Tirzepatide Pills vs Shots: What's Real & What Works (2026)
Disclosure: Some links on this site are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you. Thank you for supporting our site. For informational purposes only—not medical advice.

On this page
You've done the research. You know tirzepatide works — the clinical results are hard to ignore. But now you're stuck on a question that shouldn't be this complicated: do I take the shot, or is there a pill?
Maybe you hate needles. Maybe you've heard tirzepatide comes in tablet form and you want to know if that's even real. Maybe you're just trying to figure out which option will actually get you results without draining your bank account or making you miserable.
You're in the right place. Whether you're comparing oral tirzepatide vs injectable or just trying to figure out if a tirzepatide pill even exists, this is the comparison nobody else is giving you straight.
Here's the short version: When people compare tirzepatide pills vs shots, the active ingredient is the same — the dual-action GLP-1/GIP compound that outperformed semaglutide (Wegovy) in a head-to-head obesity trial and has produced among the largest average weight-loss results reported for prescription anti-obesity medications. Injectable tirzepatide (Mounjaro, Zepbound) is the only FDA-approved form, with clinical evidence showing about 15% to 21% average weight loss at 72 weeks (mean 20.9% at the highest dose per FDA labeling). When people say "tirzepatide pills," they typically mean compounded, non-FDA-approved sublingual tablets — preparations that use the same active ingredient but haven't been evaluated in large clinical trials the way injections have. The FDA has issued warnings about unapproved GLP-1 drugs, including compounded versions, citing dosing errors and adverse-event reports. That said, many patients access compounded tablets through licensed telehealth providers and report meaningful results. The right choice depends on your priorities — and doing your due diligence on any provider.
Below, we break it all down — efficacy, cost, side effects, dosing, convenience, and exactly who each form is best for. With real trial data, real patient experiences, and a decision framework so you can stop opening new tabs.
Tirzepatide Pills vs Shots: Quick Comparison
Before we get into the details, here's the side-by-side snapshot. This table covers the essentials — we'll go deeper on each point throughout the guide.
| Factor | Tirzepatide Shots (Injectable) | Tirzepatide Pills (Sublingual Tablets) |
|---|---|---|
| FDA Status | Yes — FDA-approved (Mounjaro, Zepbound) | No — Not FDA-approved (compounded) |
| Dosing | Once per week | Once per day |
| Clinical Evidence | Extensive — SURMOUNT 1-5, 5,000+ participants | Limited — same active ingredient, less published data |
| Average Weight Loss | 15-21% of body weight (72 weeks, FDA label) | Same drug; individual results vary |
| Bioavailability | ~80% (high, consistent absorption) | Lower and more variable (sublingual absorption) |
| How You Take It | Thin-needle injection (abdomen, thigh, or arm) | Typically sublingual (dissolve under tongue); follow your prescriber's timing instructions |
| Monthly Cost | $199-$299 (compounded) or $1,000+ (brand) | $249-$299 (compounded) |
| Needles Required | Yes | No |
| Refrigeration | Yes (before first use) | No — room temperature |
| Travel-Friendly | Requires planning (cold storage, needles) | Very easy — pack like any tablet |
| Best For | Maximum proven results, once-weekly simplicity | Needle-phobic, travelers, daily-routine preference |
Sources: FDA prescribing information for Mounjaro/Zepbound; published provider pricing February 2026; SURMOUNT clinical trial program.
That's the high-level picture. Now let's get into what actually matters.
What Is Tirzepatide — and Why Does Everyone Want It?
If you're already familiar with tirzepatide, feel free to skip ahead. But here's the quick version for anyone who needs it.
Tirzepatide is the only weight-loss medication that targets two hormones simultaneously — GLP-1 and GIP. Every other GLP-1 medication on the market (Ozempic, Wegovy, semaglutide) only hits one of those targets. Tirzepatide hits both. That dual mechanism is why the results have been so dramatically different from everything that came before it.
The FDA approved it as Mounjaro for type 2 diabetes and Zepbound for weight loss — both in injectable form.
Zepbound is FDA-approved for chronic weight management; Mounjaro is FDA-approved for type 2 diabetes (though weight loss is commonly observed).
And the results? They speak for themselves.
In the landmark SURMOUNT-5 trial published in the New England Journal of Medicine in May 2025, tirzepatide went head-to-head against semaglutide (Wegovy) — the reigning champion of weight-loss medications. Tirzepatide won. Decisively. Participants on tirzepatide lost an average of 20.2% of their body weight, compared to 13.7% for semaglutide over 72 weeks. That's roughly 50% more weight loss from the dual-action mechanism. (Aronne et al., NEJM 2025; DOI: 10.1056/NEJMoa2416394)
On Drugs.com, tirzepatide carries an 8.5 out of 10 average rating from 2,600+ patient reviews, with 79% reporting positive experiences. Those aren't pharmaceutical marketing numbers. Those are real people reporting real outcomes.
So the medication itself isn't the question. Tirzepatide works — and it works better than anything else available. The question is how you take it.
By the numbers: In SURMOUNT-1, participants on the highest dose of tirzepatide lost a mean of 20.9% of their body weight over 72 weeks (FDA label). For a 215-pound person, that's roughly 48 pounds. (Jastreboff et al., NEJM 2022; FDA Zepbound prescribing information)
Does Tirzepatide Come in Pill Form? The 2026 Reality Check
Let's clear this up immediately, because there's a lot of confusion out there — and some of it is deliberate.
The hard truth: There is no FDA-approved tirzepatide pill. As of February 2026, the only FDA-approved tirzepatide products are the injectables — Mounjaro and Zepbound.
But here's what IS available: Compounded tirzepatide sublingual tablets — rapid-dissolve tablets you place under your tongue — prescribed by licensed medical providers and prepared by FDA-registered or state-licensed compounding pharmacies.
Some compounded offerings are prescribed and dispensed through licensed channels — real prescriptions, filled by real pharmacies, with the same active ingredient as the injectable. However, the FDA has warned that some "compounded" GLP-1 products sold online are unapproved or even fraudulent, with reports of dosing errors and adverse events. Treat any seller that doesn't require a prescription as a hard "no." The difference between legitimate compounded tablets and the injectable is that the finished tablet product hasn't gone through the FDA's formal clinical trial and approval process the way Mounjaro and Zepbound did.
This is an important distinction, and you deserve to understand it clearly.

Regulatory Status in 2026: What You Need to Know
- Not FDA-approved: No tirzepatide tablet (compounded or otherwise) has FDA approval. Only injectable Mounjaro and Zepbound are FDA-approved.
- FDA warnings exist: The FDA has issued warnings about unapproved GLP-1 drugs, including compounded tirzepatide, citing dosing errors, adverse-event reports, and cases of fraudulent products. (FDA.gov)
- Shortage status has shifted: The FDA declared the tirzepatide injection shortage resolved, which affects the legal landscape for routine compounding of copies.
- Due diligence is essential. If you choose a compounded option, verify the prescriber, pharmacy, and quality standards using our legitimacy checklist below.
What People Mean When They Say "Tirzepatide Pills"
When you see the phrase "tirzepatide pills" online, it usually refers to one of three things — and they are not the same:
| What It Is | FDA-Approved? | Evidence Level | What You Should Know |
|---|---|---|---|
| Injectable tirzepatide (Mounjaro, Zepbound) | Yes | Strong — large-scale clinical trials (SURMOUNT 1-5) | The gold standard. Weekly injection. |
| Compounded sublingual tirzepatide tablets | No — prescribed by licensed providers, made by registered pharmacies | Moderate — same active ingredient, limited published data on sublingual form | Real medication, real prescriptions. Not FDA-approved as a finished product. |
| FDA-approved oral GLP-1 pill (Wegovy tablets / oral semaglutide) | Yes (since Dec 2025) | Strong — clinical trial data | This is semaglutide, NOT tirzepatide. Single-action GLP-1 only. |
Sources: FDA prescribing information; FDA concerns about unapproved GLP-1 drugs (FDA.gov)
That third category trips people up the most. The oral Wegovy pill — approved by the FDA in December 2025 — is semaglutide, not tirzepatide. It's a GLP-1-only medication. Still effective for weight loss (clinical trials showed about 13.6% average body weight loss), but it doesn't have tirzepatide's dual GLP-1/GIP mechanism that produces those 20%+ results.
Bottom line: If you want tirzepatide specifically — the dual-action powerhouse — your options right now are the FDA-approved injection or compounded sublingual tablets. There is no FDA-approved tirzepatide pill.
What About the Future?
Eli Lilly submitted orforglipron — an oral, once-daily pill — for FDA approval in late 2025; the timeline depends on FDA review. But orforglipron is a GLP-1 agonist only, not a dual GLP-1/GIP agonist like tirzepatide. It's a different medication.
There is currently no oral tirzepatide (dual agonist) in active FDA clinical trials. So if you're waiting for an FDA-approved tirzepatide pill, you may be waiting a while.
How Do Tirzepatide Pills (Sublingual Tablets) Actually Work?
Most pages either dismiss tirzepatide pills entirely or don't bother explaining the mechanics. That doesn't help you make a decision. So let's actually walk through it.
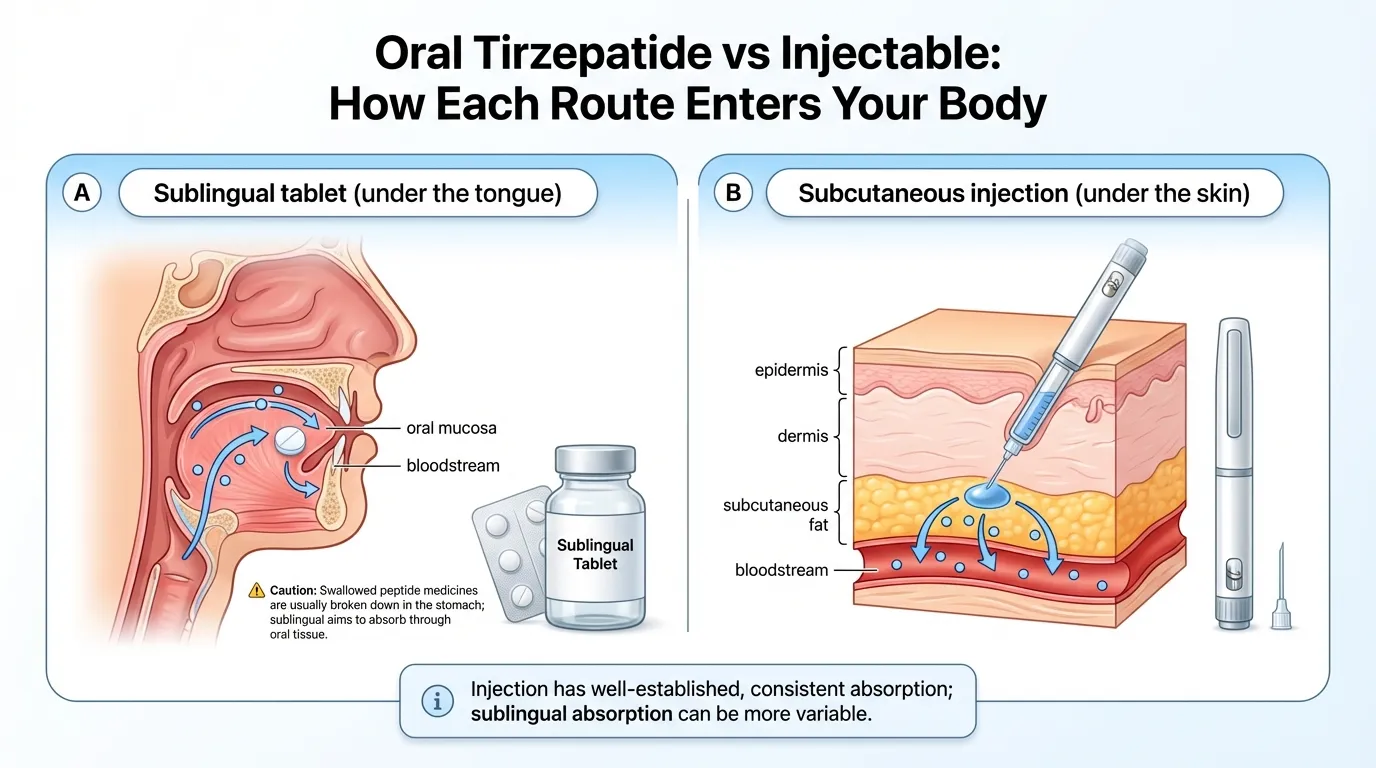
Compounded tirzepatide tablets are sublingual rapid-dissolve tablets (RDTs). "Sublingual" means under the tongue. You place the tablet beneath your tongue, let it fully dissolve, and the medication absorbs directly through the mucosal lining of your mouth — bypassing the digestive system entirely.
This matters because peptide medications like tirzepatide would be destroyed by stomach acid if you swallowed them like a regular pill. That's why there's no standard oral tirzepatide tablet you can just pop and swallow. The sublingual route gets around this problem by delivering the drug straight into your bloodstream through the tissue under your tongue.
This isn't experimental science. Sublingual delivery has been used for decades — nitroglycerin tablets for heart patients work the exact same way.
The active ingredient is the same tirzepatide that produced those remarkable clinical trial results. Same molecule. Same dual GLP-1/GIP mechanism. Different delivery method.
How to Take Sublingual Tirzepatide (General Guidance)
Compounded sublingual tablets are typically taken on an empty stomach, first thing in the morning. You place the tablet under your tongue, let it dissolve completely without chewing or swallowing, and wait before eating or drinking. The exact protocol — including timing, wait period, and any other instructions — will be specified by your prescribing clinician and printed on your prescription label.
Always follow your prescriber's specific instructions. Do not self-convert injection doses to sublingual doses or adjust your dosing without medical guidance.
Dosing
Compounded dosing protocols vary by prescriber and pharmacy. There is no FDA-approved dosing schedule for sublingual tirzepatide tablets. Your provider will determine an appropriate starting dose and titration schedule based on your individual needs and response.
Note: Sublingual daily doses are typically higher than weekly injection doses. That's because sublingual absorption is less efficient than injection — providers calibrate accordingly. This is not something you need to calculate yourself.
How Do Tirzepatide Shots (Injections) Work?
Fair is fair. Let's give injections the same thorough treatment.
Injectable tirzepatide is given subcutaneously — meaning just under the skin — once per week. It's available as pre-filled auto-injector pens (Mounjaro, Zepbound) or vials with syringes (compounded versions).
The injection delivers tirzepatide directly into the subcutaneous tissue, where it absorbs into the bloodstream with roughly 80% bioavailability. That's high. It means the vast majority of the medication reaches your system and starts working.
Because it's a once-weekly injection, you get relatively consistent blood levels throughout the week — no daily peaks and valleys.
What the Injection Is Actually Like
Let's be real: if you're reading a page called "tirzepatide pills vs shots," there's a decent chance the needle is what's holding you back.
The needle on a Mounjaro or Zepbound pen is very small and thin — much smaller than what most people picture when they think "injection." Most patients describe the sensation as a brief pinch or pressure, not pain. It takes about 10 seconds. You inject into your abdomen, thigh, or upper arm, and you rotate the site each week.
Is it fun? No. But it's much less intimidating than most people imagine before they start.
Pre-filled pens are the easiest — you click a button and the device does the work. Compounded vials require drawing the dose into a syringe, which takes a little practice but becomes routine quickly.
FDA-Approved Injectable Dosage Schedule
| Weeks | Weekly Dose | Purpose |
|---|---|---|
| Weeks 1-4 | 2.5 mg | Starting dose |
| Weeks 5-8 | 5 mg | First increase |
| Weeks 9-12 | 7.5 mg | Second increase |
| Weeks 13-16 | 10 mg | Third increase |
| Weeks 17+ | 10 mg or 15 mg | Maintenance (max tolerated dose) |
Source: FDA prescribing information for Mounjaro/Zepbound
The gradual titration is critical. Starting low and increasing slowly is what keeps side effects manageable. This schedule applies to both brand-name and compounded injectables.
Storage Note
Brand-name pens need refrigeration before first use (can be kept at room temperature for up to 21 days after). Compounded injectables also typically require refrigeration. This means planning ahead for travel and making sure your medication stays within temperature range.
Tirzepatide Pills vs Shots: The Full Side-by-Side Comparison
This is what you came for. Every meaningful factor, compared honestly.
| Category | Pills (Sublingual Tablets) | Shots (Injectable) | Edge |
|---|---|---|---|
| Proven Efficacy | Same active ingredient; limited published data on sublingual delivery specifically | Extensive clinical trial data: 15-21% weight loss (SURMOUNT 1-5, FDA label) | Shots |
| Bioavailability | Variable — depends on technique, oral mucosal absorption | ~80% — consistent subcutaneous absorption | Shots |
| Needle-Free | Yes — No needles whatsoever | No — Weekly self-injection required | Pills |
| Dosing Frequency | Daily (every morning, empty stomach) | Weekly (any day, any time) | Shots |
| Dosing Flexibility | Must take on empty stomach, wait 30 minutes | Take any time, with or without food | Shots |
| Ease of Starting | No needle anxiety, low psychological barrier | Requires getting comfortable with self-injection | Pills |
| Side Effect Onset | May be gentler — smaller daily doses vs. weekly bolus | Same side effects, may be more noticeable at start | Slight edge: Pills |
| Storage | Room temperature — no refrigeration | Typically requires refrigeration | Pills |
| Travel Convenience | Very easy — tablets pack like any supplement | Needs cold storage, needles through TSA | Pills |
| Monthly Cost (Compounded) | ~$249-$299/month | ~$199-$299/month | Similar |
| Insurance Coverage | Not covered (compounded) | May be covered if brand-name (Mounjaro/Zepbound) | Shots (if insured) |
| FDA Approval | Not FDA-approved (compounded product) | FDA-approved (Mounjaro, Zepbound) | Shots |
| Adherence | Daily habit — easier to forget | Weekly — set it and forget for 7 days | Shots |
| Privacy/Discretion | Very discreet — looks like any daily supplement | Requires a private moment for injection | Pills |
Let's be straightforward about what this table tells us.
Injectable tirzepatide wins on paper. It has the strongest evidence, the highest bioavailability, once-weekly convenience, and FDA approval behind it. If you're looking at pure clinical confidence, shots are the clear frontrunner.
But "best on paper" doesn't always mean "best for you." Sublingual tablets win on accessibility. No needles. No refrigeration. No TSA headaches. No injection-site reactions. For people who would otherwise never start tirzepatide because of needle fear, tablets remove the single biggest barrier to entry.
And here's the thing most comparison pages won't tell you: the best medication is the one you'll actually take consistently. A tablet you use every day beats a shot you keep putting off because you can't bring yourself to do the injection. Effectiveness follows adherence.
What Does the Clinical Research Actually Show?
If you're making a decision about a medication, you deserve to see the data — not just marketing claims. Here's what the largest and most rigorous obesity trials in history found.

SURMOUNT-1 (2022) — The Landmark Trial
This was the trial that changed everything. Published in the New England Journal of Medicine, it enrolled 2,539 adults with obesity (without diabetes) and followed them for 72 weeks.
The results by dose (FDA prescribing information):
- 5 mg dose: 15.0% average body weight loss
- 10 mg dose: 19.5% average body weight loss
- 15 mg dose: 20.9% average body weight loss
- Placebo: 3.1%
For context: a 20.9% average reduction means a 215-pound person losing roughly 45 pounds. And this was an average — many participants lost significantly more.
A body composition substudy confirmed that approximately 75% of the weight lost was fat mass and about 25% was lean mass — which is a favorable ratio compared to most weight-loss interventions.
(Jastreboff et al., NEJM 2022; body composition data: Diabetes Obes Metab 2025)
SURMOUNT-4 (2023) — What Happens If You Stay On It?
The follow-up studied what happened when people continued tirzepatide beyond the initial trial period. Those who stayed on the medication achieved 25.3% total body weight loss at 88 weeks. Those switched to placebo regained weight — about +14% body-weight change from week 36 to 88 — while those who continued tirzepatide kept losing.
The takeaway is clear: tirzepatide is a long-term commitment, not a quick fix. And it rewards consistency.
(Aronne et al., JAMA 2024; Horn et al., JAMA Intern Med Nov 2025)
SURMOUNT-5 (2025) — Tirzepatide vs. Semaglutide, Head-to-Head
This was the trial the industry had been waiting for. The first direct comparison between tirzepatide and semaglutide (Wegovy) for weight loss. 751 participants. 72 weeks.
Tirzepatide: -20.2% body weight Semaglutide: -13.7% body weight
Tirzepatide was statistically superior across every endpoint measured. The dual GLP-1/GIP mechanism wasn't just theoretically better — it proved it in a controlled head-to-head.
(Aronne et al., NEJM May 2025; DOI: 10.1056/NEJMoa2416394)
Meta-Analysis (2025)
A pooled analysis of 6 randomized controlled trials confirmed what the individual studies showed: tirzepatide produced a mean -16.32% body weight change compared to placebo, with significant reductions in BMI (-5.89 kg/m2) and waist circumference (-12.31 cm).
(Tirzepatide efficacy and safety meta-analysis, PMC 2025)
What About Sublingual Tirzepatide Tablet Evidence?
Here's where we have to be honest with you. And we'd rather be honest than lose your trust.
No large-scale published clinical trials have directly studied compounded sublingual tirzepatide tablets specifically. The SURMOUNT trials were all conducted with injectable tirzepatide. That's a real limitation, and you should know it.
Now — here's why that's not the end of the story.
The active ingredient in sublingual tablets is the same tirzepatide molecule that produced those trial results. Same drug. Same dual GLP-1/GIP mechanism. What's different is the delivery method, not the medication itself.
Sublingual drug delivery — absorption through the tissue under the tongue — is proven science that's been used successfully for decades. Nitroglycerin for heart patients. Certain allergy medications. Specific pain relievers. The mucous membranes under your tongue are highly vascular, meaning drugs can absorb efficiently into your bloodstream.
And we have a real-world precedent: oral semaglutide (the Wegovy pill, FDA-approved December 2025) proved that GLP-1 medications can absolutely work in oral form. The oral semaglutide trial showed about 13.6% average weight loss — meaningful, clinically significant results from a pill.
Finally, thousands of patients are currently using compounded sublingual tirzepatide tablets through legitimate telehealth providers and reporting significant results. These aren't theoretical outcomes — they're happening right now.
Is the evidence as strong as injectable tirzepatide? No. The injectable has the gold-standard clinical trials behind it. Does that mean tablets don't work? The science, the real-world patient data, and the proven sublingual delivery mechanism all say otherwise.
For many people, sublingual tablets are the path that actually gets them started — and a medication you take is infinitely more effective than one you don't.
Real Patient Experiences With Tirzepatide
Clinical trials give you averages. Real people give you context. Here are publicly attributed patient experiences from independent review platforms.
"I'm on the 5mg and have lost 56 lbs... my hip, leg, and foot pain is 95% better. I feel amazing." — User review on Drugs.com (tirzepatide: 8.5/10 average rating, 2,600+ reviews as of early 2026)
"I believe tirzepatide is the most amazing weight loss medication ever made. I had no side effects other than mild constipation." — User review on Drugs.com, 63-year-old female patient
"Just got approved for the tirzepatide pills... made me feel confident in my choice. Excited to start this journey." — Trustpilot reviewer, SkinnyRx (4.8/5 stars, 4,100+ reviews as of early 2026)
These are real people who made the same decision you're weighing right now. Their starting point was the same as yours — uncertainty, hope, and a desire for something that actually works.
Results vary, of course. No medication works identically for everyone. But the pattern across thousands of reviews is remarkably consistent: significant weight loss, reduced appetite, improved health markers, and a quality of life that many patients describe as transformative.
Check if You Qualify Through SkinnyRxTirzepatide Side Effects: Pills vs. Shots
Both forms use the same medication, so they share the same core side effect profile. The most common side effects are gastrointestinal — which makes sense, because tirzepatide works partly by slowing gastric emptying and changing how your body processes food signals.
| Side Effect | Frequency (FDA Label) | Pills vs. Shots | How to Manage It |
|---|---|---|---|
| Nausea | Up to 29% (dose-dependent) | Similar; may be spread more evenly with daily pills | Eat small meals, avoid greasy foods, ginger tea |
| Diarrhea | Up to 23% | Similar for both forms | Stay hydrated, bland foods during adjustment |
| Constipation | Up to 17% | Similar | Increase fiber, water, and daily movement |
| Decreased appetite | 20%+ | This IS the mechanism working — not really a "side effect" | Prioritize protein (60-80g/day minimum) |
| Vomiting | Up to 13% | Similar for both forms | Report to your provider if persistent |
| Injection-site reactions | Up to 8% | Shots only — minor redness, bruising, itching | Rotate injection sites weekly |
| Fatigue | Up to 7% | Similar for both | Usually resolves within 1-2 weeks |
Source: FDA prescribing information for Mounjaro/Zepbound; SURMOUNT clinical trial data
Here's what most people discover: the side effects are front-loaded. They're most noticeable during the first few weeks as your body adjusts, especially when you increase your dose. By weeks 3-4 at any given dose level, most patients report that side effects have faded significantly or disappeared entirely.
The gradual dose titration schedule exists specifically to manage this. Starting low and stepping up slowly gives your body time to adapt. Skipping ahead or increasing too fast is the most common reason people have a rough time — which is why working with a licensed provider who manages your titration properly matters so much.
One potential advantage of daily sublingual tablets: because you're taking a smaller dose each day rather than one larger weekly dose, some patients experience a gentler side effect curve. Think of it as a steady stream vs. a weekly wave. This isn't confirmed in published studies (remember — no large-scale trials on sublingual tablets specifically), but it's a reasonable pharmacological hypothesis that many providers and patients have observed anecdotally.
Important Safety Information
Tirzepatide carries a boxed warning about thyroid C-cell tumors, which were observed in animal studies. You should not use tirzepatide if you have:
- A personal or family history of medullary thyroid carcinoma (MTC)
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- A known allergy to tirzepatide
Discuss your full medical history with your provider, especially if you have a history of pancreatitis, gallbladder problems, or kidney disease. For a complete list of contraindications, see our guide.
(Source: FDA prescribing information, Zepbound/Mounjaro)
How Much Do Tirzepatide Pills vs Shots Cost in 2026?
Cost is often the deciding factor. Here are the real numbers as of February 2026 — no hedging, no "contact us for pricing" games.
| Option | Monthly Cost | What's Included | Key Notes |
|---|---|---|---|
| Brand Mounjaro (injection) | ~$1,070–$1,080/mo list price | FDA-approved, pharmacy pickup | Insurance may cover; Medicare price set at $245/mo with ~$50/mo copay (Implementation details and eligibility can vary by plan and may change) |
| Brand Zepbound (injection) | ~$1,086/mo list price | FDA-approved for weight loss | Insurance/savings card programs may apply; same Medicare pricing |
| Compounded injectable tirzepatide | $199-$299 | Telehealth consult + medication + shipping | Cash-pay, no insurance required |
| Compounded sublingual tablets | $249-$299 | Telehealth consult + medication + shipping | Cash-pay, needle-free |
| Eli Lilly Direct (LillyDirect) | $299–$449 | Brand-name single-dose vials, self-pay via LillyDirect | Requires provider consultation |
Pricing as of February 2026. Costs vary by provider, dosage tier, and location.
A few things worth noting here.
Brand-name is best if your insurance covers it. If you have commercial insurance that includes Mounjaro or Zepbound, you could be paying significantly less — sometimes as low as $25/month with manufacturer savings cards. Under a federal pricing initiative announced in November 2025, the Medicare price for Zepbound was set at $245/month, with a stated Medicare beneficiary copay of about $50/month (Implementation details and eligibility can vary by plan and may change).
Compounded options are how most people access tirzepatide affordably. Without insurance coverage (which many people don't have for weight-loss medications), brand-name tirzepatide at ~$1,086/month list price is simply out of reach. Compounded versions — both injectable and tablets — bring the monthly cost down to $199-$299, which includes the provider consultation, the medication itself, and typically free shipping.
The price difference between compounded pills and shots is minimal. Tablets typically run $249-$299/month while compounded injectables are $199-$299/month. This means cost probably shouldn't be the deciding factor between forms — it comes down to your preference on the other factors we've covered.
FSA and HSA cards are accepted by most telehealth providers, which means you may be using pre-tax dollars.
For example, telehealth providers like SkinnyRx offer compounded tirzepatide tablets starting at $299/month — that includes the licensed provider consultation, the medication, and free overnight shipping from U.S. pharmacies. No hidden fees, no surprise rebilling. (We'll cover what to look for in a provider further down.)
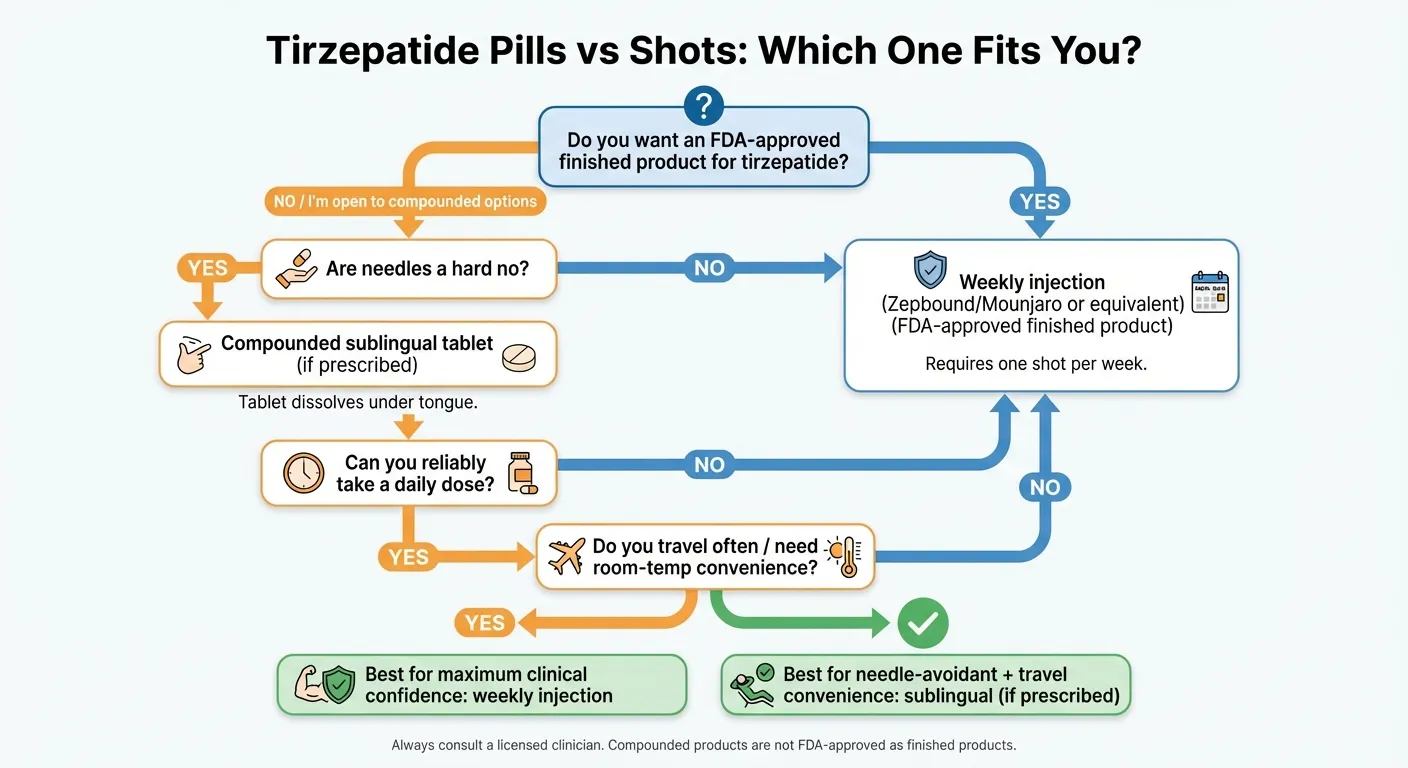
Who Should Choose Tirzepatide Pills Over Shots — And Vice Versa?
You've seen the data. You've seen the costs. Now let's make this personal.
We've put together a decision framework based on the factors that actually matter for long-term success. Not "which is technically superior on a spreadsheet" — but which one you'll stick with.
Choose Tirzepatide PILLS (Sublingual Tablets) If You:
- Have needle fear or injection anxiety. This is more common than people admit, and it's a legitimate reason. There's no point choosing the "better" option on paper if the needle prevents you from starting. If injections are a hard no, pills remove that barrier completely.
- Travel frequently. Tablets don't need refrigeration, don't require syringes or needles, and don't raise eyebrows at airport security. You toss them in your bag and go.
- Value privacy and discretion. A daily sublingual tablet looks and feels like taking any other supplement. No injection supplies, no autoinjector pens, no explaining what's in your sharps container.
- Are comfortable with a daily routine. If you already take a morning vitamin or medication, adding a sublingual tablet to that routine is seamless. First thing in the morning before breakfast — set it and forget it.
- Want a lower barrier to get started. Psychologically and practically, starting with a pill is easier for most people. No learning how to self-inject, no watching injection tutorials, no anxiety before your first dose.
Choose Tirzepatide SHOTS (Injectable) If You:
- Want the strongest clinical evidence backing your treatment. The SURMOUNT trials enrolled over 5,000 participants in the obesity program and are among the most rigorous obesity studies ever published. That level of evidence provides a confidence that compounded tablets can't match yet.
- Prefer once-weekly dosing. One injection per week — any time of day, with or without food — and you don't think about it again for seven days. For people who struggle with daily medication adherence, this is a genuine advantage.
- Don't mind the needle (or can get past it). Most patients are surprised by how painless the actual injection is. The anticipation is almost always worse than the reality. The pen needle is very thin — many patients describe it as a quick pinch.
- Want the option of FDA-approved brand-name products. If having the FDA stamp matters to you — or if you might qualify for insurance coverage — injectable is the way to go.
- Value consistent bioavailability. With ~80% absorption directly into subcutaneous tissue, injectable tirzepatide delivers the most reliable drug levels.
The Honest Truth
There's a line we keep coming back to because it's the most important thing on this page:
The best form of tirzepatide is the one you'll actually use consistently. A pill you take every day beats a shot you never start because of needle fear. And a weekly shot beats a pill you keep forgetting on an empty stomach. Effectiveness follows adherence.
Don't let perfect be the enemy of good. Both forms deliver the same powerful medication. Pick the one that fits your life, start, and give yourself the chance to see what tirzepatide can do.
"Is This Tirzepatide Pill Legit?" — The Safety Checklist
This is the section we wish existed when we started researching this topic. The FDA has raised legitimate concerns about unapproved GLP-1 products being sold online, and you should take that seriously. Not all providers are created equal.
Before you pay for any compounded tirzepatide — tablets or injectable — run through this checklist. If a provider checks all these boxes, you're in solid territory. If they don't, keep looking.
The 10-Point Legitimacy Checklist
1. Requires a prescription. Any provider selling tirzepatide without a prescription from a licensed clinician is a red flag. Period.
2. Discloses the pharmacy name and location. You should be able to verify where your medication is being compounded. Legitimate providers are transparent about this.
3. Pharmacy is verifiable. The compounding pharmacy should be either a 503B outsourcing facility (registered with the FDA) or licensed by their state board of pharmacy. You can verify this.
4. Includes a licensed medical provider. A real clinician reviews your medical history, determines if tirzepatide is appropriate for you, writes the prescription, and provides ongoing follow-up. Not just an algorithm.
5. Clear labeling on the medication. Active ingredient, dosage, lot number, expiration date, and pharmacy contact information should all be present on your medication when it arrives.
6. No "FDA-approved tablet" claims. Any provider claiming their tirzepatide tablets are FDA-approved is being dishonest. Only the injectable form has FDA approval. Transparency here is a trust signal.
7. No unrealistic weight-loss promises. "Lose 50 lbs in 2 weeks" is a scam. Legitimate providers set realistic expectations based on clinical evidence.
8. Transparent pricing with no surprise charges. You should know exactly what you're paying before you commit. Monthly cost, what's included, cancellation policy — all clear upfront.
9. Provides adverse-event guidance. A real provider tells you what to do if you experience side effects, when to contact them, and what constitutes a medical emergency.
10. Offers ongoing clinical follow-up. Weight-loss medication isn't set-and-forget. Your provider should be checking in regularly to monitor your progress, adjust dosing, and manage any issues.
Red Flag Phrases to Watch For
If you see any of these on a provider's website, proceed with extreme caution:
- "No prescription needed"
- "FDA-approved tirzepatide tablets" (these don't exist)
- "Identical to Zepbound/Mounjaro" (compounded products are not identical to brand-name)
- "Guaranteed weight loss"
- "Research-grade peptides"
- Any site selling tirzepatide without requiring a medical assessment
(Source: FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss — FDA.gov)
The legitimacy landscape can feel overwhelming, but it doesn't have to be. Reputable telehealth providers exist, and the checklist above makes it straightforward to tell the difference.
SkinnyRx checks all 10 boxes. Licensed providers, FDA-registered U.S. pharmacies, transparent pricing, clear labeling, and ongoing clinical support.
See if You Qualify Through SkinnyRxTirzepatide vs. Semaglutide: How Do They Compare?
If you're comparing forms of tirzepatide, you've probably also wondered how tirzepatide stacks up against semaglutide (Ozempic, Wegovy). Quick answer: tirzepatide wins on weight loss. Here's the data.
| Factor | Tirzepatide | Semaglutide |
|---|---|---|
| Mechanism | Dual GLP-1 + GIP agonist | GLP-1 agonist only |
| Avg. Weight Loss (Head-to-Head) | 20.2% at 72 weeks | 13.7% at 72 weeks |
| FDA-Approved Forms | Injectable only (Mounjaro, Zepbound) | Injectable (Ozempic, Wegovy) + Oral pill (Rybelsus, Wegovy tablets) |
| Brand Names | Mounjaro (diabetes), Zepbound (weight loss) | Ozempic (diabetes), Wegovy (weight loss), Rybelsus (oral) |
| Compounded Available? | Yes — injectable + sublingual tablets | Yes — injectable + sublingual + oral tablets |
| Head-to-Head Winner | Yes — Superior weight loss in SURMOUNT-5 | — |
Source: SURMOUNT-5 trial, Aronne et al., NEJM May 2025
The SURMOUNT-5 head-to-head trial settled the debate: tirzepatide's dual mechanism produces roughly 50% more weight loss than semaglutide in a direct comparison. Both are effective. Both change lives. But if maximizing weight loss is the priority, tirzepatide has the clinical edge.
Semaglutide's advantage? It has an FDA-approved oral pill (Wegovy tablets, approved December 2025). If you specifically want a pill form that's been through the full FDA approval process, oral semaglutide is currently the only option. But you'd be trading some weight-loss efficacy for that FDA stamp.
For a deeper dive, see our full semaglutide vs tirzepatide comparison.
Oral Tirzepatide vs Injectable: Bioavailability and What It Means
This is the technical factor that comes up most often, so let's address it directly.
Bioavailability is the percentage of a medication that actually reaches your bloodstream and becomes active. Injectable tirzepatide has roughly 80% bioavailability — very high, because the drug goes directly into subcutaneous tissue.
Sublingual absorption is inherently more variable. Not all of the medication makes it through the mucosal lining, and factors like technique (how long you hold it under your tongue), saliva production, and whether you accidentally swallow some of the tablet all affect how much gets absorbed.
This is why sublingual tablet doses are higher than injection doses — to compensate for the lower absorption rate. A daily 15mg sublingual tablet and a weekly 10mg injection aren't equivalent because of dose size; they're calibrated by providers to achieve comparable therapeutic effects through different delivery pathways.
Why this matters but isn't a dealbreaker: Lower bioavailability doesn't mean "doesn't work." It means providers adjust the dosing to compensate. It's the same principle behind oral semaglutide — the FDA-approved pill version uses a 25mg daily dose to achieve effects comparable to the 2.4mg weekly injection. The mg numbers look wildly different, but the clinical outcomes are comparable because the dosing accounts for the absorption difference.
Your provider manages this calculation. You just take your medication as prescribed.
Daily Pill vs. Weekly Shot: What the Routine Actually Looks Like
Comparison tables tell you the facts. But they don't tell you what it's actually like to live with each option day-to-day. Here's a practical look at both routines — because the one you can stick with long-term is the one that'll get you results.
The Weekly Injection Routine
Pick a day. Any day. That's your injection day.
Most people choose Sunday evening or Monday morning — start the week fresh. You pull the pen out of the fridge (or wherever you've been storing it), pick your injection site (rotate between abdomen, thigh, and upper arm each week), clean the area, and inject. Ten seconds. Done. You don't think about it again for seven days.
No timing restrictions around food. No waiting period. If you're busy, you can do it at 7 AM or 11 PM — doesn't matter. Some patients describe it as the most "set and forget" medication they've ever taken.
The tradeoff: you need to keep it refrigerated (pre-filled pens) or at least temperature-controlled. If you're traveling, that means a cooler pack or insulated bag. And yes, taking needles through airport security requires a prescription label — not complicated, but it's one more thing to remember.
The Daily Tablet Routine
Wake up. Before coffee, before breakfast, before anything — place the tablet under your tongue. Let it dissolve. Follow your prescriber's guidance on food/drink timing, since protocols can vary by program and formulation. Then start your day normally.
That 30-minute waiting period is the adjustment most people mention. It means your morning routine shifts slightly — you can't grab coffee immediately when you roll out of bed. But most patients say they adapt within the first week. Some use the time to shower, get dressed, or scroll through email. It integrates naturally once you find your rhythm.
The upside: no needles, no refrigeration, no special storage. If you travel, you throw the tablets in your bag the same way you'd pack vitamins. No cooler packs, no explaining injection supplies to TSA.
The tradeoff: daily dosing means daily discipline. Skip a day and you've missed a dose. With the weekly shot, even if you're a day late, you're still covered. Some people genuinely struggle with daily medication consistency — and that's worth being honest with yourself about before choosing.
Which Routine Wins?
Neither. Both are manageable. Both become second nature within 2-3 weeks. The question isn't which is objectively easier — it's which matches your existing habits and lifestyle better. If you already take a daily morning supplement, adding a tablet feels natural. If you prefer a once-and-done approach, the weekly injection fits seamlessly.
How to Get Started With Tirzepatide (Pills or Shots)
You've read the research. You've compared the options. If you're ready to move forward, here's exactly what the process looks like.
The Typical Telehealth Process
Step 1: Complete an online health assessment. You'll answer questions about your medical history, current medications, weight, height, BMI, and weight-loss goals. This takes about 5-10 minutes. Be thorough and honest — this is how your provider determines if tirzepatide is safe and appropriate for you.
Step 2: Provider review. A licensed, board-certified medical professional reviews your assessment. Most providers complete this within 24 hours. If anything needs clarification, they'll reach out directly.
Step 3: Consultation. Some providers schedule a phone or video consultation. Others review your intake and communicate through the platform. Either way, a real clinician is evaluating your case — not an algorithm.
Step 4: Prescription and shipping. If approved, your prescription is sent to a licensed compounding pharmacy and shipped to your door — often with free overnight shipping. Your medication arrives with clear instructions, dosing information, and everything you need to start.
Step 5: Ongoing support. Monthly check-ins to monitor your progress, adjust your dosing as you titrate up, and manage any side effects. Good providers don't disappear after the first shipment.
General Qualifying Criteria
You'll typically need to meet one of these thresholds:
- BMI of 30 or higher (clinical obesity), OR
- BMI of 27 or higher with at least one weight-related condition (high blood pressure, high cholesterol, pre-diabetes, sleep apnea, PCOS)
And you should not have any contraindications — personal or family history of medullary thyroid carcinoma, MEN 2 syndrome, or allergy to tirzepatide.
Choosing a Provider
Remember that legitimacy checklist from earlier? Now's when you use it.
If you're looking for a place to start, SkinnyRx is one of the more established telehealth platforms offering compounded tirzepatide in both tablet and injectable form. Here's what they bring to the table:
- Tirzepatide tablets starting at $299/month
- Tirzepatide injectables starting at $299/month
- Licensed, board-certified medical providers
- Medications compounded by FDA-registered U.S. pharmacies
- Free overnight shipping
- No hidden fees or surprise rebilling
- Ongoing clinical support and dosage management
- FSA/HSA accepted
They carry a 4.8 out of 5 rating on Trustpilot from over 4,100+ verified reviews — the kind of track record that's built over time, not manufactured overnight. Patients frequently mention the personalized onboarding process, direct access to support staff, and the confidence they felt getting started. For more details, see our SkinnyRx review.
For a broader view of best compounded tirzepatide providers, see our full comparison, and check out our guide to best oral tirzepatide for weight loss.
The most important step is the first one. Every week you spend researching instead of starting is a week of potential results you don't get back. You already know tirzepatide works. Now it's about choosing your path and beginning.
Check if You Qualify for Tirzepatide Through SkinnyRxFAQ: Tirzepatide Pills vs Shots
Are tirzepatide tablets real?
Yes. Compounded tirzepatide sublingual tablets are real, prescribed medications prepared by licensed compounding pharmacies. They use the same active tirzepatide ingredient found in FDA-approved Mounjaro and Zepbound. However, the finished tablet product itself is not FDA-approved — only injectable tirzepatide has completed the full FDA approval process.
Do tirzepatide pills work as well as the shots?
Both forms use the same active ingredient targeting the same GLP-1 and GIP receptors. Injectable tirzepatide has stronger published clinical evidence from large-scale trials (SURMOUNT 1-5) showing 15-21% average weight loss (FDA label). Sublingual tablets deliver tirzepatide through oral mucosal absorption, which has variable bioavailability — providers compensate with adjusted dosing. Many patients report meaningful results with tablets, though they haven't been studied in the same rigorous clinical trial setting as injectables.
How much weight can I expect to lose on tirzepatide?
Clinical trials of injectable tirzepatide show average weight loss of 15-21% of body weight over 72 weeks, depending on dose (FDA label). For a 200-pound person, that's approximately 30-42 pounds. Individual results vary based on dose, adherence, diet, exercise, genetics, and starting weight. Most patients begin noticing appetite changes within the first 2-4 weeks, with visible weight loss typically becoming apparent by weeks 4-8.
What are the most common side effects?
The most common side effects for both forms are gastrointestinal: nausea (up to 29%), decreased appetite (20%+), diarrhea (up to 23%), and constipation (up to 17%). These are typically mild to moderate and improve within 2-4 weeks at each dose level. Gradual dose titration is the most important factor in keeping side effects manageable.
How much do tirzepatide pills cost per month?
Compounded tirzepatide sublingual tablets typically cost $249-$299 per month through telehealth providers, including the medication, provider consultation, and shipping. Brand-name injectable tirzepatide (Mounjaro/Zepbound) costs $1,000+ per month without insurance. Compounded injectables run $199-$299/month. FSA and HSA cards are accepted by most telehealth providers.
Can I switch from shots to pills (or vice versa)?
Yes. Many providers support transitions between forms. If switching from weekly injections to daily tablets, your provider will adjust your dosage to account for the different delivery method and dosing frequency. Always work with your prescribing provider when changing forms — don't try to convert doses on your own. If you're considering switching from a different GLP-1 medication like semaglutide, see our comprehensive guide on how to switch from semaglutide to tirzepatide.
Is there an FDA-approved tirzepatide pill coming?
Not directly for tirzepatide. Eli Lilly submitted orforglipron — an oral daily GLP-1 pill — for FDA approval in late 2025 (the timeline depends on FDA review), but orforglipron is GLP-1 only, not the dual GLP-1/GIP mechanism of tirzepatide. Oral semaglutide (Wegovy tablets) was FDA-approved for weight loss in December 2025 — but again, that's semaglutide, not tirzepatide. No oral tirzepatide (dual agonist) is currently in active FDA clinical trials.
Do I need a prescription for tirzepatide?
Yes — all forms. Both injectable and sublingual tirzepatide require a prescription from a licensed medical provider. Any seller offering tirzepatide without requiring a medical assessment and prescription should be avoided. Telehealth platforms make the prescription process accessible online, but a real clinician is always involved.
How long do I need to take tirzepatide?
The SURMOUNT-4 trial answered this directly: patients switched to placebo after 36 weeks experienced about +14% body-weight change (regain), while those who continued tirzepatide reached 25.3% total weight loss at 88 weeks and kept it off. Tirzepatide is intended as a long-term treatment for most patients. The goal is sustained weight management combined with lifestyle changes — not a temporary course.
Is compounded tirzepatide safe?
Compounded tirzepatide uses the same active ingredient that's been extensively studied in clinical trials involving over 5,000 participants in the SURMOUNT obesity program (plus additional data across other tirzepatide studies). The safety concerns specific to compounded products relate to the compounding process itself — whether the pharmacy follows proper quality standards. This is why choosing a provider that works with FDA-registered or state-licensed compounding pharmacies matters. Run through our legitimacy checklist to verify any provider you're considering.
Which has fewer side effects — pills or shots?
Both forms share the same fundamental side effect profile because they use the same medication. Some patients and providers have observed that daily sublingual tablets may produce a gentler side effect curve compared to weekly injections, potentially because a smaller daily dose creates less of a "bolus effect" than a larger weekly dose. This hasn't been confirmed in published studies, but it's a plausible pharmacological observation.
What if tirzepatide doesn't work for me?
While tirzepatide has the highest average efficacy of any weight-loss medication available, individual responses vary. If you don't see meaningful results after 12-16 weeks at a therapeutic dose, your provider can discuss dose adjustments, switching forms, or considering alternative medications. The key is working with a provider who monitors your progress and adapts your treatment plan.
The Bottom Line: Our Verdict on Tirzepatide Pills vs Shots
Both tirzepatide pills and shots deliver the most powerful weight-loss medication available today — a dual GLP-1/GIP agonist that outperformed semaglutide by roughly 50% in head-to-head clinical trials.
If you want the strongest clinical backing: Choose injectable tirzepatide. The SURMOUNT trials are among the most rigorous and impressive obesity studies ever published, with over 5,000 participants in the obesity program and results that reshaped the entire field.
If needles are a dealbreaker: Sublingual tirzepatide tablets put the same medication in your hands without a single injection. For many people, the best medication is the one they'll actually use — and if needle fear is the reason you haven't started, tablets eliminate that barrier entirely.
If cost is the deciding factor: Compounded options (both forms) bring tirzepatide within reach at $199-$299/month — a fraction of the brand-name price.
What matters most is that you start. Tirzepatide has helped hundreds of thousands of people achieve weight loss they never thought possible — improved blood pressure, reduced joint pain, better sleep, more energy, and a relationship with food that finally feels manageable. The people who get those results are the ones who stop researching and start.
The question isn't whether tirzepatide works. The data settled that. The question is whether today is the day you give yourself the chance to find out.
Check if You Qualify for Tirzepatide Through SkinnyRxSources and References
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. "Tirzepatide Once Weekly for the Treatment of Obesity." N Engl J Med. 2022;387(3):205-216. (SURMOUNT-1)
- Aronne LJ, Sattar N, Horn DB, et al. "Continued Treatment With Tirzepatide for Maintenance of Weight Reduction in Adults With Obesity." JAMA. 2024;331(1). (SURMOUNT-4)
- Aronne LJ, Horn DB, le Roux CW, et al. "Tirzepatide as Compared with Semaglutide for the Treatment of Obesity." N Engl J Med. 2025;393(1). DOI: 10.1056/NEJMoa2416394 (SURMOUNT-5)
- FDA Prescribing Information: Zepbound (tirzepatide) injection. accessdata.fda.gov
- FDA Prescribing Information: Mounjaro (tirzepatide) injection. accessdata.fda.gov
- Body Composition Changes During Weight Reduction With Tirzepatide in SURMOUNT-1. Diabetes Obes Metab. 2025.
- Horn DB, et al. "Cardiometabolic Parameter Change by Weight Regain on Tirzepatide Withdrawal." JAMA Intern Med. November 2025. (SURMOUNT-4 post-hoc)
- Efficacy and Safety of Tirzepatide on Weight Loss: Systematic Review and Meta-Analysis. PMC, 2025.
- Tirzepatide Patient Reviews. Drugs.com. (8.5/10 average, 2,600+ reviews)
- SkinnyRx Reviews. Trustpilot.com. (4.8/5 stars, 4,100+ reviews)
- FDA: Concerns with Unapproved GLP-1 Drugs Used for Weight Loss. FDA.gov
- FDA: Tirzepatide Injection Shortage Resolved / Compounding Policy Updates. FDA.gov
- White House Fact Sheet: Most-Favored-Nation Pricing — GLP-1 Drug Agreements. WhiteHouse.gov, November 2025.
- Eli Lilly. Orforglipron ATTAIN-MAINTAIN Phase 3 Results. Investor.lilly.com. Dec 2025.
- FDA Approves Oral Wegovy (Semaglutide) for Weight Management. Novo Nordisk / FDA. Dec 22, 2025.
- Prime Therapeutics. GLP-1 Pipeline Update: November 2025.
- FDA Prescribing Information: Wegovy Tablets (oral semaglutide). accessdata.fda.gov
Affiliate Disclosure
WeightLossProviderGuide.com may earn a commission when you use affiliate links on this page, at no additional cost to you. We recommend providers based on our published legitimacy criteria, not partnership agreements.
Medical Disclaimer
This article is for informational purposes only and does not constitute medical advice. Always consult a licensed healthcare provider before starting any medication, including tirzepatide in any form. Individual results vary. Compounded medications are not FDA-approved as finished products.
Last Updated: February 2026 | Last Verified: February 2026
How we rank + verify
Last verified: February 13, 2026
What we verified: FDA prescribing information for Mounjaro/Zepbound, SURMOUNT 1-5 clinical trial data (NEJM, JAMA), FDA warnings on unapproved GLP-1 drugs, provider pricing as of February 2026, Drugs.com patient reviews, Trustpilot provider ratings.
Sources: FDA labels, NEJM (SURMOUNT-1, SURMOUNT-5), JAMA (SURMOUNT-4), FDA.gov safety communications, PubMed meta-analysis, Drugs.com reviews, Trustpilot
Related Articles
Oral GLP-1 is here. Compare FDA-approved Wegovy tablets vs compounded programs—pricing, safety, side effects, and how to buy online.
How GLP-1 medications like Ozempic and Wegovy work for weight loss. Mechanism map, clinical trial results, side effects, and decision framework.
Complete GLP-1 side effects guide from FDA labels: what's normal, what's serious, and how to manage symptoms on Ozempic, Wegovy, Mounjaro, Zepbound.
Best compounded tirzepatide providers compared on pharmacy quality, real pricing, clinical oversight, and verified reviews. MEDVi ranked #1 for 2026.
This content is educational only and does not replace professional medical advice. Always consult a qualified healthcare provider before starting any medication.