503A vs 503B Compounding Pharmacy: Differences & Safety (2026)
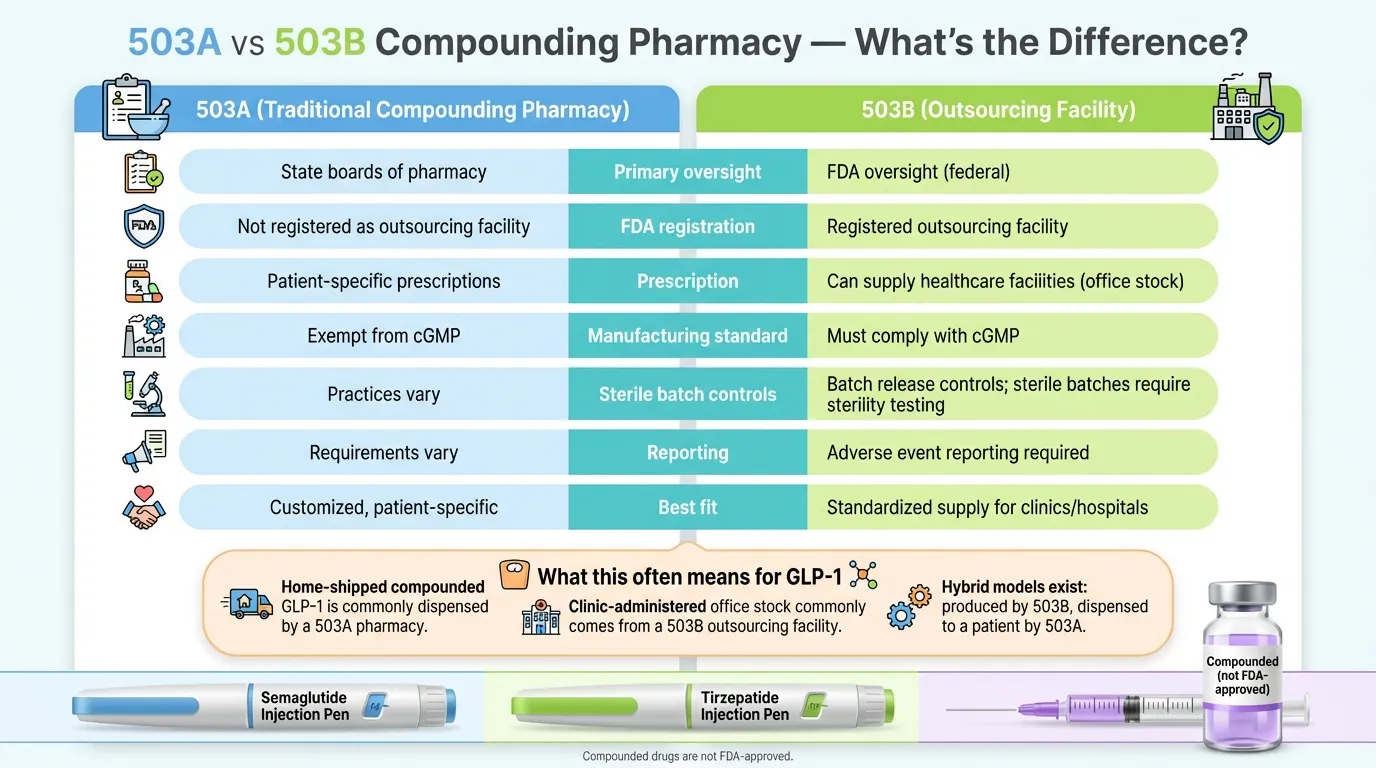
In a 503A vs 503B compounding pharmacy comparison, the core difference is regulation and testing. A 503A compounding pharmacy prepares custom medications for individual patients based on a doctor's prescription and is regulated primarily by state boards of pharmacy. A 503B compounding pharmacy — officially called an outsourcing facility — manufactures medications in large batches under direct FDA oversight and must comply with cGMP, including batch release testing and sterility testing for sterile drug products. 503A pharmacies are exempt from cGMP. Neither type produces FDA-approved medications.
Disclosure: Some links on this site are affiliate links. If you purchase through these links, we may earn a commission at no extra cost to you. Thank you for supporting our site. For informational purposes only—not medical advice.
The bottom line
- 503A pharmacies are state-regulated and prepare patient-specific medications. 503B outsourcing facilities are FDA-registered and follow cGMP manufacturing standards.
- Neither type produces FDA-approved medications — but 503B facilities must comply with cGMP, including batch release testing and sterility testing for sterile drug products.
- The 503B category was created in 2013 after the NECC disaster killed 64 people from contaminated compounded injections.
- Compounding GLP-1 medications (semaglutide, tirzepatide) is now restricted since shortages were resolved — only narrow exceptions remain.
- Verify any compounding pharmacy using the 5-step checklist: confirm designation, check licensing, search for red flags, ask the right questions, and watch for warning signs.

On this page
503A vs 503B Compounding Pharmacy: Side-by-Side Comparison
Before we explain anything, here's the comparison table. If you're short on time, this is what you need.
| Feature | 503A Compounding Pharmacy | 503B Outsourcing Facility |
|---|---|---|
| What it is | Traditional compounding pharmacy | FDA-registered outsourcing facility |
| Created by | Section 503A, FD&C Act (FDAMA, 1997) | Section 503B, Drug Quality and Security Act (2013) |
| Primary regulator | State Board of Pharmacy | FDA (federal) |
| FDA registration required? | No | Yes |
| Routine FDA inspections? | No (risk-based only) | Yes (risk-based schedule) |
| Manufacturing standard | USP 795 / 797 | cGMP (21 CFR Parts 210 & 211) + USP |
| Prescription required? | Yes — patient-specific | Not required for all products |
| Batch size | Small (individual patient quantities) | Large (bulk manufacturing) |
| Who receives the medication? | Patient (home use) | Hospitals, clinics, physician offices, patients (via 503A partners) |
| Batch testing (identity, strength, sterility)? | Not required under federal law | Required under cGMP — including sterility testing for each batch of sterile drug product |
| Process validation? | Not required | Required per cGMP |
| Adverse event reporting to FDA? | Not required | Required |
| Interstate distribution? | Limited (5% cap or state MOU) | Yes |
| Beyond-Use Dating (BUD) | Often USP-default or literature-based unless supported by stability data | Can be supported by a cGMP stability program (testing an adequate number of batches) |
| Can compound during drug shortages? | Yes (with limitations) | Yes (while drug is on FDA shortage list) |
| Can compound "essentially a copy" of FDA-approved drug? | Only when prescriber documents a significant difference for the individual patient | Only while drug is on the FDA drug shortage list |
| Best for | Personalized, patient-specific medications | Standardized bulk supply to healthcare facilities |
Sources: FDA FD&C Act Provisions, FDA Human Drug Compounding Laws, 21 CFR Parts 210–211
So what does this mean for you?
If you're a patient receiving compounded medication shipped to your home, it most likely came from a 503A pharmacy (or was manufactured by a 503B and dispensed through a 503A). If you're a clinic buying pre-made medications for in-office administration, those come from a 503B outsourcing facility.
Either way, the table above is just the starting point. The real question most people are here to answer is: "Is the pharmacy making my medication legitimate and safe?"
That requires verification — not just labels. We'll get there.
What Is a 503A Compounding Pharmacy?
Think of a 503A pharmacy as the traditional neighborhood compounding pharmacy. Your doctor writes a prescription, and the pharmacist prepares a medication specifically for you — adjusting the dose, removing an allergen, or creating a formulation that isn't commercially available.
The name comes from Section 503A of the Federal Food, Drug, and Cosmetic Act (FD&C Act), added by the FDA Modernization Act of 1997. This section created a legal framework for pharmacies that compound medications on a small scale for individual patients.
How a 503A pharmacy works
A licensed pharmacist (or physician) receives a valid, patient-specific prescription. They compound the medication using approved ingredients, following United States Pharmacopeia (USP) standards for quality and sterility. The finished medication goes to the patient, typically for home use.
That's the core of it. One patient, one prescription, one custom medication.
Who regulates 503A pharmacies?
Primarily state boards of pharmacy. Each state sets its own rules for compounding practices, facility standards, and inspections. The FDA does not routinely inspect 503A pharmacies the way it inspects 503B facilities, though it can and does inspect them on a risk-based basis — particularly when there's reason to believe products may be unsafe or conditions may be insanitary.
503A pharmacies must also register with the DEA (if handling controlled substances) but are not required to register with the FDA.
This is a critical distinction. It means the level of oversight varies significantly from state to state. A 503A pharmacy in one state may face rigorous inspections. In another state, oversight may be lighter.
Source: FDA — Compounding Inspections and Oversight FAQ
Quality standards for 503A
503A pharmacies must comply with:
- USP 795 — standards for non-sterile compounding
- USP 797 — standards for sterile compounding (injections, eye drops, etc.)
- USP 800 — standards for handling hazardous drugs
- State-specific regulations
What they are not required to follow: Current Good Manufacturing Practices (cGMP). This means 503A pharmacies are not required to validate their compounding processes, test finished products for potency or sterility, or submit adverse event reports to the FDA.
Some quality-conscious 503A pharmacies voluntarily go beyond minimum requirements — conducting their own testing, implementing quality management systems, and pursuing accreditation from organizations like the National Association of Boards of Pharmacy (NABP). But these measures are voluntary, not mandatory.
Source: FDA — FD&C Act Provisions
Limitations of 503A pharmacies
There are specific legal boundaries:
- Cannot operate as a manufacturer. 503A pharmacies compound for identified individual patients and may compound limited quantities in advance based on a history of valid prescriptions — but cannot manufacture at scale or supply general "office use" stock.
- Cannot distribute for "office use." 503A pharmacies generally cannot supply medications to clinics or hospitals to keep on hand for administration to patients (this is a 503B function).
- Limited interstate distribution. Generally limited to 5% of total prescriptions distributed out of state, unless the state has entered a Memorandum of Understanding (MOU) with the FDA.
- Cannot compound "essentially a copy" of a commercially available drug — unless the prescriber determines and documents a significant difference for the individual patient (e.g., allergen removal, different route of administration).
- Cannot use components from drugs removed from the market for safety or efficacy reasons.
Real-world example
If your telehealth provider prescribes compounded semaglutide with vitamin B12 added — specifically for you, based on your medical evaluation — and a pharmacy fills that individual prescription and ships it to your home, that's a 503A pharmacy at work.
What Is a 503B Outsourcing Facility?
A 503B outsourcing facility operates on a different scale entirely. These are compounding operations that function more like pharmaceutical manufacturers — producing large batches of medications for hospitals, clinics, and physician offices.
The correct technical term is "503B outsourcing facility," not "503B pharmacy." In fact, a 503B facility is not required to be a licensed pharmacy at all (though many are). It must, however, be supervised by a licensed pharmacist.
Why 503B exists: the NECC disaster
Section 503B was created by the Drug Quality and Security Act (DQSA) of 2013, and the reason it exists is grimly specific.
In 2012, the New England Compounding Center (NECC) in Framingham, Massachusetts shipped contaminated steroid injections to clinics across the country. The result: a nationwide fungal meningitis outbreak that killed 64 people and sickened more than 750 others across 20 states.
The investigation revealed NECC was operating as a large-scale manufacturer under the guise of a traditional compounding pharmacy — with virtually no federal oversight. State regulators had missed critical failures. The contaminated medications were never tested for sterility before they shipped.
Congress responded by passing the DQSA, which created a new category of compounder: the 503B outsourcing facility. The idea was simple — if you're compounding at manufacturing scale, you should be held to manufacturing-level standards.
Source: CDC — Multistate Fungal Meningitis Outbreak Investigation, Drug Quality and Security Act (H.R. 3204, 2013)
How a 503B facility works
A 503B outsourcing facility can compound medications in large batches — with or without patient-specific prescriptions. It can sell these medications directly to healthcare facilities (hospitals, clinics, physician offices) for office use. It can also distribute compounded medications through 503A pharmacy partners for patient-specific dispensing.
This flexibility is why 503B facilities play a critical role during drug shortages. When a commercial manufacturer can't meet demand, 503B facilities can scale up production of the needed medication.
Who regulates 503B facilities?
The FDA. Directly.
503B outsourcing facilities must:
- Register with the FDA as an outsourcing facility
- Comply with FDA outsourcing facility registration requirements and maintain any required state licensure; DEA registration applies if the facility handles controlled substances
- Submit to routine FDA inspections on a risk-based schedule
- Report adverse events to the FDA
- Submit product lists to the FDA twice a year
- Follow current Good Manufacturing Practices (cGMP) — the same manufacturing standards that apply to pharmaceutical companies (21 CFR Parts 210 and 211)
- Label products with specific required information, including a statement that the drug is compounded
This is the most important regulatory difference: cGMP compliance is mandatory, not optional.
Source: FDA — Information for Outsourcing Facilities, FDA — Registered Outsourcing Facilities
What cGMP means in practice
"Current Good Manufacturing Practices" is a specific set of FDA-enforced standards. In practice, cGMP requires written procedures for every step of the compounding and testing process, appropriate testing and release controls for each batch, validated or qualified analytical methods, investigations of deviations, and robust recordkeeping. For sterile drug products, each batch must be tested to confirm it meets sterility requirements. Raw material suppliers must be qualified, and environmental monitoring must be conducted regularly in compounding areas.
This is a fundamentally different level of quality assurance than what's required of a 503A pharmacy. It costs more, takes more time, and produces a more standardized, documented product.
Source: 21 CFR Parts 210 and 211, FDA — Compounding Inspections and Oversight FAQ
Real-world example
If a weight loss clinic has pre-filled syringes of compounded tirzepatide ready to administer during your office visit, those likely came from a 503B outsourcing facility. The clinic purchased them in bulk, and each syringe came from a batch that was tested before release.
The Differences That Actually Matter
The table above covers the regulatory framework. But most people reading this aren't looking for a law school summary. They want to know what these differences mean in real, practical terms. Let's break it down by the things that affect you most.
Oversight and accountability
This is the foundational difference.
A 503A pharmacy answers primarily to its state board of pharmacy. The rigor of that oversight depends on the state. Some states conduct thorough inspections with meaningful consequences for violations. Others have limited inspection resources and less aggressive enforcement.
A 503B outsourcing facility answers to the FDA at the federal level. Inspections follow a consistent, risk-based schedule. The FDA publishes inspection findings (Form 483 observations) and warning letters publicly. If a 503B facility is cutting corners, there's a documented federal trail you can look up.
Neither system is perfect. But the 503B model offers a more consistent, publicly transparent oversight structure.
Source: FDA — Compounding Inspections and Oversight FAQ
Testing: what happens before the medication reaches you
This is the difference that matters most for the medication you inject, swallow, or apply.
503A: The finished product is generally not subject to mandatory release testing under federal law. The pharmacy relies on proper compounding technique, appropriate ingredient sourcing, and compliance with USP standards. Quality depends heavily on the individual pharmacy's practices, the pharmacist's skill, and applicable state requirements.
503B: Batch release controls are required under cGMP — including identity and strength testing, and sterility testing for each batch of sterile drug product. If a batch fails, it doesn't ship. The results are recorded and documented.
Does this mean every 503B product is guaranteed safe? No. FDA warning letters and product recalls prove that some 503B facilities have failed to meet cGMP standards — sometimes seriously. But the testing requirement creates a systemic check that doesn't exist in the 503A model.
Does this mean every 503A product is risky? Also no. Many 503A pharmacies operate with rigorous internal quality programs. The point is that you can't assume quality based on the designation alone.
Distribution and access
503A pharmacies fill individual prescriptions and ship to patients (home use). This is the model most telehealth weight loss programs use for direct-to-consumer compounded medications.
503B facilities supply healthcare organizations — hospitals, clinics, surgical centers — with ready-to-use medications. They can also supply 503A pharmacies, which then dispense to individual patients with prescriptions. FDA has issued draft guidance explaining this distribution model.
Many telehealth programs now use a hybrid: a 503B facility manufactures the compounded medication under cGMP, and a 503A pharmacy dispenses it to you with a patient-specific prescription. If your provider uses this model, you're getting a product that was batch-tested at the manufacturing level and dispensed through a licensed pharmacy. Ask your provider which model they use. It matters.
Beyond-Use Dating (shelf life)
503A pharmacies assign Beyond-Use Dates (BUDs) based on published scientific literature or USP default limits — not their own stability testing (unless they choose to conduct it). This typically results in shorter expiration windows for many sterile preparations.
503B facilities can conduct formal stability programs under cGMP, testing an adequate number of batches to support longer expiration dating. This allows them to assign longer BUDs (often months), because they've demonstrated through testing that the product remains stable and potent for that period.
For patients, this means a 503B-sourced medication is more likely to have a longer usable window. For a 503A medication, pay close attention to the BUD on the label and store it exactly as directed.
Cost
Cost is one of the reasons people seek compounded medications in the first place — especially for GLP-1 drugs where brand-name versions can exceed $1,000 per month.
503A pharmacies have lower overhead (no FDA registration fees, less testing infrastructure). Per-unit costs can be lower for simple preparations but may be higher for complex sterile formulations due to the small-batch model.
503B facilities have significantly higher overhead (FDA compliance, batch testing, quality systems). But economies of scale can drive per-unit costs down for standardized products manufactured in volume.
For patients: the price you pay is determined by your provider and pharmacy, not directly by the 503A/503B designation. What matters is understanding why an unusually low price might be a warning sign. More on that in the verification section below.
Which Is Safer: 503A or 503B?
This is the question most searchers actually want answered. And the honest answer requires nuance.
The regulatory reality
503B outsourcing facilities are held to higher regulatory standards — cGMP compliance, mandatory batch testing, FDA inspections, and adverse event reporting. These structural safeguards reduce the risk of contamination, dosing errors, and quality failures at a systemic level.
503A pharmacies operate under less stringent federal requirements. Quality depends more on the individual pharmacy's practices, the competence of the pharmacist, and the effectiveness of state oversight.
But designation is not a guarantee
The NECC disaster that killed 64 people? That was a facility operating under the 503A framework, which is why 503B was created in the first place. The 503B designation exists specifically to prevent that kind of failure at scale.
But 503B facilities are not immune to problems either. In recent years, the FDA has issued warning letters to 503B outsourcing facilities for cGMP violations, sterility failures, and inadequate quality systems. FDA has documented quality problems — including sterility assurance concerns — at some compounding facilities, including outsourcing facilities, through inspections, warning letters, and recalls.
Source: FDA — Warning Letters, FDA Enforcement Reports (2025)
What actually determines safety
The designation is a floor, not a ceiling. What matters more:
- Is the facility licensed and in good standing? (Check state board + FDA registration for 503B)
- Does it test finished products? (Required for 503B; voluntary for 503A — ask either way)
- Has it received FDA warning letters or state enforcement actions? (Publicly searchable)
- Does it use pharmaceutical-grade ingredients from FDA-registered suppliers? (Ask for confirmation)
- Is it transparent about its processes? (A pharmacy that won't answer your questions is a red flag regardless of its designation)
The safest compounded medication comes from a facility that is properly licensed, conducts batch testing, sources quality ingredients, and operates transparently. That can be a 503A or a 503B — but 503B facilities are structurally required to meet more of these criteria.
The FDA's Position
"Compounded drugs are not FDA-approved. This means that FDA does not verify the safety, effectiveness, or quality of compounded drugs before they are marketed or reach patients." — This applies to both 503A and 503B. The difference is that 503B facilities must follow cGMP and submit to FDA inspection.
Source: FDA — Compounding and the FDA: Questions and Answers
How to Verify a Compounding Pharmacy (Your 5-Step Checklist)
This is the part most guides skip. They explain what 503A and 503B mean — then leave you to figure out the rest on your own. Here's how to actually verify whether a specific pharmacy is legitimate.
Step 1: Confirm the designation
Ask the pharmacy or your healthcare provider directly: "Are you a 503A compounding pharmacy or a 503B outsourcing facility?"
If they can't answer immediately and clearly, that's a concern. Every compounding pharmacy knows its own regulatory classification.
Step 2: Verify licensing and registration
For 503B outsourcing facilities:
The FDA maintains a public, searchable list of all registered 503B outsourcing facilities:
If a facility claims to be a 503B but doesn't appear on this list, it isn't one.
For 503A pharmacies:
There is no single federal list of 503A compounding pharmacies. 503A pharmacies are licensed at the state level through their respective state board of pharmacy. To verify a 503A pharmacy, look up its license through your state board's website.
Every state board of pharmacy maintains a license lookup tool. Search "[your state] board of pharmacy license verification" to find it. You can also use the NABP Boards of Pharmacy directory to locate your state board's website. Confirm the pharmacy's license is active, current, and in good standing.
Step 3: Check for red flags in the public record
For 503B facilities:
- Search the FDA Warning Letters database for the facility name.
- Search the FDA Recall database for any product recalls.
- Check for Form 483 observations (inspection findings) — these are available through FOIA requests or often reported in trade publications.
For 503A pharmacies:
- Check your state board of pharmacy for disciplinary actions, violations, or consent orders.
- Search news reports for the pharmacy name + "recall," "warning," or "violation."
Step 4: Ask the right questions
Whether your pharmacy is 503A or 503B, ask:
- "Do you test each batch before release?" For 503B facilities, ask about lot-specific batch release testing (identity, strength) and, if the product is sterile, confirm sterility testing and sterility assurance controls. For 503A, voluntary testing is a strong quality signal.
- "Where do you source your active pharmaceutical ingredients (APIs)?" APIs should come from FDA-registered manufacturers. Ask if they can confirm this.
- "Can you provide a Certificate of Analysis (COA) for my specific batch?" A COA verifies identity, purity, and potency of the specific batch. 503B facilities should have this; some 503A pharmacies can provide it on request.
- "What is your Beyond-Use Date, and how was it determined?" Stability-tested BUDs (503B) are more reliable than literature-based or USP-default BUDs (503A).
- "Does the product use semaglutide base or a salt form?" If you're receiving compounded semaglutide, this matters. The FDA has stated it is not aware of a lawful basis for compounding with semaglutide salt forms (sodium or acetate).
- "Are you accredited by NABP or any other quality organization?" Voluntary but meaningful, especially for 503A pharmacies.
Step 5: Watch for warning signs
Red Flags That Should Stop You
- The pharmacy won't name its specific compounding facility
- Prices are dramatically lower than every competitor (why?)
- The website uses stock photos of medications and doesn't show real facility images
- No verifiable pharmacy license number
- Products are labeled "for research use only" or "not for human consumption"
- The provider claims their compounded medication is "FDA-approved" (it's not — ever)
- No clear process for reporting adverse effects
- Medications arrive without proper labeling, BUD, or lot numbers

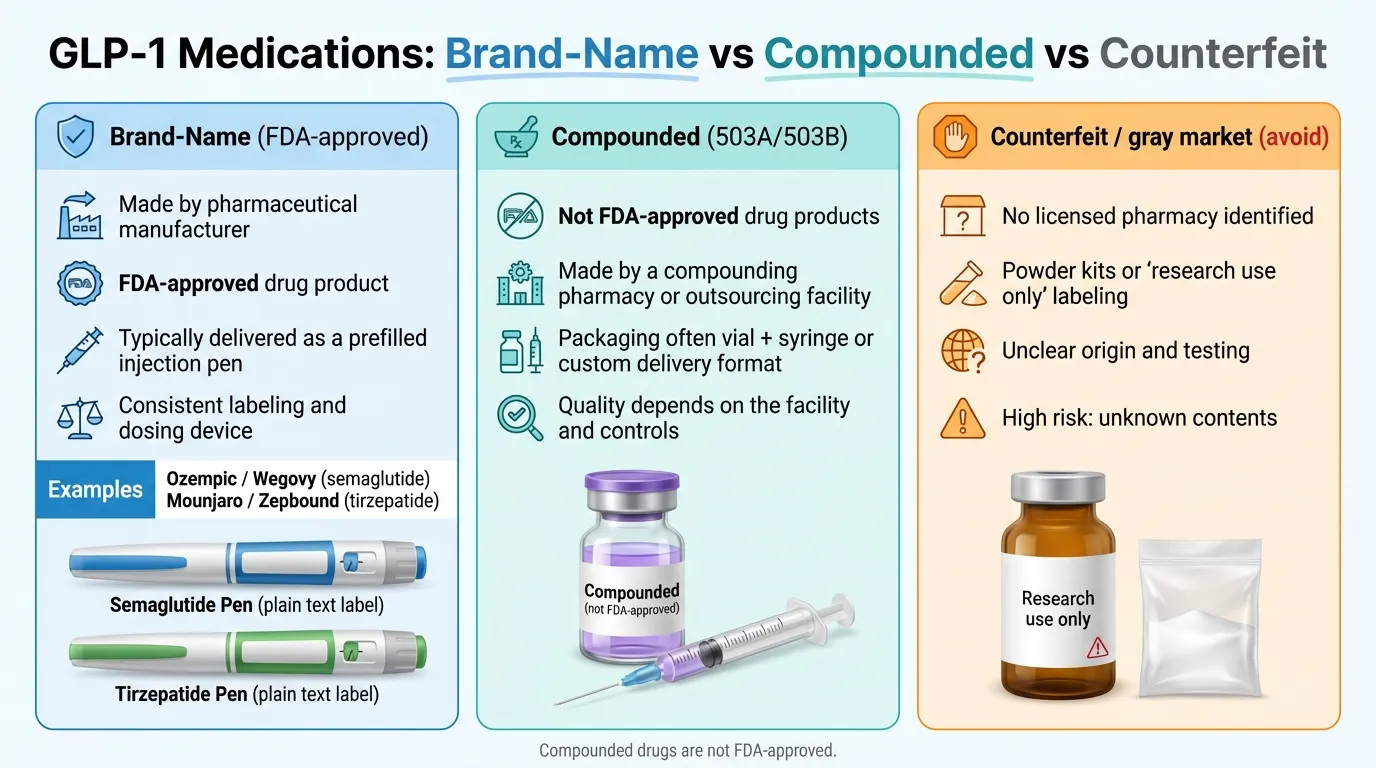
What 503A vs 503B Means for Compounded GLP-1 Medications
The 503A vs 503B distinction has become especially important for compounded GLP-1 medications like semaglutide and tirzepatide. Here's the full picture.
How we got here
Starting in 2022, severe shortages of semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro, Zepbound) created enormous demand for compounded alternatives. The FDA placed both drugs on the drug shortage list, which opened the door for both 503A and 503B compounders to legally produce copies.
During this period, hundreds of telehealth weight loss programs launched, most using 503A pharmacies to compound and ship semaglutide and tirzepatide directly to patients. The market exploded.
What changed
In late 2024 and early 2025, Novo Nordisk and Eli Lilly resolved their supply issues. The FDA initially determined the tirzepatide shortage was resolved and removed it from the shortage list on October 2, 2024. After a legal challenge by the Outsourcing Facilities Association, the FDA re-evaluated its decision and issued a declaratory order on December 19, 2024, re-confirming that the tirzepatide shortage was resolved. Semaglutide was removed from the shortage list on February 21, 2025.
Once a drug is off the shortage list, the legal basis for compounding "essentially a copy" narrows significantly:
- 503B facilities can no longer compound copies of drugs that are not on the shortage list (unless the drug appears on the 503B Bulks List, which semaglutide and tirzepatide do not).
- 503A pharmacies can still compound these drugs — but only if the prescriber determines and documents a significant difference for the individual patient that justifies compounding over the commercial product.
The legal challenges
The Outsourcing Facilities Association (OFA) filed lawsuits challenging both shortage resolution decisions. In the tirzepatide case, the FDA voluntarily agreed to re-evaluate its decision while the case was stayed — it was not blocked by a court order. In both cases, courts denied preliminary injunctions (tirzepatide: March 5, 2025, upheld May 7, 2025; semaglutide: April 24, 2025), clearing the way for full FDA enforcement of compounding restrictions on GLP-1 medications.
Sources: FDA — Clarifies Policies for Compounders as National GLP-1 Supply Begins to Stabilize, OFA v. FDA court rulings (N.D. Tex., 2025)
Can pharmacies still compound GLP-1 medications?
As of early 2026:
- 503A pharmacies can compound semaglutide and tirzepatide if the prescriber determines and documents a significant difference between the compounded version and the commercially available product for the individual patient — meaning the compounded version is not "essentially a copy."
- 503B facilities face tighter restrictions. Without a shortage listing or appearance on the 503B Bulks List, the legal path to compound these drugs at scale is limited.
What 'Significant Difference' Means in Practice
Under FDA guidance, a 503A compounded drug is not considered "essentially a copy" when the prescriber determines there is a "significant difference" between the compounded version and the commercially available product for the individual patient. Examples include: a dose not commercially available, an allergen-free formulation, a different route of administration, or a specific medical need that cannot be met by the approved product. Simply wanting a lower price does not constitute a significant difference.
The September 2025 warning letters
In September 2025, the FDA issued warning letters to multiple compounding pharmacies and telehealth companies for marketing compounded semaglutide and tirzepatide as "generic" versions of Wegovy, Ozempic, Zepbound, and Mounjaro. The FDA specifically targeted:
- Claims that compounded versions were "equivalent" or "the same as" FDA-approved products
- Use of brand names in marketing materials
- Failure to comply with compounding regulations
- Misleading safety claims
State enforcement is ramping up
State attorneys general have also taken action. In August 2025, the Connecticut Attorney General reached a settlement with a company selling "bootleg" GLP-1 products — research-grade peptides marketed for weight loss that were not prepared by licensed pharmacies.
Multiple other states have opened investigations into companies selling compounded or research-grade GLP-1 products without proper licensing.
Source: CT Attorney General — Bootleg GLP-1 Settlement (Aug 2025)
February 2026: FDA escalates enforcement
On February 6, 2026, the FDA announced its intent to take "decisive steps" to restrict GLP-1 active pharmaceutical ingredients (APIs) intended for use in non-FDA-approved compounded drugs that are being mass-marketed as alternatives to approved medications. The announcement specifically named companies including Hims & Hers.
FDA Commissioner Martin Makary stated that the agency will "use all available compliance and enforcement tools" to address violations, including the possibility of seizure and injunction. The same day, the HHS General Counsel referred Hims & Hers to the Department of Justice for investigation of potential federal law violations.
The FDA reiterated that companies cannot claim compounded products are "generic versions" of approved drugs, cannot state they use "the same active ingredient," and cannot claim compounded drugs are "clinically proven to produce results for the patient."
This represents the most aggressive FDA posture on compounded GLP-1 products to date.
Source: FDA — FDA Intends to Take Action Against Non-FDA-Approved GLP-1 Drugs (Feb 6, 2026)
FDA safety concerns specific to compounded GLP-1s
The FDA has documented specific safety concerns with compounded GLP-1 products:
- Dosing errors — confusion between units, milligrams, and milliliters when patients draw medication from vials using syringes
- Sterility failures — contamination risks from multi-use vials and improper compounding practices
- Adverse events — as of July 31, 2025, the FDA had received 605 adverse event reports for compounded semaglutide and 545 for compounded tirzepatide (1,150 total), including some requiring hospitalization
- Potency issues — under-dosing or over-dosing due to compounding errors or lack of batch testing
Source: FDA — Dosing Errors Associated with Compounded Injectable Semaglutide Products, FDA — FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss
FDA Alert: Semaglutide Salt Forms
The FDA has warned that some compounded products use semaglutide salt forms (such as semaglutide sodium or semaglutide acetate) rather than semaglutide base. These salt forms are chemically different from the semaglutide used in FDA-approved products like Wegovy and Ozempic. The FDA considers products containing semaglutide salts to be different active ingredients — meaning they are not simply compounded copies of the approved drug. Products made with semaglutide salts have not been evaluated for safety or efficacy by the FDA.
What to do if you're currently taking compounded GLP-1 medication
If you're currently using compounded semaglutide or tirzepatide:
- Don't panic and don't stop abruptly. Sudden discontinuation can cause side effects. Talk to your prescriber about a transition plan.
- Ask your provider which pharmacy compounds your medication and verify it using the 5-step checklist above.
- Ask your prescriber about the documented significant difference that justifies compounding for your specific situation.
- Explore FDA-approved alternatives. Brand-name GLP-1 medications are now more widely available, and insurance coverage and manufacturer savings programs may reduce out-of-pocket costs for eligible patients. Ask your prescriber or pharmacist about current program terms.
- Monitor for any unusual symptoms — injection site reactions, unusual pain, fever, or other signs of contamination — and report them immediately.
For more on how compounded GLP-1 medications compare to brand-name options, see our complete comparison guide.
FREE ASSESSMENT
Find Your GLP-1 Match
Answer a few questions to see which verified provider fits your needs and budget
Take the Free Quiz503A vs 503B: Which Type Is Right for Your Situation?
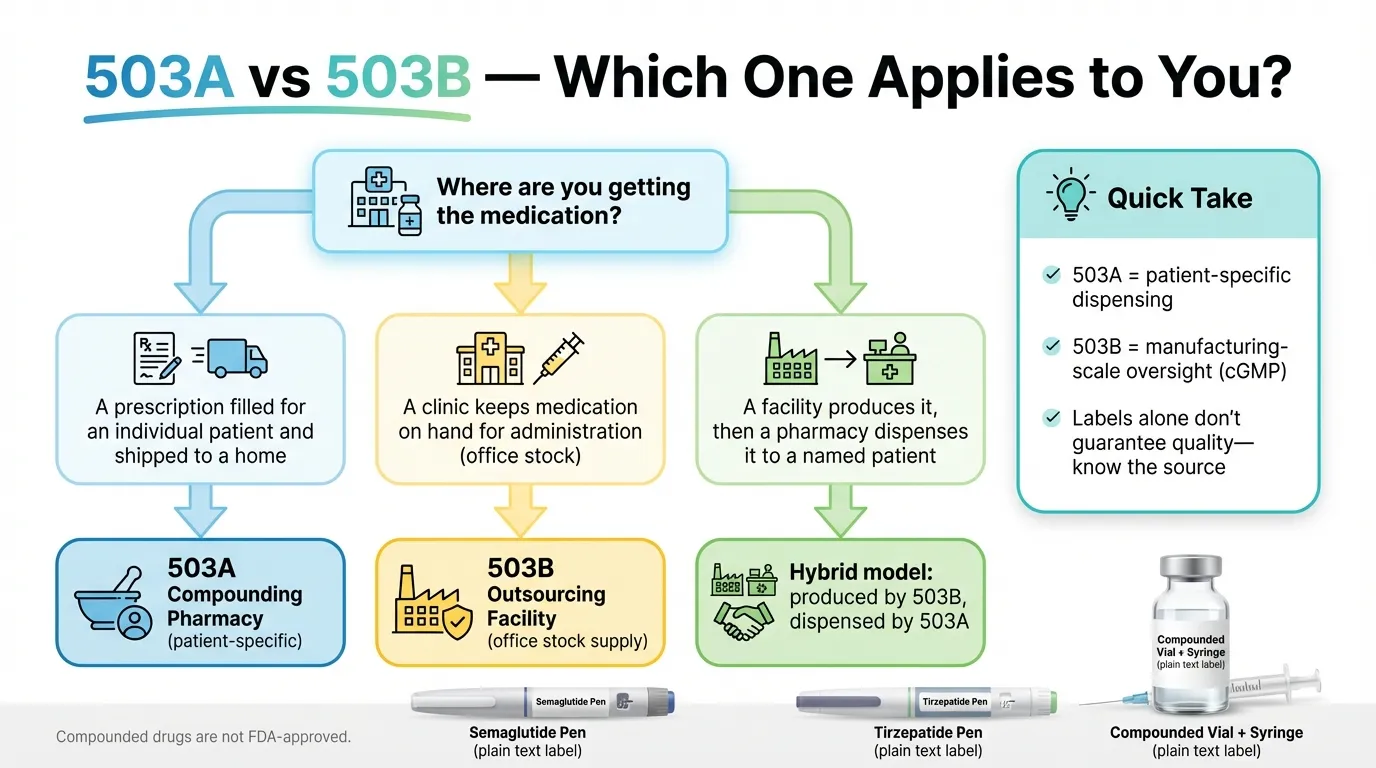
The answer depends on how you're accessing compounded medication and what your specific needs are.
Choose a 503A pharmacy when
- Your doctor writes a patient-specific prescription for a compounded medication
- You need a custom formulation (specific dose, allergen-free, different route of administration)
- You're receiving compounded medication shipped directly to your home
- Your telehealth provider uses a 503A pharmacy to fill individual prescriptions
A 503B outsourcing facility is typically involved when
- A clinic or hospital purchases pre-made medications for in-office administration
- You receive injections at a healthcare facility (the medication was likely sourced from a 503B)
- Your provider needs standardized, batch-tested products for multiple patients
- Large-volume supply is required (surgical centers, hospital pharmacies)
The hybrid model
Many telehealth weight loss programs use the 503B-to-503A model: a 503B outsourcing facility manufactures the medication under cGMP (with batch testing), and a 503A pharmacy dispenses it to you with your individual prescription.
This hybrid model combines the quality advantages of 503B manufacturing with the patient-specific dispensing framework of 503A. If your provider uses this model, ask for confirmation — it's a positive quality signal.
Common Myths About 503A and 503B Pharmacies
Myth: "503B is FDA-approved"
Reality: No. 503B outsourcing facilities are FDA-registered and FDA-inspected. The facility itself is subject to federal oversight and must follow cGMP. But the medications produced by a 503B facility are not FDA-approved. They have not been evaluated by the FDA for safety, effectiveness, or quality through the drug approval process. "FDA-registered" and "FDA-approved" are fundamentally different things.
Myth: "503A pharmacies are unregulated"
Reality: 503A pharmacies are regulated — primarily by state boards of pharmacy. They must comply with USP standards, state compounding regulations, and applicable federal requirements under Section 503A of the FD&C Act. What they lack is federal manufacturing-level oversight (cGMP, FDA inspections, mandatory batch testing). The level of effective oversight varies by state.
Myth: "All compounding is legal"
Reality: Compounding is legal only when specific conditions are met. 503A pharmacies must operate within the limits of Section 503A (patient-specific prescriptions, no essentially copies unless clinically justified, limited interstate distribution). 503B facilities must meet all 503B requirements (registration, cGMP, shortage list rules for certain drugs). Compounding a medication in violation of these conditions is not legal — even if the pharmacy is licensed.
Myth: "Compounded semaglutide is a generic version of Wegovy"
Reality: No. Generic drugs go through an abbreviated FDA approval process (ANDA) that demonstrates bioequivalence to the brand-name product. Compounded semaglutide does not go through any FDA approval process. The FDA explicitly warns against calling compounded GLP-1 products "generic" — this was a primary target of the September 2025 warning letters.
Myth: "Research-grade peptides are the same thing as compounded medications"
Reality: Products labeled "for research use only" or "not for human consumption" are not compounded medications. They are not prepared by licensed pharmacies, are not subject to any pharmacy or manufacturing regulations, and should never be injected or consumed. Multiple state attorneys general have taken enforcement action against companies selling these products for human use.
For Clinics and Prescribers: What to Verify Before Partnering with a Compounder
If you're a healthcare provider, understanding the operational differences between 503A and 503B is essential for both compliance and patient safety.
When you need a 503B outsourcing facility
- Office stock. If you need medications on hand to administer to patients in your clinic (not based on individual prescriptions written in advance), you need a 503B source. 503A pharmacies cannot legally supply medications for office use.
- Bulk orders. Large-volume medication needs for hospital pharmacies, surgical centers, or multi-provider practices.
- Standardized preparations. When you need consistent, batch-tested products across a patient population.
When a 503A pharmacy is appropriate
- Patient-specific prescriptions. When each patient's medication is individually prescribed and dispensed for home use.
- Custom formulations. When the patient requires a unique preparation not available as a standardized product.
Vendor audit considerations
When selecting a compounding partner, evaluate:
- FDA inspection history (for 503B) or state board inspection record (for 503A)
- Quality management systems — SOPs, deviation handling, corrective action processes
- Product testing protocols — what's tested, how often, and by whom
- API sourcing — certificates of analysis, supplier qualification
- Recall and adverse event procedures
- Insurance and liability coverage
For 503B facilities, review any publicly available FDA Form 483 observations or warning letters. For 503A pharmacies, consider whether they hold voluntary accreditation from NABP or equivalent organizations.
Source: NABP — Compounding Pharmacy Accreditation
Questions to ask before partnering
- "Which pharmacy compounds my medication — can you give me the name and state?" You need a specific name to verify licensing.
- "Is it a 503A compounding pharmacy or a 503B outsourcing facility?" This tells you what regulatory framework applies.
- "Is the pharmacy licensed in my state and in good standing?" A pharmacy must be licensed in the state where it dispenses or ships medication.
- "Does the pharmacy perform lot-specific batch release testing (identity, strength) and, for sterile products, sterility testing?" Required for 503B under cGMP. Voluntary but important for 503A.
- "Where does the pharmacy source its active pharmaceutical ingredients?" APIs should come from FDA-registered manufacturers.
- "If the compounded GLP-1 would otherwise be considered 'essentially a copy' of a commercially available FDA-approved product, has the prescriber determined and documented a significant difference for this individual patient?" This is the legal basis for continued 503A compounding now that shortages are resolved.
Questions to Ask Your GLP-1 Provider
If you're considering or currently using a compounded GLP-1 medication, these are the questions that matter most. Print this list or screenshot it before your next appointment.
Before starting treatment
- "Which pharmacy compounds my medication?" Get the specific name, state, and whether it's a 503A or 503B facility. Then verify it yourself using the 5-step checklist above.
- "Why are you prescribing a compounded version instead of the FDA-approved product?" Your prescriber should be able to explain the clinical rationale — a dose adjustment, allergen removal, route change, or other documented medical need.
- "Is this product made with semaglutide base or a salt form?" The FDA has warned that semaglutide salt forms (sodium, acetate) are not the same active ingredient as the semaglutide in Wegovy/Ozempic. Ask for confirmation.
- "What happens if I have a side effect or adverse reaction?" Understand the reporting process. With FDA-approved drugs, adverse events are tracked through established systems. With compounded drugs, reporting may be less structured.
While on treatment
- "Can I see the label and lot number on my medication?" Legitimate compounded medications include proper labeling with the pharmacy name, drug name, strength, BUD, lot number, and storage instructions.
- "How should I store this, and what's the expiration date?" Follow BUD and storage instructions exactly. Compounded medications — especially sterile injectables — can degrade or become contaminated if stored improperly.
- "What should I do if the medication looks cloudy, discolored, or has particles?" Never inject a product that looks abnormal. Contact your pharmacy immediately.
If considering a switch
- "What are my options for transitioning to an FDA-approved GLP-1?" Ask about insurance coverage, manufacturer savings programs, and dosing transition plans.
For a full breakdown of how compounded GLP-1 medications compare to brand-name versions, see our compounded vs. brand-name comparison guide. To compare verified providers offering FDA-approved GLP-1 medications, visit our provider comparison page.
Frequently Asked Questions
What is a 503A compounding pharmacy?
A 503A compounding pharmacy is a traditional pharmacy that prepares customized medications for individual patients based on valid prescriptions. It's regulated primarily by state boards of pharmacy under Section 503A of the Federal Food, Drug, and Cosmetic Act. 503A pharmacies are limited to small-batch, patient-specific compounding for home use and are not required to register with the FDA.
What is a 503B compounding pharmacy (outsourcing facility)?
A 503B outsourcing facility is an FDA-registered entity that compounds medications in large batches under federal oversight. Created by the Drug Quality and Security Act of 2013, 503B facilities must follow current Good Manufacturing Practices (cGMP), submit to routine FDA inspections, comply with batch release testing requirements — including sterility testing for sterile drug products — and report adverse events. They can supply medications to healthcare facilities without patient-specific prescriptions.
What is the difference between 503A and 503B compounding pharmacy?
The core differences are regulation, scale, and testing. 503A pharmacies are state-regulated, prepare medications for individual patients, and are exempt from federal cGMP requirements. 503B outsourcing facilities are FDA-regulated, can manufacture in bulk, and must comply with cGMP — including batch release testing and sterility testing for sterile drug products. See the full comparison table above for a detailed breakdown.
Is a 503A or 503B pharmacy FDA-approved?
No. Neither 503A nor 503B compounded medications are FDA-approved. A 503B outsourcing facility is FDA-registered and FDA-inspected, which means the facility itself is monitored for compliance with manufacturing standards. But the medications it produces have not been evaluated by the FDA for safety, effectiveness, or quality through the drug approval process.
Source: FDA — Compounding and the FDA: Q&A
How do I look up a 503B outsourcing facility?
The FDA maintains a public list of all registered 503B outsourcing facilities at fda.gov/drugs/human-drug-compounding/registered-outsourcing-facilities. If a facility claims to be a 503B but doesn't appear on this list, it isn't one.
Is there a list of 503A pharmacies?
There is no single federal list of 503A compounding pharmacies because they are licensed at the state level, not registered with the FDA. To verify a 503A pharmacy, look up its license through your state board of pharmacy's website. Most state boards have online license verification tools.
Can a 503A pharmacy ship medications across state lines?
Limited. Under Section 503A, a pharmacy generally cannot distribute compounded medications interstate beyond 5% of its total prescriptions — unless the state has entered a Memorandum of Understanding (MOU) with the FDA that establishes a different threshold. Some states have signed these MOUs; many have not.
Source: FDA — FD&C Act Provisions
Can compounding pharmacies still make GLP-1 medications like semaglutide and tirzepatide?
As of early 2026, compounding of semaglutide and tirzepatide is permitted only under narrow conditions. 503A pharmacies can compound these drugs if the prescriber determines and documents a significant difference between the compounded version and the commercially available product for the individual patient — meaning the compounded version is not "essentially a copy." 503B facilities face tighter restrictions because neither drug appears on the 503B Bulks List, and both have been removed from the FDA drug shortage list.
Are compounded GLP-1 medications safe?
The FDA does not evaluate compounded drugs for safety, effectiveness, or quality. The agency has documented specific concerns with compounded GLP-1 products, including dosing errors (confusion between units/mg/mL), sterility failures, and adverse events including hospitalizations. Compounded GLP-1s can be used safely when prepared by licensed, properly regulated pharmacies using pharmaceutical-grade ingredients — but the risk profile is different from FDA-approved products.
Source: FDA — FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss
What does cGMP mean?
Current Good Manufacturing Practices (cGMP) are FDA-enforced standards for manufacturing, testing, and quality assurance of drugs. For compounding, cGMP requires batch release testing and controls, process validation, supplier qualification, and robust recordkeeping. For sterile drug products, each batch must meet sterility testing requirements. 503B outsourcing facilities must comply with cGMP (21 CFR Parts 210 and 211). 503A pharmacies are exempt from cGMP requirements.
What is a Certificate of Analysis (COA)?
A Certificate of Analysis is a document that verifies specific attributes of a medication batch — typically identity, potency, purity, and/or sterility. 503B facilities should have lot-specific release testing data under cGMP. Some pharmacies may provide a Certificate of Analysis or lot testing summary on request. You can ask your pharmacy whether this documentation is available for your specific medication batch.
What is Beyond-Use Dating (BUD)?
Beyond-Use Dating (BUD) is the date after which a compounded medication should not be used. 503A pharmacies assign BUDs based on published scientific literature or USP default limits. 503B facilities assign BUDs based on their own stability testing of the specific product. Longer BUDs from 503B facilities indicate that the product has been demonstrated (through testing) to remain stable and potent for that period.
What was the NECC disaster and why does it matter?
In 2012, the New England Compounding Center (NECC) shipped contaminated steroid injections that caused a nationwide fungal meningitis outbreak — 64 deaths and over 750 infections across 20 states. NECC was operating at manufacturing scale without adequate oversight. Congress responded by passing the Drug Quality and Security Act (DQSA) in 2013, which created the 503B outsourcing facility category with federal FDA oversight. The NECC disaster is the direct reason the 503B designation exists.
Source: CDC — Multistate Fungal Meningitis Outbreak
What is the 503B-to-503A model?
Under FDA draft guidance, 503A pharmacies can purchase compounded medications from 503B outsourcing facilities and dispense them to patients with patient-specific prescriptions. This model combines 503B manufacturing standards with 503A patient-specific dispensing. Many telehealth weight loss programs use this hybrid approach.
Source: FDA — Draft Guidance: Prohibition on Wholesaling Under Section 503B (PDF)
Glossary
503A pharmacy — A traditional compounding pharmacy that prepares custom medications for individual patients based on prescriptions, regulated primarily by state boards of pharmacy under Section 503A of the FD&C Act.
503B outsourcing facility — An FDA-registered facility that compounds medications in large batches under cGMP standards, subject to FDA inspections and adverse event reporting, created by the DQSA of 2013.
cGMP (Current Good Manufacturing Practices) — FDA-enforced manufacturing standards (21 CFR Parts 210 & 211) requiring batch testing, process validation, supplier qualification, and quality systems. Required for 503B facilities and pharmaceutical manufacturers.
DQSA (Drug Quality and Security Act) — Federal law enacted in 2013 that created the 503B outsourcing facility category and strengthened oversight of drug compounding following the NECC disaster.
FD&C Act — The Federal Food, Drug, and Cosmetic Act, the primary federal law governing drugs in the United States. Sections 503A and 503B define the two types of compounding entities.
Beyond-Use Date (BUD) — The date after which a compounded medication should not be used. Assigned based on stability data (503B) or published literature/USP defaults (503A).
Certificate of Analysis (COA) — A document verifying the identity, potency, purity, and/or sterility of a specific medication batch.
USP 795 / 797 / 800 — United States Pharmacopeia standards for non-sterile compounding (795), sterile compounding (797), and hazardous drug handling (800). Many states reference USP standards in their pharmacy compounding regulations. 503A pharmacies are exempt from federal cGMP requirements, while 503B outsourcing facilities must comply with cGMP.
FDA Form 483 — A form issued by FDA investigators after an inspection, listing observations of conditions that may violate the FD&C Act. Form 483s may be publicly available through FDA public posting, trade reporting, or Freedom of Information Act (FOIA) requests. They can provide clues about quality-system issues at inspected facilities.
Essentially a copy (503A) — FDA guidance generally considers a 503A compounded drug "essentially a copy" if it has the same active ingredient(s), route of administration, and a similar strength (e.g., within 10%), unless the prescriber documents that a compounded version is necessary for an identified patient.
Essentially a copy (503B) — FDA guidance generally treats a 503B compounded drug as "essentially a copy" when it is identical or nearly identical to an FDA-approved drug, unless that approved drug is on the FDA drug shortage list.
Sources: FDA 503A Guidance (PDF), FDA 503B Guidance (PDF)
Enforcement discretion — A period during which the FDA announces it does not intend to take action against compounders for certain violations (e.g., compounding drugs on the shortage list). These periods are temporary and have specific end dates.
Sources, Methodology, and Update Policy
How this guide was built
This guide is based on primary sources from the FDA, federal law, court records, and USP standards. We reviewed:
- FDA regulatory publications on human drug compounding (503A and 503B)
- The full text of the Drug Quality and Security Act of 2013
- FDA enforcement actions, warning letters, and recall notices through February 2026
- Court filings and rulings in Outsourcing Facilities Association v. FDA (N.D. Tex., 2024–2025)
- State attorney general enforcement actions related to GLP-1 compounding (2025)
We do not accept payment from compounding pharmacies, outsourcing facilities, or pharmaceutical manufacturers for placement or favorable coverage in this guide.
Primary sources cited
- FDA — Human Drug Compounding Laws
- FDA — FD&C Act Provisions That Apply to Human Drug Compounding
- FDA — Compounding Inspections and Oversight FAQ
- FDA — Compounding and the FDA: Questions and Answers
- FDA — Information for Outsourcing Facilities
- FDA — Registered Outsourcing Facilities (searchable list)
- FDA — FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss
- FDA — Dosing Errors Associated with Compounded Injectable Semaglutide Products
- FDA — Clarifies Policies for Compounders as National GLP-1 Supply Begins to Stabilize
- FDA — Understanding the Risks of Compounded Drugs
- FDA — Warning Letters Database
- Drug Quality and Security Act (H.R. 3204, 2013)
- CDC — Multistate Fungal Meningitis Outbreak Investigation
- 21 CFR Parts 210 & 211 — Current Good Manufacturing Practice
- NABP — Compounding Pharmacy Accreditation
- FDA — FDA Intends to Take Action Against Non-FDA-Approved GLP-1 Drugs (Feb 6, 2026)
- FDA — Draft Guidance: Prohibition on Wholesaling Under Section 503B (PDF)
- FDA — Essentially a Copy Under Section 503A Guidance (PDF)
- FDA — Essentially a Copy Under Section 503B Guidance (PDF)
- FDA — Compounding: Inspections, Recalls, and Other Actions
- 21 CFR 211.166 — Stability Testing
- CT Attorney General — Bootleg GLP-1 Settlement (Aug 2025)
Update policy
The GLP-1 compounding regulatory landscape is changing rapidly. We review this guide for accuracy whenever:
- The FDA updates its compounding policies or drug shortage list
- New court rulings are issued in compounding-related cases
- FDA warning letters or enforcement actions create new consumer-relevant information
- Significant state-level enforcement actions occur
The "Last Updated" date at the top of this page reflects the most recent editorial review. If you notice an error or outdated information, contact us.
How we rank + verify
Last verified: February 13, 2026
What we verified: FDA regulatory publications on 503A and 503B compounding, the Drug Quality and Security Act of 2013, FDA enforcement actions and warning letters through February 2026, court rulings in OFA v. FDA, and state enforcement actions.
Sources: FDA primary sources (21 publications), CDC outbreak investigation, Congressional legislation, court filings, state attorney general enforcement actions
Related Articles
Compounded GLP-1 vs name brand: FDA approval, safety, real costs, and legality explained. Unbiased 2026 decision guide with pricing verified January 2026.
GLP-1 cost breakdown for 2026. Real prices for Wegovy, Zepbound, Ozempic with insurance, cash-pay, and Medicare options. Updated January 2026.
Learn how to get GLP-1 prescribed online safely. Eligibility, costs, insurance, pharmacy verification, and step-by-step guide.
A comprehensive guide to GLP-1 medication side effects, how to manage them, and when to contact your doctor. Based on FDA prescribing information.
GLP-1 contraindications explained: FDA Section 4 lists MTC/MEN2 history and serious hypersensitivity. Learn what's a real contraindication vs warning.
This content is educational only and does not replace professional medical advice. Always consult a qualified healthcare provider before starting any medication.