GLP-1 Medications Available in 2026: The Complete Guide to Pills, Shots, Cost, and What Changed
Last verified: May 25, 2026 · By: WPG Research Team at Weight Loss Provider Guide
Weight Loss Provider Guide is an independent comparison resource for GLP-1 telehealth providers. This page contains affiliate links — if you sign up through them, we may earn a commission at no extra cost to you. This does not affect our editorial conclusions.
As of April 2026, there are more GLP-1 medications available in the U.S. than at any point in history — and three of them were approved in the last four months alone. You can now take a GLP-1 weight-loss pill with no food restrictions (Foundayo), get roughly 19% average body weight loss from a higher-dose injection (Wegovy HD), and for the first time, eligible Medicare Part D beneficiaries will be able to access select GLP-1 weight-loss medications starting July 2026 at a $50/month copay.
But more options also means more confusion. Which ones are approved for weight loss vs. diabetes? What’s actually a pill? What do they really cost? And where does “compounded” fit in?
We built this page to answer all of that in one place — verified against FDA approval letters, manufacturer pricing pages, and CMS documentation. Here is the landscape, clearly separated by what is FDA-approved and what is not, with honest tradeoffs for every path.
Quick navigation
At a Glance: The Medications Most People Are Choosing Between in 2026
All pricing reflects publicly posted manufacturer or self-pay signals as of May 25, 2026. Prices change — verify directly with each source before purchasing. Intro offer windows have expiration dates noted in the cost section.
| Medication | Best for | Form | FDA-approved for weight loss? | Starting self-pay price signal |
|---|---|---|---|---|
| Zepbound (tirzepatide) | Strongest average weight loss | Weekly injection | Yes | $299/mo via LillyDirect (multi-dose KwikPen) |
| Wegovy injection (semaglutide 2.4mg) | Proven injectable with CV and MASH benefit data | Weekly injection | Yes | $199/mo intro for starter doses (through June 30, 2026), then $349/mo |
| Wegovy HD (semaglutide 7.2mg) | More weight loss for those already on Wegovy | Weekly injection | Yes | NovoCare self-pay pricing currently lists Wegovy HD 7.2 mg at $399/month, subject to eligibility and program terms. |
| Wegovy pill (semaglutide 25mg) | Needle-averse, want proven semaglutide | Daily pill (30-min fast required) | Yes | $149/mo for 1.5mg and 4mg doses¹ |
| Foundayo (orforglipron) | Easiest daily routine, no restrictions | Daily pill (anytime, with food) | Yes | $149/mo starting dose via LillyDirect |
| Ozempic (semaglutide) | Type 2 diabetes first (not weight loss) | Weekly injection | No — diabetes only | $199/mo intro for starter doses, then $349–$499/mo |
| Mounjaro (tirzepatide) | Type 2 diabetes first (not weight loss) | Weekly injection | No — diabetes only | List price: ~$1,112/fill without savings programs |
¹ Wegovy pill 4 mg is $149/month through August 31, 2026, then moves to $199/month. Wegovy pill 9 mg and 25 mg are $299/month. Source: wegovy.com/pill.
What this guide does differently
What GLP-1 Medications Are Available in 2026?
The 2026 market is no longer “Ozempic vs. Wegovy.” There are now FDA-approved GLP-1 medications across three formats (weekly injection, daily injection, daily pill), two primary goals (weight loss and type 2 diabetes), and two receptor types (GLP-1 only and dual GLP-1/GIP). Getting these categories straight is the most useful thing you can do before comparing providers or prices.
FDA-Approved Options for Weight Management
These medications carry a current FDA label specifically for chronic weight management (obesity or overweight with a weight-related condition):
| Medication | Active ingredient | Form | Avg weight loss | Key 2026 update |
|---|---|---|---|---|
| Zepbound | Tirzepatide (dual GLP-1/GIP) | Weekly injection | ~20.9% at 15mg (SURMOUNT-1, 72 wks) | KwikPen self-pay expansion; also approved for OSA |
| Wegovy HD | Semaglutide 7.2mg | Weekly injection | ~19% (STEP UP, 72 wks) | FDA approved March 19, 2026 — launching April 2026 |
| Wegovy | Semaglutide 2.4mg | Weekly injection | ~15% (STEP 1, 68 wks) | Also approved for CV risk reduction and noncirrhotic MASH (F2–F3) |
| Wegovy pill | Semaglutide 25mg | Daily pill | ~13.6% (OASIS 4, 64 wks) | FDA approved Dec 22, 2025; launched January 2026 |
| Foundayo | Orforglipron | Daily pill | ~11% at 17.2mg (ATTAIN-1 FDA label) | FDA approved April 1, 2026 — first GLP-1 pill with no food/water restrictions |
| Saxenda | Liraglutide 3mg | Daily injection | ~8% (SCALE, 56 wks) | Generic (Teva) available since August 2025 |
Clinical trial results as reported in published trial data and FDA prescribing information. Individual results vary significantly. Tirzepatide is technically a dual GIP/GLP-1 receptor agonist, not a pure GLP-1 — we include it because that’s how consumers and most of the internet group it.
Diabetes-First GLP-1 Medications People Still Ask About
These do not carry an FDA-approved weight-loss indication, but they come up constantly in weight-loss searches because doctors sometimes prescribe them off-label and because consumers confuse the brand names:
| Medication | Active ingredient | Form | FDA-approved for | Why it confuses people |
|---|---|---|---|---|
| Ozempic | Semaglutide | Weekly injection | Type 2 diabetes, CV risk reduction, CKD | Same active ingredient as Wegovy — but NOT approved for weight loss |
| Rybelsus / Ozempic tablets | Semaglutide tablets | Daily pill | Type 2 diabetes, CV risk reduction | People confuse it with Wegovy pill — different doses, different indication |
| Mounjaro | Tirzepatide | Weekly injection | Type 2 diabetes | Same active ingredient as Zepbound — but NOT approved for weight loss |
| Trulicity | Dulaglutide | Weekly injection | Type 2 diabetes, CV risk reduction | Older GLP-1; still prescribed but lower weight-loss effect |
| Victoza | Liraglutide 1.8mg | Daily injection | Type 2 diabetes | Same ingredient as Saxenda but lower dose and different indication |
Why this matters
Note (Jan 30, 2026): The FDA approved new Ozempic tablets (1.5mg, 4mg, 9mg) for T2D with improved bioavailability — expected at pharmacies Q2 2026. Rybelsus (3mg, 7mg, 14mg) remains on the market. Neither product is approved for weight loss.
Also worth knowing: Byetta (exenatide, twice daily), Bydureon BCise (exenatide ER, weekly), and Adlyxin (lixisenatide) are still technically on the market but rarely used for new starts in 2026.
What Changed for GLP-1 Medications in 2026?
2026 is the most consequential year for GLP-1 medications since semaglutide was first approved. If a page about GLP-1 medications still reads like it was written in 2024, it is already dangerously stale.
| Date | What happened | Why it matters |
|---|---|---|
| Jan 5, 2026 | Wegovy pill launched in U.S. | First oral GLP-1 specifically approved for weight loss; self-pay starting at $149/mo |
| Jan 13, 2026 | FDA requested removal of suicidal ideation/behavior warning from GLP-1 labels | FDA review found no increased risk — removes a barrier that worried some patients and providers |
| Jan 30, 2026 | FDA approved new Ozempic tablets (1.5mg, 4mg, 9mg) for T2D | Improved bioavailability oral semaglutide for diabetes; NOT approved for weight loss; expected at pharmacies Q2 2026 |
| Mar 3, 2026 | FDA warned 30 telehealth companies against illegal marketing of compounded GLP-1s | Targeted misleading claims that compounded products are interchangeable with FDA-approved brands |
| Mar 19, 2026 | Wegovy HD (7.2mg injection) FDA approved | Highest semaglutide dose ever; ~19% avg weight loss; launching in pharmacies April 2026 |
| Apr 1, 2026 | Foundayo (orforglipron) FDA approved | First non-peptide oral GLP-1 for weight loss; no food/water restrictions; fastest NME approval since 2002 |
| May 2026 | BALANCE Model — state Medicaid agencies can begin opting in | First Medicaid access to GLP-1s specifically for weight management |
| Jul 1, 2026 | Medicare GLP-1 Bridge launches | First-ever Medicare coverage for weight-loss medications; $50/mo copay for eligible beneficiaries (through Dec. 31, 2026) |
Sources: FDA press announcements and approval letters (fda.gov), Novo Nordisk press releases, Eli Lilly investor relations, CMS.gov (BALANCE Model and Medicare GLP-1 Bridge FAQ, March 3, 2026).
That is seven major events in seven months. If you haven’t checked the GLP-1 landscape since late 2025, nearly everything has changed.
See which 2026 changes affect your situation
Take the Free 60-Second Quiz →Which GLP-1 Medication Is Best for Your Situation?
There is no single “best” GLP-1. There is a best path for your specific combination of goal, route preference, cost situation, and medical history. Find yourself below.
If you want the strongest average weight loss from an FDA-approved brand
Start with Zepbound (tirzepatide injection) or Wegovy HD (semaglutide 7.2mg injection). Zepbound showed ~20.9% average body weight loss at its highest dose. Wegovy HD showed ~19%. In the head-to-head SURMOUNT-5 trial, tirzepatide at maximum tolerated dose beat semaglutide 2.4mg by a meaningful margin. Both require weekly self-injections.
These are injection-only options. If needles are a dealbreaker, jump to the pill section →
If you want a pill instead of a shot
Compare Foundayo vs. Wegovy pill. Both start at $149/month self-pay for the lowest dose. The practical difference is real:
- Foundayo: Take it any time of day, with or without food, with whatever you want to drink. No restrictions.
- Wegovy pill: Must be taken first thing in the morning on an empty stomach with a small sip of water, then wait 30 minutes before eating, drinking anything else, or taking other oral medications.
Rybelsus is also a semaglutide pill, but it is NOT approved for weight loss — it’s a diabetes medication with different dosing. Don’t confuse the two.
If you have type 2 diabetes and weight loss is secondary
Talk to your endocrinologist about Ozempic, Mounjaro, or Rybelsus first. Insurance is far more likely to cover a diabetes-indicated medication. Our GLP-1 for diabetes guide covers this path in detail →
If cost is the main barrier
- Brand-name pills (Foundayo or Wegovy pill) start at $149/month through manufacturer programs
- Ro membership starts at $39 first month, then $149/mo (or $74/mo with annual plan). Medication billed separately.
- Medicare coverage begins July 2026 at $50/month for qualifying beneficiaries
- Compounded semaglutide starts ~$99–$149/month — but these are NOT FDA-approved products (see compounded section)
If you’re afraid of needles
2026 is the first year you have real options. Both Foundayo and Wegovy pill are FDA-approved oral GLP-1 medications for weight loss. They produce meaningful weight loss (11–14% average), just somewhat less than the strongest injectables (15–21%). You don’t have to settle for a less effective medication class just because you don’t want injections.
If you may become pregnant or are planning pregnancy soon
- Wegovy: Stop 2 months before a planned pregnancy
- Zepbound: Discontinue when pregnancy is recognized
- Foundayo: Discontinue when pregnancy is recognized — and it may reduce effectiveness of oral hormonal birth control during the first weeks of treatment or when the dose is increased. Discuss backup contraception with your provider.
Get a personalized GLP-1 medication recommendation
Free 60-Second Matching Quiz →GLP-1 Pills vs. Injections: How Different Are They Really?
The pill-vs-shot decision is now a first-order question, not a niche curiosity. But “pill” does not mean the same experience across all products. There are meaningful daily-routine differences that most comparison pages bury under the same “oral option” label.
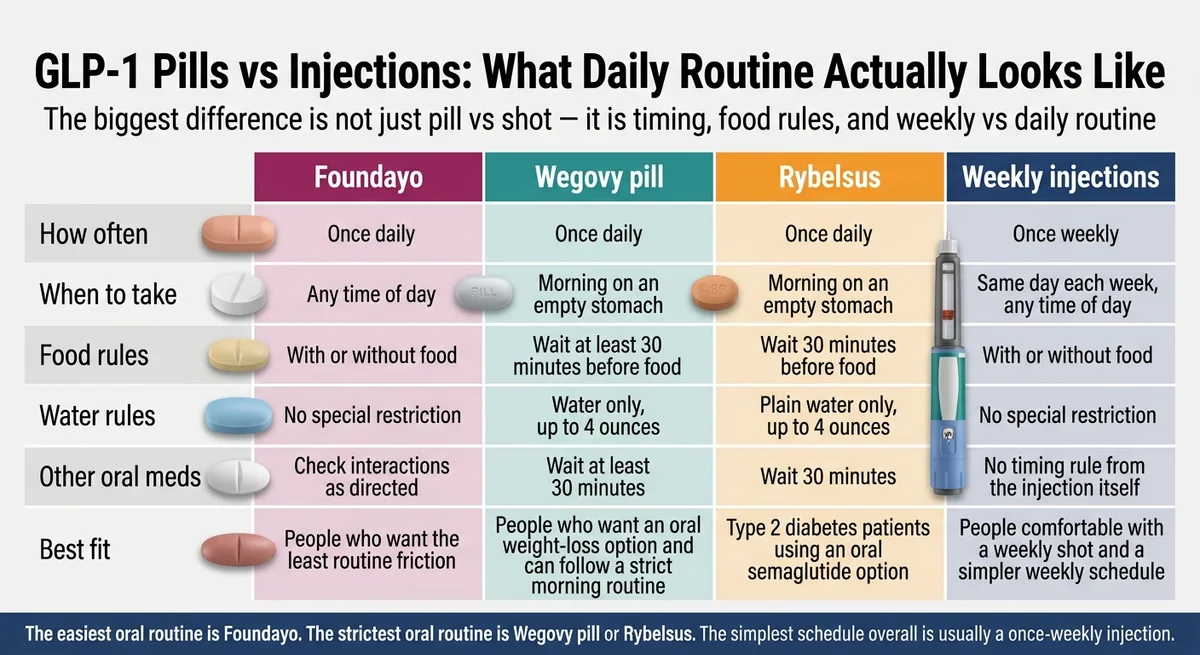
The routine friction table
| Factor | Foundayo | Wegovy pill | Rybelsus | Injectable GLP-1s |
|---|---|---|---|---|
| When to take | Any time of day | Morning, on empty stomach | Morning, on empty stomach | Same day each week, any time |
| Food rules | None — eat whenever | Wait 30 min after taking | Wait 30 min after taking | No food rules |
| Water rules | None | Small sip of water only | Small sip of water only | N/A |
| Other oral meds | Check interactions (may affect some drugs’ absorption) | Wait 30 min after Wegovy pill | Wait 30 min after Rybelsus | No oral med conflicts from injection itself |
| Frequency | Once daily | Once daily | Once daily | Once weekly |
| Best for | People who want zero routine friction | People fine with strict morning protocol | Diabetes patients (not weight-loss indicated) | People comfortable with weekly self-injection |
Do pills work as well as injections?
Currently, no. The clinical trial data is clear on averages:
~20.9%
Zepbound 15mg injection
~13.6%
Wegovy pill 25mg
~11%
Foundayo 17.2mg pill
Who should NOT pick an oral route first: If you need the maximum possible weight loss for a specific medical reason (preparing for surgery, managing severe obesity-related complications), your provider may recommend starting with an injectable. The weight-loss gap between pills and shots is meaningful enough that some clinical situations warrant the more potent option.
Compare all oral GLP-1 options side by side →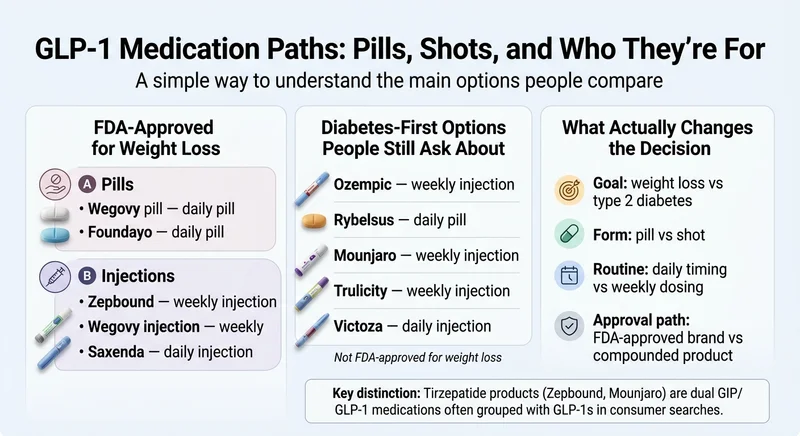
Zepbound vs. Wegovy vs. Foundayo: The Three-Way Comparison Most People Actually Need
For weight-loss searchers in 2026, the real decision set has narrowed to three brands. Here they are head-to-head — honestly.
| Factor | Zepbound | Wegovy (injection + HD) | Foundayo |
|---|---|---|---|
| Active ingredient | Tirzepatide | Semaglutide | Orforglipron |
| Receptor target | Dual GLP-1 + GIP | GLP-1 only | GLP-1 only (partial agonist) |
| Form | Weekly injection | Weekly injection (+ daily pill option) | Daily pill |
| Peak avg weight loss | ~20.9% (SURMOUNT-1) | ~19% at HD 7.2mg (STEP UP); ~15% at 2.4mg (STEP 1) | ~11% (ATTAIN-1, FDA label) |
| Take with food? | N/A (injection) | N/A (injection); pill requires 30-min fast | Yes — anytime, no restrictions |
| Self-pay price signal | $299/mo multi-dose KwikPen starting dose | $199/mo intro injection; $149/mo pill starting dose | $149/mo starting dose |
| Medicare Bridge eligible (Jul 2026)? | Yes (KwikPen formulation) | Yes (all formulations) | Lilly states eligible Medicare Part D patients may access ~$50/mo starting July 2026 |
| Manufacturer | Eli Lilly | Novo Nordisk | Eli Lilly |
| Biggest advantage | Highest weight-loss results; dual receptor mechanism | Largest evidence base; most FDA indications; both shot and pill forms | Easiest routine; only GLP-1 pill with zero restrictions |
| Biggest tradeoff | Injection only; no pill form | Pill form requires strict morning timing; injection has standard needle requirement | Lower avg weight loss than injectables; newest product with shortest track record |
When each one is the wrong choice
Zepbound is wrong if
you can’t do injections and there’s no one to help you. It has no oral form.
Zepbound patients who hate needles → Foundayo
Wegovy is wrong if
your mornings are already medication-heavy and you want the pill — the fasting requirement is non-negotiable and causes real adherence friction.
Wegovy pill patients who can’t do morning timing → Foundayo
Foundayo is wrong if
your primary goal is maximum weight loss and you’re comfortable with injections. At ~11% average (FDA label), it trails the top injectables by 8–10 percentage points.
Foundayo patients who want maximum results → Zepbound or Wegovy HD
See which of these three fits your routine
Take the Quiz →What About Ozempic, Rybelsus, Mounjaro, and Saxenda?
These medications are real and legitimate, but they serve different primary purposes. Grouping them all together — as most competitor pages do — creates confusion that costs readers real money and real outcomes.
Ozempic: why it’s still searched like a weight-loss drug
Ozempic is semaglutide, the same active ingredient as Wegovy. But Ozempic is FDA-approved for type 2 diabetes and cardiovascular risk reduction — not weight loss. Doctors sometimes prescribe it off-label for weight loss, and insurance may cover it more readily if you have a diabetes diagnosis. But if weight loss is your primary goal and you’re paying cash, there’s no reason to pursue Ozempic over Wegovy, which is specifically approved and dosed for weight management.
Rybelsus: the pill that is NOT the same as Wegovy pill
This is one of the most common confusions in the GLP-1 space. Rybelsus is an oral semaglutide tablet for type 2 diabetes. The Wegovy pill is an oral semaglutide tablet for weight loss. They contain the same active ingredient but at different doses with different FDA indications. If someone offers you “the semaglutide pill” for weight loss, make sure it’s Wegovy (25mg dose) and not Rybelsus (3mg, 7mg, or 14mg doses).
Mounjaro: the diabetes-first tirzepatide
Mounjaro contains tirzepatide, same as Zepbound. But Mounjaro is approved for type 2 diabetes, not weight loss. Same ingredient, different indication, different insurance coverage path. If you have T2D, Mounjaro may be the more insurance-friendly route to tirzepatide.
Saxenda: still valid, but daily injections hurt its case
Saxenda (liraglutide 3mg) is still FDA-approved for weight management, and a generic version from Teva became available in August 2025. But it requires daily injections (vs. weekly for Wegovy or Zepbound) and produces lower average weight loss (~8%). In a market that now has once-weekly injections and daily pills, Saxenda’s convenience profile is a hard sell unless you specifically respond well to liraglutide or your insurance only covers it.
How Much Do GLP-1 Medications Cost in 2026 Without Insurance?
The cost picture changed dramatically in 2025–2026. New manufacturer direct pricing, government agreements, and oral formulations have created real entry points below $200/month for the first time — but those numbers come with fine print.
Brand-name price signals (verified April 15, 2026)
| Medication | Source | Price | Important catch |
|---|---|---|---|
| Foundayo | LillyDirect self-pay | $149/mo (0.8mg) → up to $349/mo at higher doses | Price increases as dose escalates; savings card may reduce to $25/mo for eligible commercially insured patients |
| Wegovy pill | Novo Nordisk offer | $149/mo for 1.5mg and 4mg doses | 4 mg at $149 available through August 31, 2026, then $199/mo. Maintenance doses (9mg, 25mg) are $299/mo. Source: wegovy.com/pill |
| Wegovy injection | Novo Nordisk intro offer | $199/mo for starter doses (0.25mg, 0.5mg) through June 30, 2026 | After intro window, maintenance doses start at $349/mo. Wegovy HD 7.2 mg self-pay is $399/month via NovoCare. |
| Zepbound | LillyDirect | Multi-dose KwikPen starts at $299/mo; higher doses up to $449/mo | Single-dose pen fills may be $499. Pricing is formulation- and refill-dependent — verify at zepbound.lilly.com |
| Ro (facilitates access to Zepbound & Foundayo) | Ro.co | Membership: $39 first month, then $149/mo or $74/mo with annual plan | Medication billed separately from membership. Total cost = membership + medication. Verify full pricing at ro.co |
| Generic Saxenda (liraglutide) | Retail pharmacy | Lower than brand — check GoodRx for current pricing | Daily injection; less convenient than weekly or pill options |
Why starter pricing doesn\u2019t answer your real annual cost
Before committing, ask the provider: “What will I pay at each dose level through maintenance, and are there any additional fees?” That single question saves more money than any coupon.
What real customers notice about cost and access
“Easy upfront pricing and interaction.”
— Ro customer, Trustpilot (individual experience, not typical)
“Doctors are knowledgeable, responsive and very careful with details.”
— Eden customer, Trustpilot (individual experience, not typical)
“Getting the medication shipped after the first shipment was a hassle each time.”
— SHED customer, Trustpilot (individual experience, not typical)
See current brand-name pricing matched to your situation
See Highest-Rated GLP-1 Providers →Does Insurance, Medicare, or Medicaid Cover GLP-1 Medications in 2026?
Coverage is still fragmented — but 2026 created a genuinely new access story for tens of millions of Americans.
Commercial insurance
According to the 2025 KFF Employer Health Benefits Survey, 19% of firms with 200+ workers and 43% of firms with 5,000+ workers cover GLP-1 medications for weight loss. Coverage for diabetes indications is much broader.
Steps to check: (1) Look up Wegovy, Zepbound, or Foundayo on your plan’s formulary. (2) If listed, ask about prior authorization requirements. (3) If denied, appeal — our prior authorization guide walks through the process →
Medicare: the biggest change in 20 years
Medicare GLP-1 Bridge: July 1, 2026 — December 31, 2027
Medications covered:
- Foundayo — all formulations
- Wegovy — all formulations, including injection and tablets
- Zepbound — KwikPen formulation only
- Zepbound single-dose vial and single-dose pen: excluded
Copay & enrollment:
- $50/month (does not count toward Part D annual out-of-pocket cap)
- Prescriber submits prior auth to CMS central processor
- Your Part D plan does NOT have to opt in for you to participate
Medicare GLP-1 Bridge eligibility tiers
| Tier | BMI requirement | Plus one of these conditions |
|---|---|---|
| Tier 1 | BMI ≥ 35 | No additional condition required |
| Tier 2 | BMI ≥ 30 | Heart failure with preserved ejection fraction (HFpEF), OR uncontrolled hypertension on 2+ antihypertensive medications, OR CKD stage 3a or above |
| Tier 3 | BMI ≥ 27 | Prediabetes (per ADA guidelines), OR prior myocardial infarction, OR prior stroke, OR symptomatic peripheral artery disease |
BMI is measured at the time of initiating GLP-1 therapy, not at the time of the Bridge application. Source: CMS Medicare GLP-1 Bridge FAQ, released March 3, 2026 (cms.gov).
After December 2027: BALANCE in Medicare Part D has been delayed
Medicaid
State Medicaid agencies can opt in to the BALANCE Model starting May 2026. As of early 2026, only 13 states covered GLP-1 medications for weight loss through Medicaid (down from 16 in 2025, per KFF). Coverage timing and eligibility will vary by state. Check with your state Medicaid office for current status.
Need help with prior authorization or insurance appeals?
Read the Insurance Guide →Are Compounded GLP-1 Medications Still an Option in 2026?
Yes, compounded semaglutide and tirzepatide are still available from licensed pharmacies in April 2026. But the regulatory picture has become significantly more complicated, and this section draws a bright line you need to understand before spending money.
What “compounded” means
FDA-Approved Brand-Name Medications
- Manufactured under strict FDA oversight
- Clinical trials completed before approval
- Manufacturing processes regularly inspected
- FDA has verified what’s in the product
Compounded GLP-1 Products
- Prepared by licensed pharmacies
- Legal and widely prescribed
- NOT FDA-approved as finished products
- Not evaluated by FDA for safety, efficacy, or quality before dispensing
What actually happened with FDA enforcement in 2026
How to evaluate a compounded GLP-1 provider
If you choose the compounded path, verify these minimum standards:
Red flags to watch for
A compounded option that meets basic vetting criteria
ShedRx — Flexible Formats + Needle-Free Options
From $199/mo · Semaglutide & tirzepatide · Injections, sublingual drops, or lozenges
- Multiple delivery formats including needle-free options (sublingual drops and lozenges)
- Licensed U.S. pharmacy network, medically supervised
- Labs billed separately · some users report shipping friction at refill
- Compounded only — not FDA-approved as a finished product
Side Effects, Contraindications, and Practical Downsides That Matter
All GLP-1 medications share a similar core side-effect profile because they work on the same receptor system. Writing a generic “nausea, vomiting, diarrhea” list and moving on is what every other page does. Here is what actually changes a decision.
What to realistically expect during dose escalation
GI effects — nausea, vomiting, diarrhea, constipation, abdominal pain — are most common in the first 4–8 weeks at each new dose level. Every approved GLP-1 uses gradual dose escalation specifically to minimize these effects. Most people tolerate them well after the initial adjustment period. Eating smaller meals, staying hydrated, and avoiding high-fat foods during escalation are the most commonly recommended strategies.
Differences that matter by medication
Wegovy HD (7.2mg): dysesthesia
22% of trial participants reported dysesthesia (altered skin sensation — tingling, sensitivity, burning) vs. 6% at the 2.4mg dose and 0.3% on placebo. Most cases were mild and resolved on their own, but this is a meaningful new signal for this higher dose. Source: FDA-approved Wegovy prescribing information.
Foundayo: hair loss & postmarket safety studies
Hair loss was reported in approximately 5% of trial participants. The FDA has also requested additional postmarketing safety studies from Lilly, including liver safety data (as of April 14, 2026). Foundayo remains approved and available, but this is worth monitoring. Foundayo may also reduce the effectiveness of oral birth control during the first weeks of treatment or during dose increases. Source: FDA-approved Foundayo prescribing information.
All GLP-1 medications: boxed warning & anesthesia
- Boxed warning for thyroid C-cell tumors (based on animal studies). Do NOT use if you have a personal or family history of medullary thyroid carcinoma (MTC) or MEN2.
- GLP-1s slow gastric emptying — tell your anesthesiologist before any procedure requiring sedation. Some guidelines recommend stopping GLP-1s before surgery.
The suicidal ideation question is resolved
In January 2026, the FDA completed its review and requested removal of the suicidal behavior and ideation warning from GLP-1 receptor agonist labels (Wegovy, Zepbound, Saxenda). The review found no increased risk. Source: FDA Drug Safety Communication, January 13, 2026.
How to Choose a GLP-1 Medication in 3 Steps
By this point, you should not still be deciding at the brand-logo level. The decision reduces to three filters in this order:
Choose your goal first
- Weight loss is primary goal → Wegovy, Zepbound, Foundayo, or Saxenda (FDA-approved weight-loss indication)
- Type 2 diabetes with weight loss secondary → Ozempic, Mounjaro, or new Ozempic tablets. Insurance coverage is broader for diabetes indications.
- Both diabetes and weight loss are priorities → Tirzepatide (Mounjaro/Zepbound) addresses both powerfully.
Choose your route second
- Fine with weekly self-injection → Zepbound or Wegovy give you the strongest weight-loss data
- Want a pill with no routine friction → Foundayo
- Want a pill and don’t mind morning timing → Wegovy pill
Choose your access path third
- Commercial insurance covers weight-loss meds → Start with your formulary. Use manufacturer copay cards for lowest out-of-pocket.
- On Medicare and qualify → Prepare now for the July 2026 GLP-1 Bridge at $50/month.
- Paying cash, want FDA-approved → Foundayo or Wegovy pill starting at $149/month; Ro membership is another path to FDA-approved options.
- Paying cash, budget is main constraint → Compounded providers ~$99–$149/month, with the understanding these products are not FDA-approved.
If two options still seem equally good — get the final filter tailored to your specific insurance, budget, and medical details.
Get Your Personalized GLP-1 Action Plan →What We Actually Verified for This Page
Last verified: May 25, 2026
Weight Loss Provider Guide is an independent comparison resource for GLP-1 telehealth providers. We don’t manufacture, prescribe, or compound medications.
Frequently Asked Questions About GLP-1 Medications in 2026
Still not sure which GLP-1 program is right for you?
Answer 5 quick questions about your insurance, budget, needle preference, and health goals — and get a personalized medication and provider recommendation with real next steps. No email required. No commitment.
Find My GLP-1 Path — Free Quiz →Written by the WPG Research Team. Weight Loss Provider Guide is an independent comparison resource for GLP-1 telehealth providers. Last verified: May 25, 2026. Medical disclaimer: This content is for informational purposes only and is not medical advice. GLP-1 medications carry risks and side effects, and some are not FDA-approved for weight loss. Always consult a licensed healthcare provider before starting any medication. Individual results vary.