How to Appeal a Wegovy or Zepbound Denial
Disclosure: Some links on this site are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you. Thank you for supporting our site.·For informational purposes only—not medical advice.
Bottom line:
Yes, you can appeal a Wegovy or Zepbound denial — and appeals do work. But the smartest first move depends on the reason printed on your denial notice. If the denial was caused by missing paperwork, a bad ICD-10 code, or an incomplete prior authorization, a corrected resubmission is usually faster than a formal appeal — and these are resolved in days, not weeks.
If the denial says “not medically necessary,” requires step therapy, or involves a formulary or plan exclusion, a formal appeal with a Letter of Medical Necessity is the right move — and in Medicare Advantage, more than 80% of prior authorization denials that were appealed were overturned.
This guide walks you through every scenario, with word-for-word scripts, a documentation checklist, real case outcomes, and what to do when appeals fail. Start with the denial reason table below.

What your denial type means — and the best first move
Before you write a single word of an appeal, figure out what kind of denial you're dealing with. The wrong response wastes your limited chances.

| Your denial says… | What it actually means | Best first move | Who should lead |
|---|---|---|---|
| "Incomplete documentation" or "criteria not met" | Missing BMI records, lab work, chart notes, or wrong ICD-10 code | Corrected PA resubmission | Your doctor's office |
| "Not medically necessary" | Insurer questions whether you need this specific drug | Formal appeal + Letter of Medical Necessity | You + your doctor together |
| "Must try other treatments first" (step therapy) | Insurer wants you on a cheaper drug before approving this one | Step-therapy exception with documented prior failures | Your doctor |
| "Not on formulary" or "preferred alternative required" | Your plan covers a different GLP-1, not this one | Formulary exception + clinical differentiation argument | Your doctor |
| "Weight-loss medications excluded" | Your employer or plan excludes the entire drug category | Check for alternate FDA-approved indication (CV or OSA) or employer escalation | You + HR + your doctor |
| Medicare Part D exclusion | Federal law limits weight-loss drug coverage under standard Part D | OSA indication (Zepbound) or CV indication (Wegovy) may apply; CMS GLP-1 Bridge begins July 2026 | Your doctor + Medicare plan |
“I was still clueless on where to even begin.”
Zepbound patient before filing a successful appeal (Reddit)
Individual experience. Not a guarantee of approval.
If you already know your denial type, skip to the section that matches. If you're not sure, keep reading.
What should you do first after a Wegovy or Zepbound denial?
The first 48 hours matter — not because the deadline is short (you usually have 180 days), but because momentum matters and details are fresh.
Your Explanation of Benefits (EOB) or denial notice contains the specific reason, the clinical criteria the insurer used, and the deadline and instructions for filing. Don't skim it. The exact wording determines your entire strategy.
If you only got a verbal denial at the pharmacy, call your insurer and request the written denial. You're legally entitled to it.
Call the number on your denial letter and say this:
“I'm calling about a denial for [Wegovy/Zepbound]. Can you tell me the exact denial reason, the clinical criteria that were used, and whether this should be handled as a corrected prior authorization, a formulary exception, or a formal appeal? Can you also confirm my deadline for filing?”
Ask them to send the criteria in writing. This call takes 15 minutes and can save you weeks of fighting the wrong battle.
Keep every version of your prior authorization, denial letter, EOB, portal messages, and pharmacy communications. If your appeal goes to external review, the independent reviewer sees the full paper trail.
If you're currently on the medication and the denial is interrupting active treatment, you may qualify for an expedited appeal. Under ACA rules, expedited external reviews can be decided within 72 hours. Ask your insurer about expedited processing if a gap in treatment would harm your health.
How to appeal a Wegovy or Zepbound denial in 7 steps
This is the core sequence for most Wegovy and Zepbound denials, though the exact forms, deadlines, and escalation path depend on your plan and denial reason.
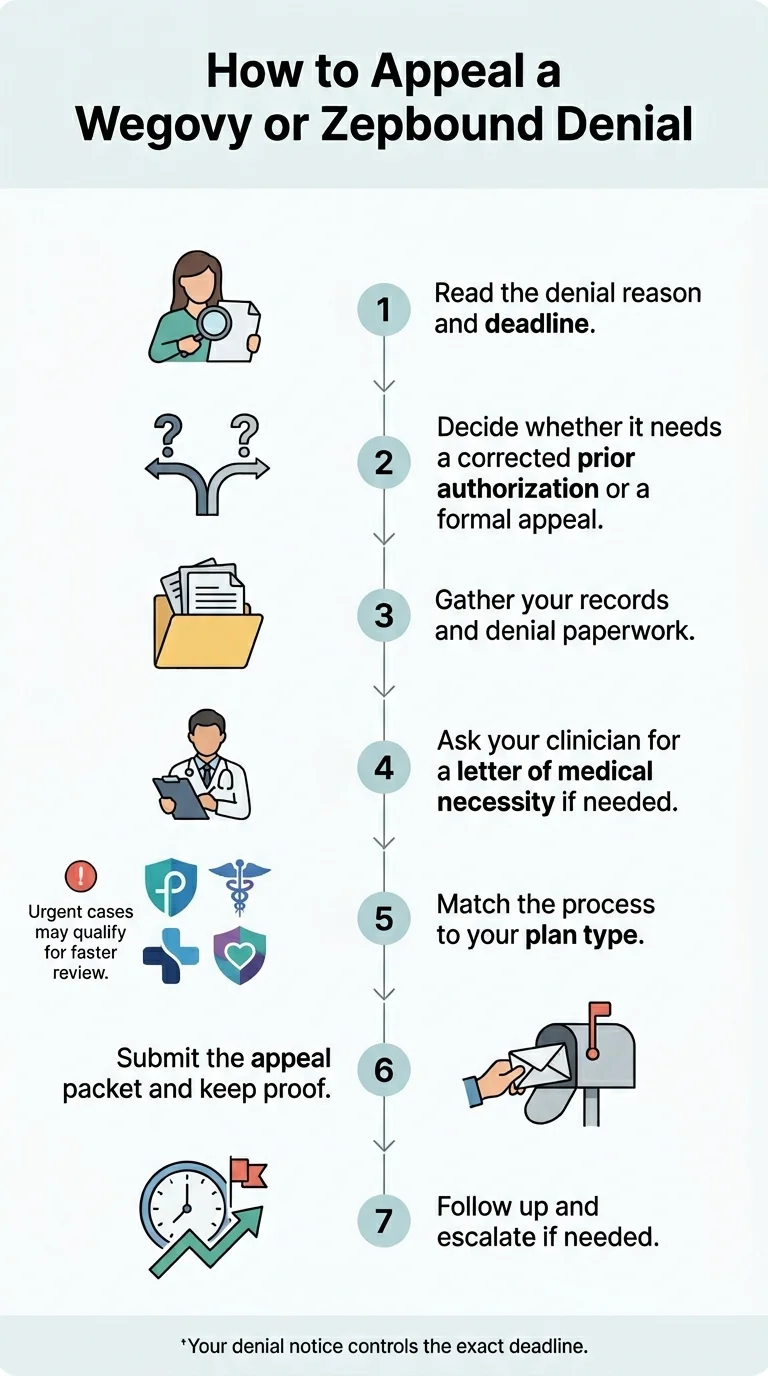
Identify the exact denial reason and your deadline
The denial reason IS the strategy. “Not medically necessary” and “not on formulary” require completely different responses. Get it in writing. Confirm the deadline.
For most commercial and ACA marketplace plans, you have 180 days from the denial notice to file an internal appeal. External review is typically available within 4 months of a final internal denial. Medicare drug appeals use different terminology and shorter timelines. Your denial letter specifies your deadline — use that number, not a generic one from the internet.
Fix a bad PA before filing a formal appeal
This is where most guides get it wrong.
If the denial happened because documentation was incomplete, a lab result was missing, the wrong ICD-10 code was submitted, or the prior authorization form wasn't fully filled out, a corrected PA resubmission is faster and more effective than a formal appeal. Your doctor's office can often resubmit within days.
Ask your doctor's office: “Was the PA submission complete? Were BMI records, chart notes, comorbidity documentation, and the correct diagnostic codes all included?” If anything was missing, fix and resubmit first.
Time-saving insight
A corrected PA resubmission can be approved in days. A formal appeal typically takes 30 days or more. Don't spend a month writing an appeal letter when the real problem was a missing lab result.
Gather the evidence your insurer actually wants
A winning appeal is not a persuasive essay. It's a case file that makes it easy for the reviewer to check boxes and approve.
Appeal documentation checklist:
For every appeal type:
- Copy of the denial letter with the reason code
- Your member ID, policy number, and group number
- Prescriber's name, NPI, and contact info
For “not medically necessary” denials, add:
- BMI measurements from at least two clinical visits (actual height and weight)
- Full list of comorbidities with ICD-10 codes (type 2 diabetes, hypertension, sleep apnea, PCOS, cardiovascular disease, etc.)
- Lab results: A1C, lipid panel, blood pressure readings
- Documentation of previous weight loss attempts (diet programs, exercise, prior medications and outcomes)
- Clinical notes explaining why this specific medication is appropriate
For step-therapy denials, add:
- Records of prior medications tried, doses, duration, and documented outcomes
- Documentation of side effects, intolerances, or contraindications
- Clinical rationale for why the required alternative is inappropriate for you
For formulary exceptions, add:
- Evidence of clinical differentiation between the requested drug and the preferred alternative
- Documentation of prior failure on or intolerance to the preferred drug
- Prescriber's explanation of why this specific medication is medically necessary

Get a Letter of Medical Necessity from your doctor
This is the document that does the heavy lifting. A strong Letter of Medical Necessity (LMN) from your prescribing clinician is the foundation of most successful appeals.
A good LMN should:
- Mirror the insurer's own criteria language (this is critical — use their words back at them)
- State your specific diagnoses, BMI, and relevant comorbidities
- Reference clinical trial evidence (STEP trials for Wegovy, SURMOUNT trials for Zepbound)
- Document what you've already tried and why it was insufficient
- Explain the medical consequences of not receiving treatment
- Make the "risk of delay" concrete — not "obesity is bad," but specific A1C, blood pressure, and failed intervention data
Both manufacturers provide sample appeal letter templates:
- Wegovy: Novo Nordisk's sample appeal letter and denials guide is available through novoMEDLINK
- Zepbound: Lilly's appeals guide and LMN template are downloadable from zepbound.lilly.com/access-coverage
How to ask your doctor:
“My insurance denied coverage for [Wegovy/Zepbound]. The denial reason is [exact reason from letter]. I'd like to file an appeal, and the strongest part would be a Letter of Medical Necessity from you. I've gathered my documentation — can we discuss what should go in the letter? I can share the insurer's clinical criteria so we can address them point by point.”
Match the appeal to your plan type
The appeal process changes based on what kind of insurance you have.
Commercial / ACA marketplace plans
File an internal appeal within 180 days. If denied, request external review within 4 months. The external reviewer's decision is binding on the insurer. You can also request concurrent external review for urgent situations.
Self-insured employer plans (ERISA)
Governed by federal law, not state insurance regulations. Typically one or two levels of internal appeal. If your employer controls the plan design, the appeals process has limits that marketplace plans don't. May need to escalate through HR or benefits department.
Medicare Part D and Medicare Advantage
Uses different terminology: coverage determination → redetermination → reconsideration. Timelines are shorter. Standard Medicare Part D does not cover GLP-1 drugs prescribed only for weight loss. CMS has announced a Medicare GLP-1 Bridge starting July 1, 2026.
Medicaid
Varies significantly by state. California's Medi-Cal ended adult weight-loss coverage for Wegovy, Zepbound, and Saxenda effective January 1, 2026. Medi-Cal may still review prior authorizations for cardiovascular and OSA indications.
Not sure what type of plan you have?
If you get insurance through a large private employer, it's likely a self-insured ERISA plan. If you bought coverage on HealthCare.gov or your state marketplace, it's an ACA plan. Call your insurance company or HR department to confirm.
Submit the appeal and document everything
Organize your appeal packet in this order:
- 1Cover sheet: Your name, member ID, denial reference number, prescriber info, and a one-sentence statement of what you're requesting
- 2Your appeal letter: Short, organized, criterion-by-criterion response to the denial reason
- 3Letter of Medical Necessity from your clinician
- 4Supporting exhibits: Lab results, BMI records, chart notes, prior treatment history, clinical trial references — labeled clearly
Submit through your insurer's preferred channel (online portal, fax, or mail). Keep a copy of everything you send and proof of your submission date.
Follow up and escalate if needed
Don't submit and forget. Mark your calendar:
Peer-to-peer review: Some insurers offer a peer-to-peer discussion between your clinician and the plan's medical reviewer. Check the denial notice or call the insurer to ask whether peer-to-peer review is available.
External review: If your internal appeal is denied, you have the right to external review by an independent organization. Their decision is binding on the insurer for most non-grandfathered plans under the ACA. You generally have 4 months from the final internal denial to request it.
“I appealed the denial via letter, and was approved about 30 days after I mailed the appeal.”
Real patient experience (Reddit)
Individual experience. Not a guarantee of approval.
Should you resubmit the prior authorization or file a formal appeal?
This is the question most pages skip — and it's the one that saves the most time.
- The denial letter mentions missing documentation, incomplete information, or criteria not met due to insufficient evidence
- Your doctor's office confirms that BMI records, lab results, or chart notes were not included
- The ICD-10 code was wrong or missing
- The denial says "not medically necessary" despite complete documentation
- You're required to complete step therapy you've already tried (or that's clinically inappropriate for you)
- The drug is not on formulary and you need an exception
- Your plan excludes weight-loss medications and you want to argue an alternate indication
A corrected PA resubmission can be approved in days. A formal appeal typically takes 30 days or more. Don't spend a month writing an appeal letter when the real problem was a missing lab result.
How to appeal a Wegovy denial specifically
Wegovy (semaglutide 2.4 mg) has unique advantages in the appeal process — most notably, its FDA-approved cardiovascular indication.
If Wegovy was denied for weight management
This is the most common scenario. Build your appeal around medical necessity: BMI documentation, comorbidities, prior treatment history, and clinical evidence.
Key clinical evidence to reference:
- STEP 1 trial: Adults taking semaglutide 2.4 mg achieved a mean weight reduction of 14.9% vs. 2.4% with placebo over 68 weeks
- STEP 3 and STEP 4 trials: Additional evidence for long-term efficacy and consequences of treatment discontinuation
Don't argue “Wegovy works for everyone.” Argue: “This patient meets the clinical criteria, has documented need, and the evidence supports this treatment for their specific condition.”
The cardiovascular reframe — Wegovy's strongest appeal card
In March 2024, the FDA approved Wegovy to reduce the risk of heart attack, stroke, and cardiovascular death in adults with established cardiovascular disease who also have obesity or overweight. This was based on the SELECT trial, which showed a 20% reduction in major adverse cardiovascular events.
The critical nuance
This argument only works if you actually have documented established cardiovascular disease (prior heart attack, stroke, or peripheral artery disease). In a publicly available Michigan external-review case, a patient's Wegovy appeal failed because they argued the cardiovascular indication but did not have documented established CVD. The reviewer upheld the denial.
If you have established heart disease, use the CV angle aggressively. If you don't, build your appeal on the weight management indication instead.
Novo Nordisk patient support
- Injectable self-pay: $199/month introductory (first 2 months), then $349/month
- Oral Wegovy: $149–$299/month depending on dose
- Patient assistance: for eligible uninsured patients
- NovoCare Support: 1-888-809-3942
Tuyo Health
from $155/mo
Compounded semaglutide · Labs included · No membership fees
Medically supervised · Licensed U.S. pharmacy · No prior authorization needed
Start Without Insurance →How to appeal a Zepbound denial specifically
Zepbound (tirzepatide) faces unique challenges — fewer years of formulary history, removal from major PBM formularies in 2025, and more limited coverage data. But it also has a unique advantage: an FDA-approved indication for obstructive sleep apnea.
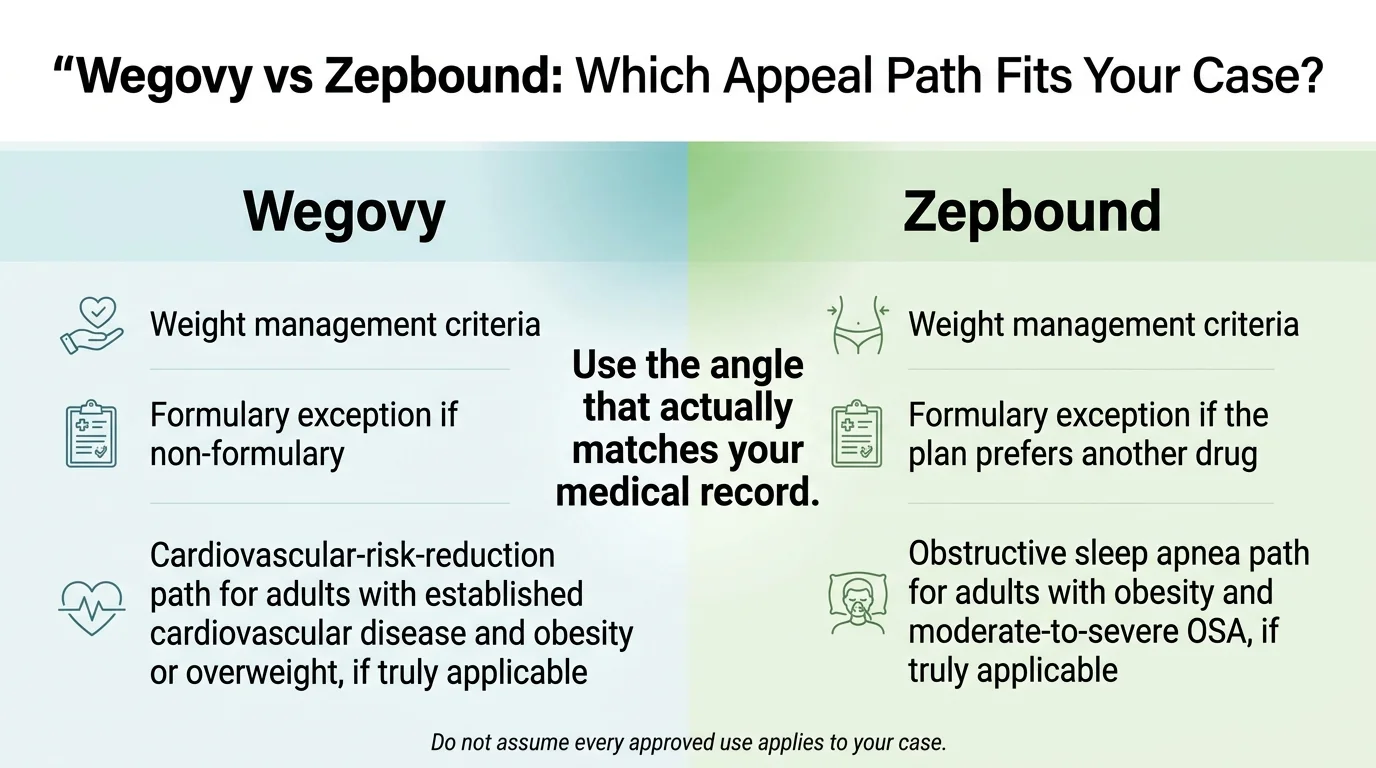
If Zepbound was denied because your plan prefers Wegovy
CVS Caremark removed Zepbound from its Standard Control, Advanced Control, and Value formularies effective July 1, 2025, while Wegovy remained preferred. If your denial came through one of those formularies, a formulary exception is the relevant next step.
The winning argument is clinical differentiation: Zepbound is a dual GIP/GLP-1 receptor agonist, while Wegovy targets GLP-1 only. These are different mechanisms of action. If you've responded well to Zepbound, or if you tried a semaglutide-based medication without adequate results, that distinction is your leverage.
Your appeal should document:
- Your clinical response to Zepbound (weight loss achieved, improvement in comorbidities)
- Any prior experience with GLP-1-only medications and why they were insufficient
- Side effects experienced on alternatives
- Your prescriber's clinical rationale for why Zepbound specifically is appropriate
The sleep apnea pathway — Zepbound's strongest coverage angle
In late 2024, the FDA approved Zepbound for moderate-to-severe obstructive sleep apnea (OSA) in adults with obesity. This creates a real coverage opportunity because many insurers — including some Medicare plans — cover medications for OSA even when they exclude weight loss drugs entirely.
If you have OSA symptoms (loud snoring, daytime fatigue, breathing pauses during sleep, morning headaches), talk to your doctor about a formal sleep study. A documented moderate-to-severe OSA diagnosis (typically AHI ≥ 15 events per hour) combined with obesity creates a strong foundation for coverage under the OSA indication.
The FDA indication is specifically for moderate-to-severe OSA, not mild. If your sleep study shows mild OSA, this pathway is weaker. Be straightforward with your doctor about whether this route genuinely fits your medical situation.
Lilly patient support
- LillyDirect self-pay: $299–$449/month for Zepbound vials (no insurance needed)
- Zepbound Savings Card: for patients with qualifying commercial insurance
- Appeals guide: downloadable from zepbound.lilly.com/access-coverage
What does your denial letter actually mean? (Plain-English decoder)
Insurance companies use language that sounds definitive but often isn't. Here's what each phrase actually means — and how final it really is.
What they mean: The reviewer decided the documentation doesn't prove you need this specific drug. This does NOT mean you don't need it — it means the paperwork didn't convince them.
How final is it? Not very final. Medical necessity denials are among the most commonly overturned on appeal. A strong Letter of Medical Necessity with complete documentation frequently changes the outcome.
What they mean: Your insurer wants you to try (and fail on) a cheaper or preferred medication first. Common requirements include Wegovy before Zepbound, or older drugs like Saxenda, Contrave, or metformin.
How final is it? Moderate. If you've already tried the required alternatives, document the results and request a step-therapy exception. If you haven't, your doctor can argue for an exception if there are medical reasons the required alternatives are inappropriate for you.
What they mean: Your plan has a list of approved drugs, and yours isn't on it.
How final is it? Moderate. A formulary exception request with clinical justification can work, especially with documented failure on or intolerance to the preferred alternative.
What they mean: Your employer or plan has decided not to cover any medications classified as weight-loss drugs.
How final is it? Toughest denial to overturn. But it's not always a dead end: if you have established cardiovascular disease → Wegovy's CV indication may create a path. If you have moderate-to-severe OSA → Zepbound's OSA indication may be covered. If your plan is employer-controlled → HR or benefits may be able to modify the exclusion.
What they mean: The paperwork was missing something. This is usually a documentation problem, not a medical rejection.
How final is it? Least final of all. Often fixed by resubmitting a complete PA.
What they mean: You were on the medication, it was working, and the insurer is refusing to continue coverage — often because renewal criteria weren't met or the formulary changed.
How final is it? Moderate to good appeal odds if you can document clinical benefit. Treatment response data, improved lab values, and your doctor's clinical notes about the risks of discontinuation become critical here.
What if your plan excludes weight-loss drugs entirely?
We need to be straight with you here.
If your employer or plan has a blanket exclusion on weight-loss medications, and you don't have qualifying cardiovascular disease (for Wegovy) or moderate-to-severe obstructive sleep apnea (for Zepbound), this is the most difficult denial to overturn.
That doesn't mean you should never try. It means you should:
- 1
Check for alternate indications first.
If you have heart disease history, the Wegovy cardiovascular pathway is real. If you have sleep apnea, the Zepbound OSA pathway is real. Talk to your doctor.
- 2
Escalate to your employer if it's a self-insured plan.
The exclusion is a decision your company made — not the insurance company. Some employers add GLP-1 coverage when employees make a clear case. Contact HR or your benefits department.
- 3
Know when to redirect your energy.
If the exclusion is firm, the indication angles don't apply, and HR won't budge, spending months on a long-shot appeal may not be the best use of your time — especially when there are ways to start treatment without insurance coverage at all.
What do real-world appeal wins and losses look like?
Public external-review decisions from state insurance departments show exactly why some cases win and others fail. We include a loss here because the pages that only tell you what you want to hear are the same ones that send you into an appeal unprepared.
A BCBS Michigan member had lost 9.6% of body weight on Zepbound when the insurer denied continued coverage, demanding proof of lifestyle modification through Teladoc. The patient appealed with clinical documentation of weight loss, comorbidity improvement, and provider notes. The independent reviewer overturned the denial, ruling the insurer's additional requirements were not consistent with standard of care.
Why it worked: Documented clinical response + strong provider support + reviewer found the insurer's criteria unreasonable.
A patient lost almost 50 pounds on Zepbound but developed side effects at the higher dose, requiring a step back to 2.5 mg. The insurer denied continued coverage at the lower dose. On external review, the independent reviewer overturned the denial, citing the patient's documented weight loss and the medical appropriateness of the lower dose.
Why it worked: Clear treatment benefit + medical rationale for the specific dose + reviewer prioritized clinical outcomes.
A Michigan patient challenged a Wegovy plan exclusion by arguing the cardiovascular risk reduction indication. The appeal failed because the patient did not have documented established cardiovascular disease as defined by the SELECT trial evidence basis. The independent reviewer upheld the denial.
Why it failed: The patient used the cardiovascular angle without meeting the actual clinical criteria.
The lesson across all three cases
Appeals succeed when the documentation clearly shows the patient meets the specific criteria being argued. They fail when the argument stretches beyond what the record supports. Reviewers are clinicians — they can tell the difference.
“I used yours as a template for my appeal, and as of this week my appeal was approved.”
Real patient experience (Reddit)
Individual experience. Not a guarantee of approval.
What to say to your doctor, insurer, and HR
You need words, not just strategy. Here are scripts you can actually use.
Message to your doctor
“Dr. [Name], my insurance denied coverage for [Wegovy/Zepbound]. The denial reason is [exact reason]. I'd like to file an appeal, and the strongest part would be a Letter of Medical Necessity from you. Could you include my BMI history, relevant comorbidities with ICD-10 codes, documentation of prior weight-loss attempts, and your clinical rationale for why this specific medication is necessary? I can share the insurer's clinical criteria so we can address each one directly. When would be a good time to discuss?”
Phone script for your insurer
“I'm calling about a denial for [Wegovy/Zepbound], reference number [from denial letter]. I'd like to understand the exact clinical criteria used to deny my claim, confirm whether I should resubmit the prior authorization or file a formal appeal, and get the submission instructions and deadline in writing. Can you also tell me whether a peer-to-peer review between my doctor and your medical reviewer is available?”
HR/benefits escalation email (for employer plan exclusions)
“Hi [HR contact], I recently learned that our health plan excludes GLP-1 medications for weight management. I'm writing because I believe this exclusion may be worth revisiting. The American Medical Association recognizes obesity as a chronic disease, and FDA-approved GLP-1 medications have demonstrated significant reductions in cardiovascular events, type 2 diabetes risk, and long-term healthcare costs. I'd appreciate the opportunity to discuss whether our plan design could be updated to include coverage for these treatments. Would you be open to a conversation about this?”
Follow-up if you don't hear back
- After 7 daysCall and reference your submission date and confirmation number
- After 30 daysSend a written follow-up requesting a status update and citing the applicable response timeframe from your plan
- If still no responseFile a complaint with your state insurance department (for fully insured plans) or the Department of Labor (for ERISA plans)
What if your first appeal fails?
A first denial is not the end. Here's the escalation path.
If your internal appeal is denied, you have the right to external review by an independent third-party organization. The reviewer is not employed by your insurance company, and their decision is binding on the insurer for most non-grandfathered ACA plans. You generally have 4 months from the final internal denial to request external review. For urgent situations, expedited external review can produce a decision within 72 hours.
Some plans offer a second level of internal appeal before external review. If available, use it — but strengthen your case with new evidence or a stronger LMN. Submitting the same packet twice rarely changes the outcome.
If you haven't already used this option, ask whether your prescriber can speak directly with the insurer's medical reviewer. This gives your doctor a chance to explain the clinical rationale in real time and address specific concerns the reviewer may have.
If you've exhausted appeals and believe the denial was handled improperly — delayed responses, contradictory explanations, failure to provide required documentation — you may have grounds for a regulatory complaint or legal action. This is more common with ERISA plans. An attorney specializing in insurance denials can evaluate your situation.
What if you need to start treatment while your appeal is pending?
Here's the honest reality most appeal guides skip entirely:
You don't have to wait for the insurance company to say yes before you start treatment. Your appeal could take 30 days. It could take longer. Meanwhile, your health doesn't pause.
Brand-name manufacturer pricing (no insurance needed)
- • Injectable: $199/month introductory, then $349/month
- • Oral Wegovy: $149–$299/month depending on dose
- • Patient assistance for eligible uninsured patients
- • 1-888-809-3942
- • Single-dose vials: $299–$449/month depending on dose
- • Brand-name, FDA-approved medication
- • No insurance needed, no prior authorization
- • lillydirect.com
Compounded GLP-1 medications
If brand-name self-pay pricing is more than your budget allows, compounded GLP-1 medications are another option. Licensed compounding pharmacies can prepare semaglutide and tirzepatide formulations under physician prescription, and several telehealth providers specialize in prescribing and delivering them.
Important distinctions to understand
Compounded GLP-1 products are not FDA-approved, are not the same as FDA-approved brand-name finished drugs, and the FDA has noted that some compounded semaglutide products may use salt forms that differ from the approved products. That said, compounded medications are legally prepared by state-licensed pharmacies under valid prescriptions, and they provide access to GLP-1 treatment at a significantly lower price point — typically $149–$299/month with a provider consultation, ongoing oversight, and home delivery included.
Compounded providers may be right for you if:
- Your plan excludes weight-loss medications and the alternate-indication angles don't apply to your medical history
- Brand-name self-pay pricing is beyond your budget
- You want to start treatment now while your insurance appeal processes — and can transition to brand-name if the appeal succeeds
Start treatment while your appeal is pending
Compare non-insurance GLP-1 options — price, clinical oversight, and what's included, side by side.
Can you appeal AND start treatment at the same time?
Yes. There's no rule that says you have to wait for an appeal decision before beginning treatment through another path. Many people start treatment immediately through a cash-pay or compounded provider, continue their appeal in the background, and transition to insurance-covered brand-name medication if the appeal succeeds.
This is often the smartest play — because you don't lose months of progress while bureaucracy grinds forward.
Tuyo Health
from $155/mo
Compounded semaglutide · Labs included every 3 months
Start now, continue your appeal, switch to brand-name if approved
Check Eligibility with Tuyo →Or take the free quiz to find the right match for your specific situation:
Not sure which path is right for you?
Take our free 60-second GLP-1 matching quiz and get a personalized recommendation based on your insurance situation and budget.
Frequently asked questions
Your next step
You now have a step-by-step guide covering every major denial type, both drugs, all plan types, and what to do when appeals don't work.
If your denial is a paperwork problem:
Contact your doctor's office today and ask them to resubmit a complete prior authorization. This is often resolved in days.
If you need to file a formal appeal:
Start gathering documentation using the checklist above, request a Letter of Medical Necessity from your doctor, and submit well before your deadline.
If your plan excludes weight-loss drugs and the alternate-indication angles don't apply:
You have two good options — manufacturer self-pay pricing or compounded GLP-1 providers that include a licensed provider visit and home delivery.
Why you should trust this page
We built this guide from primary sources: FDA announcements, HealthCare.gov and CMS appeal process documentation, Novo Nordisk's Wegovy denials and appeals guide, Eli Lilly's Zepbound access materials, KFF insurance data, AMA prior authorization surveys, public state external-review decisions from the Michigan Department of Insurance and Financial Services, and official CVS Caremark formulary information.
We included a real appeal failure alongside the wins. Not to discourage you — but because if we show you exactly why one case lost, you can avoid the same mistake.
What this page does NOT claim:
- We don't claim any specific success rate is guaranteed for your individual appeal
- We don't claim compounded medications are the same as FDA-approved brand-name products
- We don't claim the cardiovascular or OSA indication angles will work for everyone
- We don't offer legal or medical advice — work with your prescriber and insurance plan
Advertising disclosure: This page contains affiliate links to GLP-1 telehealth providers. If you sign up through our links, we may earn a commission at no additional cost to you. Our editorial recommendations are based on price, clinical quality, provider licensing, and patient experience — not commission rates. If we removed every affiliate link, this page would still be the most useful appeal resource we could build. Learn more
Sources
- [1]HealthCare.gov, "How to appeal a health insurance company decision". https://www.healthcare.gov/appeal-insurance-company-decision/internal-appeals/
- [2]CMS, "Medicare GLP-1 Bridge". https://www.cms.gov/medicare/coverage/prescription-drug-coverage/medicare-glp-1-bridge
- [3]HealthCare.gov, "External Review". https://www.healthcare.gov/appeal-insurance-company-decision/external-review/
- [4]Novo Nordisk, "Wegovy Denials and Appeals Guide" (novoMEDLINK PDF). https://www.novomedlink.com/content/dam/novonordisk/novomedlink/new/obesity/product-information/resources/library/documents/US25SEMO02476_DenialsandAppealsGuide.pdf
- [5]Eli Lilly, "Zepbound Access & Coverage". https://zepbound.lilly.com/access-coverage
- [6]FDA, "FDA Approves First Treatment to Reduce Risk of Serious Heart Problems Specifically in Adults with Obesity or Overweight" (March 2024). https://www.fda.gov/news-events/press-announcements/fda-approves-first-treatment-reduce-risk-serious-heart-problems-specifically-adults-obesity-or
- [7]FDA, Zepbound OSA approval announcement. https://www.fda.gov/news-events/press-announcements/fda-approves-new-medication-chronic-weight-management
- [8]Medi-Cal Rx, "GLP-1 Changes". https://medi-calrx.dhcs.ca.gov/cms/medicalrx/static-assets/documents/member/GLP1_Changes.pdf
- [9]CMS, "Appeals Fact Sheet". https://www.cms.gov/cciio/resources/fact-sheets-and-faqs/appeals06152012a
- [10]UnitedHealthcare Provider, "Appeals". https://www.uhcprovider.com/en/claims-payments-billing/appeals.html
- [11]Michigan DIFS, BCBSM External Review Case 233408. https://www.michigan.gov/difs/-/media/Project/Websites/difs/PRIRA/2025/March/BCBSM_233408.pdf
- [12]CVS Caremark, "Enabling Wider Access to Effective Weight Management". https://business.caremark.com/what-we-do/cost-management/formulary/glp-1s.html
- [13]Eli Lilly, LillyDirect self-pay pricing. https://www.lillydirect.com/
- [14]Michigan DIFS, BCBSM External Review Case 229915. https://www.michigan.gov/difs/-/media/Project/Websites/difs/PRIRA/2024/November/BCBSM_229915.pdf
- [15]FDA, "FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss". https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/fdas-concerns-unapproved-glp-1-drugs-used-weight-loss
- [16]KFF, "Medicare Advantage Insurers Made Nearly 53 Million Prior Authorization Determinations in 2024". https://www.kff.org/medicare/medicare-advantage-insurers-made-nearly-53-million-prior-authorization-determinations-in-2024/