Mounjaro Side Effects: What’s Normal, What’s Serious, and When to Get Help
Most Mounjaro side effects are gut-related, temporary, and manageable. According to the FDA-approved prescribing information, the most common are nausea (up to 18%), diarrhea (12–17%), decreased appetite, vomiting, constipation, indigestion, and stomach pain. They tend to show up when you first start or raise your dose — and they tend to settle down.
But not every symptom is a “wait it out” symptom. Some are red flags for pancreatitis, gallbladder disease, severe dehydration, or allergic reactions that need medical attention fast.
This guide covers both. We pulled every number from FDA trial data, separated the Mounjaro diabetes numbers from the Zepbound weight-loss numbers (because they’re meaningfully different and most articles lump them together), built a first-week timeline, and created a clear “normal vs. call your doctor vs. ER” checklist you can reference at 2 AM when something feels off.
Here’s something else most pages won’t tell you: if you’re using compounded tirzepatide from an online clinic, your side-effect picture may look different than what’s in these tables — and that matters. We cover that too.
For informational purposes only—not medical advice.

Affiliate disclosure: We earn commissions from qualifying provider referrals. This never influences our reporting of side-effect data, which is sourced exclusively from FDA labels and published clinical trials.
What Are the Most Common Mounjaro Side Effects?
Here’s the straight answer from the FDA label. These are the side effects that showed up in at least 5% of people during Mounjaro’s clinical trials for type 2 diabetes:
Official Mounjaro Side-Effect Rates (Type 2 Diabetes Trials)
| Side Effect | 5 mg | 10 mg | 15 mg | Placebo |
|---|---|---|---|---|
| Nausea | 12% | 15% | 18% | 4% |
| Diarrhea | 12% | 13% | 17% | 9% |
| Decreased appetite | 5% | 10% | 11% | 1% |
| Vomiting | 5% | 5% | 9% | 2% |
| Constipation | 6% | 6% | 7% | 1% |
| Dyspepsia (indigestion) | 8% | 8% | 5% | 3% |
| Abdominal pain | 6% | 5% | 5% | 4% |
Source: FDA Prescribing Information for Mounjaro (tirzepatide), Table 1 — pooled placebo-controlled adult trials (SURPASS-1 and SURPASS-5). The 2.5 mg dosage is for treatment initiation and is not intended for glycemic control.
A few things jump out. Side effects climb with the dose. The 15 mg column is notably higher than 5 mg across the board. And the most important pattern: most nausea, vomiting, and diarrhea occurred during dose escalation and decreased over time. That’s straight from the label, and it’s the single most reassuring sentence in this entire guide.
But these numbers only tell part of the story — especially if you’re using tirzepatide for weight loss.
Mounjaro vs. Zepbound Side Effects: Why This Page Shows Both
Here’s something most “Mounjaro side effects” articles get wrong: they ignore the weight-loss data entirely.
Mounjaro is the brand name for tirzepatide approved for type 2 diabetes. Zepbound is the brand name for the exact same medication approved for weight management. Same drug. Same manufacturer (Eli Lilly). But the clinical trial populations were different — and the side-effect rates are meaningfully different too.
If you’re taking tirzepatide for weight loss (whether through Zepbound, off-label Mounjaro, or an online provider), the Zepbound numbers are more relevant to your experience.
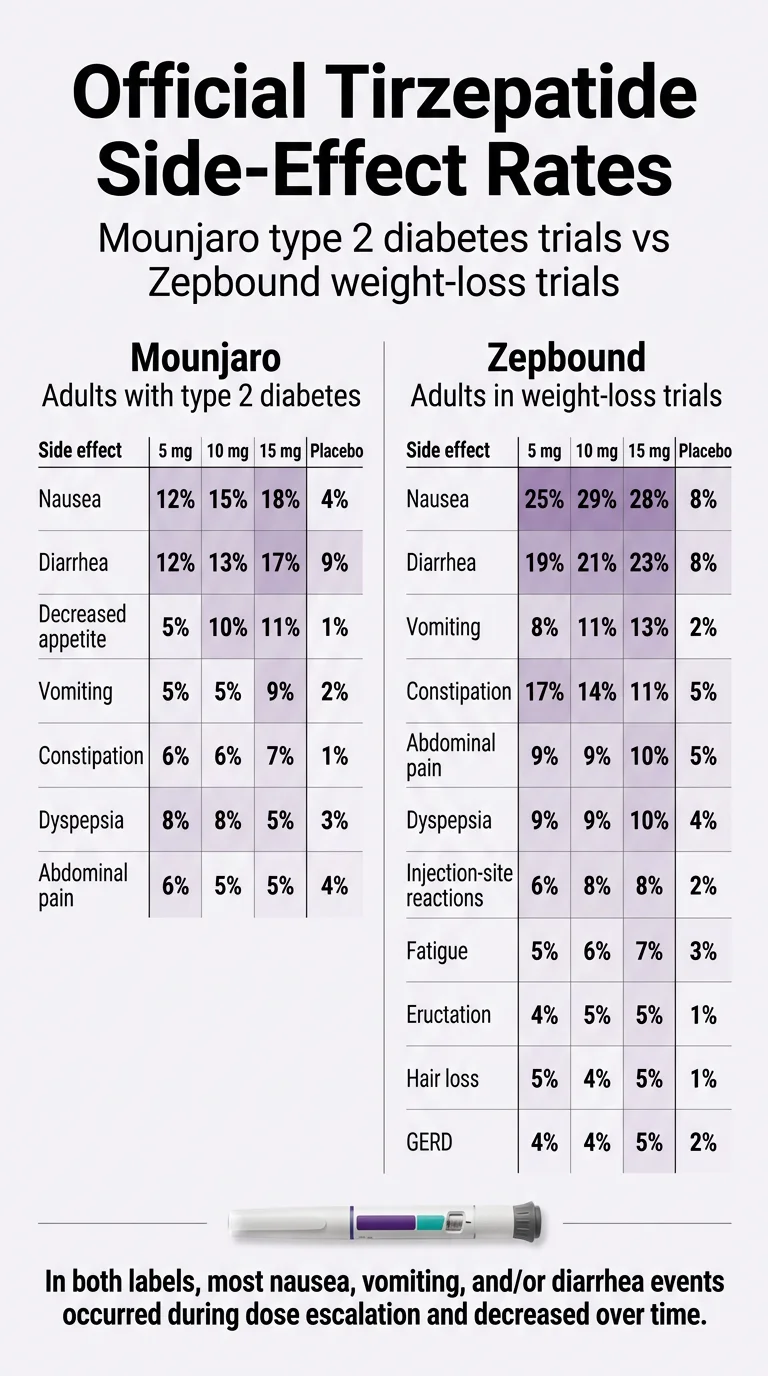
Selected Common Adverse Reactions — Zepbound (Weight-Loss Trials)
| Side Effect | 5 mg | 10 mg | 15 mg | Placebo |
|---|---|---|---|---|
| Nausea | 25% | 29% | 28% | 8% |
| Diarrhea | 19% | 21% | 23% | 8% |
| Vomiting | 8% | 11% | 13% | 2% |
| Constipation | 17% | 14% | 11% | 5% |
| Abdominal pain | 9% | 9% | 10% | 5% |
| Dyspepsia | 9% | 9% | 10% | 4% |
| Injection-site reactions | 6% | 8% | 8% | 2% |
| Fatigue | 5% | 6% | 7% | 3% |
| Hypersensitivity reactions | 5% | 5% | 5% | 3% |
| Eructation (burping) | 4% | 5% | 5% | 1% |
| Hair loss (alopecia) | 5% | 4% | 5% | 1% |
| GERD / acid reflux | 4% | 4% | 5% | 2% |
Source: FDA Prescribing Information for Zepbound (tirzepatide), pooled placebo-controlled weight-reduction trials (Study 1 and Study 2). This is a selection of common adverse reactions; the full label includes additional rows.
Notice the differences. Nausea hits up to 29% in the weight-loss trials versus 18% in the diabetes trials. The Zepbound label also lists fatigue, burping, hair loss, reflux, and injection-site reactions as common — none of which made the “common” list on the Mounjaro label.
Why does this matter to you? Because if you’re reading this page after hearing about Mounjaro on social media or getting it for weight loss, the numbers from the weight-loss trials are the ones that will match your experience more closely. And if you’re reading articles that only cite the Mounjaro diabetes numbers, you’re getting an incomplete picture.
Not sure which GLP-1 path fits your body, budget, and goals?
Our free 60-second matching quiz compares your options based on your situation — not just price.
Take the GLP-1 Match QuizI Have a Symptom Right Now. Is It Normal, Dangerous, or a Reason to Stop?
This is the section you might need at midnight when something feels wrong. We organized every major tirzepatide-related symptom by what to do about it.

Quick Triage Guide
| Symptom | Usually Normal? | Call Your Doctor | Go to Urgent Care / ER |
|---|---|---|---|
| Mild nausea after meals | ✓ | If it persists and does not go away | If you can’t keep any food or fluids down |
| Vomiting after overeating | ✓ | If it persists beyond several days | If you can’t stop vomiting or see blood |
| Loose stools / mild diarrhea | ✓ | If it does not go away | If severe, bloody, or you’re getting dehydrated |
| Constipation | ✓ | If persistent with no relief from home remedies | If paired with severe abdominal pain |
| Decreased appetite | ✓ | If you’re consistently eating very little | If you’re losing consciousness or very weak |
| Burping / sulfur burps | ✓ | If persistent and bothersome | — |
| Mild heartburn / reflux | ✓ | If worsening or not responding to OTC antacids | If chest pain or trouble breathing |
| Bloating / gas | ✓ | If severe or persistent | — |
| Mild fatigue | ✓ | If it doesn’t improve after a few weeks | If paired with confusion or rapid heartbeat |
| Injection-site redness/itch | ✓ | If it spreads or worsens | If spreading hives, breathing trouble |
| Hair thinning (after 2–3 months) | ✓ | Worth mentioning at next visit | — |
| Dizziness / lightheadedness | ⚠ | Yes — may signal low blood sugar or dehydration | If you faint or feel severely disoriented |
| Severe stomach pain → back | ✘ | — | Yes — possible pancreatitis |
| Upper right abdominal pain + fever | ✘ | — | Yes — possible gallbladder disease |
| Face/lip/throat swelling | ✘ | — | Yes — allergic reaction. Call 911. |
| Difficulty breathing or swallowing | ✘ | — | Yes — call 911. |
| Neck lump, persistent hoarseness | ✘ | Yes — get evaluated promptly | If breathing/swallowing affected |
| Dark/reduced urine + swelling | ✘ | Yes — possible kidney issue | If severe or sudden |
| Vision changes | ✘ | Yes — same day if possible | If sudden loss of vision |
| Signs of severe low blood sugar | ✘ | — | Yes — confusion, seizure, unconsciousness |
This is informational guidance, not a replacement for medical advice. When in doubt, call your provider.
Now let’s walk through the major symptoms in detail.
Nausea and Vomiting
This is the side effect people worry about most — and the one most likely to hit you.
Nausea is reported by up to 18% of Mounjaro users (diabetes trials) and up to 29% of Zepbound users (weight-loss trials). Vomiting is less common — up to 9% and 13%, respectively.
Here’s the pattern that matters: it’s worst when you first start and each time you increase your dose. The official label confirms this. Your body is adjusting to a medication that fundamentally changes how fast your stomach empties food, and that process takes time.
For most people, nausea at the 2.5 mg starting dose is mild or nonexistent. The first real test usually comes at the 5 mg increase (week 5). By the time your body acclimates to each dose, symptoms typically ease.
What actually helps:
- Eat smaller portions, more often. Four to six mini-meals beats three large ones.
- Stop eating the instant you feel full. On tirzepatide, your “full” signal arrives much earlier and means business.
- Cut way back on greasy, fried, and high-fat foods. They already digest slowly — slower gastric emptying makes this worse.
- Eat slowly. Seriously. Put your fork down between bites.
- Ginger tea or ginger chews before meals can help.
- Stay upright for at least 30 minutes after eating.
- Talk to your prescriber about anti-nausea support if home measures aren’t enough.
Red flag:
Severe vomiting that won’t stop, vomiting with intense abdominal pain radiating to your back, or inability to keep any fluids down. These warrant urgent medical evaluation — persistent vomiting can lead to dehydration and kidney injury.
Diarrhea
Reported by 12–17% (Mounjaro) and 19–23% (Zepbound) of trial participants. Like nausea, it’s most common during dose escalation and usually improves.
What helps: Stay hydrated — at least 64 ounces of water daily, more if you’re losing fluids. Temporarily ease off high-fiber foods. Stick with bland, easy-to-digest options. Avoid dairy if it worsens things. A BRAT-style approach (bananas, rice, applesauce, toast) can help during flare-ups.
Red flag:
Diarrhea that persists for weeks without improvement, bloody stool, or signs of dehydration (dark urine, dizziness when you stand, dry mouth that won’t go away).
Constipation
About 6–7% in diabetes trials and 11–17% in weight-loss trials. This one makes physiological sense: tirzepatide slows your entire digestive tract, and what goes in slower comes out slower.
What helps: Drink more water than you think you need. Add fiber back gradually (not all at once). Walk daily — even 20 minutes helps. A gentle stool softener can help, but check with your prescriber first.
Red flag:
No bowel movement for several days with increasing pain, or blood in your stool.
Stomach Pain and Indigestion
Abdominal pain shows up in 5–10% of users depending on the trial. Indigestion (dyspepsia) in 5–10%.
What helps: Smaller meals, avoiding carbonated beverages, not lying flat after eating, and wearing loose-fitting clothing around your midsection. Sounds simple. It genuinely makes a difference.
Red flag:
This is the one to pay attention to. Severe pain in your upper abdomen that radiates to your back and doesn’t go away could be pancreatitis. Upper right-side pain with fever could be gallbladder disease. Both need medical evaluation — don’t push through severe abdominal pain on this medication.
Sulfur Burps, Gas, and Bloating
Eructation (the fancy word for burping) is listed as common in the Zepbound label: 4–5%. The sulfur burps specifically aren’t broken out in trial data, but they’re a hallmark complaint in patient communities. They’re unpleasant, sometimes embarrassing, and entirely related to slower digestion.
What helps: Eat slowly. Avoid carbonated drinks. Reduce high-sulfur foods (eggs, cruciferous vegetables, garlic) during the worst of it. Gas-reducing products like simethicone can take the edge off. Most people find these improve as their body adjusts.
Heartburn and Acid Reflux (GERD)
Listed as common in the Zepbound label (4–5%). Makes sense — food staying in your stomach longer increases the chance of acid backing up into your esophagus, especially if you eat too much or lie down after meals.
What helps: Don’t eat close to bedtime. Elevate the head of your bed. Avoid spicy, acidic, and fatty trigger foods. Over-the-counter antacids can help. If reflux is persistent or worsening, talk to your prescriber — you may need a proton pump inhibitor or a dose adjustment.
Fatigue and Low Energy
Fatigue is listed as a common side effect in the Zepbound label (5–7%). It is not listed among the common adverse reactions in the Mounjaro label, but it is frequently reported by patients. People report it. It’s real.
Why it happens: You’re likely eating significantly fewer calories than before. Your metabolism is shifting. Your blood sugar dynamics are changing. All of that can leave you feeling drained, especially in the first few weeks.
What helps: Make sure you’re eating enough — reduced appetite is not permission to starve yourself. Focus on nutrient-dense meals. Prioritize protein. Stay hydrated. Gentle exercise (even walking) often helps more than rest. And get your sleep — your body is doing a lot of adjusting.
Red flag:
Fatigue paired with confusion, rapid heartbeat, or shakiness could signal low blood sugar, especially if you’re also taking insulin or a sulfonylurea. Check your glucose and talk to your prescriber.
Injection-Site Reactions
Reported in 3.2% of Mounjaro trial participants and 6–8% of Zepbound users. Symptoms: redness, swelling, itching, or mild pain where you injected.
What helps: Rotate your injection site every week (abdomen, thigh, or upper arm). Let the alcohol swab dry completely before injecting. Don’t use the exact same spot twice in a row.
Red flag:
Redness that keeps spreading, warmth or heat at the site (possible infection), or hives spreading beyond the injection area (allergic reaction — seek care immediately).
Hair Loss
This one scares people, so let’s be precise.
Hair loss was not listed as a common side effect in the original Mounjaro diabetes trials. But it IS listed as common in the Zepbound weight-loss label: 4–5% overall. In female participants specifically, the rate was 7.1%. In males, 0.5%.
What’s happening: This is almost certainly telogen effluvium — a temporary form of hair shedding triggered by rapid weight loss, not the drug attacking your hair follicles. When your body loses weight quickly, it redirects resources away from non-essential functions like hair growth. There’s a 2–3 month delay between the weight loss and the shedding, which is why it often catches people off guard.
What helps: Prioritize protein at every meal (25–30 grams). Take a quality multivitamin. Don’t crash-diet on top of the medication’s appetite suppression — your hair needs nutrients. Stay hydrated. Be gentle with your hair (avoid tight styles, excessive heat).
The reassurance: For most people, this is temporary. Hair typically regrows once your weight stabilizes and your nutritional intake is consistent. If shedding is severe or doesn’t improve, see your doctor to rule out thyroid issues or iron deficiency.
Dizziness and Lightheadedness
Not prominently listed in the label as a standalone common side effect, but commonly reported — and often a secondary signal.
It could mean:
- Low blood sugar (especially if you’re also on insulin or a sulfonylurea)
- Dehydration (from not drinking enough or from GI fluid losses)
- Blood pressure drop (some people experience lower blood pressure as they lose weight)
What to do: If you feel dizzy, sit down. Check your blood sugar if you have a monitor. Drink water. If it’s recurring, bring it up with your prescriber. If you feel faint, confused, or it gets worse, seek medical evaluation.
Low Blood Sugar (Hypoglycemia)
Tirzepatide itself doesn’t typically cause low blood sugar when used alone. The mechanism is glucose-dependent — it only signals your pancreas to release insulin when your blood sugar is elevated.
But if you’re taking Mounjaro alongside insulin or a sulfonylurea, the risk is real: 10–19% with insulin, 10–14% with sulfonylureas in clinical trials.
Watch for: Shakiness, sweating, rapid heartbeat, confusion, irritability, blurred vision, weakness.
What to do: Your prescriber may need to adjust your insulin or sulfonylurea dose when you start Mounjaro. Don’t adjust these medications on your own.
What to Expect Your First Week on Mounjaro (and After Each Dose Increase)
This is one of the most-searched questions around this medication, and most articles don’t give you a clear timeline. Here’s what we’ve pieced together from trial data, FDA labeling, and clinical guidance.
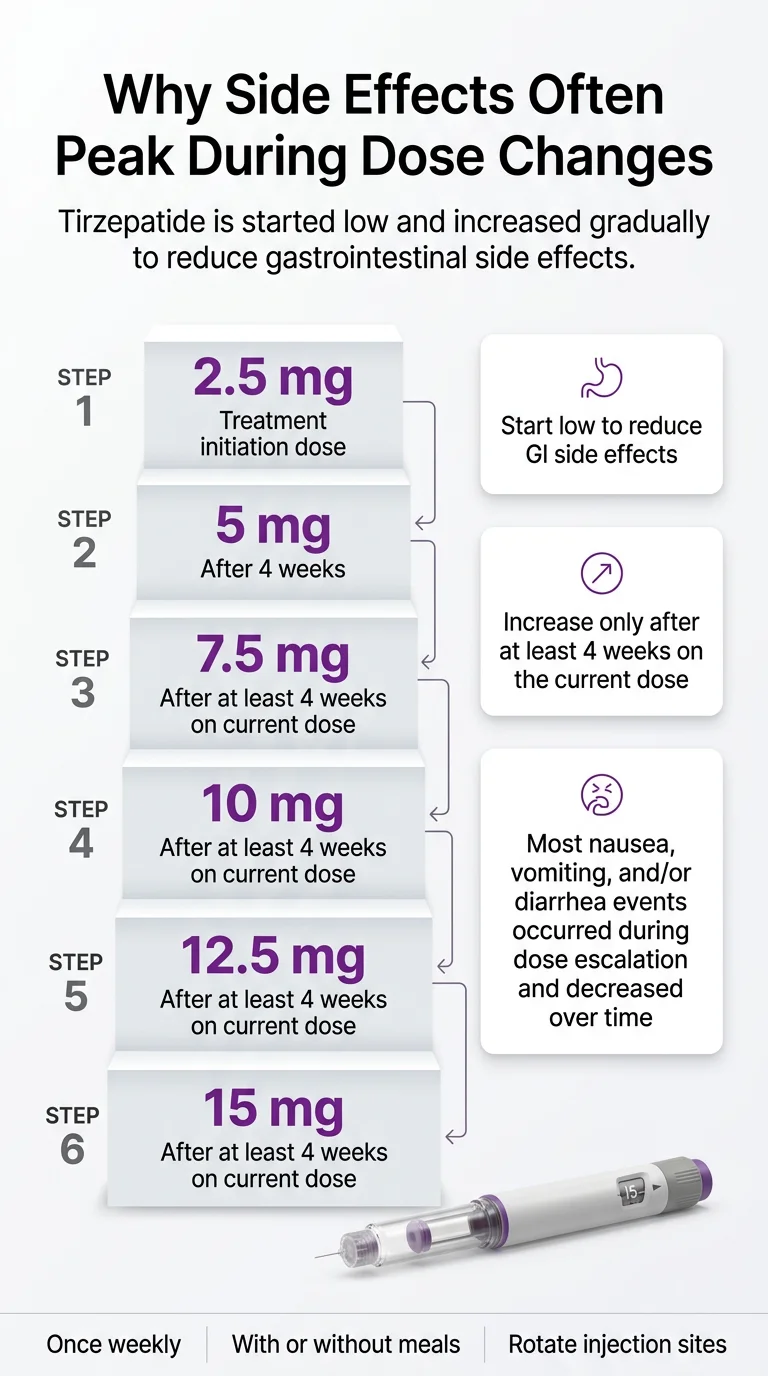
Days 1–3 (First Injection at 2.5 mg)
Many people feel nothing. The 2.5 mg dose is a starter dose — it exists to let your body begin adjusting, not to control blood sugar or cause significant weight loss. Some people notice a slight decrease in appetite or mild nausea within the first day or two. Your injection site may be mildly sore. Some people feel a bit tired. Others feel completely normal.
Don’t panic if nothing happens. Don’t panic if something does. Both are normal at this stage.
Week 1
Appetite reduction is usually the first noticeable change. You may find yourself getting full after half a sandwich when you’d normally eat the whole thing. Mild nausea is possible, particularly around meals. Energy may dip slightly as your body processes fewer calories.
Weeks 2–4 (Still at 2.5 mg)
Your body continues acclimating. Whatever mild symptoms appeared in week one often start fading. You’re building a new baseline. Weight change at this stage is typically minimal.
Week 5 (Dose Increase to 5 mg)
This is where many people notice a bigger shift. The move from the starter dose to the first maintenance dose is commonly reported as a more noticeable adjustment period. Nausea may return or appear for the first time. Your stomach will need to readjust to a stronger signal to slow down. GI symptoms like loose stools, bloating, or constipation may show up.
The label supports what matters here: the majority of nausea, vomiting, and diarrhea occurred during dose escalation and decreased over time.
Each Subsequent Dose Increase
The pattern tends to repeat — a temporary flare of GI symptoms that settles as your body acclimates. The official labels confirm that GI adverse reactions are dose-related and concentrated during escalation periods.
A Note on Timing
Some patients and prescribers have found that injecting in the evening (so the initial hours of adjustment happen while you sleep) reduces the daytime impact of nausea. This isn’t in the official label — it’s practitioner observation. Worth discussing with your provider if you’re struggling.
How Long Do Mounjaro Side Effects Last?
The honest answer: it depends on the side effect.
| Side Effect | Typical Duration | When to Worry |
|---|---|---|
| Nausea | Often improves as body adjusts after starting or increasing dose | Severe or does not go away |
| Diarrhea | Often improves over time | Persistent or worsening |
| Constipation | Variable — may persist at some level | Severe pain, no movement for days |
| Decreased appetite | Often ongoing (this is partly how the drug works) | If caloric intake drops dangerously low |
| Fatigue | Usually improves as body adjusts | Severe or worsening |
| Burping / reflux | Variable — weeks to months for some | Persistent GERD that needs clinical management |
| Injection-site reactions | Typically brief | Spreading redness, signs of infection |
| Hair loss | May begin months into treatment, can last weeks to months | Severe or doesn’t improve as weight stabilizes |
| Severe abdominal pain | Should not be waited out | Seek immediate medical evaluation |
The most important takeaway: most GI side effects are front-loaded. The labels confirm that the majority of nausea, vomiting, and diarrhea occurred during dose escalation and decreased over time. If your symptoms don’t improve — or if they get worse — that’s a conversation for your prescriber, not something to push through alone.
Why Does Mounjaro Cause These Side Effects?
Understanding the “why” makes the symptoms less frightening and helps you manage them.
Mounjaro (tirzepatide) is a dual GIP and GLP-1 receptor agonist. In plain language: it mimics two gut hormones your body already produces naturally. These hormones do three major things:
- Tell your brain you’re full — which reduces appetite and food intake.
- Slow down how fast your stomach empties food — which keeps blood sugar from spiking after meals.
- Help your pancreas release the right amount of insulin at the right time — which improves blood sugar control.
That second one — slower gastric emptying — is the root cause of almost every common side effect on this list. When food sits in your stomach longer than your body is accustomed to, you get nausea, bloating, fullness after small meals, reflux, sulfur burps, and changes in bowel habits. Your gut is recalibrating to a new normal.
This is also why portion size suddenly matters so much. Before tirzepatide, your stomach emptied food at one speed. Now it’s slower. If you eat the same portions you ate before, your stomach literally can’t process it as fast — and it lets you know.
The dose titration schedule (starting low at 2.5 mg, increasing every 4+ weeks) exists specifically to give your digestive system time to adjust gradually rather than all at once. People who escalate too quickly tend to have worse side effects. This is one of the most important things to understand about this medication.
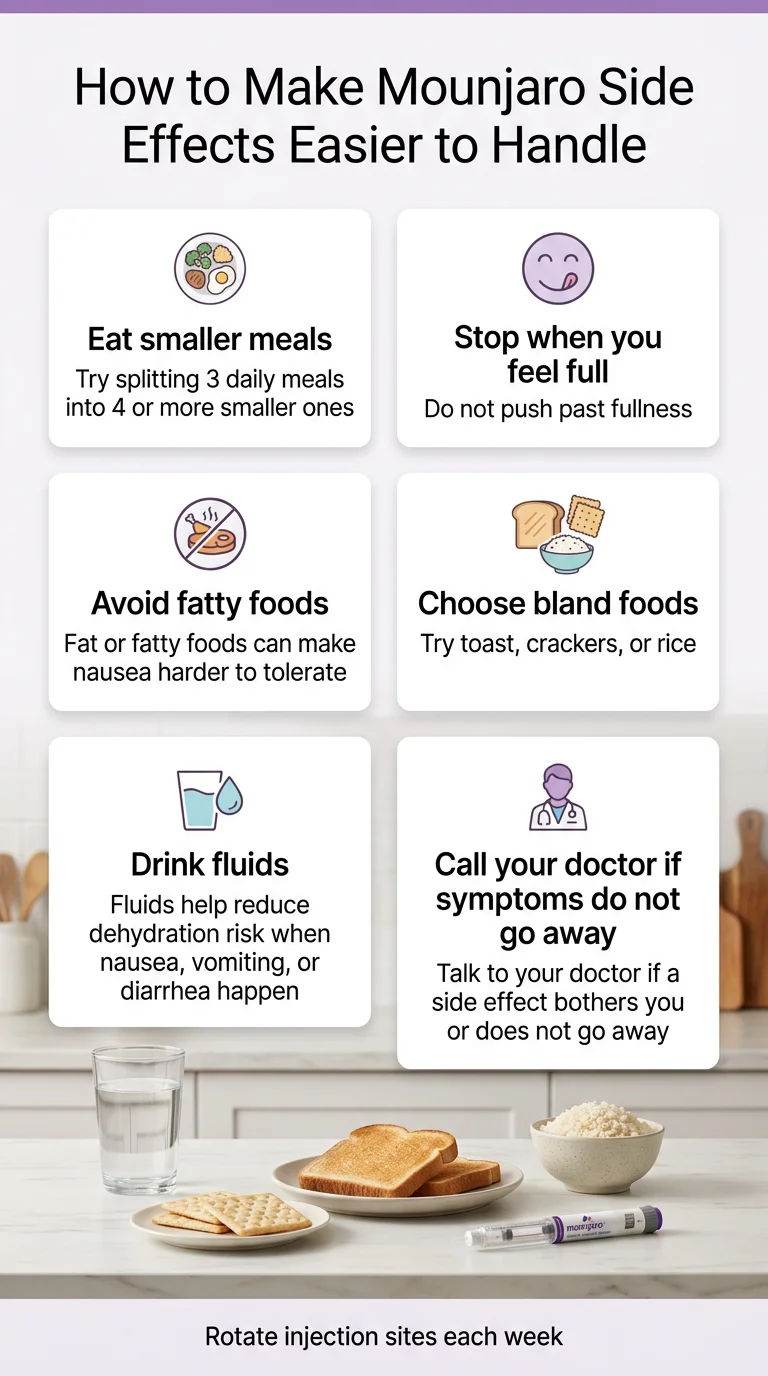
How to Make Mounjaro Side Effects Easier to Handle
These aren’t generic “talk to your doctor” tips. These are specific, practical, and backed by what the manufacturer, clinical guidance, and experienced prescribers consistently recommend.
Respect the titration.
Don’t ask to jump doses faster because the weight isn’t coming off fast enough yet. The 2.5 mg dose exists for a reason. Each step up should be at least 4 weeks. Some prescribers go even slower — and slower is almost always better than sicker.
Eat small.
We mean it. Think snack-sized meals, 4–6 times a day, rather than 3 standard meals. Your stomach is processing food at a different pace now.
Stop when you’re full.
This sounds obvious, but it’s the #1 mistake new users make. The medication creates a strong satiety signal. Eating past it is the fastest path to nausea and vomiting.
Drop the greasy food.
Fatty, fried foods digest slowly under normal conditions. Add slowed gastric emptying on top of that and you’re asking for trouble. Lean proteins, vegetables, and simpler preparations will treat you better.
Hydrate like it’s your job.
Minimum 64 ounces of water daily — ideally more. This prevents constipation, protects your kidneys if you do get diarrhea or vomiting, and helps with fatigue. Sip throughout the day rather than chugging large amounts.
Prioritize protein.
Aim for 25–30 grams per meal. This protects your muscle mass during weight loss, supports hair health, and keeps you feeling stronger on fewer total calories.
Stay upright after meals.
At least 30 minutes. Lying down too soon worsens nausea and reflux.
Keep a symptom journal.
Track what you ate, when symptoms hit, and how severe they were. Patterns emerge quickly — and this data is genuinely useful when you talk to your prescriber about dose adjustments.
Move your body.
Even gentle walking after meals helps digestion and reduces bloating. You don’t need intense workouts — a 15–20 minute walk does more than you’d expect.
Communicate early.
Don’t wait until you’re miserable to contact your prescriber. A simple message about persistent nausea at week two might result in a slower titration, a supportive medication, or reassurance that what you’re feeling is temporary. The worst move is suffering in silence and then quitting.
What NOT to Do
A few mistakes we see repeatedly:
- Don’t keep escalating through severe symptoms without your prescriber’s guidance. More is not always better. A dose that makes you miserable is not a dose that’s working for you.
- Don’t rely on social media dosing advice. The person telling you to “push through” the 10 mg nausea because “it gets better” doesn’t know your medical history.
- Don’t skip meals entirely. Reduced appetite doesn’t mean zero appetite is healthy. Your body still needs fuel — especially protein.
- Don’t ignore dehydration. This is how manageable GI side effects become kidney problems. If you’re losing fluids to diarrhea or vomiting, replacing them is not optional.
- Don’t combine alcohol heavily with tirzepatide, especially if you’re also on diabetes medications. Alcohol can worsen low blood sugar risk and amplify GI symptoms.
When to Call Your Doctor, Go to Urgent Care, or Call 911
We built this section to be blunt and skimmable. Save it on your phone.
Normal — Monitor at Home
- Mild nausea that improves with smaller meals
- Reduced appetite (this is partly how the drug works)
- Mild constipation that responds to water and fiber
- Mild fatigue in the first few weeks
- Slight soreness or redness at the injection site that fades within a day
- Occasional burping or gas
- Mild bloating after meals
Message Your Prescriber
- Nausea or vomiting that persists and does not improve
- Diarrhea that does not go away
- Constipation that persists with no improvement from home measures
- Noticeable hair shedding (not urgent, but worth discussing)
- Signs of dehydration: dark urine, dry mouth, dizziness when standing
- Fatigue that does not improve
- Mood changes, unusual anxiety, or depression
- Persistent or worsening heartburn/reflux
- New vision changes in anyone with a history of diabetic retinopathy
- Possible gallbladder symptoms: pain in upper right abdomen after fatty meals
- Any side effect that’s making you consider stopping
Urgent Care or ER — Don’t Wait
- Severe abdominal pain, especially radiating to your back — possible pancreatitis
- Vomiting that won’t stop — dehydration risk and possible kidney injury
- Swelling of face, lips, tongue, or throat — allergic reaction. Call 911.
- Difficulty breathing or swallowing — call 911
- Severe or spreading rash or hives — allergic reaction
- Lump in your neck + persistent hoarseness + trouble swallowing — thyroid warning signs; get evaluated promptly
- Severe upper right abdominal pain with fever or jaundice — possible gallbladder emergency
- Signs of severe low blood sugar: confusion, seizure, loss of consciousness — call 911 (most relevant if taking insulin or a sulfonylurea alongside Mounjaro)
- Sudden vision loss — seek emergency eye evaluation
This list is sourced from the FDA Medication Guide for Mounjaro and the official label’s warnings section. When in doubt, call. It’s always better to call and be reassured than to wait and wish you hadn’t.
Who Is More Likely to Have Stronger or Riskier Side Effects?
Mounjaro is well-tolerated by most people who use it. But certain groups carry higher risk for specific complications. If any of these describe you, make sure your prescriber knows before you start.
People Taking Insulin or Sulfonylureas
Tirzepatide plus insulin or a sulfonylurea significantly increases your risk of hypoglycemia. In trials, 10–19% of people on Mounjaro plus a long-acting insulin experienced low blood sugar, versus single digits on Mounjaro alone. Your prescriber may need to reduce your insulin or sulfonylurea dose when starting tirzepatide.
People With Kidney Issues or Dehydration Risk
The FDA label warns about acute kidney injury, mostly linked to severe GI side effects (vomiting, diarrhea) that cause dehydration. If you have existing kidney disease, your prescriber should monitor your kidney function more closely. Your job: hydrate aggressively, and don’t push through severe vomiting without calling your provider.
People With a History of Gallbladder Problems
Rapid weight loss — from any cause — increases gallstone risk. Tirzepatide’s clinical trials showed gallbladder events in about 0.6% of Mounjaro users and roughly 1–2% over two years in weight-loss trials. If you have a history of gallstones or gallbladder issues, be vigilant about upper right abdominal pain, especially after fatty meals.
People With Diabetic Retinopathy
Rapid improvement in blood sugar has been associated with a temporary worsening of diabetic retinopathy. Mounjaro has not been studied in patients with nonproliferative diabetic retinopathy requiring acute therapy, proliferative diabetic retinopathy, or diabetic macular edema. If you have a history of diabetic retinopathy, your doctor should monitor you for progression when starting tirzepatide.
People With Severe Gastroparesis
The FDA label says Mounjaro has not been studied in patients with severe gastroparesis and is not recommended for them. Tirzepatide slows gastric emptying by design — layering that on top of a stomach that already empties too slowly can cause serious problems.
People Facing Surgery, Anesthesia, or Sedation Procedures
This is a newer concern and an important one. Because tirzepatide slows gastric emptying, there’s a risk of pulmonary aspiration (inhaling stomach contents) during sedation or anesthesia. The label notes rare postmarketing reports of this in patients receiving GLP-1 receptor agonists who had residual gastric contents despite following preoperative fasting recommendations. The label also states that available data are insufficient to know whether temporarily discontinuing tirzepatide or modifying fasting recommendations would reduce this risk. If you have a scheduled surgery, endoscopy, or colonoscopy, tell your surgical and anesthesia team that you’re on tirzepatide. Do not skip this conversation.
Pregnant People, People Trying to Conceive, and Oral Birth Control Users
Mounjaro’s label says to use it during pregnancy only if the potential benefit justifies the risk — animal studies showed fetal abnormalities at human-relevant doses. Zepbound is even blunter: weight loss offers no benefit to pregnant people and may cause fetal harm. Discontinue when pregnancy is recognized.
Planning pregnancy? If you are pregnant, may become pregnant, or are trying to conceive, talk with your prescriber before using tirzepatide. The Mounjaro label states it may cause fetal harm based on animal data, and the Zepbound label says to discontinue when pregnancy is recognized.
On the pill? This is frequently missed: tirzepatide’s slowing of gastric emptying can reduce the absorption of oral contraceptives. Both labels recommend switching to a non-oral birth control method (IUD, implant, condom, etc.) or adding a barrier method for 4 weeks after starting and for 4 weeks after each dose increase.
People Taking Other Oral Medications
Because tirzepatide slows gastric emptying, it can affect how quickly your body absorbs other oral medications. This doesn’t mean you need to stop other drugs, but your prescriber should know everything you take so they can adjust timing or monitoring if needed. This is especially relevant for medications with narrow therapeutic windows — where small absorption changes can matter.
Are Mounjaro Side Effects Different in Women?
The most evidence-backed sex-based difference is hair loss. In the Zepbound weight-loss trials, 7.1% of female participants experienced hair loss compared to just 0.5% of males. This was associated with weight reduction.
Beyond hair loss, there are three female-specific considerations worth calling out:
Oral contraceptive interaction: Covered above — if you’re on the pill, this is not optional reading. The absorption issue is real.
Pregnancy timing: The 2-month pre-conception washout period matters. If you’re of reproductive age and not using reliable contraception, discuss this with your prescriber before starting.
Menstrual changes: This isn’t studied in the tirzepatide trials, and we can’t point to label-backed data here. However, significant weight loss and body composition changes from any cause can affect your cycle. If you experience major menstrual irregularities, bring it up with your doctor to rule out other causes.
Can Mounjaro Cause Cancer, Pancreatitis, or Other Long-Term Problems?
This is the section where we separate verified facts from internet fear.
The Thyroid Black Box Warning — What It Actually Means
Every Mounjaro pen comes with an FDA black box warning — the most serious warning category — about thyroid C-cell tumors.
Here’s exactly what the evidence says: In a two-year study in rats, tirzepatide caused thyroid C-cell tumors (both noncancerous and cancerous) at doses comparable to human use. The higher the dose and the longer the treatment, the higher the tumor incidence.
Here’s what we don’t know: whether this happens in humans. The label states: “It is unknown whether MOUNJARO causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of tirzepatide-induced rodent thyroid C-cell tumors has not been determined.” The FDA has required Eli Lilly to run a registry-based study of at least 15 years to monitor MTC rates in the population now that millions of people are using tirzepatide.
This same black box warning applies across the entire GLP-1 drug class — Ozempic, Wegovy, Saxenda — not just Mounjaro.
Who should NOT take Mounjaro: Anyone with a personal or family history of medullary thyroid carcinoma (MTC), or anyone with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). This is an absolute contraindication.
What to watch for: A lump or swelling in your neck, persistent hoarseness, trouble swallowing, or shortness of breath. Report these to your doctor promptly.
Our honest read: The warning reflects appropriate regulatory caution based on animal data. Whether this risk extends to humans remains unknown. If you don’t have the specific risk factors listed above, the warning does not mean you will get thyroid cancer — but it does mean you and your prescriber should be aware of the symptoms to watch for.
Pancreatitis
Acute pancreatitis (inflammation of the pancreas) has been reported in people taking tirzepatide and other GLP-1 drugs. In clinical trials, it was rare — well under 1%.
Symptoms: Sudden, severe abdominal pain (often radiating to the back) that doesn’t go away, often accompanied by vomiting.
What to do: Stop taking the medication and seek emergency care. Pancreatitis can become life-threatening.
If you have a history of pancreatitis, your prescriber should factor that into the decision to prescribe tirzepatide.
Gallbladder Disease
Gallstones and gallbladder inflammation showed up in roughly 0.6% of Mounjaro trial participants (versus 0% on placebo). In longer-duration weight-loss trials, the rate was approximately 1–2% over two years, with events peaking in months 6–18.
This isn’t specific to tirzepatide — rapid weight loss from any cause increases gallstone risk. The body mobilizes fat quickly, which raises cholesterol concentrations in bile and promotes stone formation.
Watch for: Pain in the upper right abdomen, especially after fatty meals. Fever. Yellowing of the skin or eyes. Clay-colored stools.
Gastroparesis: The Real Story
Let’s be precise here, because this topic generates a lot of fear online.
The Mounjaro label says tirzepatide is not recommended in people with severe gastroparesis. The drug slows gastric emptying by design — that’s a core part of how it works. In the vast majority of users, this slowing is temporary and tolerable.
True gastroparesis (stomach paralysis) is a separate medical condition where the stomach loses the ability to empty itself. Some lawsuits allege GLP-1 medications caused this, but the FDA label does not list gastroparesis as a confirmed side effect of Mounjaro. The label states that Mounjaro “is not recommended in patients with severe gastroparesis” and warns about severe gastrointestinal adverse reactions. Postmarketing reports also include ileus (bowel obstruction), intestinal obstruction, and severe constipation including fecal impaction — these are rare but belong on a page about what to watch for.
If you experience persistent inability to eat, prolonged vomiting, or severe bloating that isn’t improving, contact your prescriber. That’s different from the temporary GI adjustment most people experience.
Kidney Injury
Rare, and almost always secondary to dehydration. The mechanism: severe vomiting or diarrhea → fluid loss → kidneys don’t get enough blood flow → acute injury.
Prevention is straightforward: stay hydrated, and don’t push through persistent vomiting without calling your provider. Monitor for signs of kidney trouble: dark or reduced urine output, swelling in legs or ankles, and unusual fatigue.
Diabetic Retinopathy Complications
Relevant primarily if you have type 2 diabetes with pre-existing retinal disease. Rapid blood sugar normalization has been associated with a temporary worsening of diabetic retinopathy. Mounjaro has not been studied in patients with nonproliferative diabetic retinopathy requiring acute therapy, proliferative diabetic retinopathy, or diabetic macular edema. If you have a history of diabetic retinopathy, your doctor should monitor you for progression.
What’s Still Being Studied
| Topic | Label Warning? | Clinical Trial Signal? | Postmarketing Reports? | Still Under Study? |
|---|---|---|---|---|
| Thyroid C-cell tumors | Yes (black box) | Yes (in rats) | Human relevance unknown | Yes — 15+ year registry |
| Pancreatitis | Yes | Rare events in trials | Yes | Ongoing surveillance |
| Gallbladder disease | Yes | Yes — ~0.6% (Mounjaro) / ~1–2% (Zepbound) | Yes | Ongoing surveillance |
| Gastroparesis | Not recommended in severe gastroparesis | Not formally quantified | Lawsuits pending | Under investigation |
| Ileus / intestinal obstruction | Yes (postmarketing) | Not reported in trials | Yes | Ongoing surveillance |
| Suicidal ideation | Removed from GLP-1 weight-loss labels (Jan 2026) | No signal found | Reports investigated | FDA concluded no association |
On suicidal thoughts: FDA completed its investigation of GLP-1 drugs and suicidal ideation in January 2026. It found no increased risk and requested removal of that warning from affected GLP-1 weight-loss labels. If you see articles from 2024 still flagging this as an active concern, they’re outdated.
How Do Mounjaro Side Effects Compare to Ozempic?
This is the natural next question. Here’s the short version.
Mounjaro (tirzepatide) and Ozempic (semaglutide) are both GLP-1 medications, but Mounjaro also activates GIP receptors — making it the only dual-action drug in its class. The side-effect profiles overlap substantially: both cause GI symptoms (nausea, diarrhea, vomiting, constipation), both carry the same thyroid C-cell tumor black box warning, and both follow the same general pattern of symptoms peaking during dose escalation and improving over time.
An important note about comparing the numbers directly: The FDA labels for both drugs explicitly state that adverse reaction rates from one drug’s trials cannot be directly compared to rates from another drug’s trials, because the trials were conducted under different conditions with different patient populations. We’re not going to put two sets of percentages side by side and pretend that’s an apples-to-apples comparison — because it isn’t.
What we can say: the general side-effect landscape is similar. Both drugs are well-tolerated by most people, and both can cause meaningful GI discomfort during the adjustment period. An important clinical observation that prescribers frequently report: patients who can’t tolerate one drug sometimes do well on the other. The receptor pathways aren’t identical, and your gut may respond differently to tirzepatide versus semaglutide. If you’re having a rough time on one, switching is a legitimate conversation to have with your prescriber — not a failure.
For a deeper comparison of these medications, see our full GLP-1 provider comparison guide.
Do Compounded Tirzepatide Products Change the Side-Effect Picture?
This section matters — especially for people getting tirzepatide from online clinics or compounding pharmacies.
Important distinction: We are talking about compounded tirzepatide, which is pharmacy-prepared from bulk ingredients. This is different from FDA-approved Mounjaro or Zepbound made by Eli Lilly. Compounded drugs are not approved by FDA, and the agency does not review them for safety, effectiveness, or quality before they are marketed.
Why This Affects Side Effects
The FDA has specifically warned about adverse events associated with compounded GLP-1 products. The agency has received reports of adverse events — some requiring hospitalization — related to dosing errors with compounded injectable semaglutide and tirzepatide, as well as reports tied to doses beyond the FDA-approved label schedule.
If you’re experiencing side effects that seem unusually severe — worse than what the trial data above would predict — it’s worth considering whether your source is the issue, not just the medication itself.
Common compounded-product risk factors:
- Incorrect dosing — without standardized pen delivery, measuring errors are more common
- Too-rapid titration — some online clinics escalate faster than clinical guidelines recommend
- Unknown product quality — without FDA-approved manufacturing processes, batch-to-batch consistency isn’t guaranteed
- Products that aren’t what they claim — FDA has warned about fraudulent products marketed as compounded semaglutide or tirzepatide
How to Protect Yourself
If you’re using compounded tirzepatide:
- Verify the compounding pharmacy is licensed and accredited (look for PCAB accreditation or state board licensing)
- Ask your prescriber specifically about titration pacing — slower is safer
- Monitor your symptoms carefully and compare them to the FDA trial rates above
- If your experience is dramatically worse than what the data predicts, your product or dosing may be the problem
We’ve reviewed dozens of online GLP-1 providers and their sourcing practices. For help finding a provider with transparent medication sourcing, see our guide to getting GLP-1 medications safely online.
What If Mounjaro Side Effects Aren’t Worth It?
Let’s be direct: this medication isn’t right for everyone, and side effects that don’t improve are a valid reason to reassess.
Before you quit:
Talk to your prescriber about a slower titration. Many people who struggled at 10 mg do fine when they step back to 7.5 mg for an extra cycle. The goal is to find the dose that balances results with tolerability — not to race to the maximum dose.
Ask about supportive medications. Prescription anti-nausea drugs, proton pump inhibitors for reflux, or stool softeners for constipation can make the adjustment period much more manageable.
Consider whether the source is the issue. If you’re on compounded tirzepatide and having an unusually rough time, switching to an FDA-approved product (or a different provider with better dosing protocols) might resolve the problem.
Some people do better on a different GLP-1 medication. Someone who can’t tolerate tirzepatide may do well on semaglutide (Ozempic/Wegovy), or vice versa. The two drugs work on overlapping but different receptor pathways, and prescribers frequently report that tolerability varies between them.
Some people need more clinical supervision, not less. If you’re managing side effects through an asynchronous texting app and struggling, a provider that offers real-time video visits or in-person care may be the difference between quitting and succeeding.
Don’t guess your next move while feeling awful.
Our free quiz matches you with the GLP-1 path that fits your goals, budget, and side-effect tolerance — whether that’s a different provider, a different medication, or a different approach entirely.
Get My GLP-1 MatchHow We Verified This Page
We’re an editorial team, not a medical practice. We don’t prescribe medications and we don’t give medical advice. What we do is research, compile, verify, and explain — and we’re transparent about exactly where our data comes from.
Primary sources for this guide
- FDA Prescribing Information for Mounjaro (tirzepatide), revised December 2025
- FDA Prescribing Information for Zepbound (tirzepatide), revised February 2026
- FDA Medication Guides for Mounjaro and Zepbound
- SURPASS clinical trial program (type 2 diabetes)
- SURMOUNT clinical trial program (weight management)
- FDA Warning Letters and Safety Communications (including September 2025 letter to Eli Lilly; January 2026 GLP-1 suicidal ideation update)
- FDA safety communication on unapproved compounded GLP-1 products
- NCBI/StatPearls tirzepatide monograph
- MedlinePlus tirzepatide drug information
What we did NOT use as medical-fact sources: Reddit posts, TikTok anecdotes, or unverified patient testimonials. We read those to understand how people describe their experience and what language they use — but every medical claim in this guide traces back to an FDA label, a published clinical trial, or a peer-reviewed source.
Affiliate disclosure: WeightLossProviderGuide.com earns commissions when you sign up with a provider through our links. This financial relationship never influences our reporting of side-effect data, which comes from FDA and clinical trial sources. We would rather lose a commission than lose your trust.
Last updated: March 2026 · Last verified against FDA labels: March 2026
Frequently Asked Questions About Mounjaro Side Effects
Does everyone get side effects on Mounjaro?
No. In clinical trials, a meaningful percentage of people had no notable side effects at all. But GI symptoms are common enough that you should be prepared for the possibility, especially during the first few weeks and after dose increases.
What are Mounjaro side effects in the first week?
At the 2.5 mg starter dose, many people feel little to nothing. Mild appetite reduction is the most common early effect. Some experience mild nausea. Injection-site soreness is possible. Severe symptoms during the first week at the starter dose are uncommon.
How long do Mounjaro side effects last?
The labels confirm that the majority of nausea, vomiting, and diarrhea occurred during dose escalation and decreased over time. Most people find GI side effects improve as their body adjusts to a stable dose.
Can side effects start later, after months on the same dose?
Uncommon for new GI symptoms to appear months into a stable dose. If they do, talk to your prescriber — it may indicate something else going on, like gallbladder issues, dietary changes, or a medication interaction.
Does Mounjaro cause sulfur burps?
Burping (eructation) is listed as a common side effect in the Zepbound label (4–5%). Sulfur-flavored burps specifically are widely reported by patients, likely related to slowed digestion of sulfur-containing foods. Eating more slowly and reducing high-sulfur foods can help.
Does Mounjaro cause hair loss?
About 4–5% of tirzepatide users in weight-loss trials experienced hair loss — 7.1% in women, 0.5% in men. This is almost certainly telogen effluvium from rapid weight loss, not direct follicle damage. It’s usually temporary.
Does Mounjaro cause body aches or muscle pain?
Body aches and muscle pain are not among the commonly listed label side effects for Mounjaro or Zepbound. However, some patients report these symptoms. They may be related to caloric restriction, electrolyte changes, or the body adjusting to rapid composition changes. Mention persistent aches to your prescriber.
Can Mounjaro cause stomach paralysis (gastroparesis)?
Mounjaro slows gastric emptying by design — that’s not the same as gastroparesis (a chronic motility disorder). The FDA label says Mounjaro is not recommended in people with severe gastroparesis. There have been legal claims about GLP-1 drugs and gastroparesis, but the label does not list it as a confirmed side effect.
Can Mounjaro cause cancer?
Mounjaro carries a black box warning based on thyroid C-cell tumors found in rats. No confirmed human cases of tirzepatide-caused thyroid cancer exist as of March 2026. A 15+ year monitoring study is ongoing.
Are side effects worse in women?
Hair loss is significantly more common in women (7.1% vs 0.5% in men per Zepbound trials). Women should also be aware of the oral contraceptive interaction and pregnancy considerations.
Can I drive after taking Mounjaro?
Mounjaro can cause dizziness or lightheadedness, particularly if your blood sugar drops. If you’re on insulin or a sulfonylurea alongside Mounjaro, use caution until you know how the combination affects you.
What should I eat to reduce Mounjaro side effects?
Small, frequent, low-fat, protein-rich meals. Avoid greasy and fried foods. Stop eating when full. Stay hydrated. Bland foods during nausea flare-ups (toast, rice, crackers, broth). Avoid carbonated beverages if burping or bloating is an issue.
Can I skip a dose because of side effects?
Talk to your prescriber before skipping. If you miss a dose and fewer than 4 days (96 hours) have passed, take it as soon as you can. If more than 4 days, skip it and take the next one on schedule. Never take two doses within 3 days.
Are Mounjaro and Zepbound side effects the same?
They use the same medication (tirzepatide) from the same manufacturer. But side-effect rates differ between the diabetes and weight-loss trial populations — weight-loss trials report higher rates for several GI symptoms, and the Zepbound label lists additional common effects (fatigue, burping, hair loss, reflux) that the Mounjaro label does not.
What if I'm using compounded tirzepatide — are side effects the same?
We can’t tell you that compounded products will produce the same experience as FDA-approved Mounjaro or Zepbound. Compounded medications aren’t FDA-approved and don’t go through the same manufacturing process. If your side effects seem unusually severe, the product or dosing may be a factor worth investigating with your prescriber.
When should I call a doctor instead of waiting it out?
Mild, improving GI symptoms are usually fine to manage at home. Persistent symptoms that aren’t getting better deserve a prescriber call. Severe abdominal pain, inability to keep fluids down, allergic reaction signs, or any “something is really wrong” feeling should get urgent or emergency evaluation. Trust your instincts.
Still Not Sure Which GLP-1 Program Is Right for You?
You just read one of the most thorough Mounjaro side-effect resources on the internet. If you made it this far, you’re serious about making an informed decision — and that’s exactly the kind of person we built our matching tool for.
Our free 60-second GLP-1 matching quiz considers your medical situation, insurance status, budget, state, and yes — your side-effect tolerance — to recommend the providers and medication paths most likely to work for you.
No spam. No commitment. Just clarity.
Medical Disclaimer: This content is for informational purposes only. It is not medical advice and does not replace consultation with a qualified healthcare provider. Always consult your doctor or prescriber before starting, stopping, or changing any medication.
Affiliate Disclosure: WeightLossProviderGuide.com is an affiliate site. We earn commissions when readers sign up with providers through our links. Our side-effect reporting is sourced from FDA labels and published clinical trials and is never influenced by affiliate relationships. See our advertising disclosure for details.
Last Updated: March 2026 · Last Verified Against FDA Labels: March 2026