indicate: whether this is a side effect, product problem, medication error, or problem after switching; whether the outcome was serious; the date of the event; and what happened in your own words.
Include:
- What you took — product name, dose, how often.
- What happened — symptoms, severity, duration.
- When — dates, and hours after the injection if relevant.
- What care you got — self-managed, urgent care, ER, hospital.
- What the doctors found — labs, imaging, diagnoses.
- What happened when you stopped — improvement is a strong signal for FDA.
Sample narrative you can adapt:
“Started weekly 0.25 mg compounded semaglutide subcutaneous injection on January 3, 2026, sourced from [telehealth company], prescribed by [clinician name or ‘a telehealth provider through the company’]. Dose increased to 0.5 mg on January 31. On February 14, within 6 hours of the injection, I developed severe upper-abdominal pain radiating to my back, nausea, and vomiting that wouldn’t stop. I went to [hospital] ER. Lipase was 1,240 (normal <60). Admitted for 3 days with a diagnosis of acute pancreatitis. No alcohol in the 30 days prior. No gallstones on abdominal ultrasound. No other new medications or supplements. Stopped the compounded semaglutide. Pain resolved over 5 days. Have not restarted any GLP-1.”
Section B — Product availability
Simple but important: do you still have the product? If yes, the FDA may ask for it. Keep it refrigerated and sealed. You’ll also indicate whether you have photos — a yes answer here is always better.
Section C — About the products
This is where GLP-1-specific detail really matters. You’ll enter the product name as printed (including every name on the vial or pen), the manufacturer or compounding pharmacy (for compounded products, this is the pharmacy on the label — not the telehealth company that prescribed it), strength, formulation, lot number, expiration date, NDC (for approved brands), dose and schedule, start and stop dates, why it was prescribed, and where you purchased it.
Section D — About the medical device
Most GLP-1 injection events don’t involve a reportable medical device — skip or indicate not applicable unless the delivery device itself was the problem (a defective pen, a malfunctioning auto-injector). If the pen malfunctioned, describe it here: brand, device name, what went wrong, whether you still have it.
Section E — About the person who had the problem
This is the person the report is about — you, or someone you’re filing for (caregivers, spouses, and parents file 3500B all the time). You’ll enter initials (not a full name), age or date of birth, sex, weight, ethnicity/race, and pregnancy status. Be thorough with medications: GLP-1s slow stomach emptying, which changes how everything else you take is absorbed. FDA can’t tell a drug interaction from a drug effect without the full picture.
Example: “D.K., 44, female, 185 lb, not pregnant. History of PCOS and prediabetes. No personal or family history of pancreatitis or gallbladder disease.”
Section F — About the person filling out this form
This is the reporter section — the person doing the filing. Unlike Section E, this section asks for your last name, first name, address, phone, and email. You can’t be completely anonymous to FDA on the current form.
Online: submit and save the PDF confirmation. Keep that PDF — it’s your proof you filed and you’ll need it if you also file a state Board of Pharmacy complaint. Mail or fax: keep a photocopy before you send it.
Special scenarios: compounded GLP-1s, warm shipments, and counterfeits
This is where most generic “how to file a MedWatch report” articles stop being useful. GLP-1s have their own problem patterns the FDA has publicly flagged. Here’s how to handle each.
“I took a compounded GLP-1 and had a reaction”
- • As of July 31, 2025, FDA had received approximately 605 adverse event reports involving compounded semaglutide and 545 involving compounded tirzepatide — with the agency noting real numbers are almost certainly higher because of the reporting gap.
- • FDA declared the semaglutide shortage officially resolved on February 21, 2025. Enforcement discretion for compounded semaglutide ended April 22, 2025 (503A pharmacies) and May 22, 2025 (503B outsourcing facilities).
- • Products using “semaglutide sodium” or “semaglutide acetate” are different ingredients from the active in FDA-approved drugs and have not been shown to be safe or effective in that form.
To file the state complaint:
- Identify your state’s Board of Pharmacy (search “[your state] Board of Pharmacy”).
- Most have an online complaint form. If not, a phone number and PDF to mail.
- Include: the pharmacy name as it appeared on the label, the product, the dates, what happened, and your MedWatch confirmation number.
- Usually 10–15 minutes on top of your MedWatch filing.
Before your next refill, it’s also worth reading our guide to getting GLP-1 medications safely online — the same vetting checklist would have caught most of the issues the FDA now sees in compounded-product complaints.
“My vial or pen arrived warm”
If you already injected a dose from a warm shipment and something’s wrong: Poison Help first (1-800-222-1222), then MedWatch, then your prescriber.
“My syringe said ‘units’ but my vial is in mg/mL”
This is one of the most dangerous dose-conversion errors the FDA has flagged with compounded semaglutide. An insulin syringe marked in “units” doesn’t correspond to a milligram dose unless someone has done the math for you correctly. People have ended up injecting 5× to 20× their intended dose, some requiring hospitalization.
“I think my product is fake”
FDA’s published red flags for counterfeit GLP-1s:
Your actions if you think you have a counterfeit:
- Don’t use it.
- MedWatch Form 3500B — mark it as a product problem. Upload photos.
- FDA unlawful-online-sales report at
fda.gov/safety/report-problem-fda/reporting-unlawful-sales-medical-products-internet— or call 1-866-300-4374. - NABP Suspicious Online Pharmacy at
nabp.pharmacy. - Novo Nordisk product complaint line: 1-800-727-6500 (for Ozempic®, Wegovy®, Rybelsus®, Saxenda®).
- Eli Lilly product complaint line: 1-800-545-5979 (for Mounjaro®, Zepbound®, Foundayo™).
We check these numbers quarterly. Verify before relying on them.
“Something changed when I switched manufacturers”
If you moved between brand-name products, between compounded providers, or from brand-name to compounded (or vice versa), and your experience changed materially — new side effects, loss of efficacy, different tolerability — that’s worth reporting. In the narrative, explicitly note the switch: “Was on [prior product] for [duration] with [outcome]. Switched to [new product] on [date]. Within [timeframe], experienced [change].”
MedWatch vs. Poison Help vs. Board vs. FTC
| Your problem | Primary channel |
|---|---|
| Side effect, serious reaction, medication error | MedWatch Form 3500B |
| Active overdose or wrong dose happening now | Poison Help (1-800-222-1222) |
| Product quality problem (warm shipment, defective pen, wrong label) | MedWatch Form 3500B |
| Suspected counterfeit or fake pharmacy | MedWatch Form 3500B + FDA unlawful online sales + NABP |
| Deceptive ad, surprise billing, impossible cancellation, "free trial" scam | FTC at reportfraud.ftc.gov |
| Clinical practice concern (prescribing that felt wrong, no real clinician evaluation) | Your state Medical Board |
What MedWatch can — and can’t — do
What your report can contribute to
- Identification of a side-effect pattern the clinical trials missed. Your formal FDA report is the version of a signal the agency can act on.
- FDA or state-level review of a specific pharmacy's practices. Compounded-product reports can contribute to state board action and FDA inspection decisions.
- Label updates. The FDA routinely updates GLP-1 prescribing information based on post-market data.
- Regulatory action against a manufacturer. In March 2026, the FDA issued a warning letter to Novo Nordisk after inspectors identified problems with how the company was tracking and reporting side effects for semaglutide-containing drugs.
- In the most serious cases, a recall.
What your report can’t do
- ✕Get you a refund. MedWatch isn't a consumer-protection system for billing disputes.
- ✕Prove causation by itself. Submission of a report does not constitute an admission that the product caused the event.
- ✕Guarantee you'll hear back. Most consumer reports don't get a follow-up call unless FDA needs specific clarification.
“Even one report can make a difference.” — U.S. Food and Drug Administration
What happens after you submit
- 11. Intake: Your report is received and assigned a case number.
- 22. Coding: Events are coded into standardized medical terms (MedDRA) so they're searchable alongside other reports.
- 33. Review: A safety evaluator reads the narrative and cross-references it with other reports on the same product.
- 44. Aggregation: Patterns are monitored over time. A single unusual report may be the first of a trend.
- 55. Potential action: If a signal emerges, the FDA may request a label change, issue a "Dear Health Care Provider" letter, send a warning letter to the manufacturer, or in serious cases initiate a recall.
fis.fda.gov lets anyone see how many adverse-event reports exist for any drug. Two caveats: the number of reports does not establish causation, and the totals undercount real incidence.Your privacy, honestly explained
- What FDA always has: Your name and contact info from Section F. Used only if FDA needs follow-up.
- What you can withhold from the manufacturer: Check the "do not disclose my identity to the manufacturer" option in Section F.
- What's never public: Your name, address, doctor, and hospital. Not released under FDA disclosure rules.
- What the affected person reports as: Initials only in Section E.
What to do about your ongoing treatment
First: don’t stop abruptly without talking to someone
GLP-1s have metabolic and appetite effects that don’t disappear the moment you stop. For type 2 diabetics, stopping abruptly can cause blood-sugar spikes. For weight-loss patients, appetite and weight regain often happen quickly. None of this means you should keep taking something that hurt you. It means the decision belongs in a conversation with your prescriber.
If you’re considering switching from compounded to FDA-approved
This is a common outcome after a bad compounded experience. Your options:
- Insurance, if your plan covers Wegovy®, Zepbound®, Ozempic®, Mounjaro®, or Foundayo™. Most plans require prior authorization.
- Published-price cash-pay channels. LillyDirect (Eli Lilly) and NovoCare Pharmacy (Novo Nordisk) sell FDA-approved GLP-1s at published cash prices.
- A licensed telehealth clinician. Several FDA-approved-focused services carry the brands above and can handle insurance and prior authorization.
Disclosure: we may earn a commission if you sign up through some of the links below. It doesn’t change what we recommend or what you pay.
Switching to an FDA-approved GLP-1 program
Ro carries Wegovy® pill, Wegovy® pen, Zepbound® pen, Zepbound® KwikPen, and Foundayo™, and includes a free GLP-1 insurance coverage checker that shows what your plan covers before you pay anything.
Check eligibility with Ro →$39 for the first month · Then $149/mo ongoing (or $74/mo annual) · Medication billed separately · Verified April 21, 2026
Still figuring out what’s next for your treatment?
If you’d rather step back and think through all your options — staying, switching, pausing, using insurance — our quiz asks about your situation and outputs a plan in about a minute.
Take the free 60-second GLP-1 matching quiz →Frequently asked questions
What we actually verified for this page
Weight Loss Provider Guide is an independent comparison resource for GLP-1 telehealth providers. We built this page from primary sources. Here’s what we verified and when:
- MedWatch online portal at fda.gov/medwatch — verified April 21, 2026
- Form 3500B current version (OMB No. 0910-0291, expires September 30, 2027) — six sections A–F — verified against the current FDA PDF, April 21, 2026
- FDA MedWatch phone (1-800-FDA-1088 / 1-800-332-1088) — verified April 21, 2026
- FDA MedWatch fax (1-800-FDA-0178) — verified April 21, 2026
- MedWatch mailing address (White Oak Campus, Building 22, G0207, Silver Spring, MD 20993) — verified April 21, 2026
- Poison Help national hotline (1-800-222-1222) — verified April 21, 2026
- FDA unlawful-online-sales hotline (1-866-300-4374) — verified April 21, 2026
- Retirement of Consumer Complaint Coordinator phone lines — verified against FDA's current CCC page, April 21, 2026
- 21 CFR §314.80 as the mandatory postmarketing reporting rule for approved drug applicants — verified April 21, 2026
- FDA's statement that state-licensed pharmacies are not federally required to report adverse events — verified against FDA's 'Concerns with Unapproved GLP-1 Drugs Used for Weight Loss' page, April 21, 2026
- Compounded adverse-event totals (~605 compounded semaglutide / ~545 compounded tirzepatide as of July 31, 2025) — FDA source page
- Semaglutide shortage resolution date (February 21, 2025) and enforcement discretion end dates (April 22, 2025 for 503A; May 22, 2025 for 503B) — FDA source
- SI/B warning removal request (Saxenda, Wegovy, Zepbound) — FDA Drug Safety Communication, early 2026
- March 2026 Novo Nordisk FDA warning letter for pharmacovigilance failures — FDA Warning Letters database
- December 5, 2025 counterfeit Ozempic seizure by FDA (lot PAR1229 counterfeits) — FDA announcement
- Novo Nordisk product complaint line (1-800-727-6500) and Eli Lilly product complaint line (1-800-545-5979) — verified April 21, 2026
- Ro Body membership pricing ($39 first month, $149/month ongoing, as low as $74/month annual prepay) — verified against Ro pricing pages, April 21, 2026
If you notice anything out of date or inaccurate, email [email protected] — we take factual corrections seriously and update the same day.
Closing
Filing a MedWatch report is one of the most useful things a patient can do that also happens to take almost no effort. You don’t need a lawyer. You don’t need your doctor’s permission. You don’t need to be sure the drug was the cause — the FDA’s job is to figure that out, not yours. You need about 30 minutes, the label off your product, and a clear head.
Sources
Every factual claim on this page traces to one of the sources below. Click through to verify anything directly.
- FDA: MedWatch — The FDA Safety Information and Adverse Event Reporting Program
- FDA: Reporting Serious Problems to FDA
- FDA: FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss
- FDA: Compounding Risk Alerts
- FDA: FAERS/AEMS Public Dashboard
- FDA Drug Safety Communication: Request to Remove Suicidal Ideation Warning from GLP-1 RA Medications
- FDA Warning Letter to Novo Nordisk (March 2026)
- FDA announcement on counterfeit Ozempic seizure (December 5, 2025)
- NABP: Suspicious Online Pharmacy reporting
- Poison Help national hotline information
- 21 CFR §314.80 (postmarketing reporting of adverse drug experiences)
Related guides
Weight Loss Provider Guide is an independent comparison resource for GLP-1 telehealth providers. Advertising disclosure. Editorial standards.
Last verified against FDA sources: April 21, 2026. Next scheduled review: July 2026, or immediately after any new FDA Drug Safety Communication for GLP-1 receptor agonists.