What Is Tirzepatide? Uses, Side Effects & Cost
Tirzepatide is the prescription drug inside both Mounjaro and Zepbound. It’s a once-weekly injection that activates two gut hormone receptors — GIP and GLP-1 — to reduce appetite, improve blood sugar control, and drive significant weight loss. In clinical trials, adults without diabetes taking the highest dose lost an average of 20.9% of their body weight over 72 weeks. For someone starting at 230 pounds, that’s about 48 pounds.
Here’s what most pages won’t tell you upfront: tirzepatide is the drug name, not the brand name. Mounjaro is the FDA-approved brand for type 2 diabetes. Zepbound is the FDA-approved brand for weight loss and obstructive sleep apnea. Same molecule, different labels, different insurance rules, very different price tags. And the compounded versions you’ve seen advertised online? Those are not FDA-approved and are not the same regulatory product — a distinction that matters more than most websites admit.
We built this page to be the only resource you need. Below, we break down how tirzepatide actually works in your body, what the clinical data really shows, every FDA-approved use, the full side effect profile (including the serious ones), real 2026 pricing across every access path, and exactly who should — and shouldn’t — consider it.
If you’ve been hearing this word everywhere and want the straight answer, you’re in the right place.
For informational purposes only—not medical advice.

This page contains affiliate links. We earn commissions when you sign up through our links — this never affects our recommendations or the accuracy of our content. Full disclosure →
Tirzepatide Decoded: Quick Reference
Before we go deeper, here’s the table that clears up the single biggest confusion around this drug.

| Term | What It Is | FDA-Approved? | What It’s For | Common Confusion |
|---|---|---|---|---|
| Tirzepatide | The active ingredient (drug molecule) | Yes — as Mounjaro and Zepbound | See brand-specific uses below | People think it’s a brand name. It’s not. |
| Mounjaro | Brand name made by Eli Lilly | Yes (approved May 2022) | Improve blood sugar in adults and children 10+ with type 2 diabetes | People assume it’s only for diabetes. Doctors sometimes prescribe it off-label for weight loss. |
| Zepbound | Brand name made by Eli Lilly | Yes (approved Nov 2023; OSA indication Dec 2024) | Weight management in qualifying adults; moderate-to-severe obstructive sleep apnea in adults with obesity | People assume it’s a different drug than Mounjaro. It’s the same molecule. |
| Compounded tirzepatide | Versions made by compounding pharmacies | No. Not FDA-approved for safety, effectiveness, or quality. | Varies by provider claims | People assume it’s a cheaper “generic.” It is not a generic, and FDA has warned against marketing it as equivalent to the approved products. |
In one line: Tirzepatide is the drug. Mounjaro and Zepbound are the FDA-approved brands. Compounded products are a separate regulatory category entirely.
Sources: FDA prescribing information for Mounjaro (NDA 215866) and Zepbound (NDA 217806); FDA press announcement on non-FDA-approved GLP-1 marketing claims.
Why Is Everyone Suddenly Talking About Tirzepatide?
If it feels like this word appeared out of nowhere, that’s because the timeline moved fast.
Tirzepatide was originally approved in May 2022 for type 2 diabetes. Then in November 2023, it picked up a second FDA approval for weight loss under a new brand name (Zepbound). In December 2024, it got a third indication — obstructive sleep apnea in adults with obesity. That’s three FDA approvals in under three years.
Add to that: the broader GLP-1 conversation exploded. Ozempic became a cultural phenomenon. Insurance companies started debating coverage. Telehealth platforms began advertising compounded versions at lower prices. Suddenly, people who’d never thought about injectable medications were hearing “tirzepatide” from their doctor, their coworker, the news, and their social media feed — often without any context about what it actually is.
And here’s the thing that made this especially confusing: unlike semaglutide (where Ozempic became the household name), tirzepatide entered public awareness under three different names — the molecule name, Mounjaro, and Zepbound — and most people have no idea these are all the same drug. That confusion is exactly why you’re here. Let’s clear it up.
Is Tirzepatide the Same as Mounjaro or Zepbound?
Yes and no. This is the question that trips up nearly everyone, so let’s break it down carefully.
Tirzepatide vs. Mounjaro
Tirzepatide is the active ingredient. Mounjaro is the FDA-approved brand name that Eli Lilly uses for the type 2 diabetes indication. When your doctor prescribes Mounjaro, you’re getting tirzepatide — manufactured by Eli Lilly, in FDA-approved doses, through FDA-regulated supply chains.
Mounjaro is specifically approved for improving glycemic control (blood sugar management) in adults and pediatric patients age 10 and older with type 2 diabetes, used alongside diet and exercise.
Source: FDA prescribing information, Mounjaro (NDA 215866, revised January 2026).
Tirzepatide vs. Zepbound
Same molecule, different label. Zepbound is the FDA-approved brand name for two indications:
- Chronic weight management — for adults with a BMI of 30 or greater (obesity), or BMI of 27 or greater with at least one weight-related condition like high blood pressure, high cholesterol, type 2 diabetes, or cardiovascular disease. Must be used with a reduced-calorie diet and increased physical activity.
- Moderate-to-severe obstructive sleep apnea — in adults with obesity. This approval came in December 2024, making Zepbound the first medication approved for moderate-to-severe obstructive sleep apnea in adults with obesity.
The key practical difference? Insurance coverage. Many insurance plans cover Mounjaro for diabetes but won’t cover Zepbound for weight loss. Same drug, different rules. We cover cost details further down.
Source: FDA prescribing information, Zepbound (NDA 217806, revised January 2026); Eli Lilly press release, December 2024.
Tirzepatide vs. Compounded Tirzepatide
This is where it gets critical, and where we need to be very precise.
Compounded tirzepatide is not FDA-approved. It has not undergone FDA review for safety, effectiveness, or quality before being sold. It is not a “generic” version. It is not interchangeable with the approved products from a regulatory standpoint. The FDA has specifically warned that companies cannot market non-FDA-approved compounded products as generic versions of, the same as, or claim they use the same active ingredient as, the approved drugs in their promotional materials.
We cover the compounding situation in full detail in a dedicated section below — including what changed with the FDA shortage status, what’s legally permitted in 2026, and what to verify before using any online source. It’s one of the most important sections on this page.
Source: FDA press announcement, “FDA Intends to Take Action Against Non-FDA-Approved GLP-1 Drugs”; FDA, “Concerns About Unapproved GLP-1 Drugs Used for Weight Loss.”
What Is Tirzepatide Used For?
Tirzepatide has three FDA-approved uses as of 2026. Each one goes through a different brand name, which is part of why the landscape feels so tangled.
Type 2 Diabetes (as Mounjaro)
Mounjaro is approved as an add-on to diet and exercise for improving blood sugar control in adults and children age 10 and older with type 2 diabetes. In the SURPASS clinical trial program, tirzepatide lowered A1C levels by up to 2.34% at the 15 mg dose — which is a substantial reduction. Many participants achieved A1C levels below 7%, which is the standard treatment target.
Chronic Weight Management (as Zepbound)
Zepbound is approved for long-term weight management in adults who meet specific criteria: a BMI of 30+ (obesity), or a BMI of 27+ (overweight) with at least one weight-related health condition. It must be used alongside a reduced-calorie diet and increased physical activity. This is where the headline-grabbing weight loss numbers come from. We’ll cover the clinical trial data in detail below.
Obstructive Sleep Apnea (as Zepbound)
In December 2024, the FDA approved Zepbound for moderate-to-severe obstructive sleep apnea in adults with obesity — making it the first medication ever approved specifically for this condition. This matters because OSA affects an estimated 30 million Americans, and weight is a primary contributor.
What Tirzepatide Is NOT Approved For
- Not for type 1 diabetes. Tirzepatide has not been studied in people with type 1 diabetes and should not be used for it.
- Not for cosmetic weight loss in people who don’t meet BMI eligibility criteria.
- Not for children under 18 for weight management (the pediatric approval only covers type 2 diabetes in ages 10+).
- No FDA-approved oral tirzepatide exists. If you see a pill or tablet version sold online, that is not an FDA-approved product.
Sources: FDA prescribing information for Mounjaro and Zepbound; clinical trial data from SURPASS program.
How Does Tirzepatide Work in the Body?
This is actually the most fascinating part — and understanding the mechanism helps make sense of both the benefits and the side effects.
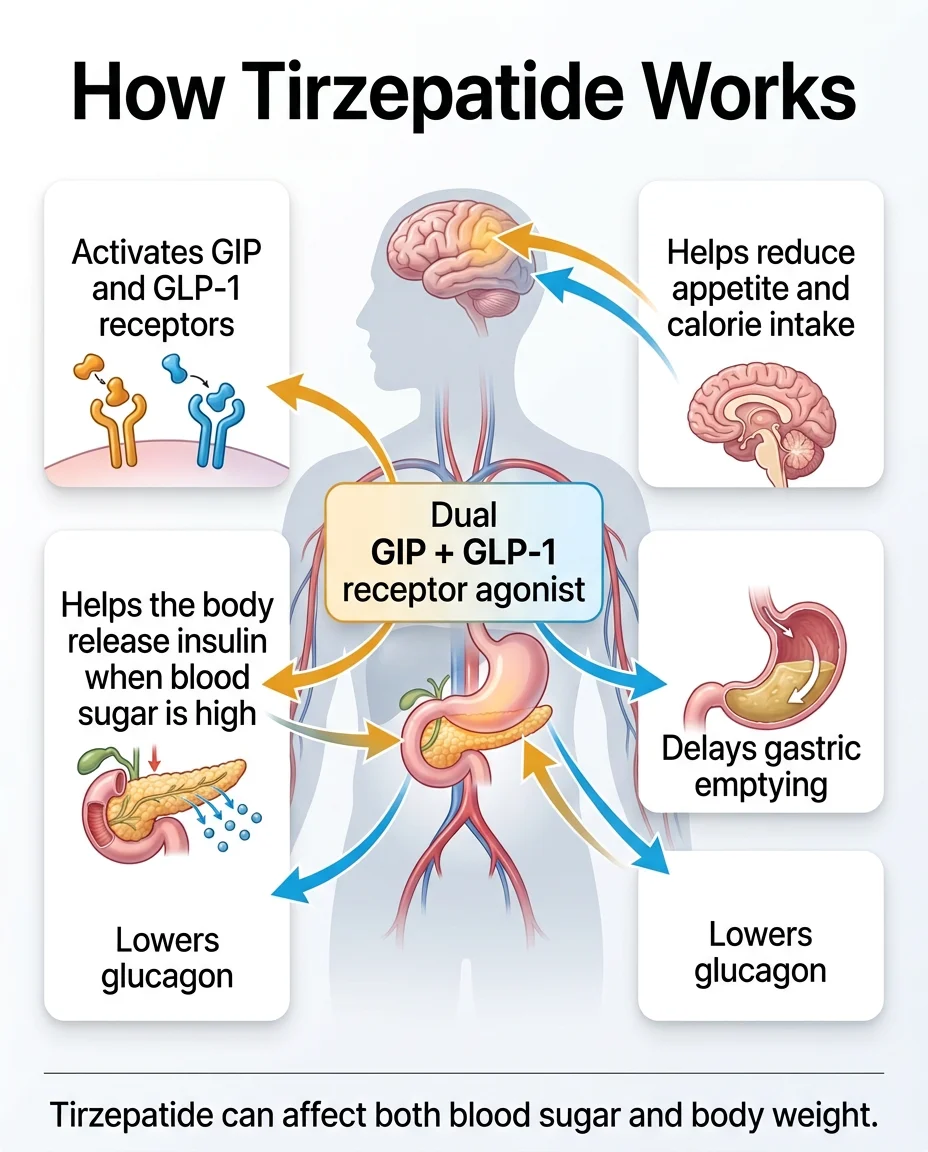
The Two-Hormone System
Your gut naturally releases hormones after you eat. Two of the most important ones for appetite and blood sugar are:
GLP-1 (glucagon-like peptide-1): Tells your pancreas to release insulin. Reduces glucagon (a hormone that raises blood sugar). Slows down how fast your stomach empties — so you feel full longer. Signals your brain that you’ve eaten enough.
GIP (glucose-dependent insulinotropic polypeptide): Also stimulates insulin release (when blood sugar is elevated). Appears to play a role in fat metabolism. Works synergistically with GLP-1 — meaning the two together produce a bigger effect than either alone.
Tirzepatide mimics both of these hormones simultaneously. That’s why it’s called a “dual GIP/GLP-1 receptor agonist” — and why the FDA called it a “first-in-class” medication. No other approved drug does both.
What This Means in Practical Terms
When you take tirzepatide, several things happen:
- Your appetite genuinely decreases. Many people describe the constant background chatter about food — sometimes called “food noise” — going quiet. You feel satisfied with smaller portions.
- Your stomach empties more slowly. Food stays in your stomach longer, which extends the feeling of fullness after meals.
- Your blood sugar stabilizes. Insulin is released more efficiently, and glucagon is suppressed — both of which pull blood sugar down.
- Your body composition shifts. The weight loss in clinical trials was primarily fat mass, not just water weight.
Why Dual Action Matters
Semaglutide (the active ingredient in Ozempic and Wegovy) only activates GLP-1 receptors. Tirzepatide activates both GLP-1 and GIP. This dual mechanism is the primary reason tirzepatide has produced greater weight loss in clinical trials — including in a direct head-to-head trial against semaglutide.
Think of it this way: semaglutide works through one hormonal pathway. Tirzepatide works through two. The second pathway doesn’t just add a little — it appears to amplify the effect.
Sources: FDA prescribing information (mechanism of action section); StatPearls, “Tirzepatide” (NCBI Bookshelf, NBK585056).
Is Tirzepatide a GLP-1? Is It a Peptide? What Is It Made From?
Is tirzepatide a GLP-1? Technically, it’s a dual GIP/GLP-1 receptor agonist. It activates GLP-1 receptors (like semaglutide does) and GIP receptors (which semaglutide does not). It’s often grouped with the “GLP-1 class” for simplicity, but it’s more accurately described as a “twincretin” — a word you’ll see in medical literature.
Is tirzepatide a peptide? Yes. It’s a synthetic polypeptide — a chain of 39 amino acids. It’s engineered to be an analog (modified version) of the naturally occurring GIP hormone, with structural modifications that also allow it to activate GLP-1 receptors. A fatty-acid moiety attached to the molecule extends its half-life, which is why a single injection lasts a full week.
What is tirzepatide made from? It’s manufactured synthetically by Eli Lilly — it’s not extracted from an animal or plant source. The FDA-approved versions (Mounjaro and Zepbound) are produced under strict pharmaceutical manufacturing standards (Current Good Manufacturing Practice, or CGMP).
Sources: FDA prescribing information (description section); StatPearls, “Tirzepatide.”
How Much Weight Can You Lose on Tirzepatide?
This is probably why you’re really here. Let’s look at what the clinical trials actually found.
The Key Trial Numbers
Tirzepatide has been studied in one of the largest clinical trial programs for obesity treatment. Here are the headline results:
SURMOUNT-1 (72 weeks, adults without diabetes):
| Dose | Average Weight Loss | % Losing 20%+ Body Weight |
|---|---|---|
| 5 mg weekly | 15.0% | 30% |
| 10 mg weekly | 19.5% | 50% |
| 15 mg weekly | 20.9% | 57% |
| Placebo | 3.1% | 3% |
To put that in perspective: at the highest dose, the average participant lost roughly one-fifth of their body weight. For a 250-pound person, that’s about 50 pounds over 72 weeks.
Source: Jastreboff AM, et al. “Tirzepatide Once Weekly for the Treatment of Obesity.” New England Journal of Medicine, 2022.
SURMOUNT-1 Three-Year Extension (176 weeks): In the published 176-week extension in adults with obesity and prediabetes, mean body-weight change was −19.7% with tirzepatide 15 mg. Type 2 diabetes developed in 1.3% of tirzepatide-treated participants versus 13.3% with placebo.
Source: Jastreboff AM, et al. Published in The New England Journal of Medicine, 2024. (SURMOUNT-1 extension)
SURMOUNT-5 (Head-to-head vs. semaglutide, 72 weeks):
| Metric | Tirzepatide | Semaglutide |
|---|---|---|
| Average % weight loss | 20.2% | 13.7% |
| Average pounds lost | 50.3 lbs | 33.1 lbs |
| Waist size reduction | 7.2 inches | 5.1 inches |
This was the first direct comparison between the two drugs. Tirzepatide produced 47% greater weight loss than semaglutide at maximum tolerated doses.
Source: Aronne LJ, et al. “Tirzepatide as compared with semaglutide for the treatment of obesity.” New England Journal of Medicine, 2025.
What These Numbers Mean in Real Life
Clinical trials are controlled environments. Participants received structured support, regular monitoring, and were following diet and exercise programs. Your results may differ based on your starting weight, dose, adherence, diet, activity level, and individual biology.
That said, these are the strongest weight loss numbers ever recorded in a large-scale obesity medication trial. Even at the lowest maintenance dose (5 mg), the average result was substantial.
What These Numbers Do NOT Mean
They don’t mean everyone loses 20%. Some people respond more, some less. A small percentage don’t respond meaningfully at all. And they don’t mean the weight loss happens overnight — the titration schedule takes months, and most of the result accumulates gradually over 6 to 12+ months.
Who Is Tirzepatide For?
Basic Eligibility for Weight Management (Zepbound)
Based on FDA labeling, you may qualify if you meet either of these criteria:
- BMI of 30 or greater (obesity) — that’s roughly 210 lbs at 5’9”, or 180 lbs at 5’4”
- BMI of 27 or greater (overweight) plus at least one weight-related health condition: high blood pressure, high cholesterol or triglycerides, type 2 diabetes, obstructive sleep apnea, or cardiovascular disease
When the Diabetes Indication Matters
If you have type 2 diabetes, your doctor may prescribe Mounjaro (not Zepbound) because insurance coverage is significantly better for the diabetes indication. You still get tirzepatide — just under a different brand label.
Signs You Might Want to Talk to a Clinician About This
- You’ve been trying to lose weight through diet and exercise and hit a wall
- Your doctor has mentioned GLP-1 medications
- You have weight-related health conditions that aren’t improving
- You’re comparing tirzepatide to other medications you’ve heard about
Who Might Be a Better Fit for a Different Path
Not everyone needs — or will benefit most from — tirzepatide specifically. Semaglutide might be a better fit if your insurance covers it and doesn’t cover tirzepatide. Lifestyle changes alone might be sufficient for people with lower BMIs. Bariatric surgery may be more appropriate for some individuals with very high BMI and multiple comorbidities. A qualified clinician can help you sort this out.
Not sure which GLP-1 path fits your situation?
Our free 60-second quiz matches your health profile, insurance, and budget to the best available options — no sales pitch, just clarity.
Find My Best GLP-1 PathTirzepatide Dosing Schedule: What to Expect
Tirzepatide follows a gradual dose-escalation protocol. You don’t start at the full dose — your body needs time to adjust.
Standard Titration Schedule
| Timeframe | Weekly Dose | Purpose |
|---|---|---|
| Weeks 1–4 | 2.5 mg | Initiation only — not a therapeutic dose. Lets your body adjust. |
| Weeks 5–8 | 5.0 mg | First maintenance dose. Therapeutic effects begin. |
| Weeks 9–12 | 7.5 mg (if needed) | Optional increase based on response and tolerability. |
| Weeks 13–16 | 10.0 mg (if needed) | A common landing point for many patients. |
| Weeks 17–20 | 12.5 mg (if needed) | Further escalation if tolerated. |
| Week 21+ | 15.0 mg (maximum) | Highest approved dose. |
Source: FDA prescribing information for Mounjaro and Zepbound.
Important dose notes: For adults, tirzepatide can be titrated up to 15 mg once weekly. For pediatric patients age 10 and older using Mounjaro for type 2 diabetes, the maximum approved dose is 10 mg. For Zepbound, recommended maintenance doses are 5, 10, or 15 mg for chronic weight management, and 10 or 15 mg for OSA. The 7.5 mg and 12.5 mg steps are escalation doses, not listed maintenance targets.
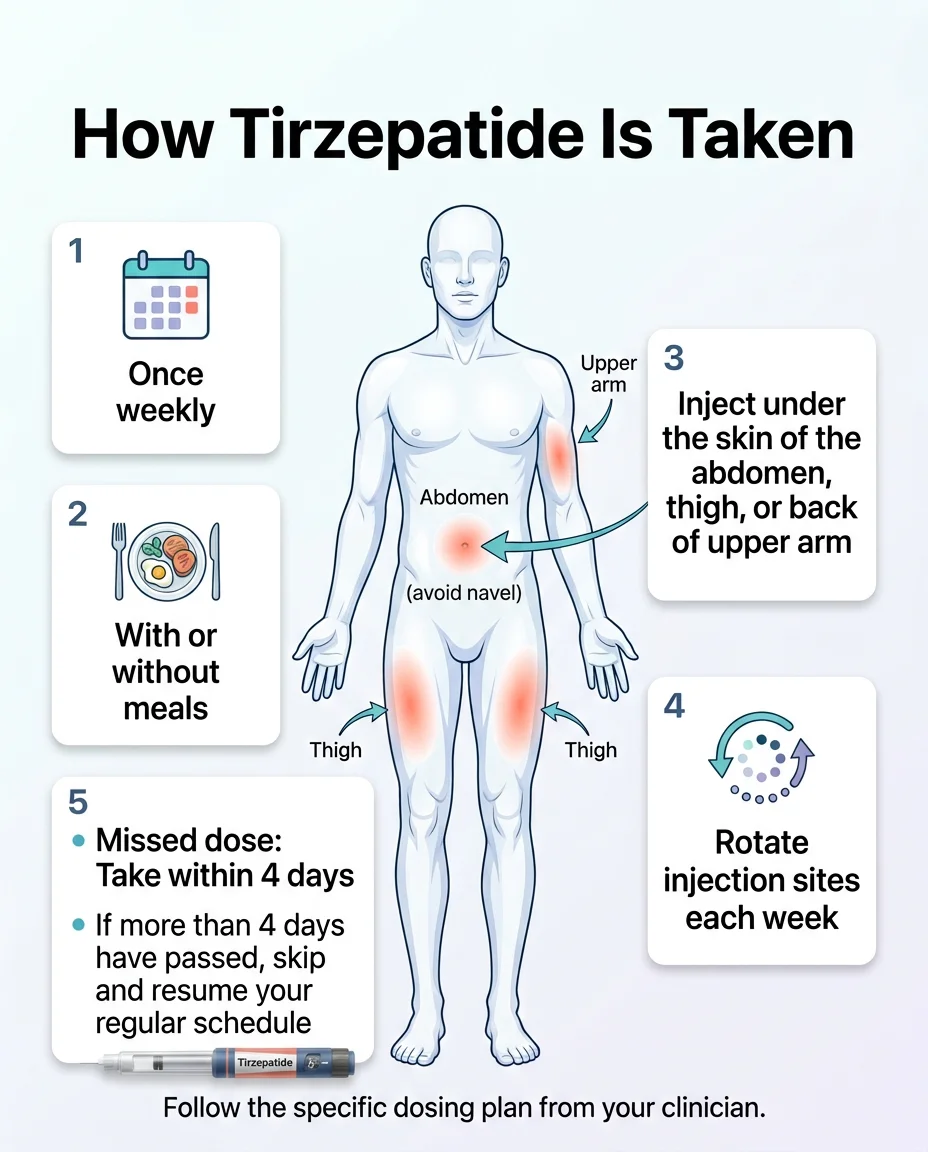
Key Dosing Details
How to take it: Inject once weekly, subcutaneously (under the skin), in your stomach, thigh, or back of upper arm. Rotate injection sites each time. Take it on the same day each week, at any time, with or without food.
If you miss a dose: Take it as soon as you remember, as long as it’s within 4 days (96 hours). If more than 4 days have passed, skip it and resume your regular schedule. Don’t double up.
An important note about the 2.5 mg dose: This is the starting dose, but it is not a treatment dose. It exists purely to help your body adjust and minimize side effects. Don’t judge the medication’s effectiveness based on how you feel at 2.5 mg.
What About “Microdosing” Tirzepatide?
You may have seen this term online. “Microdosing” tirzepatide — taking doses smaller than the FDA-approved starting dose — is not a labeled protocol. The FDA prescribing information defines a specific initiation and escalation schedule. Any deviation from that should be a conversation between you and your prescribing clinician, not something you decide based on an internet post.
What Actually Changes in Your Day-to-Day Life?
This is the section most tirzepatide pages skip entirely — and it’s the one people actually want to know about.
Your Relationship With Food Shifts
This is the change people notice first. The constant mental loop of what should I eat, when should I eat, I shouldn’t eat that but I want to — it gets quieter. Not silent. Quieter. Many people describe feeling neutral about food for the first time in years. You eat, you feel satisfied, and you move on.
You’ll Eat Less Without White-Knuckling It
Because tirzepatide slows gastric emptying and reduces appetite signals in the brain, smaller portions feel genuinely satisfying. You’re not fighting willpower. You’re working with your biology for possibly the first time.
Side Effects Often Cluster Around Dose Changes
The first few days after starting — or after each dose increase — are when nausea, digestive changes, and fatigue are most likely. For most people, these settle down within 1–2 weeks at each new dose. Knowing this upfront helps you plan accordingly (don’t increase your dose the day before a big event).
Hydration Becomes More Important
GI side effects like nausea, diarrhea, and vomiting can cause dehydration, which can then lead to more serious issues like kidney problems. Drink more water than you think you need. This isn’t a suggestion — it’s a medical priority.
Some Practical Adjustments
- Eat protein-rich meals to help preserve muscle mass during weight loss
- Avoid greasy, fried, or very high-fat foods — they tend to worsen nausea
- Alcohol tolerance may decrease; some people feel effects more quickly
- You’ll need to tell all your healthcare providers (including your dentist and anesthesiologist) that you’re on a GLP-1 medication
- If you take oral contraceptives, you may need a backup method — tirzepatide can reduce their effectiveness
This Is Not a Short-Term Fix
Tirzepatide is designed for chronic use. Clinical data shows that stopping leads to weight regain (we cover this in detail below). Going in with the right mindset — this is ongoing therapy, like blood pressure medication — helps set realistic expectations.
What Are the Side Effects of Tirzepatide?
We’re going to be thorough here, because this is information you deserve to have before making a decision.

Common Side Effects
These are the side effects that occur in more than 5% of people in clinical trials:
- Nausea — the most commonly reported side effect. Usually worst during the first weeks at each new dose. Tends to improve over time.
- Diarrhea
- Vomiting
- Constipation
- Stomach pain or discomfort
- Indigestion (dyspepsia) and heartburn (GERD)
- Decreased appetite — this is partly how the drug works, but it’s listed as a side effect
- Injection site reactions — redness, itching, or swelling at the injection site
- Fatigue
- Belching
- Hair loss — reported in Zepbound trials. Likely related to rapid weight loss itself (a condition called telogen effluvium) rather than the drug directly. Usually temporary.
- Allergic or hypersensitivity reactions
For our full breakdown of Mounjaro side effects by dose — including the Zepbound weight-loss trial numbers — see our complete Mounjaro Side Effects guide.
Serious Warnings
These are rarer but important to understand:
Thyroid C-Cell Tumor Warning (Boxed Warning): In animal studies, tirzepatide caused thyroid tumors in rats, including thyroid cancer. Whether this happens in humans is unknown. Tirzepatide is contraindicated — meaning you absolutely should not take it — if you or a family member has a history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Report any lump or swelling in your neck, hoarseness, trouble swallowing, or shortness of breath to your doctor immediately.
Pancreatitis: Acute pancreatitis, including fatal cases, has been reported. Symptoms include severe, persistent abdominal pain (sometimes radiating to the back), with or without vomiting. If suspected, tirzepatide should be discontinued immediately.
Gallbladder Disease: Gallstones and cholecystitis (gallbladder inflammation) were reported in clinical trials and are associated with rapid weight loss. Tell your doctor about upper abdominal pain, fever, or yellowing of skin/eyes.
Kidney Problems: Dehydration from nausea, vomiting, or diarrhea can lead to acute kidney injury, including cases requiring dialysis. Stay hydrated. Report severe or prolonged GI symptoms.
Hypoglycemia: When used alongside insulin or sulfonylureas, tirzepatide can cause low blood sugar. This risk is lower when used alone.
Severe Allergic Reactions: Anaphylaxis and angioedema have been reported (rare). Seek emergency care for difficulty breathing, swelling of face/throat, or severe rash.
Surgery/Anesthesia Risk: Because tirzepatide slows stomach emptying, there’s an increased risk of pulmonary aspiration during surgery under general anesthesia or deep sedation. Tell your surgical and anesthesia team you’re on a GLP-1 medication. Current FDA labeling states that data are insufficient to know whether temporarily discontinuing the drug reduces retained gastric contents.
Diabetic Retinopathy: Patients with existing diabetic retinopathy should be monitored, as rapid blood sugar improvement can temporarily worsen eye disease.
When to Call Your Doctor Urgently
Don’t wait on these:
- Severe abdominal pain that won’t go away
- Signs of an allergic reaction (swelling, difficulty breathing, severe rash)
- Signs of dehydration (dark urine, dizziness, very dry mouth) that don’t improve with fluids
- A new lump or swelling in your neck
- Yellowing of skin or eyes
Sources: FDA prescribing information for Mounjaro and Zepbound (Warnings and Precautions, Adverse Reactions sections).
Who Should NOT Take Tirzepatide?

Absolute Contraindications (Do Not Take)
- Personal or family history of medullary thyroid carcinoma (MTC)
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Known serious hypersensitivity to tirzepatide or any of its ingredients
Do not combine tirzepatide with another tirzepatide-containing product or any GLP-1 receptor agonist.
Pregnancy and Reproductive Cautions
- Tirzepatide may cause fetal harm. Tell your clinician if you are pregnant or plan to become pregnant. Discontinue Zepbound when pregnancy is recognized.
- Oral contraceptives may be less effective when starting tirzepatide or increasing the dose (because of delayed gastric emptying). Use a non-oral contraceptive or add a barrier method for 4 weeks after initiation and after each dose increase.
- Limited data on breastfeeding — discuss with your provider.
Discuss With Your Clinician First
- History of pancreatitis
- Gallbladder disease or history of gallstones
- Severe gastrointestinal disease or gastroparesis
- Kidney disease or conditions that increase dehydration risk
- Diabetic retinopathy requiring monitoring
- History of suicidal attempts or active suicidal ideation — the current Zepbound label warns about suicidal behavior and ideation and recommends avoiding use in those patients
- Taking insulin or sulfonylureas (dose adjustment needed to prevent low blood sugar)
- Type 1 diabetes (not approved, not studied)
Source: FDA prescribing information for Mounjaro and Zepbound (Contraindications, Warnings and Precautions).
Is Tirzepatide Safe Long Term?
Honest answer: we have growing evidence, but the picture is still developing.
The longest completed trial data we have is the SURMOUNT-1 176-week extension, which showed sustained weight loss (−19.7% at 15 mg) and a safety profile consistent with the shorter trials. The most common issues remained GI-related and manageable. Discontinuation rates due to adverse events ranged from 4–10% depending on dose, which is comparable to other medications in this class.
In January 2026, the FDA completed a review of suicidal behavior and ideation reports with GLP-1 receptor agonists. The conclusion: their evaluation did not identify an increased risk, and the FDA requested removal of that warning from certain GLP-1 labels. However, the current January 2026 Zepbound label still contains suicidal behavior and ideation language pending the label update propagation. This is a meaningful development, but labeling language may still be in transition — always check the most current version.
What’s still being studied: long-term cardiovascular outcomes (early data is encouraging — a heart failure trial showed a 38% reduction in major cardiovascular complications), effects on fatty liver disease (NASH/MAFLD), and outcomes in populations not heavily represented in initial trials.
The bottom line: tirzepatide’s safety profile appears consistent and manageable in the data we have so far. But “so far” is the operative phrase. This is a relatively new medication, and post-market surveillance is ongoing. Your clinician can help you weigh the known benefits against the evolving safety picture.
Sources: SURMOUNT-1 extension (176 weeks), published in NEJM 2024; FDA announcement on GLP-1 suicidal ideation review (January 2026); cardiovascular outcomes data from phase III trials.
Is Compounded Tirzepatide the Same as FDA-Approved Tirzepatide?
No. And this section might be the most important one on this page.
We know that’s a strong statement. Here’s why we’re making it.
The Short Version
Compounded tirzepatide products have not been reviewed by the FDA for safety, effectiveness, or quality before being marketed. They are not “generics.” They are not interchangeable with Mounjaro or Zepbound from a regulatory standpoint. The FDA has specifically warned that companies marketing these products cannot claim they are generic versions of, the same as, or use the same active ingredient as FDA-approved tirzepatide drugs in their promotional materials.
What Changed With the Shortage
Here’s the timeline that matters:
- December 2022: The FDA placed tirzepatide on the drug shortage list due to surging demand.
- During the shortage (2022–2024): Federal law permitted compounding pharmacies to produce versions of tirzepatide while the approved products were unavailable. Many telehealth companies began selling compounded tirzepatide at significantly lower prices.
- October 2024: The FDA determined the tirzepatide shortage was resolved — meaning Eli Lilly could now meet national demand.
- December 2024: After legal challenges from compounding groups, the FDA reaffirmed its determination and issued a declaratory order.
- February–March 2025: Enforcement discretion periods ended. State-licensed pharmacies (503A) had until February 18, 2025. Outsourcing facilities (503B) had until March 19, 2025.
Where Things Stand in 2026
Routine compounding of tirzepatide “copies” is no longer permitted under the shortage exception. Some companies have continued offering what they call “personalized” formulations — with modified doses or additional ingredients — arguing these fall under a different legal provision. Whether this approach complies with FDA regulations is actively debated, and the FDA and brand manufacturer have pushed back.
What the FDA Is Warning About
The FDA has raised specific concerns about non-approved tirzepatide products:
- Misleading marketing claims (calling products “generic” or “same as” the approved drugs)
- Dosing inaccuracies
- Unverified ingredients and quality standards
- Storage and shipping conditions that may degrade the product
- Products labeled as “research use only” being sold to consumers
- Illegal online sales without legitimate prescriptions
What to Verify Before Using Any Online Source
If you’re considering any tirzepatide product — whether brand-name or otherwise — verify:
- ✓ You have a prescription from a licensed clinician who has evaluated you
- ✓ The pharmacy dispensing your medication is state-licensed (you can verify at your state board of pharmacy)
- ✓ You know exactly what product you’re receiving — brand-name Mounjaro/Zepbound or something else
- ✓ The product hasn’t been shipped without proper cold chain/storage
- ✓ There are no “research use only” labels
- ✓ The clinician is available for ongoing monitoring, not just an initial transaction
We are not telling you what to do. We are telling you what to check. The stakes are too high for vague reassurances.
Sources: FDA declaratory order, December 19, 2024; FDA press announcement on non-approved GLP-1 marketing; Outsourcing Facilities Association v. FDA, N.D. Texas, May 7, 2025; FDA, “Concerns About Unapproved GLP-1 Drugs Used for Weight Loss.”
Want the safest path, not the sketchiest one?
Our quiz helps you navigate brand-name access, insurance options, and cost-reduction programs — based on your actual situation.
Find My Safest GLP-1 PathHow Much Does Tirzepatide Cost in 2026?
Let’s cut through the noise. Tirzepatide pricing is genuinely confusing — and intentionally so, depending on who’s selling it. Here’s what we’ve verified.
The 2026 Cost Landscape
| Access Path | Approx. Monthly Cost | Key Details |
|---|---|---|
| Zepbound pre-filled pens (WAC list price) | ~$1,086.37 | Same price at all doses. This is the wholesale acquisition cost without insurance or discounts. |
| Mounjaro pre-filled pens (WAC list price) | ~$1,079.77 | Same molecule, diabetes label. |
| Zepbound vials or KwikPen via LillyDirect (self-pay) | $299–$449 | Self Pay Journey Program pricing. $299 (2.5 mg), $399 (5 mg), $449 (7.5–15 mg). The $449 price for 7.5 mg+ requires refill within 45 days. |
| With commercial insurance + Lilly savings card | As low as $25/month | Must have commercial insurance that covers the specific brand. Government insurance excluded. |
| Commercial insurance, no Zepbound coverage + savings card | $299–$449 | KwikPen pricing for commercially insured patients whose plan does not cover Zepbound. Subject to program terms. |
Last verified: March 2026 against Zepbound.lilly.com and LillyDirect official pricing. Pricing changes frequently — confirm current prices before making decisions.
Why Prices Are So Confusing
Several things are happening simultaneously:
- Brand list price vs. actual out-of-pocket — almost nobody pays $1,086
- Insurance coverage varies widely by brand and indication. Many commercial plans cover Mounjaro for diabetes but fewer cover Zepbound for weight loss. Always check your specific plan.
- Medicare is changing. CMS has announced the Medicare GLP-1 Bridge program starting July 2026, which will provide eligible Medicare Part D beneficiaries access to Zepbound for weight loss at $50/month. The BALANCE Model follows in January 2027. Coverage depends on manufacturer participation and prior authorization criteria.
- LillyDirect vials and KwikPens are the most affordable brand-name option for self-pay patients, available through LillyDirect with free home delivery or Walmart pharmacy pickup
What to Verify Before You Believe a Price
When comparing offers, make sure you’re comparing the same things:
- Does the price include the medication, or just the consultation?
- Is it brand-name (Mounjaro/Zepbound) or something else?
- Does it include clinician access and ongoing monitoring?
- What about labs, shipping, and supplies?
- Is it a starter price or the price at your maintenance dose?
- Are there hidden subscription fees or cancellation penalties?
The cheapest price isn’t always the best value. And the most expensive price isn’t always the safest option. What matters is: are you getting an FDA-approved product from a legitimate provider with real clinical oversight?
Sources: Eli Lilly official pricing (Zepbound.lilly.com/savings, LillyDirect, Colorado WAC Disclosure Sheets); CMS Medicare GLP-1 Bridge FAQ (cms.gov, March 2026).
For our full breakdown of GLP-1 costs with and without insurance, see our comprehensive GLP-1 cost guide.
Tirzepatide vs. Semaglutide: What’s the Difference?
If you’re researching tirzepatide, you’ve almost certainly heard of semaglutide too. Here’s how they compare.
Mechanism
Tirzepatide activates two receptors: GIP and GLP-1. Semaglutide activates only GLP-1. This is the fundamental difference. The dual mechanism is why tirzepatide has outperformed semaglutide in trials.
Weight Loss (Head-to-Head Data)
In SURMOUNT-5 — the first direct comparison trial — tirzepatide produced 47% more weight loss than semaglutide over 72 weeks. Average loss: 20.2% (tirzepatide) vs. 13.7% (semaglutide).
Side-by-Side Comparison
| Feature | Tirzepatide (Mounjaro / Zepbound) | Semaglutide (Ozempic / Wegovy) |
|---|---|---|
| Drug class | Dual GIP + GLP-1 agonist | GLP-1 agonist only |
| Manufacturer | Eli Lilly | Novo Nordisk |
| Avg. weight loss (trials) | ~20–22% at highest dose | ~15% at highest dose |
| Dosing | Once weekly injection | Once weekly injection |
| Dose range | 2.5–15 mg | 0.25–2.4 mg |
| Common side effects | Nausea, diarrhea, vomiting, constipation | Nausea, diarrhea, vomiting, constipation |
| Brand list price/month | ~$1,086 | ~$1,350 (Wegovy) |
| Oral option? | No | Yes (Rybelsus for diabetes; oral Wegovy approved Dec 2025) |
| OSA approval? | Yes (Zepbound) | No |
| Years on market | Since 2022 | Since 2017 (Ozempic) |
The Honest Tradeoff
Tirzepatide produces better weight loss numbers in clinical trials. But semaglutide has a longer real-world track record, more insurance coverage pathways for some patients, and now an oral option. Some people respond better to one than the other. This is a decision to make with your clinician based on your health profile, insurance situation, and goals — not just trial data.
We have a full comparison guide if you want to go deeper: Tirzepatide vs. Semaglutide: Complete Comparison →
Source: Aronne LJ, et al. “Tirzepatide as compared with semaglutide for the treatment of obesity.” NEJM, 2025 (SURMOUNT-5).
What Happens if You Stop Taking Tirzepatide?
This is a question that deserves a straight answer, because the answer affects how you think about starting.
In the SURMOUNT-4 trial, the results were clear. From week 36 to week 88, participants switched from tirzepatide to placebo regained 14.0% of body weight, while those who continued on tirzepatide lost an additional 5.5%.
As Dr. Louis Aronne of Weill Cornell Medicine put it: if you stop the medication, you regain the weight.
That’s not unique to tirzepatide. It’s how obesity works. The body’s weight-regulation systems push toward regaining lost weight once the therapeutic pressure is removed — the same way blood pressure rises again if you stop taking blood pressure medication. This doesn’t mean the medication “failed.” It means obesity is a chronic condition that, for many people, requires ongoing treatment.
Going in with realistic expectations matters. If you start tirzepatide expecting to take it for 6 months and then stop, the data suggests most of the benefit will reverse. If you approach it as long-term therapy (like many chronic conditions require), the sustained results are genuinely impressive.
Source: Aronne LJ, et al. “Continued Treatment With Tirzepatide for Maintenance of Weight Reduction in Adults with Obesity.” JAMA, 2024 (SURMOUNT-4).
How Do You Get Tirzepatide Safely — and What Should You Do Next?
You’ve made it through the full picture. Here’s how to actually move forward.
Three Paths Based on Where You Are
Path 1: “I think I might be a candidate and want to explore options.” Take our free 60-second matching quiz. It maps your health profile, insurance, and budget to the best available GLP-1 options — whether that’s tirzepatide, semaglutide, or something else. No diagnosis, no obligation. Just a starting point.
Path 2: “I’m concerned about safety or have specific health conditions.” Start with your primary care doctor or endocrinologist. Bring this page with you if it helps. Also see our guides on GLP-1 side effects and GLP-1 contraindications for more detail.
Path 3: “I’m comparing providers and want to find the best deal.” See our Best GLP-1 Providers Compared (2026) guide, where we break down pricing, provider quality, and what’s actually included across the top platforms.
What to Ask Your Clinician Before Starting
Keep this list handy for your appointment:
- Based on my health profile, am I a better candidate for tirzepatide or semaglutide?
- Which brand name gives me the best insurance coverage?
- What’s the plan if I experience significant side effects?
- How will we monitor my progress and adjust dosing?
- What’s the long-term plan — how long should I expect to stay on this?
- Do I need to adjust any current medications?
- Are there any upcoming procedures where I’d need to pause treatment?
Still not sure which GLP-1 program is right for you?
We’ll match your health profile, budget, and insurance to the options that actually fit — with zero pressure and zero cost.
Get My Personalized GLP-1 PlanHow We Researched This Guide
We take accuracy seriously — especially on a YMYL (Your Money or Your Life) topic like prescription medications.
Sources we used: FDA prescribing information for Mounjaro (NDA 215866) and Zepbound (NDA 217806), peer-reviewed clinical trial data published in The New England Journal of Medicine and JAMA (SURPASS and SURMOUNT trial programs), StatPearls reference material, Eli Lilly official pricing and program data, FDA regulatory communications, and federal court records.
How we handle pricing: All cost figures are verified against manufacturer programs and current pharmacy data, with a “Last Verified” date on each claim. Pricing is updated as changes are confirmed.
What we don’t do: We don’t make medical diagnoses. We don’t claim compounded products are equivalent to FDA-approved drugs. We don’t use “always,” “guaranteed,” or “no risk” language. And we don’t hide our affiliate relationship — we earn commissions through our recommendation links, but this never changes what we recommend or how we present the evidence.
Medical disclaimer: This content is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional before starting, stopping, or changing any medication.
What Changed in This Update
- Verified current Mounjaro and Zepbound FDA labeling (January 2026 revisions)
- Updated compounded tirzepatide regulatory status for 2026
- Confirmed FDA’s January 2026 GLP-1 suicidal ideation review conclusion
- Verified current LillyDirect self-pay pricing (including KwikPen, effective February 2026)
- Updated Medicare access information (GLP-1 Bridge program starting July 2026)
- Checked all clinical trial citations against published records
References
- FDA Prescribing Information — Mounjaro (tirzepatide) injection, NDA 215866. Revised January 2026.
- FDA Prescribing Information — Zepbound (tirzepatide) injection, NDA 217806. Revised January 2026.
- Jastreboff AM, et al. “Tirzepatide Once Weekly for the Treatment of Obesity.” New England Journal of Medicine, 2022;387:205-216. (SURMOUNT-1)
- Aronne LJ, et al. “Continued Treatment With Tirzepatide for Maintenance of Weight Reduction in Adults with Obesity.” JAMA, 2024. (SURMOUNT-4)
- Aronne LJ, et al. “Tirzepatide as compared with semaglutide for the treatment of obesity.” New England Journal of Medicine, 2025. (SURMOUNT-5)
- Eli Lilly and Company. SURMOUNT-1 Three-Year Results press release. October 2024.
- Eli Lilly and Company. “FDA approves Zepbound for moderate-to-severe obstructive sleep apnea.” Press release, December 2024.
- FDA Declaratory Order: Resolution of Tirzepatide Injection Product Shortage. December 19, 2024.
- FDA. “FDA Intends to Take Action Against Non-FDA-Approved GLP-1 Drugs.” Press announcement.
- FDA. “FDA’s Concerns About Unapproved GLP-1 Drugs Used for Weight Loss.”
- FDA. “FDA Requests Removal of Suicidal Behavior and Ideation Warning from GLP-1 Receptor Agonist Labels.” January 2026.
- Tirzepatide. StatPearls. NCBI Bookshelf, NBK585056. Updated 2024.
- NCPA. “FDA ends compounding discretion for tirzepatide.” March 2025.
- Eli Lilly. Zepbound Self-Pay Journey Program and LillyDirect pricing.
- CMS. Medicare GLP-1 Bridge FAQ.
- Eli Lilly. Colorado WAC Disclosure Sheets for Mounjaro and Zepbound.
Frequently Asked Questions About Tirzepatide
Is tirzepatide the same as Mounjaro?
Tirzepatide is the active ingredient inside Mounjaro. Mounjaro is the brand name Eli Lilly uses for the type 2 diabetes indication. When you take Mounjaro, you're taking tirzepatide.
Is tirzepatide the same as Zepbound?
Same active ingredient, different brand label. Zepbound is the brand name for tirzepatide approved for weight management and obstructive sleep apnea. Same molecule as Mounjaro, different FDA indication and insurance coverage.
Is tirzepatide a brand name or the drug name?
Tirzepatide is the drug name (also called the active ingredient or generic name). Mounjaro and Zepbound are the brand names. Think of it like ibuprofen (drug name) vs. Advil and Motrin (brand names).
Is tirzepatide FDA approved?
Yes — under two brand names. Mounjaro was approved in May 2022 for type 2 diabetes. Zepbound was approved in November 2023 for weight management and expanded in December 2024 for obstructive sleep apnea.
Is tirzepatide only for diabetes?
No. While Mounjaro is approved specifically for type 2 diabetes, Zepbound is approved for chronic weight management and obstructive sleep apnea — neither of which requires a diabetes diagnosis.
Is tirzepatide a GLP-1?
It's a dual GIP/GLP-1 receptor agonist. It activates both GLP-1 receptors (like semaglutide does) and GIP receptors (which semaglutide does not). It's often grouped with GLP-1s, but its dual mechanism is what sets it apart.
Is tirzepatide a peptide?
Yes. It's a synthetic 39-amino-acid polypeptide — an engineered analog of the natural GIP hormone, modified to also activate GLP-1 receptors.
What is tirzepatide made from?
Tirzepatide is manufactured synthetically by Eli Lilly. It's not extracted from animals or plants. The FDA-approved versions are produced under pharmaceutical-grade manufacturing standards.
What are the most common side effects?
Nausea (most common), diarrhea, vomiting, constipation, stomach pain, indigestion, injection site reactions, fatigue, and decreased appetite. Most are GI-related and tend to improve over time, especially after your body adjusts to each dose level.
Is tirzepatide safe long term?
The longest completed trial data (176 weeks / 3+ years) shows a consistent and manageable safety profile. The most common issues remain GI-related. The FDA's January 2026 review found no increased risk of suicidal behavior with GLP-1 receptor agonists. Long-term post-market surveillance is ongoing.
Who should not take tirzepatide?
People with a personal or family history of medullary thyroid carcinoma (MTC) or MEN 2 syndrome. People with known hypersensitivity to tirzepatide. People currently on another GLP-1 medication. Pregnant women or those planning pregnancy. People with type 1 diabetes.
What's the starting dose?
2.5 mg injected subcutaneously once weekly. This is an initiation dose — not a therapeutic dose. After 4 weeks, you increase to 5 mg, then potentially higher in 2.5 mg increments every 4+ weeks up to a maximum of 15 mg.
What is tirzepatide microdosing?
"Microdosing" is not an FDA-labeled dosing strategy. The approved protocol starts at 2.5 mg weekly and follows a defined escalation schedule. Any off-label dosing approach should be discussed with your prescribing clinician.
What if I miss a dose?
If it's been fewer than 4 days since your missed dose, take it as soon as you remember. If it's been more than 4 days, skip it and resume your regular weekly schedule. Don't take two doses to make up for a missed one.
Is compounded tirzepatide the same as the FDA-approved versions?
No. Compounded tirzepatide has not undergone FDA review for safety, effectiveness, or quality. It is not a generic. The FDA has warned against marketing compounded products as equivalent to approved drugs.
Why do some products mention tirzepatide "with glycine" or other additives?
Some compounded products add ingredients like glycine or mannitol. These are not FDA-approved tirzepatide products. Added ingredients do not make a product FDA-approved, so patients should verify exactly what product they are receiving, who prescribed it, and which licensed pharmacy is dispensing it.
How much does tirzepatide cost?
It depends on your access path. Brand-name pens list at ~$1,086/month. LillyDirect self-pay vials and KwikPens start at $299/month (2.5 mg) and go up to $449/month (7.5–15 mg) with the Self Pay Journey Program. With commercial insurance and a savings card, eligible patients may pay as low as $25/month. Starting July 2026, eligible Medicare Part D beneficiaries may access Zepbound for $50/month through the Medicare GLP-1 Bridge program.
Tirzepatide vs. semaglutide: which is stronger for weight loss?
In the head-to-head SURMOUNT-5 trial, tirzepatide produced 47% greater weight loss than semaglutide (20.2% vs. 13.7% over 72 weeks). However, "stronger" doesn't always mean "better for you" — insurance, side effect tolerance, and individual response all matter.
What happens when you stop taking tirzepatide?
Clinical data (SURMOUNT-4) shows that stopping leads to significant weight regain — participants regained about half their lost weight after discontinuing. This is consistent with obesity being a chronic condition that often requires ongoing treatment.
Is there an oral tirzepatide pill?
Not currently. Tirzepatide is only available as an injection. Eli Lilly has studied oral formulations, but none are FDA-approved. Note: oral Wegovy (semaglutide) was approved in December 2025 — that is a different drug.
Is tirzepatide a generic?
Tirzepatide is the active ingredient name, not a generic product. There is currently no generic version of tirzepatide. Mounjaro and Zepbound are both brand-name products made by Eli Lilly. Compounded products should not be marketed as generic equivalents.
Can you take birth control pills while using tirzepatide?
Tirzepatide delays gastric emptying, which can reduce the absorption of oral medications — including oral contraceptives. The FDA label recommends using a non-oral contraceptive method or adding a barrier method for 4 weeks after starting tirzepatide and after each dose increase.
What should I ask my clinician before starting?
Key questions: Am I a candidate? Which brand makes sense for my insurance? What's the monitoring plan? What's the long-term strategy? Do I need to adjust other medications?
Still not sure which GLP-1 program is right for you? Take our free 60-second matching quiz. We’ll map your health profile, budget, and insurance situation to the options that actually fit — no cost, no pressure, just clarity.
Take the Free GLP-1 QuizMedical Disclaimer: This content is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional before starting, stopping, or changing any medication.
Affiliate Disclosure: WeightLossProviderGuide.com is an affiliate site. We earn commissions when readers sign up with providers through our links. This never changes what we recommend or how we present the evidence. See our advertising disclosure for details.
Last Updated: March 2026 · Last Verified Against FDA Labels: March 2026