GLP-1 Medication Storage Temperature Guide (2026): Every Major U.S. Brand
By WPG Editorial Team · Last updated: · Last verified against FDA labeling on DailyMed:
Last verified: May 6, 2026
Bottom line:
Most injectable GLP-1 pens and vials (Ozempic, Wegovy, Mounjaro, Zepbound, Trulicity, Saxenda, Victoza) live in the fridge at 36–46°F (2–8°C) until you start using them. After that, each brand has its own room-temperature window — anywhere from 14 days to 56 days, capped at 86°F (30°C) for most products and 77°F (25°C) for a few. Never freeze them. If a pen froze, touched a frozen gel pack, was left in a hot car, or arrived warm by mail — call your pharmacist before injecting.What we verified for this guide:
Every brand-name FDA-approved storage temperature, time window, and "do not use" condition in this page was checked against current FDA-approved labeling on DailyMed (the NIH's official drug label database) and manufacturer prescribing information. Compounded GLP-1 medications are handled separately because compounded products do not have FDA-approved product labeling — the dispensing pharmacy's label and Beyond-Use Date control. Last verification: May 6, 2026.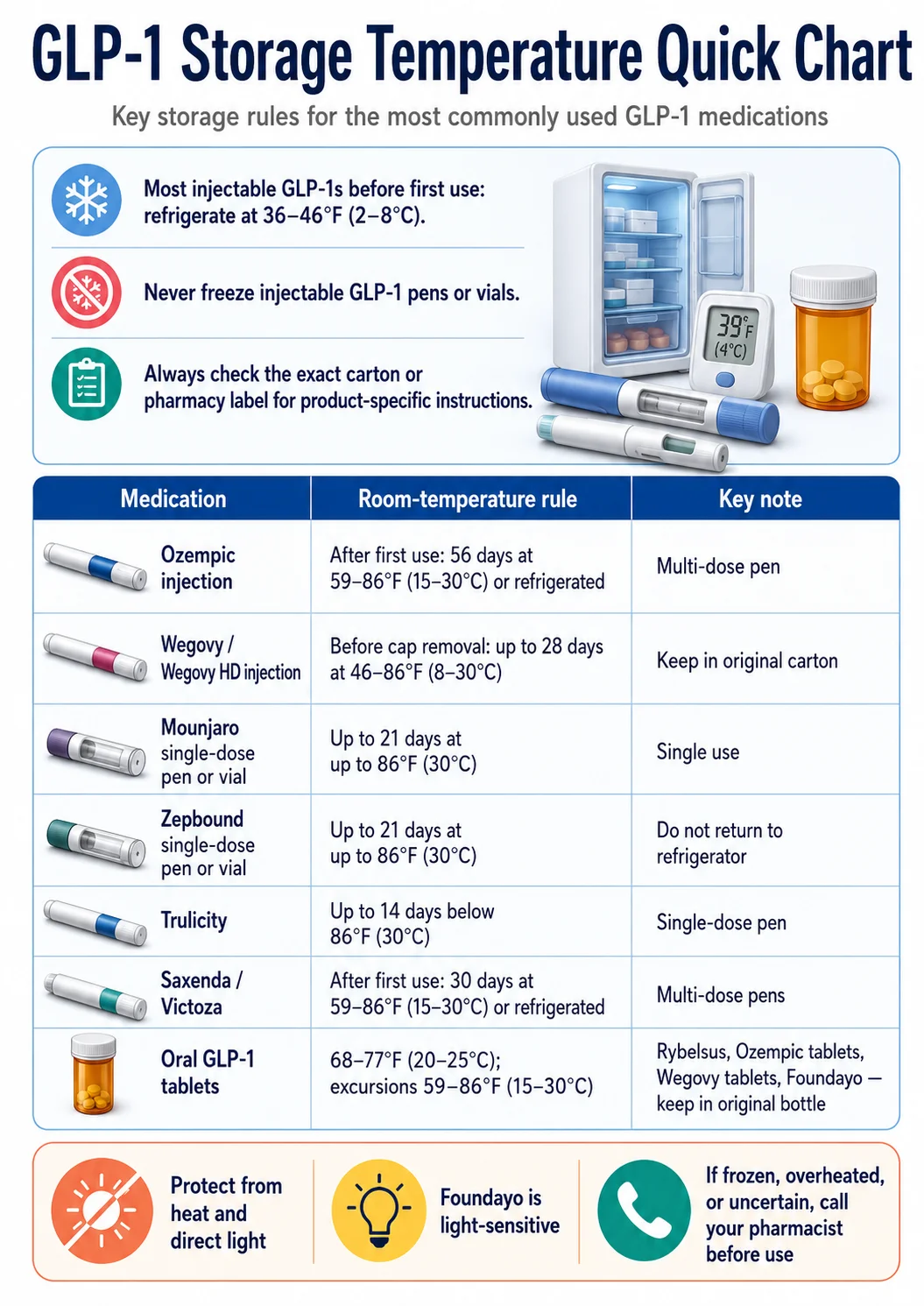
Why GLP-1 Storage Temperature Matters
GLP-1 medications come in two main forms: injectable peptide drugs (Ozempic, Wegovy injection, Mounjaro, Zepbound, Trulicity, Saxenda, Victoza, Byetta, Bydureon BCise, Adlyxin) and oral tablets (Rybelsus, Ozempic tablets, Wegovy tablets, Foundayo). The injectables are temperature-sensitive and need refrigeration. The tablets aren't.
When storage goes wrong, you do not have a home test that proves the medication still meets the conditions the label validated. You only know whether you followed the rules or not. That's the whole framework.
GLP-1 receptor agonists — the formal class name — break into two camps on storage. The injectables are peptide drugs: long peptide chains folded into a precise 3D shape that lets them bind their target receptor. Heat unfolds and degrades that shape. Freezing fractures it. Both are irreversible at the molecular level. Semaglutide, tirzepatide, liraglutide, dulaglutide, and exenatide are all in this category.
The oral GLP-1s play by different rules. Semaglutide tablets (Rybelsus, Ozempic tablets, Wegovy tablets) are formulated for oral absorption and stored at room temperature. Foundayo (orforglipron) is a small-molecule oral GLP-1 — not a peptide at all — also stored at room temperature with light-protective packaging.
The Master GLP-1 Medication Storage Temperature Chart
Every FDA-approved GLP-1 medication has slightly different storage rules. The chart below pulls every refrigerator temperature, room-temperature window, and "discard after" rule into one place, with each row sourced to the official FDA label on DailyMed. We rebuild this table whenever a label changes.
| Medication | Form | Refrigerated (before first use) | Room-temperature allowance | After first use / opened |
|---|---|---|---|---|
| Ozempic | Multi-dose semaglutide pen | 36–46°F (2–8°C) until expiration | Up to 86°F (30°C) | 56 days at 59–86°F or in fridge |
| Wegovy injection | Single-dose semaglutide pen | 36–46°F (2–8°C) | Up to 28 days at 46–86°F before cap removal | Single use — discard after dose |
| Wegovy HD | Single-dose semaglutide pen | 36–46°F (2–8°C) | Up to 28 days at 46–86°F before cap removal | Single use — discard after dose |
| Rybelsus | Oral semaglutide tablet | Room temperature 68–77°F | Excursions allowed 59–86°F | N/A — keep in original bottle |
| Ozempic tablets | Oral semaglutide tablet | Room temperature 68–77°F | Excursions allowed 59–86°F | N/A — keep in original bottle |
| Wegovy tablets | Oral semaglutide tablet | Room temperature 68–77°F | Excursions allowed 59–86°F | N/A — keep in original bottle |
| Mounjaro single-dose pen/vial | Tirzepatide | 36–46°F (2–8°C) | Up to 21 days at 86°F max | Single use — discard after dose |
| Mounjaro KwikPen / multi-dose vial | Tirzepatide multi-dose | 36–46°F (2–8°C) | Up to 30 days at 86°F max | Discard after 30 days from first use OR 30 days at room temp OR 4 weekly doses, whichever first |
| Zepbound single-dose pen/vial | Tirzepatide | 36–46°F (2–8°C) | Up to 21 days at 86°F max | Single use — discard after dose |
| Zepbound KwikPen / multi-dose vial | Tirzepatide multi-dose | 36–46°F (2–8°C) | Up to 30 days at 86°F max | Discard after 30 days from first use OR 30 days at room temp OR 4 weekly doses |
| Trulicity | Dulaglutide single-dose pen | 36–46°F (2–8°C) | Up to 14 days below 86°F | Single use — discard after dose |
| Saxenda | Liraglutide multi-dose pen | 36–46°F (2–8°C) | Up to 86°F | 30 days at 59–86°F or in fridge after first use |
| Victoza | Liraglutide multi-dose pen | 36–46°F (2–8°C) | Up to 86°F | 30 days at 59–86°F or in fridge after first use |
| Byetta | Exenatide multi-dose pen | 36–46°F (2–8°C) | Below 77°F (25°C) — lower than most | 30 days after first use |
| Bydureon BCise | Exenatide ER autoinjector | 36–46°F (2–8°C) | Up to 86°F for up to 4 weeks before use | Single use — discard after dose |
| Adlyxin | Lixisenatide multi-dose pen | 36–46°F (2–8°C) until activation | Up to 86°F | 14 days after activation |
| Foundayo | Oral orforglipron tablet | Room temperature 68–77°F | Excursions allowed 59–86°F | N/A — keep in original bottle |
| Xultophy 100/3.6 | Insulin degludec + liraglutide pen | 36–46°F (2–8°C) until expiration | Up to 86°F | 21 days at 59–86°F or in fridge |
| Soliqua 100/33 | Insulin glargine + lixisenatide pen | 36–46°F (2–8°C) | Below 77°F (25°C) | 28 days at room temperature; do not return to fridge after first use |
| Compounded semaglutide | Pharmacy-prepared vial | Per pharmacy label | Per pharmacy label | BUD on pharmacy label is authoritative |
| Compounded tirzepatide | Pharmacy-prepared vial | Per pharmacy label | Per pharmacy label | BUD on pharmacy label is authoritative |
Can I Put It Back in the Fridge? Brand-by-Brand
For most injectable GLP-1s, yes — you can move a pen between fridge and room temperature within its in-use window, and the cumulative time at room temperature is what counts. Two products are exceptions: Zepbound single-dose pens and vials should not be returned to the refrigerator once stored at room temperature, and Soliqua should not be returned to the refrigerator after first use.
| Medication / Form | Can return to fridge? | Clock starts when? | Discard rule |
|---|---|---|---|
| Ozempic | Yes — cycling allowed in 56-day window | First use (cap removal) | Discard 56 days after first use |
| Wegovy injection | Yes — within the 28-day pre-cap-removal window | Removal from refrigeration | Discard if at room temp 28+ days, frozen, above 86°F, or exposed to light |
| Wegovy HD | Yes — within the 28-day pre-cap-removal window | Removal from refrigeration | Same as Wegovy injection |
| Mounjaro single-dose pen/vial | Yes — within the 21-day window | Removal from refrigeration | Discard 21 days after removal from fridge |
| Mounjaro KwikPen / multi-dose vial | Yes — cycling allowed in 30-day in-use window | First use | 30 days from first use, 30 days at room temp, or 4 weekly doses |
| Zepbound single-dose pen/vial | ⚠️ No — once at room temp, keep at room temp until use or discard | Removal from refrigeration | Discard 21 days after removal from fridge |
| Zepbound KwikPen / multi-dose vial | Yes — cycling allowed in 30-day in-use window | First use | 30 days from first use, 30 days at room temp, or 4 weekly doses |
| Trulicity | Yes — within the 14-day window | Removal from refrigeration | Discard 14 days after removal from fridge |
| Saxenda | Yes — cycling allowed in 30-day in-use window | First use | Discard 30 days after first use |
| Victoza | Yes — cycling allowed in 30-day in-use window | First use | Discard 30 days after first use |
| Byetta | Yes, but ceiling is 77°F | First use | Discard 30 days after first use |
| Bydureon BCise | Yes — within the 4-week pre-use window | Removal from refrigeration | Single-dose — use within 4 weeks of removal from fridge |
| Adlyxin | Yes — within the 14-day in-use window | Activation (first use) | Discard 14 days after activation |
| Soliqua 100/33 | ⚠️ No — do not return to fridge after first use | First use | Discard 28 days after first use |
| Xultophy 100/3.6 | Yes — cycling allowed in 21-day in-use window | First use | Discard 21 days after first use |
| Compounded semaglutide / tirzepatide | Per pharmacy label | Per pharmacy label | Beyond-Use Date on label is authoritative |
When Does the Room-Temperature Clock Start?
Different GLP-1 medications start their room-temperature clock at different events. Some count from removal from the refrigerator. Some count from first use, cap removal, or activation. Some count from first puncture. Track both the date the pen left the fridge and the date you first used it — different labels start different clocks.
| Medication / Form | Room-temp clock starts at | Why it matters |
|---|---|---|
| Ozempic | First use (cap removal) | A pen that sat in the fridge for 30 days before first use still has its full 56-day window |
| Wegovy injection / Wegovy HD | Removal from refrigeration | The 28-day window is pre-cap-removal — you use the pen once, then discard |
| Mounjaro single-dose pen/vial | Removal from refrigeration | The 21-day window applies whether or not you've used it yet |
| Mounjaro KwikPen / multi-dose vial | First use | Plus the cap of 30 days at room temp and 4 weekly doses, whichever first |
| Zepbound single-dose pen/vial | Removal from refrigeration | 21 days, no return to fridge |
| Zepbound KwikPen / multi-dose vial | First use | Same 30/30/4 cap structure as Mounjaro KwikPen |
| Trulicity | Removal from refrigeration | 14 days, single-dose pen |
| Saxenda / Victoza | First use | Multi-dose pens, 30-day window |
| Byetta | First use | 30 days, 77°F ceiling |
| Bydureon BCise | Removal from refrigeration | 4-week pre-use window, then single use |
| Adlyxin | Activation | 14 days post-activation |
| Soliqua 100/33 | First use | 28 days, no return to fridge |
| Xultophy 100/3.6 | First use | 21 days |
| Compounded GLP-1 | First puncture, per pharmacy label | BUD on label controls |
The Sharpie habit:
When a pen leaves the fridge, write the date on the carton. When you first use it (or activate or puncture, depending on the product), write that date too. You'll thank yourself later."I Left My Pen Out — Is It Still Good?" The 5-Question Framework
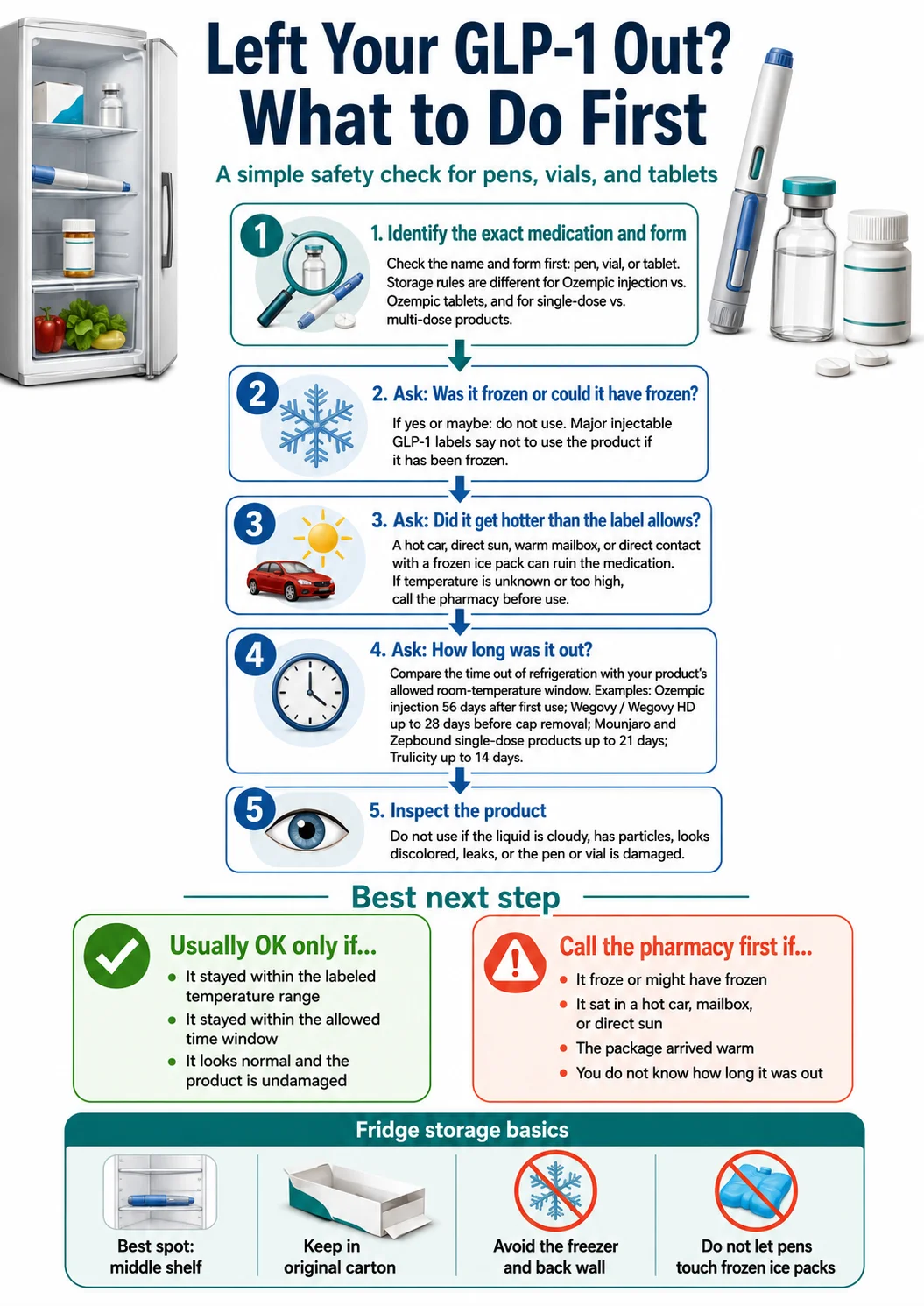
When something has gone wrong with your GLP-1 storage, work through five questions in order. If you reach a question you can't answer with confidence, stop and call the dispensing pharmacy before injecting.
Which exact medication and form?
Pull the box. Read the label. Mounjaro single-dose pen and Mounjaro KwikPen follow different rules. Zepbound single-dose pen and Zepbound KwikPen follow different rules. Ozempic injection and Ozempic tablets are completely different — one is a pen that lives in the fridge, the other is a pill at room temperature. Match what's on your carton to the row in the chart above.
Was it frozen, or could it have been frozen?
If yes — even maybe — most labels say discard. Wegovy, Mounjaro, Zepbound, Ozempic, Trulicity, Saxenda, Victoza, and Byetta all carry "do not use if frozen" language. Freezing physically rearranges the peptide structure. The medication may look perfectly clear after thawing and still be inactive. Common ways pens freeze without you realizing: stored against the back fridge wall, in a cooler touching a frozen gel pack, in checked airline luggage, in a mailbox during winter, or in a car overnight in cold weather.
Did it get hotter than the label allows?
Most GLP-1 pens are rated to 86°F (30°C). Byetta and Soliqua are tighter at 77°F (25°C). A parked car can hit 130°F+ on a moderate sunny day. A mailbox in summer sun can hit similar temperatures. A windowsill in afternoon light can run 90–100°F. If the pen got measurably above its label's max temperature, call the dispensing pharmacy or manufacturer before using it.
How long was it out, and at what temperature?
Cross-reference the master chart above. An Ozempic pen out for 18 hours at 70°F is well within its 56-day window. A Zepbound single-dose pen out for 22 days at 75°F has just crossed the 21-day cap — the label says throw it away. A Trulicity pen out for 16 days at 80°F is past its 14-day cap. Cycling between fridge and room temperature does not reset the clock — cumulative time at room temperature is what counts.
Are there any visible signs of damage?
Look through the pen window. The medication should be clear, colorless or very faintly yellow, with no particles, no cloudiness, no visible color change. The pen body should be intact — no cracks, no leaks. A clear, normal-looking pen is not proof of safety after a temperature excursion — but cloudy, discolored, or particle-containing medication is proof of compromise. Don't inject it.
Specific Mishap Scenarios
"I left my GLP-1 pen out overnight"
Probably fine — for most brands, in most home environments. Standard indoor temperatures (65–75°F) are well within every GLP-1's room-temperature range. The only question is whether you've used up your room-temperature budget.
For an Ozempic pen, an overnight excursion costs you one of 56 days. For a Mounjaro or Zepbound single-dose pen/vial, one of 21 days. For Trulicity, one of 14 days. Mark the date the pen left the fridge with a Sharpie on the carton.
"I left my pen in a hot car"
This one is harder. Car interiors heat fast. The National Highway Traffic Safety Administration notes vehicle temperatures can rise 20°F in as little as 10 minutes, and the National Weather Service notes interiors can rise 50°F in an hour even when outdoor temperatures are in the 70s. Both extremes exceed every GLP-1 label's temperature ceiling.
For Wegovy, the label explicitly says to throw away the pen if it has been exposed to temperatures above 86°F. For other GLP-1 products, heat above the labeled maximum means you're outside validated storage conditions. Call the dispensing pharmacy or manufacturer before using it.
Manufacturer hotlines for hot-car calls:
- Novo Nordisk (Ozempic, Wegovy, Saxenda, Victoza, Rybelsus): 1-800-727-6500
- Eli Lilly (Mounjaro, Zepbound, Trulicity): 1-800-545-5979
- Sanofi (Adlyxin, Soliqua): 1-800-633-1610
- AstraZeneca (Bydureon BCise): 1-800-236-9933
- Byetta: 1-800-868-1190
"My pen froze in the fridge"
Discard it. Every major injectable GLP-1 manufacturer says the same thing: do not use a GLP-1 medication that has been frozen, even if it has thawed and looks normal. Freezing physically denatures the peptide. The molecular shape that lets the drug bind its receptor is broken, and that damage isn't reversed by thawing. The pen will look identical to a working one.
"My pen looks cloudy or has particles"
Discard it. Healthy injectable GLP-1 medication is clear, colorless or very faintly yellow, with no visible particles, no cloudiness, and no color change. Anything outside that — discoloration, particles, gel-like consistency, white flecks — is a signal that the medication has degraded or been contaminated. Mayo Clinic's semaglutide patient guidance is direct: do not use it if it is cloudy, discolored, or has particles in it.
"My mail-order GLP-1 arrived warm"
Don't inject yet. Document and call. The FDA's official guidance is clear: the agency recommends patients not use any injectable GLP-1 drug that arrives warm or with insufficient refrigeration, because this can affect the drug's quality. That guidance applies whether the product is a brand-name pen or a compounded vial.
Within five minutes of opening the package: photograph the box exterior, the gel packs, the inside liner, and the medication. Note the time of delivery and the time of opening. If the gel packs are at room temperature or the inside liner feels warm, treat the shipment as a cold-chain failure.
"The fridge ran too cold and the pen feels icy"
If the pen genuinely froze — or if you're not sure — apply the freezing rules. Discard. Replace. If the pen is just very cold but never reached freezing, it should still be fine. Most home fridges should be set to 37–40°F to comfortably hold the 36–46°F GLP-1 range.
"There was a power outage — how long is my pen safe?"
The common 4-hour closed-fridge food-safety rule is a rough emergency clue, not medication-stability validation. Product-specific guidance:
- ·Ozempic in-use: validated for 56 days at room temperature — a multi-hour outage is essentially a non-event
- ·Wegovy injection unopened: has a 28-day room-temp window already built in
- ·Mounjaro / Zepbound single-dose: 21-day room-temp window
- ·Trulicity: only 14 days room-temp, so longer outages start to count against it
- ·Compounded vials: follow the pharmacy label
If the refrigerator went above 46°F and you cannot match the event to a labeled room-temperature allowance, call the dispensing pharmacy.
"I took the pen out and put it back — is that OK?"
For Ozempic, Saxenda, Victoza, Mounjaro KwikPen, Zepbound KwikPen, and most other multi-dose pens within their in-use window: yes, cycling between fridge and room temperature is fine. The total time at room temperature is what matters, and the clock doesn't reset when you re-refrigerate. For Zepbound single-dose pens and vials and for Soliqua: do not return to the refrigerator per the FDA-approved labels.
Brand-by-Brand Storage Rules
Each FDA-approved GLP-1 medication has its own labeled storage profile. Below is the detailed rule set for each — refrigeration range, room-temperature window, what to do if frozen, and common mistakes — sourced from the FDA-approved label on DailyMed.
Ozempic (Semaglutide)
Unopened pens: refrigerator at 36–46°F (2–8°C) until printed expiration date. After first use (cap removal): 56 days at 59–86°F or in the fridge. Ozempic is a multi-dose pen — this 56-day window covers all weekly injections you'll get from one pen.
Discard after 56 days regardless of how much medication remains. Discard if frozen. Discard if above 86°F. Keep the cap on between uses. Remove the needle after every injection.
Common mistake: confusing Ozempic's 56-day window with Wegovy injection's 28-day window. Same active ingredient (semaglutide), different storage profiles because of formulation and pen design.
Manufacturer (Novo Nordisk): 1-800-727-6500
Wegovy and Wegovy HD Injection (Semaglutide)
Wegovy injection is a single-dose pen. Unopened: fridge at 36–46°F (2–8°C). Before cap removal: can be kept at 46–86°F for up to 28 days. After 28 days at room temperature, throw it away. After cap removal, use it once and discard.
The Wegovy label explicitly says: throw away if frozen, exposed to temperatures above 86°F, exposed to light outside the carton, or kept at room temperature for 28+ days. Keep in the original carton until you're ready to inject — the carton blocks light.
Common mistake: assuming the 28-day window resets when you put the pen back in the fridge. It doesn't. Cumulative time at room temperature counts.
Manufacturer (Novo Nordisk): 1-800-727-6500
Mounjaro (Tirzepatide) — Single-Dose Pen and Vial
Fridge at 36–46°F (2–8°C) until expiration. Room temperature: up to 86°F for up to 21 days. After 21 days at room temperature, discard. Single-dose — one pen or vial, one weekly dose, then dispose.
Common mistake: treating Mounjaro storage as identical to Ozempic. Mounjaro single-dose gets 21 days at room temp; Ozempic gets 56. Both come from FDA labels, but they're not the same.
Manufacturer (Eli Lilly): 1-800-545-5979
Mounjaro KwikPen and Multi-Dose Vial
Refrigerated at 36–46°F (2–8°C). Once opened, discard after 30 days from first use OR 30 days at room temperature OR 4 weekly doses, whichever comes first. Can be stored in fridge or at room temperature up to 86°F during the in-use window. Most patients will use 4 weekly doses out of a KwikPen and discard, even if some medication appears to remain.
Zepbound Single-Dose Pen and Vial (Tirzepatide)
Fridge at 36–46°F (2–8°C). Room temperature: up to 86°F for up to 21 days. After 21 days at room temperature, discard.
Important: once the pen or vial has been kept at room temperature, do not return it to the refrigerator. This is stricter than Mounjaro's KwikPen rule.
Zepbound KwikPen and Multi-Dose Vial
Refrigerated until first use at 36–46°F. Discard after 30 days from first use OR 30 days at room temp OR 4 weekly doses — whichever cap is hit first. Always verify against the specific carton in your hands.
Trulicity (Dulaglutide)
Shortest room-temperature window of the major brands: 14 days below 86°F (30°C) after removal from fridge. Refrigerated at 36–46°F until first use. Each pen is single-dose. Do not freeze.
Common mistake: assuming Trulicity matches the more generous windows of Ozempic or Mounjaro KwikPen. 14 days is the cap.
Manufacturer (Eli Lilly): 1-800-545-5979
Saxenda and Victoza (Liraglutide)
Same active ingredient (liraglutide), same storage profile. Refrigerated 36–46°F before first use. After first use: 30 days at 59–86°F or in the fridge. Discard after 30 days even if medication remains. Both are multi-dose pens — Saxenda is dosed daily for weight management, Victoza daily for type 2 diabetes.
Manufacturer (Novo Nordisk): 1-800-727-6500
Byetta and Bydureon BCise (Exenatide)
Byetta: Multi-dose pen. Refrigerated at 36–46°F. After first use: store below 77°F (25°C) — note this is a tighter ceiling than the 86°F that applies to most of the class. Use within 30 days. Protect from light.
Bydureon BCise: Extended-release autoinjector. Refrigerated at 36–46°F. Can be kept at up to 86°F for up to 4 weeks before use. Each is single-dose.
Common mistake with Byetta: using the 86°F ceiling that applies to most GLP-1s. Byetta's room-temperature ceiling is 77°F — one of the rare label nuances most generic storage articles miss.
Adlyxin (Lixisenatide)
Multi-dose pen. Refrigerated at 36–46°F until activation. After activation: room temperature up to 86°F for up to 14 days. Protect from light. Replace the cap between uses.
Manufacturer (Sanofi): 1-800-633-1610
Xultophy 100/3.6 and Soliqua 100/33 (Combination Products)
Xultophy (insulin degludec + liraglutide): Refrigerated at 36–46°F until expiration. After first use: 21 days at 59–86°F or in the fridge. Manufacturer (Novo Nordisk): 1-800-727-6500.
Soliqua (insulin glargine + lixisenatide): Refrigerated at 36–46°F before first use. After first use: below 77°F for up to 28 days. Once at room temperature after first use, do not return to the refrigerator. Manufacturer (Sanofi): 1-800-633-1610.
Rybelsus, Ozempic Tablets, Wegovy Tablets, and Foundayo (Oral GLP-1s)
Oral GLP-1 medications are stored at room temperature 68–77°F (20–25°C) with brief excursions allowed at 59–86°F. They do not need refrigeration.
Critical rule: keep them in the original sealed bottle until you're ready to take them. The labels say not to transfer tablets to a pill organizer. The original bottle includes a desiccant that protects the tablet from moisture. Foundayo is also light-sensitive — keep the bottle closed and out of direct sun.
Compounded Semaglutide & Tirzepatide: The Rules Are Different
Compounded GLP-1 medications are prepared by individual compounding pharmacies, not by Novo Nordisk or Eli Lilly. They are not FDA approved — FDA does not review compounded drugs for safety, effectiveness, or quality before they are marketed. Their storage rules — including the Beyond-Use Date (BUD) — are set by the dispensing pharmacy. Use the pharmacy label, not the brand-name pen rules.
Why compounded GLP-1 storage is different
A compounded medication is prepared from active pharmaceutical ingredient by a licensed compounding pharmacy. Each pharmacy uses its own formulation, preservative system, packaging, and stability testing protocol. That means: the concentration may differ from the brand-name pen; the container is usually a multi-dose vial, not a pen; sterility and potency are validated by the pharmacy, not the original manufacturer; and the shelf life — the Beyond-Use Date (BUD) — replaces the manufacturer's expiration date.
Current FDA position on compounded GLP-1 medications
Common mistakes with compounded GLP-1 storage
- Applying Ozempic's 56-day rule to a compounded vial. The rule was set for a specific Novo Nordisk pen formulation with specific stabilizers. It doesn't generalize.
- Ignoring the BUD because the medication looks fine. A peptide vial that has lost potency looks identical to a fresh one.
- Failing to track the puncture date. Most compounded vials are multi-dose. The clock starts at first puncture. Write the date on the carton.
- Letting the vial cycle through light or heat exposure. Compounded products generally don't have manufacturer-validated room-temperature excursion data. Behave conservatively.
What "Room Temperature" Actually Means
"Room temperature" on a GLP-1 label is a specific labeled range, not a feeling. When the FDA-approved label says "room temperature," it means a specific range — not "anywhere indoors."
| Range | Fahrenheit | Celsius | Where you'll see it |
|---|---|---|---|
| Refrigerated | 36–46°F | 2–8°C | Every injectable GLP-1 before first use |
| Most pens, room temp | 59–86°F | 15–30°C | Ozempic, Saxenda, Victoza after first use |
| Wegovy unopened range | 46–86°F | 8–30°C | Wegovy injection before cap removal |
| Lower ceiling | Below 77°F | Below 25°C | Byetta after first use; Soliqua after first use |
| Oral tablets standard | 68–77°F | 20–25°C | Rybelsus, Ozempic tablets, Wegovy tablets, Foundayo |
| Oral tablets excursion | 59–86°F | 15–30°C | Brief travel allowance for oral GLP-1s |
Where "indoor" can still be too warm
- A windowsill in afternoon sun: 90–100°F
- A kitchen counter near the stove: spikes during cooking
- The top of the fridge (heat plume from the compressor)
- A bathroom during a hot shower
- A car cup-holder in summer: 130°F+ in under an hour
- A tote bag near a radiator
- A mailbox in summer sun: easily exceeds 90°F
If your home runs warm in summer and you don't have AC, store the medication in the fridge, not on the counter. The room-temperature window is a flexibility option, not a default.
How to Store GLP-1 Medication in Your Refrigerator
The middle shelf of your fridge, in the original carton, is the safest spot for GLP-1 pens. A $10 fridge thermometer is the best preventive investment a GLP-1 patient can make.
Door shelves
AvoidWarmest area, most temp swings
Back wall / top
AvoidCan dip below freezing
Crisper drawers
AvoidToo humid
Middle shelf, center
Use thisMost stable temperature
Keep the pen in its original carton. The carton blocks light, a separate degradation pathway from heat. Never warm a pen with a microwave, hot water, a heater, or sunlight — those methods can spike the temperature past the label's ceiling.
Traveling With GLP-1 Medication
Always pack GLP-1 medications in carry-on luggage, not checked bags. TSA permits medically necessary liquids and frozen gel packs through screening — declare them. Use an insulated medical pouch with a non-frozen cooling element. Frozen gel packs in direct contact can freeze the pen, ruining it.
TSA rules for GLP-1 medications
The TSA permits medically necessary liquids — including injectable medications — in carry-on luggage in quantities greater than the standard 3.4 oz limit. Pens, vials, and syringes are permitted. Frozen gel packs are permitted; if a pack is partially thawed at screening, you may need additional inspection but it's still allowed.
- Keep the medication in its original pharmacy-labeled packaging
- Declare it to the TSA officer before screening starts
- Separate it from other carry-on items so it can be screened individually
- Carry a copy of your prescription, especially for international travel
- A doctor's note is not legally required in the U.S. but can help if questioned
Choosing a travel cooler
The frozen gel pack mistake:
A frozen gel pack in direct contact with the pen can freeze the pen — the same failure mode as the back-of-the-fridge cooling element. Better options: PCM-based cooling packs (phase-change material, rated for 36–46°F), gel packs that have been refrigerated not frozen, or insulated medical pouches with separate compartments.Hotels, Airbnbs, and cruises
Confirm refrigerator access before you book. Hotel mini-fridges are notoriously inconsistent — some run too warm (45–55°F), some too cold and can freeze pens. A small refrigerator thermometer in your travel kit solves this. Carry your medication in original pharmacy-labeled packaging. For international travel, check the destination country's medication-import rules — do not rely on U.S. TSA rules outside the U.S.
What If Your Mail-Order GLP-1 Arrived Warm?
Document everything within five minutes of opening the package. The FDA recommends patients not use any injectable GLP-1 drug that arrives warm or with insufficient refrigeration, as this can affect the drug's quality.
The 5-minute cold-chain self-check
How to Tell If Your GLP-1 Has Gone Bad
Healthy GLP-1 medication is clear, colorless or very faintly yellow, with no particles, no cloudiness, and no visible damage to the pen body. Cloudy, discolored, or particle-containing medication has degraded and should not be used. A frozen pen, however, often looks completely normal after thawing — visual inspection alone cannot rule out freeze damage.
What healthy looks like
- · Clear liquid — no haze, no cloudiness
- · Colorless or very faintly yellow
- · No particles — no flecks, no specks
- · No discoloration
- · No leakage at the pen body or cap
- · No cracks in the pen housing
Discard and replace if:
- · Cloudy or hazy liquid
- · Discolored (brown, dark yellow, pinkish)
- · Visible particles or white flecks
- · Gel-like consistency
- · Leakage or cracks
- · Known freeze or heat excursion — even if it looks fine
Why visual inspection isn't enough after a temperature excursion:
A pen that has been frozen and thawed often looks identical to a pen that has never been frozen — still clear, still colorless, still particle-free. The peptide structure has been damaged at the molecular level, but you can't see it. Visual inspection can identify obvious damage, but it cannot confirm safety after a known temperature excursion.Tracking First-Use and Discard Dates: The Sharpie Protocol
Write the first-use date on the pen carton with a permanent marker the moment the pen leaves the fridge. This is the easiest habit to build and the one that prevents the most expensive mistakes.
Safe Disposal of GLP-1 Pens, Needles, and Vials
GLP-1 pens with attached needles, used needles, and used compounded vials should go into an FDA-cleared sharps container immediately after use, per FDA guidance. They should not go in regular household trash.
What goes in a sharps container
- ·Used pen needles
- ·Empty Ozempic, Saxenda, Victoza, and other multi-dose pens (residual needle-attached risk)
- ·Single-dose autoinjector pens (Wegovy, Mounjaro single-dose, Zepbound single-dose, Trulicity, Bydureon BCise) after use
- ·Used syringes from compounded vial dosing
- ·Empty compounded vials with sharps potential
When to Call: Pharmacy, Manufacturer, or Prescriber?
For storage and temperature questions, call the dispensing pharmacy first — they have your specific dispensing record. For lot-specific quality issues or pen mechanical problems, call the manufacturer. For dosing questions or symptom changes, call your prescriber.
| Situation | Who to call |
|---|---|
| Not sure if a temperature excursion was within label tolerance | Dispensing pharmacy (first) |
| The pen looks abnormal (cloudy, particles, discolored) | Dispensing pharmacy; if needed, manufacturer |
| Mail-order shipment arrived warm | Dispensing pharmacy |
| Need a replacement under temperature-excursion policy | Dispensing pharmacy |
| Pharmacy can't resolve the question | Manufacturer hotline (listed on carton) |
| Pen has a mechanical problem (dose counter stuck, leakage, won't dispense) | Manufacturer hotline |
| Lot-specific quality information needed | Manufacturer hotline |
| Storage problem caused you to miss a dose | Prescriber |
| Considering doubling up or skipping a dose | Prescriber |
| Symptoms have changed (blood sugar, weight loss, side effects) | Prescriber |
| Need a new prescription because the previous pen was discarded | Prescriber |
Manufacturer hotlines:
- Novo Nordisk (Ozempic, Wegovy, Wegovy HD, Wegovy tablets, Rybelsus, Ozempic tablets, Saxenda, Victoza, Xultophy 100/3.6): 1-800-727-6500
- Eli Lilly (Mounjaro, Zepbound, Trulicity): 1-800-545-5979
- Sanofi (Adlyxin, Soliqua 100/33): 1-800-633-1610
- AstraZeneca (Bydureon BCise): 1-800-236-9933
- Byetta: 1-800-868-1190
- Compounded medications: call the dispensing compounding pharmacy directly.
Frequently Asked Questions
Does Ozempic need to be refrigerated?
Yes — before first use. Unopened Ozempic pens must stay in the refrigerator at 36–46°F (2–8°C) until first use. After first use, the pen can be kept at 59–86°F or in the fridge for up to 56 days.
How long can Wegovy injection stay out of the fridge?
Up to 28 days at 46–86°F before cap removal, according to the FDA-approved Wegovy label. After 28 days at room temperature, throw away the pen even if you haven't used it yet.
Is my Mounjaro pen still good if it was left out overnight?
Probably yes, as long as the temperature stayed below 86°F. A Mounjaro single-dose pen or vial can be kept at room temperature up to 86°F for up to 21 days. One overnight on the counter uses one of those 21 days. Mark the date on the carton and discard by day 21.
What happens if my Zepbound pen freezes?
Discard it. The Zepbound label says do not use if frozen. Freezing damages the molecular structure of tirzepatide, and the medication may have lost potency even if it looks normal after thawing. Call the dispensing pharmacy for a replacement.
Can frozen GLP-1 medication still be used after it thaws?
No, according to every injectable GLP-1 label in this class. Wegovy, Ozempic, Mounjaro, Zepbound, Trulicity, Saxenda, Victoza, Byetta, Bydureon BCise, and Adlyxin labels all instruct patients to discard the medication if it has been frozen, regardless of whether it has thawed and looks normal.
Does compounded semaglutide need to be refrigerated?
Usually yes, though the exact storage rule comes from the dispensing pharmacy's label, not from Ozempic or Wegovy guidance. Most compounded semaglutide is stored at 36–46°F (2–8°C). Always follow the pharmacy label and the Beyond-Use Date (BUD) printed on it.
How do I travel with Ozempic on a plane?
Pack it in your carry-on, not checked baggage. TSA permits medically necessary liquids in carry-on, including injectable medications and ice packs. Use an insulated medical pouch with cooling that won't freeze the pen. Declare the medication at the security checkpoint.
What temperature is too hot for Ozempic, Wegovy, Mounjaro, and Zepbound?
86°F (30°C) is the maximum room temperature for all four. Above 86°F, the medications are no longer guaranteed within validated storage conditions. Wegovy's label specifically says to throw away the pen if it has been exposed to temperatures above 86°F.
Can I store my GLP-1 pen in the fridge door?
We recommend against it. The door is the warmest part of the fridge and has the largest temperature swings every time you open the fridge. A pen stored in the door can drift outside the 36–46°F range. Use the middle shelf, away from the door and away from the back wall.
What does cloudy Ozempic mean?
Cloudy, discolored, or particle-containing Ozempic should not be injected. Healthy semaglutide is clear and colorless. Cloudiness is a sign that the medication has degraded, been contaminated, or been exposed to conditions outside its labeled storage range.
Can you put Ozempic back in the fridge after it's been out at room temperature?
Yes, for Ozempic specifically. Ozempic pens can cycle between fridge and room temperature within the 56-day in-use window. The clock does not reset — cumulative time at room temperature is what counts. Zepbound single-dose pens cannot be returned to the fridge after room-temperature storage, and Soliqua cannot be returned after first use.
What if my GLP-1 was delivered to a hot mailbox?
Treat it as a potential cold-chain failure. Photograph the package and contents. Touch-test the gel packs and inside liner. Visual-inspect the medication. FDA recommends patients not use any injectable GLP-1 drug that arrives warm or with insufficient refrigeration. Call the dispensing pharmacy to document the excursion and ask about their replacement policy.
Does Rybelsus need to be refrigerated?
No. Rybelsus (oral semaglutide) is stored at room temperature 68–77°F (20–25°C), with brief excursions allowed at 59–86°F. Keep the tablets in the original closed bottle until use. Don't transfer to a pill organizer.
How long does compounded tirzepatide last in the fridge?
The Beyond-Use Date (BUD) on the pharmacy label is authoritative. BUDs vary by compounding pharmacy based on formulation and stability testing. Refrigerated storage at 36–46°F is standard, but follow your specific label.
Can I use GLP-1 medication that's past the expiration date?
No. The expiration date is the manufacturer's guarantee of potency and sterility under labeled storage conditions. After that date, neither is guaranteed. Discard expired pens in a sharps container and get a replacement.
Should I take an extra dose if I think my GLP-1 lost potency from bad storage?
No. Don't compensate for suspected potency loss by taking extra medication. The right step is to discard the suspect pen, replace it, and resume your regular dosing schedule. Call your prescriber if you've missed a dose because of a storage problem.
Does Foundayo need to be refrigerated?
No. Foundayo (oral orforglipron) is stored at room temperature 68–77°F, with excursions allowed at 59–86°F. Foundayo is light-sensitive — keep the bottle closed and out of direct sun.
Are oral GLP-1 tablets safer to travel with than injections?
Generally easier, yes. Oral semaglutide tablets (Rybelsus, Ozempic tablets, Wegovy tablets) and Foundayo tablets don't need refrigeration, so you don't need a cooler. The main travel rule for tablets is to keep them in the original sealed bottle, not a pill organizer, to protect from moisture and (for Foundayo) light.
Sources & What We Verified
We built this guide by checking every storage rule against the FDA-approved labels on DailyMed (the NIH's official drug label database) and the manufacturer's published prescribing information.
- ·DailyMed labels for Ozempic, Wegovy injection, Wegovy HD, Wegovy tablets, Rybelsus, Ozempic tablets, Mounjaro single-dose pen and vial, Mounjaro KwikPen, Zepbound single-dose pen and vial, Zepbound KwikPen, Trulicity, Saxenda, Victoza, Byetta, Bydureon BCise, Adlyxin, Foundayo, Xultophy 100/3.6, and Soliqua 100/33 — accessed at dailymed.nlm.nih.gov
- ·FDA: FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss — fda.gov
- ·FDA: FDA Proposes to Exclude Semaglutide, Tirzepatide, and Liraglutide on 503B Bulks List (April 30, 2026) — fda.gov
- ·TSA medication and liquid screening guidance — tsa.gov
- ·FDA sharps disposal guidance — fda.gov
- ·Mayo Clinic patient guidance on semaglutide visual inspection
- ·Novo Nordisk Scientific Exchange storage and stability page — novonordiskmedical.com
- ·Eli Lilly prescribing information — pi.lilly.com
- ·National Highway Traffic Safety Administration — vehicle temperature data
- ·National Weather Service — vehicle interior temperature guidance
Update log
| Date | What changed |
|---|---|
| Initial publication. Verified storage rules for all 21 products. Added FDA's April 30, 2026 503B bulks list proposal and current FDA adverse-event counts (605 compounded semaglutide, 545 compounded tirzepatide as of July 31, 2025). |
Next scheduled review: August 2026 — earlier if FDA updates GLP-1 prescribing labels or issues new compounding guidance.