Tirzepatide Side Effects: What's Normal, What's Serious, and What to Do
Disclosure: Some links on this site are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you. Thank you for supporting our site.·For informational purposes only—not medical advice.
Sources: FDA Prescribing Information (Zepbound, Mounjaro), SURPASS & SURMOUNT Clinical Trials, PubMed

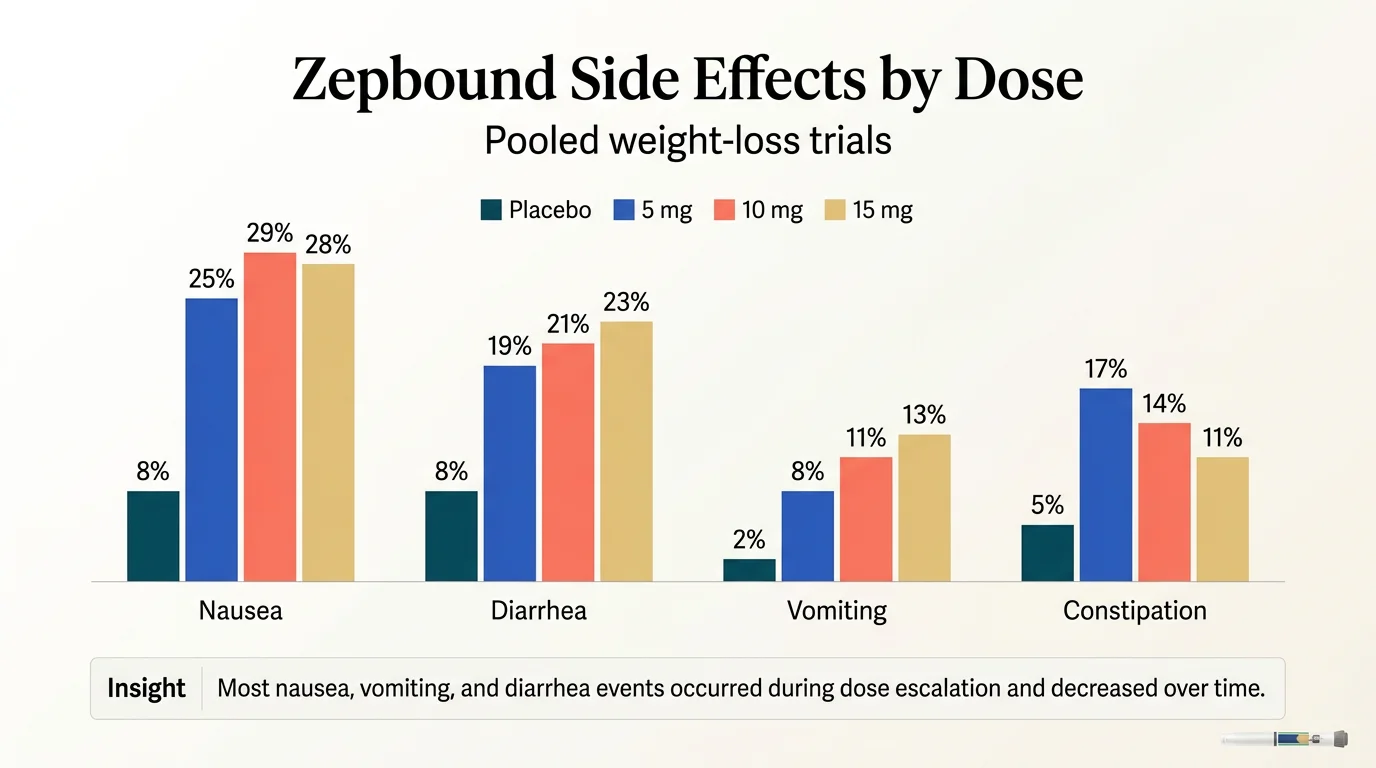
Most tirzepatide side effects are stomach-related, temporary, and worst during dose increases. In pooled Zepbound weight-loss trials, nausea occurred in 25–29% of patients, diarrhea in 19–23%, vomiting in 8–13%, and constipation in 11–17%, depending on dose. Most nausea, vomiting, and diarrhea events occurred during dose escalation and decreased over time. Between 4.8% and 6.7% of participants discontinued treatment due to adverse reactions. Serious reactions like pancreatitis and severe allergic reactions are rare but documented.
The thyroid cancer boxed warning is based on animal studies. A 2025 meta-analysis of 13 randomized trials (13,761 participants) found no increased overall or specific cancer risk over 26 to 72 weeks — though that does not answer very-long-term risk. And as of January 2026, the FDA completed a comprehensive review of 91 clinical trials (107,910 patients) and formally concluded there is no increased risk of suicidal thoughts or behavior with GLP-1 medications — and requested removal of that warning from affected labels.
Below, we break down every tirzepatide side effect with clinical trial percentages, a week-by-week timeline of what to expect, and a clear framework for when to call your doctor versus when to ride it out.
Affiliate disclosure: Some links on this page are affiliate links. This doesn't influence our analysis — everything here is sourced from FDA labels and peer-reviewed research.
Considering tirzepatide for weight loss? Take our 60-second quiz to see if you're a candidate — and find a provider matched to your needs.
Check Your EligibilityIn This Guide
How Common Are Tirzepatide Side Effects? (Clinical Trial Data by Dose)
Before the deep dives — here's what the actual clinical trials found. Not Reddit anecdotes, not someone's TikTok horror story. These numbers come from the SURPASS trials (type 2 diabetes) and SURMOUNT trials (weight loss), which together enrolled over 15,000 participants and were submitted to the FDA for approval.
Tirzepatide Side Effects: Frequency Table
| Side Effect | Zepbound 5mg | Zepbound 10mg | Zepbound 15mg | Placebo | When It Typically Starts |
|---|---|---|---|---|---|
| Nausea | 25% | 29% | 28% | 8% | Most events occur during dose escalation and decrease over time |
| Diarrhea | 19% | 21% | 23% | 8% | Most events occur during dose escalation and decrease over time |
| Constipation | 17% | 14% | 11% | 5% | May develop as gastric emptying slows |
| Vomiting | 8% | 11% | 13% | 2% | Most events occur during dose escalation and decrease over time |
| Abdominal pain | 9% | 9% | 10% | 5% | Variable; often tied to meals |
| Dyspepsia / indigestion | 9% | 9% | 10% | 4% | Variable; improves with smaller meals |
| Injection site reactions | 6% | 8% | 8% | 2% | Day of injection; usually resolves in days |
| Fatigue | 5% | 6% | 7% | 3% | Variable; rule out under-eating or dehydration |
| Hypersensitivity reactions | 5% | 5% | 5% | 3% | Variable; mostly skin reactions (rash, itching) |
| Eructation (burping) | 4% | 5% | 5% | 1% | Often during dose escalation |
| Hair loss | 5% | 4% | 5% | 1% | Months 3–6; associated with weight reduction; more common in women |
| GERD / acid reflux | 4% | 4% | 5% | 2% | Variable; manageable with dietary changes |
| Dizziness | 4% | 5% | 4% | 2% | Variable; check hydration and blood pressure |
Source: Zepbound FDA Prescribing Information, Table 1 — pooled data from Study 1 and Study 2 (weight reduction trials in adults with obesity or overweight). Mounjaro label rates differ slightly due to different patient population (type 2 diabetes).
A few things worth noting:
Side effect rates were generally higher in the Zepbound weight-loss trials than in the Mounjaro diabetes trials, reflecting different patient populations and dosing protocols. In the Zepbound trials, gastrointestinal adverse reactions occurred in about 56% of patients across all dose groups compared to 30% on placebo. The majority of nausea, vomiting, and diarrhea events occurred during dose escalation and decreased over time.
The numbers above are from FDA-approved brand-name Zepbound. Compounded tirzepatide products are not FDA-approved, and FDA does not review them for safety, effectiveness, or quality before they are marketed. FDA guidance states that compounded drugs should only be used when a patient's medical needs cannot be met by an FDA-approved drug.
Hair loss was reported more frequently in women (7.1%) than men (0.5%) among Zepbound-treated patients, and was associated with weight reduction.

When Do Tirzepatide Side Effects Start — and When Do They Stop?
This is the question that matters most when you're living it. Every other page dumps side effects into categories. But you don't experience this drug in categories. You experience it in weeks.
The Tirzepatide Side Effect Timeline
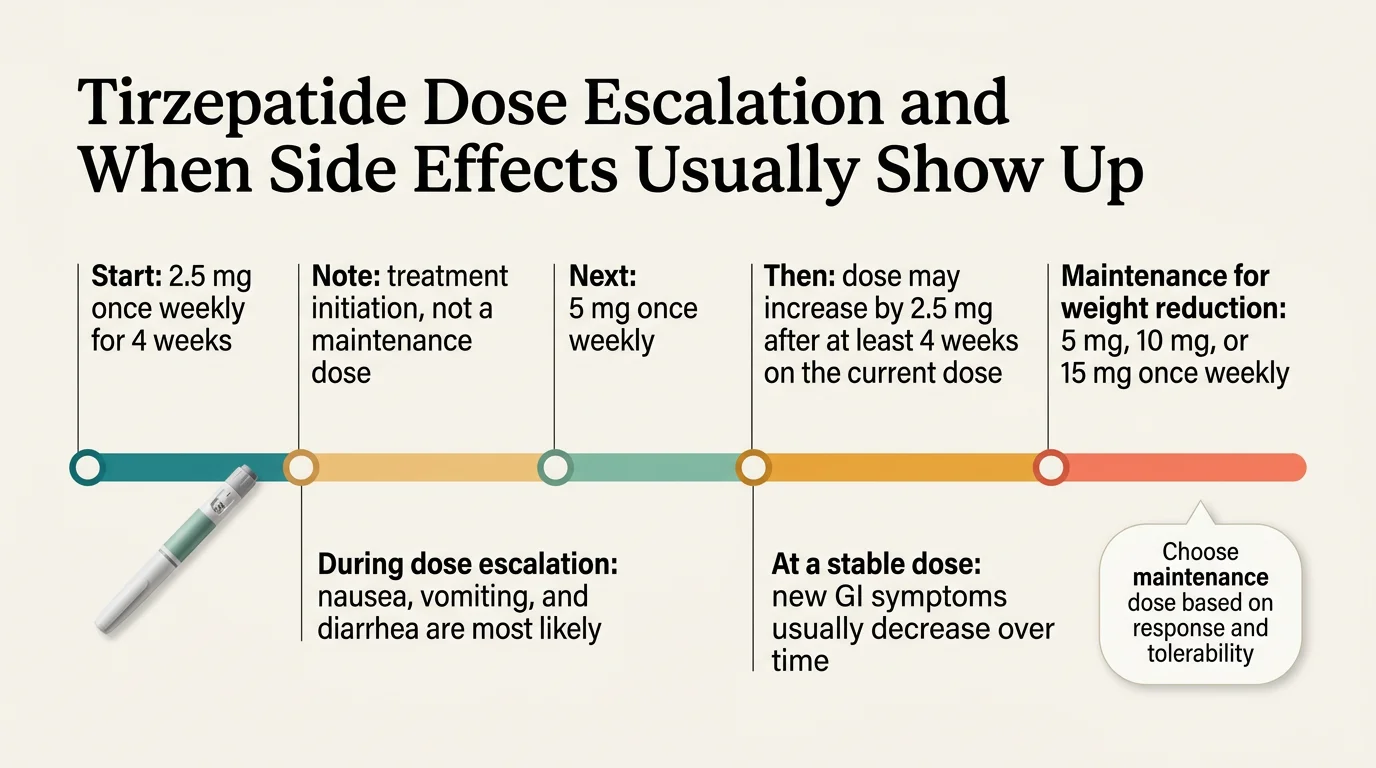
Week 1–2 (Starting dose: 2.5mg)
Your body is meeting this medication for the first time. The recommended starting dose is 2.5mg once weekly for 4 weeks — this is for treatment initiation and is not an approved maintenance dose. The dose escalation schedule exists specifically to reduce the risk of GI adverse reactions.
What's common: Mild to moderate nausea, especially in the first days after your injection. Some decreased appetite. Maybe a queasy feeling that makes you push your plate away earlier than usual. Possible injection site redness that fades in a day or two.
What's normal to feel: A general sense of "something's different." Your relationship with food starts shifting. You might notice you stop thinking about your next meal.
What's not normal: Severe vomiting where you can't keep fluids down. Intense abdominal pain. These need medical attention — don't wait.
Week 3–4 (Still at 2.5mg)
Your body is adjusting. Nausea should be easing. The appetite suppression starts feeling less foreign and more like your new baseline. Constipation may show up as gastric emptying slows. Stay hydrated.
Week 5–8 (First dose increase to 5mg)
Here's the pattern you'll see repeated: each dose increase is like a mini version of Week 1. GI symptoms may briefly return or worsen. Then they settle again, usually faster than the first time because your body already knows the drug.
Sulfur burps may make their debut. Fatigue is possible. Some people report feeling wiped out for a day or two after injecting.
Month 2–4 (Escalating through 7.5mg, 10mg)
Each dose step follows the same rhythm — temporary flare, then adjustment. By now, you know the pattern and you know what to expect. Weight loss is becoming visible.
This is also when hair shedding can start. It's not the drug causing it directly — it's the rapid weight loss triggering a normal hair cycle response called telogen effluvium. We cover this in detail below.
Post-marketing case reports have also documented body aches and headaches during this phase, typically peaking 48–72 hours after injection and resolving on their own.
Month 4–6 (Reaching target dose: 10–15mg)
For most people, GI side effects are significantly better or gone entirely. The data backs this up — clinical trials consistently show that the rate of new GI adverse events decreases over time at a stable dose.
Weight loss is progressing. Hair shedding may be most noticeable. Energy levels are usually stable if you're eating and hydrating adequately.
Month 6+ (Maintenance)
If you've made it here, the hardest part is behind you. Most people report that side effects are either resolved or background-level. The ongoing monitoring priorities shift to gallbladder health, thyroid awareness, and staying on top of your protein and nutrient intake.
After stopping
Side effects resolve after discontinuation. However, appetite typically returns to pre-treatment levels, and weight regain is common. In SURMOUNT-4, participants who switched from tirzepatide to placebo after 36 weeks regained a significant portion of lost weight over the following year. Those who continued on tirzepatide lost an additional 5.5% from their Week 36 weight.
This isn't a "side effect" — it's the natural consequence of removing the pharmacological appetite suppression. If you're planning to stop, work with your provider on a transition plan.
Which Tirzepatide Side Effects Are Serious?
We built this section as a triage framework — because when you're sitting there at 11pm wondering if what you're feeling is an emergency, you need clarity, not a list of medical terms.
Red: Go to the ER or Call 911
These are rare. But if they happen, act immediately.
Severe allergic reaction (anaphylaxis):
Swelling of face, lips, tongue, or throat. Difficulty breathing or swallowing. Rapid heartbeat. Hives spreading across your body. Dizziness or fainting. This is a medical emergency. Call 911.
Signs of pancreatitis:
Severe, persistent pain in your upper abdomen that may radiate to your back. Pain that worsens after eating. Nausea and vomiting that won't stop. Fever. Rapid pulse. Stop tirzepatide immediately and go to the emergency room. Pancreatitis has been reported in clinical trials. Tirzepatide has not been studied in people with a history of pancreatitis.
Severe hypoglycemia (primarily if combined with insulin or sulfonylureas):
Confusion, loss of consciousness, seizure, inability to function. If someone is unconscious, call 911. If conscious, give fast-acting glucose immediately.
Yellow: Contact Your Doctor the Same Day
These aren't emergencies, but they need professional evaluation soon.
You can't keep fluids down for 12+ hours. Persistent vomiting and diarrhea can cause dehydration, which can lead to kidney injury. The FDA specifically warns about this. If you're not urinating, have dark urine, feel lightheaded, or have a rapid heartbeat — your doctor needs to know today.
Upper right abdominal pain, especially after eating fatty food. This could signal gallbladder problems — gallstones or cholecystitis. Add fever, nausea, or yellowing of your skin/eyes and it's more urgent.
New lump or swelling in your neck. Persistent hoarseness. Trouble swallowing. These are the thyroid warning signs listed on the label. They're rare, but they need evaluation.
Mood changes that feel different from your baseline. New depression, worsening anxiety, thoughts of self-harm. While the FDA's 2026 review found no increased risk with GLP-1 medications, any significant mental health change deserves attention regardless of cause.
Severe or worsening constipation with bloating and pain. Could indicate gastroparesis or, rarely, intestinal obstruction — both identified in post-marketing surveillance.
Green: Mention at Your Next Appointment
These are common, expected, and usually self-resolving. Track them, but don't panic.
- Mild to moderate nausea that's trending better week to week
- Constipation manageable with OTC remedies
- Sulfur burps (unpleasant but not dangerous)
- Injection site redness that resolves in a few days
- Fatigue during dose escalation
- Decreased appetite (this is the medication working)
- Mild hair shedding starting around month 3–4
- Feeling full faster at meals
- Brief headache or body aches after injection
Common Tirzepatide Side Effects: What They Feel Like and What to Do
Nausea and Vomiting
This is the headline side effect, and for good reason. In pooled Zepbound weight-loss trials, nausea occurred in 25–29% of patients depending on dose (compared to 8% on placebo). Vomiting affected 8–13%.
Why it happens: Tirzepatide slows gastric emptying — food stays in your stomach longer. Your brain's satiety signals fire earlier and harder. The result is that familiar queasy feeling, often worst 24–48 hours post-injection.
What actually helps:
- Eat smaller meals, more often. Five small plates beats three large ones during dose escalation.
- Avoid fatty, greasy, and spicy foods — they're harder to digest when your stomach is moving slowly.
- Ginger tea, ginger chews, or ginger capsules have real anti-nausea evidence behind them.
- Stay upright for at least 30 minutes after eating.
- Ask your prescriber about ondansetron (Zofran) — it's commonly prescribed off-label for GLP-1-related nausea.
When it crosses the line: If you can't keep any fluids down for more than 12 hours, that's a yellow flag. Persistent vomiting can cause dehydration and kidney problems. Contact your provider.
Diarrhea
Up to 23% in the Zepbound weight-loss trials (compared to 8% on placebo). It tends to hit during the first couple of weeks and during dose increases, then settles.
What helps: Stay hydrated — oral rehydration solutions or electrolyte mixes are better than plain water when you're losing fluids. Avoid dairy and high-fiber foods temporarily. The BRAT diet (bananas, rice, applesauce, toast) works. OTC loperamide (Imodium) is fine short-term.
When to worry: Blood in stool. Severe dehydration signs. Diarrhea lasting more than a few days without improving. These need medical attention.
Constipation
The flip side of slowed gastric emptying. Affects 11–17% of Zepbound patients depending on dose (interestingly, it was more common at 5mg than 15mg in the trials).
What helps: Gradually increase fiber (emphasis on gradually — too much too fast makes it worse). Drink at least 64 ounces of water daily. Psyllium husk (Metamucil) or polyethylene glycol (MiraLAX) are good OTC options. Move your body — even walking helps.
When to worry: No bowel movement for 3+ days with significant pain and bloating. Post-marketing reports have identified severe constipation and ileus as rare but documented events.
Sulfur Burps
This is the one everyone Googles but nobody's doctor warned them about. The clinical trials list eructation (burping) at 4–5% across Zepbound doses. Reddit and patient forums tell a much bigger story — sulfur burps (rotten egg smell and taste) are one of the most discussed tirzepatide experiences online.
Why it happens: When your stomach empties more slowly, food can ferment. That fermentation produces hydrogen sulfide gas. Hence the sulfur.
What helps: Reduce high-sulfur foods (eggs, cruciferous vegetables like broccoli and cauliflower, garlic). Simethicone (Gas-X) may help. Peppermint tea. Probiotics. Some people find that bismuth subsalicylate (Pepto-Bismol) helps short-term.
This is annoying, sometimes embarrassing, but not dangerous. It typically improves as your body adjusts.
Hair Loss
Listed in the Zepbound label at 4–5% across dose groups and reported more frequently in women (7.1% vs. 0.5% in men). This one frightens people, and understandably so.
The important distinction: Hair loss in the Zepbound label was associated with weight reduction. Rapid weight loss — from any cause — can push hair follicles from the growth phase into the resting phase prematurely. This is called telogen effluvium, and it typically shows up 3–6 months after significant weight loss begins. The label does not establish a direct pharmacological mechanism beyond the weight-loss association.
The reassuring part: It's temporary. Hair regrowth occurs once your weight stabilizes. Most people see improvement within 6–12 months.
What helps: Prioritize protein — aim for at least 0.7 grams per pound of lean body mass daily. A quality multivitamin with biotin, iron, and zinc supports hair health. Don't crash-diet on top of the medication's appetite suppression. Your body needs adequate nutrition even as you're losing weight. See our guide to the best supplements to take with GLP-1 medications.
Fatigue
Listed in the Zepbound label as a common adverse reaction at 5–7% across dose groups (compared to 3% on placebo). Generally mild.
Possible causes: You may be eating too little. Dehydration. Blood sugar fluctuations (especially if you're diabetic and on other medications). Your body is also doing a lot of metabolic work — processing fat stores, adjusting hormones, recalibrating hunger signals.
What helps: Make sure you're actually eating enough. The appetite suppression can be powerful, and some people inadvertently go too low on calories. Hydrate. Prioritize sleep. If fatigue is severe or persistent, see your provider — there may be another cause.
Injection Site Reactions
Redness, mild swelling, itching at the injection site. Affects 6–8% of Zepbound patients. Almost always mild and self-resolving within a few days. Injection site reactions were more frequent in patients who developed anti-tirzepatide antibodies (11.3%) compared to those who did not (1%).
Tips: Rotate injection sites between your abdomen, thigh, and upper arm. Let the alcohol swab dry completely before injecting. Apply ice before or after if you're sensitive. If you notice spreading redness, warmth, or signs of infection, contact your provider.
Headache and Body Aches
These are not listed among the common adverse reactions in the current Zepbound or Mounjaro labels. However, a small 2025 case series (published in PMC) described headache, muscle pain, and palpitations after tirzepatide initiation. Frequency is unknown from this limited data. Some patients in those reports noted symptoms beginning 24 hours post-injection, peaking at 48–72 hours, and resolving spontaneously.
What helps: Hydration. Adequate nutrition. OTC pain relief. One case report noted that thigh injections were better tolerated than abdominal injections for this particular patient — worth trying if this is your experience.
Acid Reflux / GERD
Affects 4–5% of Zepbound patients. Slowed gastric emptying plus increased stomach pressure equals that burning sensation.
What helps: Don't lie down for 2–3 hours after eating. Smaller meals. Avoid trigger foods (citrus, tomatoes, coffee, chocolate, alcohol). Elevate the head of your bed. OTC famotidine (Pepcid) or antacids as needed.
When to worry: Chest pain (always rule out cardiac causes). Difficulty swallowing. Persistent symptoms despite management.
Does Tirzepatide Cause Cancer or Thyroid Problems?
This is probably why you're here. You saw the boxed warning. You Googled it. Now you want the truth. Let's be direct.
Why the boxed warning exists
In animal studies, tirzepatide caused dose-dependent thyroid C-cell tumors in rats. That's documented. The FDA's prescribing information states clearly: "It is unknown whether tirzepatide causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans." That unknown is why the boxed warning — the most serious kind of FDA warning — is there.
Why the animal finding may not apply to humans
Rat thyroid biology is meaningfully different from human thyroid biology. Rats have significantly more calcitonin-producing C-cells than humans. The mechanism by which GLP-1 stimulates C-cell proliferation in rodents has not been demonstrated to occur in human thyroid tissue. This doesn't mean the risk is zero. It means the animal model may not translate.
What human data actually show
A 2025 systematic review and meta-analysis (Kamrul-Hasan et al., published in Endocrinology and Metabolism) pooled data from 13 randomized controlled trials with 13,761 participants followed for 26–72 weeks. The findings: tirzepatide did not increase overall cancer risk or any specific cancer risk compared to controls. No cases of papillary thyroid carcinoma were reported — even though higher tirzepatide doses produced modest calcitonin elevations.
A large retrospective cohort study (presented at the Endocrine Society, 2025) analyzed over 283,000 tirzepatide patients matched against controls using the TriNetX network. Tirzepatide patients actually showed a lower incidence of malignant thyroid cancer (Relative Risk 0.348, p<0.001). Lower, not higher.
An FAERS database analysis (published in Endocrinology Diabetes and Metabolism, 2025) found tirzepatide had a reporting odds ratio of 2.09 for thyroid cancer reports. Context matters here: FAERS data reflects voluntary reports and cannot establish causation. Tirzepatide's signal was also substantially lower than liraglutide (15.59) and semaglutide (7.61), both of which have been on the market much longer.
The bottom line on cancer
Randomized-trial evidence has not shown increased overall or specific cancer risk over 26 to 72 weeks. That does not rule out very-long-term risk — the studies simply haven't run long enough to answer that question. The boxed warning remains because tirzepatide caused thyroid C-cell tumors in rats, and human relevance remains unknown.
Who should not take tirzepatide because of thyroid risk
This is non-negotiable. If you have a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2), tirzepatide is contraindicated. Do not take it.
What to watch for
Report these to your doctor immediately: a new lump or swelling in your neck, hoarseness that won't go away, difficulty swallowing, or persistent shortness of breath.
Can Tirzepatide Cause Anxiety, Depression, or Suicidal Thoughts?
This question has caused a lot of fear — partly because the media ran with it, and partly because the original Zepbound label included a precautionary warning about suicidal ideation. Let's look at where the evidence landed.
The FDA's final word (January 2026)
On January 13, 2026, the FDA issued a Drug Safety Communication requesting the removal of the suicidal behavior and ideation warning from GLP-1 RA medications — including Zepbound (tirzepatide), Wegovy (semaglutide), and Saxenda (liraglutide). The affected labels are expected to be updated to reflect this change.
This wasn't a casual decision. The FDA conducted a meta-analysis of 91 placebo-controlled GLP-1 RA trials encompassing 107,910 patients (60,338 on a GLP-1 RA, 47,572 on placebo). The results: no increased risk for suicidal ideation, suicidal behavior, or other psychiatric adverse events including anxiety, depression, irritability, or psychosis.
They also ran a retrospective cohort study through the FDA Sentinel System covering 2.2 million patients. Same conclusion — no increased risk.
Their statement: "FDA's evaluation did not identify an increased risk of suicidal ideation or behavior with the use of GLP-1 RA medications."
Why older pages may sound scarier
If you're reading a side effects page that was last updated before January 2026 — and most were — it may still reference the suicidal ideation warning as a current concern. It's not misinformation; it's just outdated. The FDA has now formally concluded its review and moved to remove that language.
What's real about mood and emotional changes
Even though the psychiatric drug effect concern has been addressed, people do report emotional shifts during GLP-1 treatment. Some possible reasons:
- Rapid body changes can affect self-image and identity.
- Your relationship with food is fundamentally changing — and food has emotional dimensions for most people.
- Caloric restriction itself can impact mood, energy, and sleep.
- Positive changes — better sleep, more energy, reduced inflammation — can also improve mental health for many people.
A 2026 post-hoc analysis of the SURMOUNT trials (Wadden et al., published in Obesity) formally assessed depressive symptoms and suicidal ideation using validated measures. Tirzepatide-treated patients had equal or lower rates compared to placebo across all measures.
A real-world study of 226,060 patients (published in Frontiers in Psychiatry, October 2025) found that tirzepatide patients had fewer suicidal ideation/attempt events than matched controls.
What to do if your mood changes
If you experience persistent low mood, loss of interest in things you usually enjoy, worsening anxiety, or any thoughts of self-harm — tell your provider. This applies regardless of what medication you're taking or not taking. Your mental health matters, full stop.
Tirzepatide Side Effects in Women

Hair loss
Hair loss was reported more frequently in women in the Zepbound clinical trials. As covered above, this is driven by rapid weight loss, not a gender-specific drug reaction. Women tend to notice and report it more because hair shedding is more visible and distressing with longer hairstyles.
Birth control interaction
This one is important and often overlooked. Tirzepatide can reduce the effectiveness of oral contraceptives. Why? Because it slows gastric emptying, which can affect how oral medications are absorbed. The FDA prescribing information recommends switching to a non-oral form of contraception (IUD, implant, patch, ring, or condoms) for 4 weeks after starting tirzepatide and 4 weeks after each dose increase. If you're relying on the pill, talk to your prescriber about this before your first injection.
Pregnancy
Tirzepatide may cause fetal harm based on animal studies. The current Zepbound label states: discontinue when pregnancy is recognized. Weight loss offers no benefit during pregnancy. If you're planning to conceive, discuss the timing of discontinuation with your provider well in advance.
Breastfeeding
The current Zepbound label states there are no data on the presence of tirzepatide in human milk, the effects on the breastfed infant, or the effects on milk production. The decision to use tirzepatide while breastfeeding should involve weighing the benefits of breastfeeding and the mother's clinical need for the medication against any potential risks.
Related: Best GLP-1 Programs for Women
Tirzepatide vs. Semaglutide: How Do Side Effects Compare?
This is one of the most common follow-up searches, and it deserves an honest answer rather than a sales pitch for either drug.
Label-to-Label Comparison Table
| Side Effect | Tirzepatide (Zepbound) | Semaglutide (Wegovy) |
|---|---|---|
| Nausea | 25–29% | 44% |
| Diarrhea | 19–23% | 30% |
| Vomiting | 8–13% | 24% |
| Constipation | 11–17% | 24% |
| Hair loss | 4–5% | 3% |
| Injection site reactions | 6–8% | 3–6% |
Sources: Zepbound FDA Prescribing Information (pooled weight-loss trials) and Wegovy FDA Prescribing Information (STEP trials). These are different trials with different designs — not a head-to-head comparison.
The key takeaway
The GI side effect profiles are broadly similar in the sense that both drugs affect the same pathways, but the label rates look quite different because the trials were designed differently. In the actual head-to-head SURMOUNT-5 trial, tirzepatide achieved greater weight loss than semaglutide (-20.2% vs. -13.7% at week 72). Serious adverse events occurred in 4.8% with tirzepatide and 3.5% with semaglutide; GI adverse events leading to discontinuation were 2.7% with tirzepatide vs. 5.6% with semaglutide.
For most people, the additional efficacy of tirzepatide is worth the slightly higher side effect burden during dose escalation. But that's an individual decision — one that depends on your tolerance, your medical history, and your goals. If semaglutide-level results are sufficient for your needs and you want the lowest possible side effect profile, that's a valid choice.
Neither drug is "better" universally. The best one is the one you'll tolerate and stay on long enough to see results.
Can Tirzepatide Affect Your Pancreas, Gallbladder, Kidneys, Heart, or Eyes?
These organ-specific fears come up repeatedly in searches. Here's what the evidence says, organ by organ.
Pancreatitis
Acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with GLP-1 receptor agonists or tirzepatide. In the pooled Zepbound weight-reduction trials, 0.2% of treated patients had adjudication-confirmed acute pancreatitis, comparable to the 0.2% rate in placebo patients. Tirzepatide has never been studied in people with a history of pancreatitis.
Warning signs: Severe, persistent upper abdominal pain — often radiating to the back. Worse after eating. Accompanied by nausea, vomiting, fever. Action: Stop the medication. Get emergency medical care. Do not restart if pancreatitis is confirmed.
A note on lipase and amylase: Tirzepatide can cause asymptomatic elevations of these pancreatic enzymes. Per StatPearls and the FDA label, monitoring these levels without symptoms has not shown clinical utility — elevated enzymes alone don't mean pancreatitis.
Gallbladder Disease
Rapid weight loss — from any method — increases the risk of gallstones. Bile composition changes when you lose weight quickly, and gallstones can form. This isn't unique to tirzepatide; it happens with bariatric surgery, very low-calorie diets, and other GLP-1 medications too.
Warning signs: Pain in your upper right abdomen, especially after fatty meals. Nausea, fever, chills, yellowing of skin or eyes (jaundice).
Kidney Injury
This isn't a direct effect of tirzepatide on your kidneys. It's a consequence of dehydration from vomiting and diarrhea. If you're losing fluids and not replacing them, your kidneys suffer.
Prevention: Hydration is non-negotiable. If you're experiencing persistent GI symptoms, increase your fluid intake proactively. Report severe, ongoing vomiting or diarrhea to your provider.
The FDA label specifically states: "Monitor renal function in patients reporting adverse reactions that could lead to volume depletion."
Heart Rate
The Zepbound label notes dose-dependent increases in heart rate — treatment resulted in a mean increase of 1 to 3 beats per minute compared to no increase in placebo-treated patients. For most people, this is clinically insignificant. However, if you have a pre-existing heart condition or notice persistent palpitations, discuss this with your cardiologist. Post-marketing case reports (PMC, 2025) have documented palpitations in some patients following tirzepatide initiation.
Vision Changes (Diabetic Retinopathy)
This applies primarily to people with type 2 diabetes. Rapid improvements in blood sugar can temporarily worsen diabetic retinopathy. This has been observed with insulin and other diabetes medications too — it's not unique to tirzepatide. If you have diabetic retinopathy, your ophthalmologist should be involved in monitoring your eye health during tirzepatide treatment.
What Happens After You Stop Tirzepatide?
This is one of the most searched follow-up questions — and the answer is important to understand before you start, not just when you're thinking about quitting.
Side effects resolve
The good news: once you stop tirzepatide, GI side effects fade relatively quickly. Nausea, constipation, and the other stomach-related issues are tied to the drug's active presence in your body. Tirzepatide has an elimination half-life of approximately 5–6 days, so it takes roughly 25–30 days after your last dose to substantially clear your system. Side effects should diminish progressively during that window.
Appetite returns to baseline
This is the part people aren't prepared for. Tirzepatide suppresses appetite through multiple hormonal pathways. When you remove the drug, those pathways revert. Hunger returns — often to pre-treatment levels. For many people, this is the most challenging aspect of stopping.
Weight regain is common
The SURMOUNT-4 trial provides the clearest data here. After 36 weeks of open-label tirzepatide treatment (during which patients lost an average of 20.9% of body weight), patients randomized to placebo experienced substantial weight regain of 14.0% from their Week 36 weight over the following 52 weeks. Those who continued on tirzepatide lost an additional 5.5% from their Week 36 weight.
This isn't a failure of willpower. It's biology. Obesity involves hormonal and neurological mechanisms that tirzepatide modifies while you're taking it. When the modification stops, the underlying biology reasserts itself.
Blood sugar returns to pre-treatment levels
For people with type 2 diabetes, HbA1c and fasting glucose typically rise back toward pre-treatment levels after stopping. This makes sense — tirzepatide improves insulin sensitivity and glucose-dependent insulin secretion while you're on it, but doesn't cure the underlying metabolic condition.
When stopping makes sense
There are valid reasons to stop: intolerable side effects that haven't improved despite dose adjustment, pregnancy planning (stop at least 2 months before conceiving), reaching a weight goal with a solid maintenance plan in place, or cost/access issues. The key is to stop intentionally with a plan rather than abruptly without one.
If you're considering stopping, talk to your provider about transitioning to a maintenance strategy that includes nutrition counseling, exercise programming, and possibly a different medication if needed. See also: How to Prevent Muscle Loss on GLP-1s.
What Are the Long-Term Side Effects of Tirzepatide?
We have to be honest here about what we know and what we don't.
What 3-year data show
The longest published data comes from the SURMOUNT-1 extension study (3 years / 176 weeks). The findings: GI side effects were most common early in treatment and during dose escalation. They decreased substantially over time. No new safety signals emerged with extended use. This is reassuring.
What hasn't been proven yet
Tirzepatide was FDA-approved in 2022. That means the longest anyone has been studied on this medication in a clinical trial is about 3.5 years. True long-term data — 5, 10, 15 years — simply doesn't exist yet.
The honest framing
"No new safety signals after 3 years" is genuinely encouraging. But "no signal seen yet" is not the same as "risk proven absent." That distinction matters. We'll update this section as longer-term data becomes available.
Who Should Not Take Tirzepatide
Do not take tirzepatide if you have:
- A personal or family history of medullary thyroid carcinoma (MTC)
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- A known serious allergic reaction to tirzepatide or any of its components
Talk to your doctor before starting if you have:
- Type 1 diabetes (tirzepatide is not approved or studied for T1DM)
- A history of pancreatitis (tirzepatide has not been studied in this population; discontinue if pancreatitis is suspected)
- Severe gastroparesis or significant gastric motility issues
- Active gallbladder disease
- Diabetic retinopathy (rapid glucose improvement can temporarily worsen eye disease)
- Current pregnancy, breastfeeding, or plans to conceive within 2 months
- You're scheduled for surgery (tirzepatide's delayed gastric emptying can affect anesthesia safety — your surgical team needs to know)
Medication combinations that need attention:
- Insulin or sulfonylureas: Increased hypoglycemia risk. Dose adjustment is usually needed.
- Oral contraceptives: Reduced effectiveness. Switch to non-oral contraception for 4 weeks after starting and after each dose increase.
- Oral medications in general: Slowed gastric emptying can affect absorption timing. Discuss all your medications with your prescriber.
How to Reduce Tirzepatide Side Effects: The Practical Playbook
Generic advice is everywhere. Here's what actually moves the needle.
Before Your First Injection
- Stock your kitchen: Ginger tea or ginger chews. An electrolyte mix (Liquid IV, LMNT, Drip Drop — whatever you prefer). Bland foods you'll actually eat: crackers, rice, bananas, broth. A fiber supplement (psyllium husk). OTC anti-nausea if your prescriber approves it.
- Set expectations: The first 2–4 weeks are the adjustment period. It gets better. Knowing this in advance helps you mentally.
- Schedule smart: Many people inject on Friday evening so the peak side effect window falls on the weekend. Experiment with timing.
During Dose Escalation
- Eat smaller, more often. Imagine your stomach just got smaller — because functionally, it has. Five small meals beat three big ones.
- Protein first, always. When your appetite is suppressed, every bite counts more. Prioritize protein (chicken, fish, eggs, Greek yogurt, protein shakes) before anything else. Aim for at least 0.7g per pound of lean body mass to protect muscle mass.
- Hydrate aggressively. 64 ounces minimum. More if you're experiencing diarrhea or vomiting. Keep a water bottle visible — when appetite is low, thirst signals can also diminish.
- Rotate injection sites. Abdomen, thigh, upper arm. Some patients report fewer side effects with thigh injections. Find what works for your body.
- Inject at the same time each week. Consistency matters for steady drug levels. Many people find evening injection helps them sleep through the peak nausea window.
If Side Effects Aren't Improving
Talk to your provider about staying at your current dose longer than the standard 4 weeks before escalating. There's no rule that says you must increase on schedule. Slower titration often means better tolerance.
Don't white-knuckle it. If side effects are significantly affecting your daily life, there are options — dose reduction, timing changes, anti-nausea medication, or even switching to a different GLP-1 if needed. A good provider will work with you on this, not just tell you to push through.
Should You Keep Taking Tirzepatide, Lower the Dose, or Stop?
This is the decision question that sits under every other question on this page. Here's a framework.
Continue and monitor if:
Your side effects are mild to moderate, trending better over time, and manageable with the strategies above. This is the majority of people. The body adjusts. The results come.
Pause and message your clinician if:
Side effects are moderate and not improving after 3–4 weeks at the same dose. You're unable to meet minimum nutrition or hydration needs. You notice anything from the "Yellow" list in our triage section above. You're emotionally struggling and need support.
Stop and seek urgent evaluation if:
You experience anything from the "Red" list. Severe allergic reaction. Signs of pancreatitis. Symptoms of intestinal obstruction. Suicidal ideation (call 988 or go to your nearest ER).
The goal isn't to endure the maximum possible discomfort. The goal is to find the dose and pace that gets you results while keeping your quality of life intact. That's what a good provider helps you calibrate.
Tirzepatide Side Effects by Dose: Does Higher Always Mean Worse?
One of the most practical questions people have — and one that clinical trial data answers clearly.
What the SURMOUNT-1 Trial Found
| Side Effect | 5mg | 10mg | 15mg | Placebo |
|---|---|---|---|---|
| Nausea | 24.6% | 33.3% | 31.0% | 9.5% |
| Diarrhea | 18.7% | 21.2% | 23.0% | 7.3% |
| Vomiting | 5.2% | 9.1% | 12.8% | 1.7% |
| Constipation | 5.9% | 5.9% | 6.3% | 3.0% |
| Discontinued due to AE | 4.3% | 7.1% | 6.2% | 2.6% |
Source: SURMOUNT-1 (Jastreboff et al., NEJM, 2022)
Side effects are dose-dependent — but the relationship isn't perfectly linear. Nausea, for example, was actually highest at 10mg, not 15mg. Vomiting climbed steadily with dose. Constipation was fairly flat across all doses.
The most important insight: the biggest side effect spike happens with your very first exposure to the drug, not necessarily at the highest dose. By the time you reach 15mg, your body has been adjusting for months. Each escalation step is typically shorter and milder than the one before.
This is why the slow titration protocol exists. Starting at 2.5mg and spending at least 4 weeks at each dose isn't bureaucratic caution — it's the strategy that makes higher doses tolerable.
What this means for you
If you're currently at a lower dose and wondering whether going higher will make things unbearable — for most people, it won't. The adjustment window shortens. Your body already knows the drug. And if a particular dose is giving you trouble, you can stay there longer or step back down. Work with your provider on the pace that fits your body.
Tirzepatide and Alcohol: What You Should Know
No formal drug-alcohol interaction is listed in the prescribing information. But that doesn't mean alcohol is a non-issue on tirzepatide. Here's the practical reality.
Nausea amplification. If you're already dealing with GI side effects, alcohol will almost certainly make them worse. It irritates the stomach lining, slows healing, and increases acid production — none of which help when your stomach is already processing food more slowly.
Dehydration risk. Alcohol is a diuretic. If you're already at risk of dehydration from GI symptoms, adding alcohol compounds the problem. And dehydration on tirzepatide isn't just uncomfortable — it can lead to kidney issues.
Blood sugar effects. For people with type 2 diabetes, alcohol can cause unpredictable blood sugar swings — including dangerous lows when combined with diabetes medications. If you're using tirzepatide for diabetes alongside insulin or sulfonylureas, be especially cautious.
Lower tolerance. Many people on GLP-1 medications report that their alcohol tolerance drops noticeably. One drink may feel like three. This has been widely reported anecdotally on forums and in patient interviews, and is likely related to delayed gastric emptying keeping alcohol in contact with your stomach lining longer.
Our practical advice: If you drink, start slower than your pre-tirzepatide normal. Eat something first. Hydrate between drinks. And pay attention to your body — it may be telling you it can handle less than it used to. There's no need to abstain completely unless your provider recommends it, but awareness goes a long way.
Can You Take Tirzepatide Before Surgery?
This is a newer concern that's become increasingly important as GLP-1 medications grow in use.
The core issue: Tirzepatide delays gastric emptying. That means food or liquid may stay in your stomach longer than expected. During procedures requiring anesthesia or deep sedation, this increases the risk of pulmonary aspiration — where stomach contents enter the lungs while you're unconscious.
Post-marketing reports submitted to the FDA have documented rare cases of pulmonary aspiration in patients receiving GLP-1 receptor agonists undergoing elective surgeries or procedures requiring general anesthesia or deep sedation, even when patients reported following preoperative fasting recommendations.
What you should do: The Zepbound label states: instruct patients to inform healthcare providers prior to any planned surgeries or procedures. The label also notes that available data are insufficient to inform specific recommendations to mitigate the risk, including whether modifying fasting or temporarily discontinuing the drug would help. Tell your surgeon and anesthesiologist that you're taking tirzepatide — ideally well before the procedure date, not the morning of.
Are Zepbound and Mounjaro Side Effects the Same?
Same molecule. Same manufacturer (Eli Lilly). Different approved uses, different clinical trial populations.
Zepbound is approved for chronic weight management in adults with obesity (BMI ≥30) or overweight (BMI ≥27) with at least one weight-related condition. Mounjaro is approved for type 2 diabetes.
The side effect profiles overlap heavily because the drug is identical. The differences in reported rates mostly reflect the different populations studied — the SURMOUNT trials (Zepbound/weight loss) generally showed higher GI side effect rates than the SURPASS trials (Mounjaro/diabetes). This is likely because weight loss trials used higher doses for longer periods and enrolled a different patient demographic.
If you're using tirzepatide for weight management, the Zepbound trial data is more directly applicable to your experience than the Mounjaro data.
Related: Best Compounded Tirzepatide Programs · Cheapest Tirzepatide Online Without Insurance
How We Verified This Guide
We take accuracy seriously because this is health information. Here's exactly how we built this page and how we keep it current.
Primary sources used
- FDA Prescribing Information for Mounjaro (2025 label) and Zepbound (2025 label) — the official, FDA-reviewed safety and efficacy data
- FDA Drug Safety Communication (January 13, 2026) — the comprehensive review of GLP-1 medications and suicidal ideation/behavior
- SURPASS-1 through SURPASS-5 clinical trial publications — the pivotal trials for Mounjaro in type 2 diabetes
- SURMOUNT-1 through SURMOUNT-5 clinical trial publications — the pivotal trials for Zepbound in weight management, including 3-year safety data
- Peer-reviewed meta-analyses and cohort studies from PubMed and PMC, including the 2025 cancer risk meta-analysis (Kamrul-Hasan et al.), the 2025 FAERS safety analysis, and the 2025–2026 psychiatric safety analyses
- StatPearls (NCBI Bookshelf) clinical reference for tirzepatide
- MedlinePlus drug information
- Memorial Sloan Kettering patient education materials
Our evidence categories
Throughout this guide, we've tried to be clear about where information comes from:
- Label-listed common: Reported in clinical trials, included in FDA-approved prescribing information
- Label-listed serious warning: Identified in the FDA Warnings and Precautions or Boxed Warning sections
- Post-marketing report: Identified after FDA approval through voluntary adverse event reporting or post-marketing surveillance studies
- Frequently searched, but current evidence is limited or indirect: Topics people ask about that don't have strong clinical trial data — we say so plainly rather than guessing
What we are not
We are not doctors. We do not provide medical advice. We compile, explain, and cite publicly available clinical trial data and FDA information so you can have better conversations with your healthcare provider. Always discuss medication decisions with a qualified professional.
How we stay current
This page is reviewed and updated when new clinical data is published, FDA labels change, or new safety communications are issued. All "Last Verified" dates reflect when we actually verified specific claims — not when we last touched the page cosmetically.
What Should You Do Next?
If you've read this far, you have a solid, evidence-based understanding of tirzepatide's side effect profile. You know what's common. You know what's serious. You know what the clinical trial data says versus what the headlines claim. And you know when to wait it out versus when to get help.
If you're currently taking tirzepatide and experiencing side effects, you have a concrete management playbook and a clear triage framework. Use them. And if something doesn't feel right — trust your instincts and contact your provider.
If you're considering tirzepatide and the side effects were your biggest hesitation, here's the honest picture: most people experience some GI discomfort during dose escalation. It almost always gets better. The serious risks are rare. And the benefit — for the right candidate — is substantial.
The key word is right candidate. Tirzepatide isn't for everyone, and the best outcomes happen when you're matched with the right medication, the right dose trajectory, and a provider who actually monitors you through the process.
Not sure if tirzepatide is right for you?
Take our 60-second eligibility quiz to find out — and get matched with a provider who monitors you through the process.
Take the Eligibility QuizTirzepatide Side Effects: Frequently Asked Questions
Do tirzepatide side effects go away?
For most people, yes. GI side effects (nausea, diarrhea, constipation) are worst during the first 8–12 weeks and during dose increases. Clinical trial data shows the rate of new adverse events decreases over time at stable doses.
How long does nausea last on tirzepatide?
Typically 1–2 weeks at each new dose level. It's usually worst 24–48 hours post-injection and improves as your body adjusts. If nausea persists beyond 3–4 weeks at the same dose, talk to your provider.
Which tirzepatide dose causes the most side effects?
Side effects are dose-dependent — higher doses generally produce higher rates. But the biggest adjustment usually happens with your first dose, not subsequent escalations, because your body has never seen the drug before. Each escalation tends to be milder than the last.
Is tirzepatide hair loss permanent?
No. Hair shedding from tirzepatide treatment is telogen effluvium — triggered by rapid weight loss. It's temporary. Hair typically regrows once weight stabilizes, usually within 6–12 months. Adequate protein intake helps minimize it.
Does tirzepatide increase cancer risk?
The boxed warning exists because of thyroid C-cell tumors found in rat studies. In humans, a 2025 meta-analysis of 13 randomized trials (13,761 participants) found no increased cancer risk over 26 to 72 weeks — though very-long-term data is still needed. If you have a history of MTC or MEN 2, tirzepatide is contraindicated.
Can tirzepatide cause anxiety or depression?
The FDA completed a comprehensive review in January 2026 — analyzing 91 clinical trials with 107,910 patients — and concluded there is no increased risk of suicidal ideation, behavior, depression, anxiety, or other psychiatric events with GLP-1 medications. They've requested removal of the suicidal ideation warning from affected labels. However, report any mood changes to your provider regardless of cause.
What happens when you stop taking tirzepatide?
Side effects resolve. But appetite typically returns to pre-treatment levels and weight regain is common. SURMOUNT-4 data showed significant weight regain after stopping. If you're considering stopping, develop a transition plan with your provider.
Can tirzepatide affect birth control pills?
Yes. Tirzepatide slows gastric emptying, which can reduce absorption of oral contraceptives. The FDA recommends using non-oral contraception for 4 weeks after starting and 4 weeks after each dose increase.
Is fatigue normal on tirzepatide?
It's listed as a common adverse reaction in the Zepbound label at 5–7%. It's typically mild and resolves. If you're experiencing significant fatigue, check that you're eating and hydrating adequately — under-eating is a common culprit.
Is constipation normal on tirzepatide?
Yes, it affects 11–17% of Zepbound patients depending on dose. It's a consequence of slowed gastric emptying. Gradually increasing fiber, staying hydrated, and using OTC remedies like MiraLAX usually help. Severe or persistent constipation should be reported to your provider.
Are Zepbound and Mounjaro side effects the same?
Very similar. They contain the same medication made by Eli Lilly. Side effect rates differ slightly between their clinical trials because the patient populations (weight loss vs. diabetes) and dosing protocols were different.
When should you go to the ER for tirzepatide side effects?
Severe allergic reaction (throat swelling, difficulty breathing). Signs of pancreatitis (severe abdominal pain radiating to back). Severe hypoglycemia (confusion, loss of consciousness). Suicidal ideation. These are emergencies — don't wait.
Can tirzepatide cause vision problems?
In people with diabetic retinopathy, rapid blood sugar improvement can temporarily worsen eye disease. This is not unique to tirzepatide — it happens with insulin too. If you have diabetes-related eye disease, your ophthalmologist should be monitoring you.
Can tirzepatide cause body aches?
Body aches and musculoskeletal pain have been documented in post-marketing case reports (PMC, 2025). They typically occur 24–72 hours post-injection and resolve on their own. Hydration, adequate nutrition, and OTC pain relief generally help.