GLP-1 Headache: What’s Normal, What’s Not, and What to Do Right Now
By WPG Research Team · · ·
Disclosure: Some links on this page are affiliate links. If you buy through them, we may earn a commission at no extra cost to you. Medical safety always comes before any commercial mention. This page is informational, not medical advice.
🚨 Stop and call 911 immediately if your headache is:
- Sudden and severe — "the worst headache of my life"
- Accompanied by neck stiffness, fever, confusion, or slurred speech
- Accompanied by vision changes, weakness on one side, or facial droop
- Accompanied by fainting, seizure, or loss of consciousness
- A new severe headache during pregnancy or right after delivery
These are not GLP-1 side effects. These are emergencies on any medication.
Overview
A GLP-1 headache is most often mild, manageable, and tied to your first dose, a recent dose increase, or low fluid and food intake from nausea. Headache rates vary a lot by drug — from rates very close to placebo on tirzepatide trials, up to 17% in Wegovy adolescent trials. For most people, symptoms ease in the first few weeks as the body adjusts.
We pulled headache numbers straight from each medication’s official FDA label on DailyMed so you can see exactly what’s expected for your drug, figure out what’s likely causing your headache, and know whether to manage it at home, message your prescriber, or pause the medication. We are Weight Loss Provider Guide, an independent comparison resource for GLP-1 telehealth providers — we don’t sell drugs, we don’t run a clinic, and we have no reason to talk you into staying on something that’s hurting you.
Which “bucket” are you in right now?
Find the row that fits you and act on it first.
| Your situation | What it likely means | What to do now |
|---|---|---|
| 🚨 Sudden "worst headache ever," confusion, slurred speech, weakness on one side, vision loss, fever with stiff neck, fainting, seizure, or recent head injury | Possible neurological emergency (not a GLP-1 effect) | Call 911 or go to the ER. Don’t text your prescriber. Don’t take a quiz. Go. |
| 🟠 Headache + vomiting that won't stop, can't keep fluids down, dark urine, dizziness when you stand up | Possible dehydration | Call your prescriber or urgent care today. GLP-1 labels specifically warn about kidney problems from severe dehydration. |
| 🟡 Headache + shakiness, sweating, hunger, or confusion — and you take insulin or a sulfonylurea for diabetes | Possible low blood sugar | Treat the low blood sugar first. If you can safely swallow, the standard 15-15 rule is 15 g of fast-acting carbs, wait 15 minutes, then recheck. Then call your prescriber. |
| 🟢 Mild-to-moderate headache that started after your first dose or a dose bump, no red flags, you can drink and eat | Common adjustment-period headache | Hydrate, eat a real meal, rest, take an OTC pain reliever if it's safe for you. Track the timing. Keep reading — the protocol below works for most people in this bucket. |
If you landed in the green row, the rest of this page is for you. If you landed anywhere else, act on it first, then come back when you’re stable.

What we actually verified for this page
- ✅ Drug-by-drug headache rates pulled from current FDA prescribing information on DailyMed for Wegovy (injection and tablets), Ozempic, Rybelsus, Zepbound, Mounjaro, Saxenda, Victoza, Trulicity, and Foundayo. Re-verified May 4, 2026.
- ✅ FDA safety communications on compounded GLP-1 products and the February 20, 2026 FDA warning letter to MEDVi (Letter #721455).
- ✅ Mechanism evidence from peer-reviewed reviews (PMC12558699, PMC11823005) and the Braca et al. liraglutide migraine pilot (PMC12638507).
- ✅ Red-flag guidance based on Cleveland Clinic, NHS, and standard emergency-medicine criteria.
- ⚠️ Editorial conclusions — which mechanism is most likely for which symptom pattern, the 48-hour protocol structure, the prescriber message template — are our framework based on the verified facts above. They are flagged as such.
We have no financial relationship with any GLP-1 manufacturer. We do have affiliate relationships with some telehealth platforms — clearly marked when they appear later on this page — and they do not influence the medical content above.
When a GLP-1 headache is actually an emergency
Most GLP-1 headaches aren’t dangerous. But some are. Tell the difference by looking at what comes with the headache, not the headache alone.
Call 911 or go to the ER right now if you have any of these
These are emergencies in any person, on any medication. They are not normal GLP-1 side effects.
- Sudden, severe (“thunderclap”) headache that hits in seconds — like nothing you’ve felt before
- Headache + neck stiffness + fever (could be meningitis)
- Vision changes — sudden blurriness, double vision, partial vision loss, severe eye pain
- Confusion, slurred speech, weakness on one side, facial droop (stroke symptoms — call 911 even if it passes)
- Fainting, seizure, or loss of consciousness
- Persistent vomiting where you can’t keep down water for hours
- New severe headache during pregnancy or right after delivery
- Headache after any kind of head injury, even a minor bump
- Severe upper-belly pain that wraps around to your back — this can be pancreatitis, a known (rare) GLP-1 risk and a medical emergency
Call your prescriber within 24 hours if
- The headache is unchanged after 48 hours of trying the steps below
- It’s brand new for you and feels different from any headache you’ve had before
- It’s getting worse, not better
- It comes back every time you inject or every time you bump your dose
- You’re taking insulin or a sulfonylurea and you wake up with morning headaches
- You’re using a compounded GLP-1 and the dosing instructions, syringe, or product look different from what you expected
Does GLP-1 actually cause headaches? The drug-by-drug evidence
Quick answer: Yes — for some GLP-1 medications more than others. Wegovy, Saxenda, Victoza, and Foundayo labels list headache as a common adverse reaction with specific trial percentages. Ozempic, Rybelsus, Zepbound, Mounjaro, and Trulicity don’t show headache in their main “common adverse reactions” tables — those drugs discuss headache in postmarketing reports or as a possible low-blood-sugar symptom. So “common” depends on which drug.
The GLP-1 headache evidence matrix
| Medication | Active drug | What the FDA label shows | Headache rate (drug vs. placebo) | Source |
|---|---|---|---|---|
| Wegovy® (injection, 2.4 mg) | semaglutide | Listed as a common adverse reaction | 14% adults vs. 10% placebo; 17% adolescents vs. 16% placebo | DailyMed |
| Wegovy® (injection, HD 7.2 mg) | semaglutide | Listed as a common adverse reaction | 9% (7.2 mg) vs. 8% (2.4 mg) vs. 7% placebo | DailyMed |
| Wegovy® tablets (1.5, 4, 9, 25 mg, daily oral) | semaglutide | Adverse-reaction profile reported as similar to Wegovy injection table | Similar pattern to Wegovy injection rates | DailyMed |
| Rybelsus® / Ozempic® tablets (oral semaglutide for diabetes) | semaglutide | Not in the ≥5% common adverse-reaction table; postmarketing nervous-system reports | Frequency cannot be reliably estimated from postmarketing data | DailyMed |
| Ozempic® (injection, up to 2 mg, for diabetes) | semaglutide | Not in the ≥5% common adverse-reaction table; appears in postmarketing reports | Reported but not a top side effect | DailyMed |
| Saxenda® (injection, daily) | liraglutide | Listed as a common adverse reaction | 13.6% drug vs. 12.6% placebo | DailyMed |
| Victoza® (injection, daily, for diabetes) | liraglutide | Listed as a common adverse reaction | 11% (1.2 mg) and 10% (1.8 mg) vs. 7% placebo | DailyMed |
| Zepbound® (injection) | tirzepatide | Not in the common adverse-reaction table; listed in Medication Guide as a possible low-blood-sugar symptom | A 2025 meta-analysis found no increased headache risk vs. placebo (OR 1.00) | DailyMed; Frontiers in Endocrinology, 2025 |
| Mounjaro® (injection, for diabetes) | tirzepatide | Not in the common adverse-reaction table; listed in Medication Guide as a possible low-blood-sugar symptom | Similar to Zepbound — no consistent excess over placebo in pooled analyses | DailyMed |
| Foundayo™ (oral pill, FDA-approved April 1, 2026) | orforglipron | Listed as a common adverse reaction (≥5%) | 7% placebo, 8% at 5.5 mg, 9% at 9 mg, 9% at 17.2 mg | DailyMed |
| Trulicity® (injection, for diabetes) | dulaglutide | Listed in the Medication Guide as a possible low-blood-sugar symptom | Not in the ≥5% common adverse-reaction table | DailyMed |
| Compounded semaglutide / tirzepatide | semaglutide / tirzepatide | No FDA-approved label exists for any compounded version. FDA reports dosing errors, salt-form issues, and adverse events. | No trial-quality rate data exists | FDA safety communications |
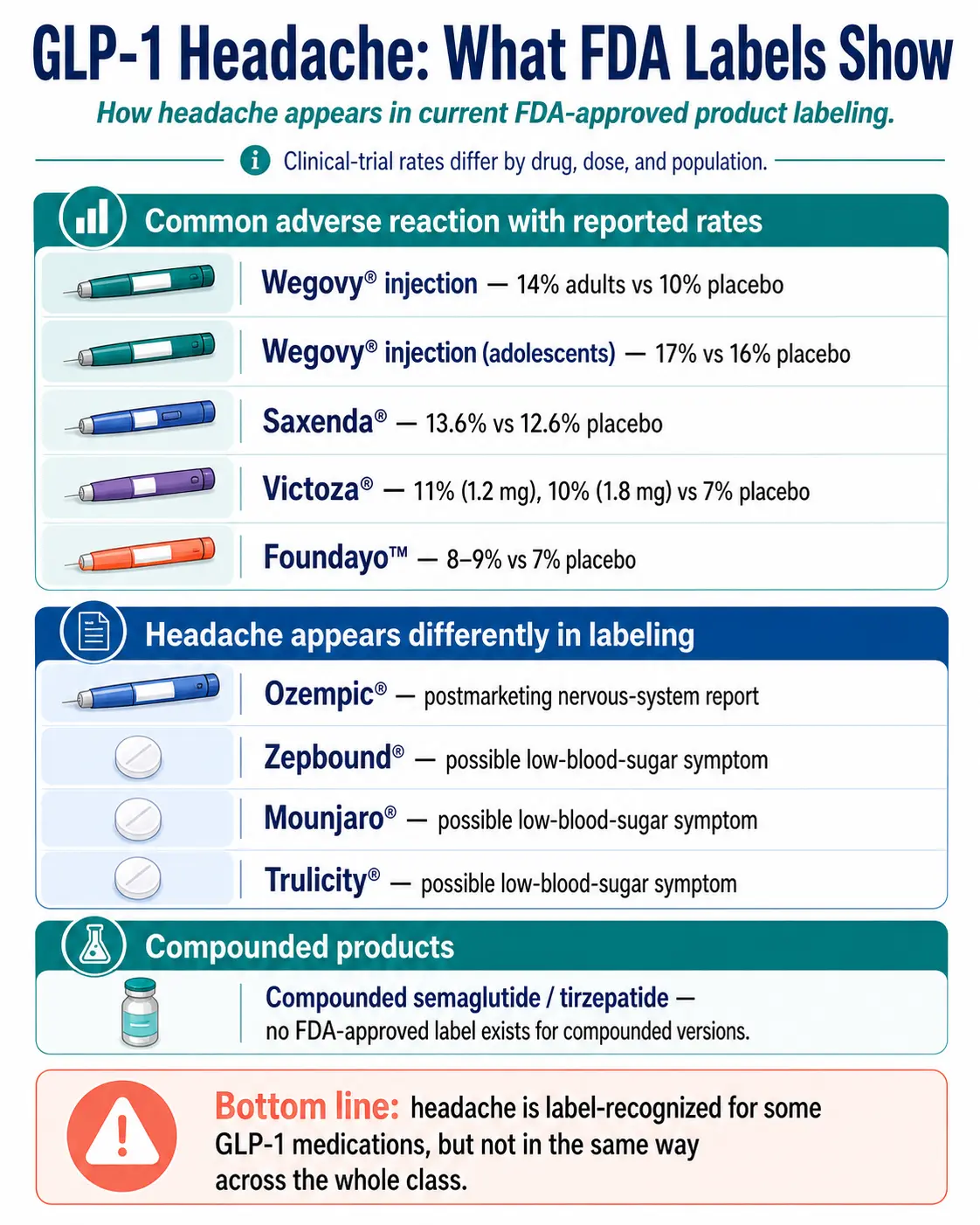
How to read this table without freaking out
- “Common adverse reaction” with specific percentages (Wegovy, Saxenda, Victoza, Foundayo) means the drug company actually counted headaches in trials and reported them. The numbers are what they are.
- “Postmarketing report” or “hypoglycemia symptom” (Ozempic, Rybelsus, Zepbound, Mounjaro, Trulicity) means headaches happen, but they weren’t common enough in trials to land in the main side-effects table. For these drugs, your headache might really be coming from low blood sugar, dehydration, or skipped meals — especially if you also take insulin or a sulfonylurea.
- For compounded products, there is no FDA-approved label for the compounded version. The FDA has flagged real concerns including dosing errors, salt-form questions, refrigeration failures, and counterfeit or fraudulently labeled products. Brand-label rates should not be treated as compounded-product rates.
The big takeaway: a headache on Wegovy, Saxenda, Victoza, or Foundayo is well-documented and somewhat expected. A headache on Zepbound, Mounjaro, Ozempic, or Rybelsus is more often a clue to look at hydration, food intake, and blood sugar than at the drug itself. That changes what you do about it.
Semaglutide headache rates: Wegovy vs. Ozempic vs. Wegovy tablets vs. Rybelsus
Semaglutide is the same molecule across these products, but the labels look different because the indication, dose range, and trial populations differ. Wegovy injection (for weight management) has the clearest headache data — 14% in adults, 17% in adolescents — because it was studied in obesity trials. Ozempic (semaglutide for diabetes, up to 2 mg) doesn’t list headache as a common adverse reaction; it shows up only in postmarketing reports. Rybelsus is the same: postmarketing reports, not the main reactions table. Wegovy tablets list adverse-reaction profiles similar to the Wegovy injection.
So if you’re on Wegovy and have a headache, the label expects it. If you’re on Ozempic or Rybelsus, your headache is less likely to be a direct semaglutide effect and more likely to be a hydration, low-blood-sugar, or skipped-meal issue.
Tirzepatide headache rates: Zepbound vs. Mounjaro
Tirzepatide is the active drug in both Zepbound (for weight loss and obstructive sleep apnea) and Mounjaro (for type 2 diabetes). Neither product’s current DailyMed label lists headache as a common adverse reaction. Both list headache only as a possible low-blood-sugar symptom in the Medication Guide. A 2025 systematic review and meta-analysis in Frontiers in Endocrinology found no increased headache risk with tirzepatide vs. placebo (odds ratio 1.00, 95% CI 0.84–1.20). That doesn’t mean you can’t get a headache on Zepbound or Mounjaro — you absolutely can — but the most likely cause is something downstream (low fluid intake, low blood sugar with diabetes meds, skipped meals, or rapid weight loss), not the drug acting directly on your brain.
Why GLP-1 medications cause headaches
Quick answer: For non-emergency GLP-1 headaches, the most practical buckets to consider are dehydration and low fluid intake, low blood sugar risk (especially with insulin or sulfonylureas), dose-escalation timing, migraine susceptibility, and less-established CNS or vascular mechanisms still under study. The evidence is strongest for the first three.
1. Dehydration and low intake (the best-supported cause)
What’s happening: GLP-1s slow gastric emptying, suppress appetite, and can cause nausea — especially the first few weeks. You drink and eat less without realizing it. Mild dehydration is one of the most common headache triggers in the general population, and several GLP-1 labels (including Wegovy, Saxenda, Foundayo, and Zepbound) specifically warn about kidney problems from severe dehydration.
How to recognize it: Dry mouth, dark yellow urine, feeling tired, headache that gets noticeably better within an hour of drinking 16+ ounces of water.
Fix: Drink steadily through the day — about 64 to 80 ounces (2 to 2.5 liters) is a reasonable target for most adults without fluid restrictions. Add electrolytes if you’re losing fluids to nausea or diarrhea.
2. Low blood sugar (a real risk for some, not most)
What’s happening: GLP-1s on their own rarely cause low blood sugar. But if you’re also taking insulin or a sulfonylurea (drugs like glipizide, glimepiride, or glyburide), the combination can drop your blood sugar in ways that show up as headache, shakiness, sweating, sudden hunger, irritability, or confusion.
How to recognize it: Headache hits before a meal, with shakiness or sweating, especially in the morning. You’re on diabetes medications other than just the GLP-1.
Fix: If you can safely swallow, the standard 15-15 rule: 15 grams of fast carbs (juice, glucose tabs, regular soda), wait 15 minutes, then recheck. After you’re stable, call your prescriber — the dose of your other diabetes medications often needs to come down once you start a GLP-1.
3. Dose escalation and adjustment
What’s happening: GLP-1 dosing schedules typically bump you up every four weeks (semaglutide goes 0.25 → 0.5 → 1.0 → 1.7 → 2.4 mg; tirzepatide goes 2.5 → 5 → 7.5 → 10 → 12.5 → 15 mg; Foundayo goes 0.8 → 2.5 → 5.5 → 9 → 14.5 → 17.2 mg). Each new dose is a small adjustment. Headaches that hit for a few days after each escalation and then settle are common.
How to recognize it: Headache started within 1–7 days of a dose increase, settled before or around the next week.
Fix: Track the pattern. If it’s severe, ask your prescriber about extending titration intervals from 4 weeks to 6 or 8 weeks per step. That’s a totally normal request.
4. Migraine or headache susceptibility
What’s happening: Some people already have migraine or tension-headache patterns. GLP-1-related changes in appetite, sleep, hydration, and nausea can overlap with migraine triggers. Some research even suggests GLP-1s affect the trigeminal pain system and CGRP (a key migraine pathway) — and counterintuitively may help some chronic migraine sufferers (more on that paradox below).
How to recognize it: One-sided throbbing pain, light or sound sensitivity, nausea that feels different from your GLP-1 GI nausea, sometimes visual aura.
Fix: This is a “talk to your prescriber and your neurologist” scenario, not a self-management one.
5. Possible CNS or vascular mechanism (still under study)
What’s happening: A 2025 peer-reviewed review (PMC12558699) describes how GLP-1 receptors in the brain may influence cerebrovascular tone and regional blood flow. However, at least one randomized human GLP-1 infusion study found no cerebral hemodynamic change and did not induce headache compared with placebo. So this mechanism is proposed, not proven.
How to recognize it: Headache hits 24–72 hours after your injection, without a clear hydration, food, or blood-sugar explanation. A published case series (PMC12395549) documented this timing pattern in three tirzepatide patients.
What to try: Injecting in the evening (so the peak window happens during sleep) and rotating injection sites (abdomen, thigh, upper arm). Track the pattern and discuss with your prescriber.
How long do GLP-1 headaches last?
Quick answer: There’s no single label-backed duration that applies to every GLP-1 headache. The pattern most people experience is that adjustment headaches show up in the first 1–2 weeks, return briefly for a few days after each dose increase, and ease as the body settles at a stable dose.
| Time point | What usually happens |
|---|---|
| Day 0 | First injection or dose increase |
| Day 1–3 | Some users get a "post-injection" headache, sometimes peaking 24–72 hours after the shot |
| Week 1–2 | Initial adaptation phase — headaches most common here |
| Week 4 | First standard dose escalation — headache may briefly return for 1–7 days |
| Stable maintenance dose | Most people find symptoms have faded; new headaches at this point usually have a non-GLP-1 cause |
| Persistent at stable dose | Worth a clinical workup for other causes — don't keep pushing |
Why it’s worse the day after your shot. Subcutaneous semaglutide and tirzepatide hit their peak blood levels roughly 24 to 72 hours after injection. The “I’m fine when I shoot, headache on Wednesday” pattern is real and common. Shifting your shot to evening helps a lot of people, because the pharmacological peak then lines up with sleep.
Why each dose increase brings it back. Each new dose is a small adjustment period for your body. Headaches that hit for a few days after each step and then settle are extremely common. If yours isn’t settling, that’s a conversation with your prescriber about slowing the titration.
About FAERS data. The FDA’s FAERS pharmacovigilance database reported a median time-to-onset of 16 days across the neuropsychiatric adverse events it studied, and found reporting signals for both headache and migraine with GLP-1 medications. Important caveat: FAERS measures reports, not incidence. It can’t prove causation or true frequency. But the 16-day median lines up with what most users describe.
How do you get rid of a GLP-1 headache? The 48-hour action protocol
Quick answer: For a GLP-1 headache without red flags, the right protocol is hydrate aggressively in the next 30 minutes, eat a real meal, rest, take an OTC pain reliever that’s safe for you, track when it started — and reassess at the 4-hour, 24-hour, and 48-hour marks. If it’s not significantly better at 48 hours, message your prescriber.
The next 30 minutes
- Drink 16–20 ounces of water (about 500 ml). Sip, don’t chug, especially if you’re nauseated.
- Eat something with protein and complex carbs. Greek yogurt with berries. Peanut butter on whole-grain toast. Hard-boiled egg with crackers. Whatever you can keep down.
- Get to a quiet, low-light environment for 30 minutes if you can.
- Do NOT take an extra dose to “make up” for anything. Do NOT skip your next scheduled dose without talking to your prescriber.
The next 4 hours — pain reliever decision tree
General guidance only — confirm with your pharmacist or prescriber if you are unsure.
- Acetaminophen (Tylenol) is commonly used for adjustment-period headaches; no GLP-1-specific interaction was identified in the labels we reviewed. Follow the product label. Avoid if you have liver disease, heavy alcohol use, or take other acetaminophen-containing products.
- Ibuprofen / NSAIDs (Advil, Motrin, Aleve) are effective for many headaches but have bleeding, stomach, and kidney considerations. Don’t take them if you’re dehydrated. Skip if you have a history of GI ulcers, kidney disease, or are on blood thinners.
- Aspirin has bleeding and stomach-risk considerations and isn’t right for everyone. Use only if it’s already safe for you.
- Avoid prescription migraine drugs (triptans, ergotamines) without checking with your prescriber. Interactions vary.
Keep sipping water. Aim for 1.5 to 2 liters total over the day. Skip alcohol. Skip the extra coffee — caffeine swings can make headaches worse.
The next 24 hours
- If it’s improving: keep going. Don’t change your dose.
- If it’s unchanged: start a log — when it started, time of last dose, what you’ve eaten, what you’ve drunk, what you’ve tried, and any other symptoms.
- If injection day is the trigger pattern: plan to inject at a different time of day next week (evening usually works best), and pick a different injection site.
The 48-hour mark — prescriber message template
Send your prescriber a message. Not a panicked one — a specific one:
Hi, I'm taking [medication] at [dose]. My last dose was on [day] at [time]. I've had a headache since [day]. It feels [throbbing / pressure / one-sided / dull / sharp] and is about a [X]/10 in pain. What's going on alongside it: [nausea, vomiting, dizziness, diarrhea, vision issues, low fluid intake — list everything]. Today I've had [amount of water] and eaten [brief food list]. I'm [taking / not taking] insulin or a sulfonylurea. This is the [first time / 2nd time / it's happened after every dose] this has happened. Should I change anything about my dose schedule, hydration plan, or anything else before my next dose?
Copy that, fill in the blanks, send it. You’ll get a useful answer in hours instead of days, because you’ve given them everything they need to actually help you.
Is it dehydration, low blood sugar, migraine, or the medication itself?
It’s a pattern question, not a single-cause question. Here’s the symptom-pattern decoder.
| What you’re feeling | Most likely cause | Your move |
|---|---|---|
| Headache + dry mouth + dark urine + dizzy when standing + reduced peeing | Dehydration | Fluids if you can keep them down; urgent care if you can't |
| Headache + shakiness + sweating + hunger + you're on insulin or a sulfonylurea | Low blood sugar | 15 g fast carbs immediately, recheck blood sugar in 15 min, then call prescriber |
| Headache 24–72 hours after your injection, otherwise feeling fine | Possible CNS or post-injection pattern | Track pattern; ask prescriber whether evening injection or site rotation makes sense |
| Headache 1–7 days after a dose increase | Adjustment / dose escalation | Use the 48-hour protocol; ask prescriber about slower titration if it's severe |
| One-sided throbbing + light sensitivity + migraine history | Migraine triggered by GLP-1-related changes | Use your usual clinician-approved migraine plan; loop in your neurologist |
| Sudden severe headache + neurological symptoms (any) | Emergency — not a GLP-1 effect | 🚨 911 / ER. Now. |
The three-question self-check
- Is it dangerous? (Red flags from the emergency section above.) → If yes, get help.
- Is there a pattern? (Tied to dose, time of day, food, hydration, dose increases.) → If yes, that pattern points to the cause.
- Is it solvable? (Hydration, meals, dose timing, prescriber adjustment.) → For most green-bucket users, yes — within 48 hours of the right move.
Should you skip a dose, lower your dose, or stop your GLP-1?
Quick answer: Don’t change your GLP-1 dose based on internet advice (including this page) unless it’s an emergency. For mild-to-moderate headaches in the first weeks at a stable dose, continue tracking and use the 48-hour protocol if you’re improving. For severe or persistent headaches that don’t respond, slowing your dose or switching to a different GLP-1 is a reasonable conversation — with your prescriber.
The micro-commitment that beats panic
Before you decide GLP-1 is “not for you,” do exactly two things:
- Run one full 48-hour protocol.
- Send one specific prescriber message (template above).
Many people who quit at the first headache quit unnecessarily — and miss results that would have come with another two weeks of patience.
The paradox: GLP-1 medications may also reduce migraines for some people
Quick answer: Counterintuitive but true. Early clinical research suggests GLP-1 medications may help some chronic migraine sufferers. A 2025 pilot study at the University of Naples found that liraglutide cut monthly headache days nearly in half (from ~20 to ~11) in people with chronic migraine and obesity, even when weight loss was minimal. The proposed mechanism: GLP-1s may lower intracranial pressure and influence the trigeminal pain system. This is investigational — GLP-1s are not FDA-approved for migraine. But it explains why some people with migraine history actually feel better on a GLP-1 over time, while feeling worse during the early titration phase.
The 2025 Braca et al. pilot study (published as PMC12638507) followed adults with high-frequency or chronic migraine plus obesity who took liraglutide as an add-on to their usual migraine prevention. Over 12 weeks:
- Monthly headache days dropped from about 20 to 11
- Migraine disability scores improved by 35 points on a standard scale
- Weight loss was minimal (BMI dropped from 34.0 to 33.7 — not statistically significant)
- The headache benefit was independent of weight loss, pointing to a direct neurological mechanism
GLP-1 receptors in the choroid plexus help regulate intracranial pressure — and elevated intracranial pressure is now thought to play a role in chronic migraine that doesn’t respond to standard prevention. GLP-1 receptor agonists also reduce CGRP expression in chronic migraine animal models — and CGRP is the exact pathway targeted by the newest migraine prevention drugs.
If you have a migraine history and you’re afraid to start a GLP-1: the early-phase risk is real, but it’s not the whole picture. Talk to your neurologist before starting, and don’t quit at week 2 if the headaches are titration-related. None of this is a treatment recommendation. It’s context most pages don’t give you.
Does it matter if your GLP-1 is compounded or FDA-approved?
Quick answer: Yes, and the reason matters. Compounded GLP-1 products are NOT FDA-approved finished medications. They do not have FDA-approved product labels, and the FDA has not reviewed them for safety, effectiveness, or quality before they reach you. Brand-label headache rates should NOT be treated as compounded-product rates.
The regulatory reality
- Compounded semaglutide and tirzepatide are prepared by licensed compounding pharmacies under a clinician’s prescription.
- They are not FDA-approved as finished products.
- They are not generic drugs — generics go through FDA review; compounded drugs don’t.
- The FDA has issued multiple safety communications flagging dosing errors, questionable salt forms (semaglutide sodium and acetate aren’t FDA-approved active ingredients), refrigeration failures, and counterfeit or fraudulently labeled products.
- The FDA’s February 20, 2026 warning letter to MEDVi (Letter #721455) specifically called out language implying compounded products were equivalent to FDA-approved medications — because they are not.
What this means for your headache:
- If you’re on compounded and your headache is unusually severe, don’t assume the brand-name label rates apply.
- Ask your prescriber to verify the concentration, dose in milligrams, and syringe markings match what they intended.
- Ask whether the pharmacy is state-licensed.
- Follow the storage instructions for the specific product you received.
- If the answer to any of those is “I’m not sure,” that’s worth flagging.
If you’d rather just be on FDA-approved brand-name medication and skip the compounded ambiguity entirely:
Ro carries FDA-approved Zepbound® (tirzepatide) and Foundayo™ (orforglipron). They include an insurance concierge to handle prior authorization paperwork and offer a free GLP-1 Insurance Coverage Checker. Ro Body membership is $39 for the first month, then as low as $74/month with annual prepay or $149/month on a monthly plan; GLP-1 medication is billed separately.
Check Your GLP-1 Insurance Coverage with Ro (Free) →Does your provider matter when you have GLP-1 headaches?
Quick answer: Your medication is your medication — switching telehealth platforms won’t change the molecule. But provider quality dramatically affects how fast you get help when side effects happen. If your current program won’t return your messages or won’t discuss titration adjustments, that’s a fixable problem independent of the medication.
Most “GLP-1 headache” complaints we see online aren’t really about the medication. They’re about programs that take 5 days to answer a side-effect question, no clear written dose-escalation schedule, pharmacy source not disclosed with compounded products, pricing that jumps when you titrate up, and no path to a real prescriber when something goes wrong. If you’re nodding: you don’t have a GLP-1 problem. You have a program-fit problem.
| Feature | Why it matters when you have headaches |
|---|---|
| Clinician messaging that responds in <24 hours | You need answers fast when a side effect hits |
| Written dose-escalation schedule, in advance | Removes a major source of "is this normal?" anxiety |
| Pharmacy source named on the product | Especially critical with compounded products |
| Flat-rate pricing across doses | Means you can request slower titration without it costing more |
| Easy cancellation, transparent billing | Reduces pressure to push through unsustainable side effects |
| 24/7 support access, not just a portal queue | Side effects don't follow business hours |
We are Weight Loss Provider Guide, an independent comparison resource for GLP-1 telehealth providers. We don’t run a clinic. We don’t sell GLP-1 medications. We’ve reviewed over 30 telehealth programs. The shortlist below is the one we most commonly recommend for accessible clinician communication and clear dose handling. We have affiliate relationships with these — that’s why this section is toward the end of the page, after the medical content, instead of at the top.
Eden — flat-pricing, FDA-approved brand-name access
Strongest broad-default option for FDA-approved brand-name access. Eden’s published prices for FDA-approved medication are $1,695/month for Wegovy and $1,399/month for Zepbound, both HSA/FSA eligible, with no membership fees on top and same price every dose. Messaging-based clinical support. Best fit if you want predictable pricing, FDA-approved medication, and no surprise charges when you titrate up.
Check Eden’s Flat-Rate FDA-Approved Pricing →Ro — FDA-approved + insurance concierge
The right pick if you specifically want FDA-approved brand-name medication (Zepbound®, Foundayo™) and insurance is in play. Insurance concierge handles prior authorization paperwork. Free GLP-1 Insurance Coverage Checker. Ro Body membership is $39 first month, then as low as $74/month with annual prepay or $149/month monthly; GLP-1 medication is billed separately.
Check GLP-1 Insurance Coverage with Ro (Free) →MEDVi — broad menu including compounded options
Broad menu including both FDA-approved and compounded options. 24/7 support team access. Compounded pricing starts at $179 for the first month. Important context: MEDVi received an FDA warning letter on February 20, 2026 (Letter #721455) regarding website language about its compounded GLP-1 products — the letter cited marketing claims that suggested FDA approval or evaluation of compounded products. This was part of an industry-wide enforcement action affecting more than 30 telehealth companies. The letter addressed marketing language, not product safety. If you’re considering MEDVi for compounded products, verify current pharmacy source disclosure, dosing instructions, and labeling before enrolling.
Check Eligibility With MEDVi →Not sure which fits your situation, medication, and insurance?
60-second matching quiz. Accounts for your current medication, budget, insurance status, and side-effect history. No commitment.
Take the Free 60-Second Matching Quiz →If your current program is fine and you just need to manage a headache: skip the providers and use the protocol above. We’re not going to push you to switch when you don’t need to.
Can you prevent GLP-1 headaches from happening in the first place?
Quick answer: You can lower the odds. The four moves with the strongest evidence behind them: hydrate consistently from day one, don’t skip meals even when appetite drops, ask for slower titration if you’re prone to headaches, and inject in the evening with site rotation.
1. Hydrate from day one
- Target at least 64 ounces of water daily (~2 liters), if you don’t have fluid restrictions
- Add electrolytes if you’re losing fluids to nausea or diarrhea
- Headache is one of the earliest dehydration signals. Catching it early is the easiest fix.
2. Don’t skip meals
- GLP-1s suppress appetite. You’ll forget to eat, and skipped meals are a top headache trigger.
- Aim for three modest meals + one snack, even if you’re not “hungry”
- Protein at every meal stabilizes blood sugar and prevents the hangry-headache combo
3. Ask for slower titration
- Standard schedule: 4 weeks per dose step
- Acceptable to ask for 6 or 8 weeks per step if you’ve had headaches at the previous bump
- A good clinician will say yes. A program that won’t accommodate it is a program-fit issue.
4. Inject in the evening, rotate sites
- Evening injection means the 24–72 hour pharmacological peak lines up with your sleep window
- Rotate between abdomen, thigh, and upper arm
- One documented case series found headaches resolved after site rotation
What real GLP-1 users say about headaches
Real patient quotes from public discussion forums, used for language and emotional context only — not as medical evidence. Anecdotes are not proof of cause or cure.
"Without fail, after each injection I have been hit with a debilitating headache."— r/Semaglutide
"Anyone else feel tired with headaches during the next 24-36 hours after each injection?"— r/Semaglutide
"The headaches were always the first few days after injection."— r/Semaglutide
The pattern in all three: 24–72 hours post-injection. That’s the pharmacological peak window. The fix for that pattern isn’t quitting the medication — it’s tracking the timing, trying evening injection, rotating sites, hydrating preemptively the day after, and looping in your prescriber if it persists.
Frequently asked questions
- Is headache a common GLP-1 side effect?
- It depends on the medication. Wegovy lists headache in 14% of adults and 17% of teens (vs. 10% and 16% on placebo). Saxenda lists 13.6% vs. 12.6% placebo. Victoza lists 11% vs. 7%. Foundayo lists 8–9% vs. 7% placebo. Ozempic, Rybelsus, Zepbound, Mounjaro, and Trulicity do not show headache in their main common-adverse-reaction tables — those drugs list headache as a possible low blood sugar symptom or in postmarketing reports.
- Why do I get a headache the day after my GLP-1 shot?
- Subcutaneous semaglutide and tirzepatide hit peak blood levels 24 to 72 hours after injection. Some users report a headache pattern during that window that may relate to pharmacological peak, hydration, or other factors. Hydrate the day before and after, try injecting in the evening, rotate injection sites, and use an OTC pain reliever that is safe for you. If the pattern persists, message your prescriber.
- How long do GLP-1 headaches last?
- For most people, adjustment headaches show up in the first 1–2 weeks and ease as the body settles at a stable dose. Headaches that recur briefly after each dose increase are also common. Headaches that are severe, persistent past several weeks of stable dosing, or interfering with normal life warrant a clinical workup for other causes.
- Can I take Tylenol or ibuprofen with my GLP-1?
- Acetaminophen (Tylenol) is commonly used for adjustment-period headaches, with no GLP-1-specific interaction identified in the labels reviewed; follow the product label and avoid it if you have liver disease, heavy alcohol use, or take other acetaminophen-containing products. Ibuprofen and other NSAIDs work too, but skip them if you are dehydrated, have a history of GI ulcers, kidney issues, or are on blood thinners. Confirm with your pharmacist if you have other medications or chronic conditions.
- Are headaches worse with semaglutide or tirzepatide?
- Wegovy (semaglutide) lists headache as a common adverse reaction at 14% in adults. Tirzepatide (Zepbound, Mounjaro) does not list headache in its common adverse-reaction table, and a 2025 meta-analysis found no increased headache risk vs. placebo. Individual response varies — some people tolerate one molecule far better than the other. A switch between molecules is a reasonable conversation with your prescriber if headaches do not resolve.
- Does Foundayo (orforglipron) cause headaches?
- Yes — Foundayo's FDA label lists headache as a common adverse reaction (≥5%) at 7% with placebo, 8% at 5.5 mg, 9% at 9 mg, and 9% at 17.2 mg. As a once-daily oral pill rather than an injection, Foundayo does not have the post-injection peak pattern that injectable GLP-1s do.
- Can a GLP-1 cause a migraine?
- GLP-1s can be associated with headache and migraine reports, especially during titration. FAERS pharmacovigilance data shows reporting signals for both. Counterintuitively, an early clinical trial showed liraglutide actually reduced monthly migraine days for people with chronic migraine and obesity. Both findings can be true: short-term titration headache and longer-term migraine modulation are different mechanisms. Talk to your neurologist if you have migraine history.
- Should I stop my GLP-1 because of the headache?
- For mild-to-moderate headaches in the first weeks at a stable dose: usually no. Run the 48-hour protocol and let your body adjust. For severe, persistent, or red-flag headaches: stopping or switching is reasonable, and that decision belongs with your prescriber, not the internet. If your current provider is not responsive about side effects, that is a separate fixable problem.
- What if I am using compounded semaglutide or tirzepatide?
- Contact your prescriber or pharmacy if you have headache plus severe GI symptoms, dehydration, or any uncertainty about your dose, syringe markings, or product appearance. The FDA does not review compounded GLP-1 products before they reach you and has flagged real safety concerns including dosing errors and salt-form issues. Compounded products are not FDA-approved finished medications.
- Can dehydration from GLP-1s cause headaches?
- Yes — dehydration and low fluid intake are among the best-supported contributing factors for GLP-1 headaches. GLP-1 medications cause nausea and reduce appetite, which means many users drink less without realizing it. Headache plus dark urine, dry mouth, or dizziness when standing strongly suggests dehydration. Severe dehydration needs medical care because GLP-1 labels specifically warn about kidney problems in this scenario.
- Can a GLP-1 headache be a sign of low blood sugar?
- It can be, particularly if you also take insulin or a sulfonylurea (drugs like glipizide, glimepiride, or glyburide). Multiple FDA Medication Guides — including Zepbound's, Mounjaro's, Foundayo's, and Trulicity's — list headache as a possible hypoglycemia symptom. If your headache comes with shakiness, sweating, sudden hunger, or confusion and you can safely swallow, the standard 15-15 rule is 15 g of fast carbs, wait 15 minutes, then recheck. Then call your prescriber.
- What should I do if my GLP-1 headache happens during pregnancy?
- Don't manage it on your own. GLP-1 medications are not recommended during pregnancy. Wegovy labeling advises stopping at least 2 months before a planned pregnancy. Zepbound and Foundayo labels advise stopping when pregnancy is recognized. New severe headaches during pregnancy or postpartum can indicate serious conditions like preeclampsia. Contact your obstetric provider or go to urgent care.
Still not sure what to do?
If you’ve made it this far, you have more information than 95% of people searching this. You know which bucket you’re in, what’s likely causing your headache, what to do for the next 48 hours, and when to escalate. That’s a complete answer to “GLP-1 headache.”
If you’re still unsure whether your current GLP-1 program is the right fit — particularly if your provider hasn’t been responsive when side effects happen — the simplest next step is the matching quiz.
Still not sure which GLP-1 program is right for you?
The quiz accounts for your current medication, budget, insurance, and side-effect history, and gives you a personalized recommendation. It takes 60 seconds and routes you to options where side-effect support is part of the program, not an afterthought.
Take the Free 60-Second Quiz →No commitment. No spam.
Sources & methodology
We pulled headache rates from each medication’s official FDA prescribing information on DailyMed (the National Library of Medicine’s official label database), the original peer-reviewed trial publications, and FDA safety communications.
Drug labels (DailyMed):
- Wegovy® (semaglutide injection and tablets)
- Ozempic® (semaglutide injection)
- Rybelsus® / Ozempic® tablets (oral semaglutide)
- Zepbound® (tirzepatide injection)
- Mounjaro® (tirzepatide injection)
- Saxenda® (liraglutide injection)
- Victoza® (liraglutide injection)
- Trulicity® (dulaglutide injection)
- Foundayo™ (orforglipron tablets)
Trial publications and meta-analyses:
- Wilding JPH et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. NEJM, 2021 (STEP-1).
- Jastreboff AM et al. Tirzepatide Once Weekly for the Treatment of Obesity. NEJM, 2022 (SURMOUNT-1).
- Efficacy and safety of tirzepatide for weight loss. Frontiers in Endocrinology, 2025.
- Eli Lilly and Company. FDA approves Foundayo™ (orforglipron). April 1, 2026.
Mechanism and pharmacovigilance reviews:
- Headache and GLP-1 receptor agonists. PMC12558699.
- Neuropsychiatric adverse events associated with GLP-1 receptor agonists: a pharmacovigilance analysis of the FDA FAERS database. PMC11823005.
- Braca S, et al. Effectiveness and tolerability of liraglutide as add-on treatment in patients with obesity and high-frequency or chronic migraine. PMC12638507.
- Colorado M, et al. Unanticipated Adverse Events With Tirzepatide: Three Cases. PMC12395549.
FDA safety communications:
- FDA’s Concerns with Unapproved GLP-1 Drugs Used for Weight Loss. FDA.gov.
- FDA Warning Letter to MEDVi, LLC — #721455, dated February 20, 2026. FDA.gov.
Red-flag and emergency-care guidance:
- Cleveland Clinic. Headache: What It Is, Types, Causes, Symptoms & Treatment.
- NHS Inform. Headaches.
- CDC. Treatment of Low Blood Sugar (Hypoglycemia).
Methodology: Where two sources reported different headache rates for the same drug, we used the FDA-label number. DailyMed warns that clinical-trial rates shouldn’t be compared directly across different trials. Editorial conclusions are flagged as such and aren’t represented as clinical guidelines. We re-verify all rates and label data quarterly. Last verification: May 4, 2026. We have no financial relationship with any GLP-1 manufacturer.
Related guides
- Best GLP-1 Telehealth Providers (2026) — full comparison by cost, medication type, and legitimacy.
- GLP-1 Providers With the Best Online Reviews — Trustpilot audit with FDA warning-letter disclosures.
- Compounded vs. FDA-Approved GLP-1 — the full trade-off picture.
- GLP-1 Providers That Accept HSA/FSA — verified acceptance details.
How this page was made and why
Author: WPG Research Team · Publisher: Weight Loss Provider Guide — an independent comparison resource for GLP-1 telehealth providers.
This page was produced by reviewing current FDA/DailyMed labels for the major GLP-1 medications, primary trial publications in peer-reviewed journals, FDA safety communications about compounded GLP-1 products, and standard emergency-medicine references on headache red flags. Public user-language threads from Reddit and similar forums were used only for understanding searcher language and emotional context — they are not used as medical evidence.
We don’t add fabricated “medically reviewed by” credits or invented clinician bylines. If a real licensed clinician reviews and signs off on this page in the future, we’ll add their name and credentials with their permission.
Medical disclaimer: This page is for informational purposes only and does not constitute medical advice, diagnosis, or treatment. Always consult a licensed healthcare provider about your specific situation before changing any medication. If you are experiencing a medical emergency, call 911 or go to your nearest emergency department.