What Happens If Your Compounding Pharmacy Shuts Down or Loses 503A Status (2026 Patient Guide)
By the WPG editorial team at Weight Loss Provider Guide · Last verified: April 22, 2026 · Updated monthly.
Weight Loss Provider Guide may earn a commission if you start treatment through links on this page. On this specific continuity topic, routing prioritizes reader safety and regulatory fit over affiliate priority. This article is informational and does not constitute medical or legal advice. Compounded medications referenced are not FDA-approved as finished drug products. Full disclosure.
Who this guide is for
This guide is for: people already taking compounded semaglutide or tirzepatide who just got a closure email, a canceled refill, a warning-letter scare, or saw headlines about their provider.
This isn't for: first-time readers just shopping for a GLP-1 — our best GLP-1 online programs comparison is a better starting point.
What we actually verified
- FDA's April 1, 2026 GLP-1 compounding clarification and the underlying Section 503A guidance
- FDA Compounding: Inspections, Recalls, and other Actions page — all entries dated 2024–2026
- Official state closure rules for California, New York, Texas, Florida, and Utah (board-of-pharmacy source documents)
- DEA's 2023 prescription transfer regulation
- Current self-pay pricing on LillyDirect (Zepbound, Foundayo), NovoCare (Wegovy, Ozempic), and Ro.co
- 15+ documented 2024–2026 pharmacy enforcement actions with source links, using primary records where available
If we could not verify something, we marked it or left it out.
1. What to Do in the First 24 Hours

A practical continuity plan for people already taking compounded semaglutide or tirzepatide.
Before you do anything else — screenshot these 4 things
- 1Your subscription dashboard (medication name, dose in mg/mL, last fill date, next scheduled fill)
- 2Your most recent invoice or receipt
- 3The label on your current vial or pen (pharmacy name, NPI if shown, lot number, beyond-use date)
- 4The most recent message from your prescriber inside the telehealth portal
Portals disappear. Invoices go missing. Capture the records before you need them.
Then, in this order:
- 1Message your prescribing clinician the same day — not tomorrow. Use the telehealth portal if it still works, or email directly. Ask two questions: "Where did my prescription records go?" and "Can you re-issue to a new pharmacy if transfer fails?"
- 2Check your pharmacy against two FDA lists before your next dose. Look up your pharmacy on the FDA compounding actions page and on the FDA recall enforcement reports. If your pharmacy's name is on either, don't use any medication from an affected lot until you've talked to your prescriber.
- 3Write the refund request email today (template in section 9). Don't wait until the company goes silent.
- 4Open exactly one replacement path — the quiz takes 60 seconds and routes you to the right one based on your specific situation.
FDA verification links (open in a new tab before your next dose):
2. What Happens If Your Compounding Pharmacy Shuts Down? The 4 Scenarios
| Scenario | Fastest next step |
|---|---|
| 1. Voluntary closure | Confirm records destination; transfer prescription |
| 2. FDA warning letter | Read the specific letter directly; don't rely on vague provider reassurance |
| 3. Failed 503A conditions — cannot legally compound GLP-1 copies | Decide between Path 1 (compliant compounded) or Path 2 (brand-name) in section 11 |
| 4. Product recall (Class I, II, or III) | Check lot numbers against the FDA recall list; contact prescriber |
3. What “Loses 503A Status” Actually Means
503A in plain English
Section 503A was added to federal law in 1997 through the Food and Drug Administration Modernization Act. It lets a state-licensed pharmacy compound a drug for an individual patient based on a valid prescription — without needing FDA pre-market approval for each formulation — as long as certain conditions are met. Compounded drugs under 503A are not FDA-approved as finished products.
Why 503A conditions matter for GLP-1s specifically
One of the 503A conditions is that the compounded drug may not be, routinely or in inordinate amounts, “essentially a copy” of a drug that’s already commercially available from a manufacturer. While semaglutide and tirzepatide were on the FDA drug shortage list (2022 through early 2025), pharmacies could compound them under a shortage-related exemption. FDA removed tirzepatide from the shortage list in December 2024 and semaglutide in February 2025. With both off the shortage list, the shortage pathway closed.
What FDA clarified on April 1, 2026
FDA issued a GLP-1-specific clarification applying the existing 503A “essentially a copy” framework using current GLP-1 examples. The 2026 update made two things specific: it used the semaglutide + vitamin B12 combination as an example of a formulation that is still essentially a copy (adding B12 is not enough of a difference), and it announced a 4-or-fewer-Rx-per-month enforcement tolerance for truly patient-specific compounding. Source: Foley & Lardner
4. Does a Warning Letter Mean Your Pharmacy Is Shut Down?
Warning letter
FDA has identified violations (often after an inspection). The company responds. Fills may or may not pause. Products may or may not be recalled. This is a signal, not a verdict.
Recall
Specific lot numbers are being pulled from the market. This is an action, not an advisory. If your lot is recalled, stop using the medication immediately.
Injunction or consent decree
A court has ordered the company to stop specific activities (e.g. Pharm D Solutions in Texas, ordered to cease all sterile compounding). Strongest enforcement action short of criminal charges.
State board suspension or revocation
Your state's pharmacy board has suspended or revoked the license. The pharmacy cannot legally dispense in that state.
Two types of 2026 warning letter you’re probably hearing about
Type 1 — Pharmacy quality letters
These cite specific product safety failures. The most notable 2026 example: Boothwyn Pharmacy’s January 16, 2026 letter, which cited subpotent compounded semaglutide (tested at 79.9% of labeled strength), tirzepatide that failed sterility testing, and insanitary conditions. This is the kind of letter where your specific lot matters. Partnership for Safe Medicines
Type 2 — Telehealth marketing letters
55+ letters to online sellers in September 2025; 30+ additional in March 2026. These cited illegal marketing of compounded GLP-1s — language that falsely implied FDA approval. MEDVi received a separate February 20, 2026 letter (#721455) on similar misbranding grounds. If the letter cites only marketing claims, the medication itself may not be directly implicated — but the company’s compliance posture is worth paying close attention to.
Every FDA warning letter is public. Search your pharmacy or telehealth company on the FDA Warning Letters database . Read the letter directly.
5. Is the Medication in Your Fridge Still Safe to Use?
Run this 7-point check before your next injection
Does your pharmacy name appear on the FDA's Compounding Inspections, Recalls, and Other Actions page?(stop if YES)
Does your specific lot number appear on any Class I, II, or III recall in FDA enforcement reports?(stop if YES)
Is the pharmacy state-licensed and traceable to a real, verifiable address on your state board of pharmacy website?(stop if YES)
Has your state board of pharmacy suspended or revoked the license?(stop if YES)
Is the vial within its beyond-use date (BUD)?(stop if NO)
Has the medication been continuously refrigerated per label instructions? Did any past shipment arrive warm?(stop if NO)
Is there any visible issue with the medication itself — particulate, color change, cloudiness, broken seal?(stop if NO)
- Dosing errors. FDA has received multiple adverse event reports — some requiring hospitalization — related to dosing errors with compounded semaglutide vials, where patients miscalculated or measured incorrect doses. Unlike pre-filled manufacturer pens, vials require you to calculate the correct volume. If you’ve ever felt unsure about your injection math, ask your prescriber to walk you through it. FDA source
- Salt forms. FDA has reported some compounded products containing “semaglutide sodium” or “semaglutide acetate” — different active ingredients from the base form in FDA-approved drugs. Check your label. If it lists a salt form, that’s information your prescriber should have.
6. Verify Your Pharmacy in 3 Lookups
Lookup 1 — FDA enforcement check
Search the pharmacy's name on FDA's compounding actions page. If it appears, read the most recent 483 inspection report or warning letter directly. You're looking for: sterility failures, potency failures, insanitary conditions, and products in your specific drug line.
FDA compounding actions pageLookup 2 — State board license check
Every state has a Board of Pharmacy with a public license lookup. Search the pharmacy's name. You're looking for: active license status, disciplinary history, and recent inspection results.
National Association of Boards of Pharmacy directoryLookup 3 — Label fraud check
Compare the pharmacy name on your vial label to what's on the pharmacy's website, FDA database, and state board record. FDA has documented fraudulent compounded GLP-1 products where the pharmacy name on the label doesn't match a real pharmacy, or where the named pharmacy says it didn't make the product. Spelling errors, wrong addresses, and mismatched identities are signals to stop.
Three lookups, five minutes, zero guesswork.
7. Can Your Compounded GLP-1 Prescription Be Transferred?
State-by-state closure rules (spot-check, 5 major states)
This is a spot-check of five major states, not a complete 50-state legal survey. Your state may have additional rules.
| State | What this means for you |
|---|---|
| California | California explicitly acknowledges you may still hit refill friction. Plan for a new Rx just in case. Source: California Board of Pharmacy |
| New York | Your records go somewhere specific — find out where. Source: NY Office of the Professions |
| Texas | Record-location disclosure is part of compliance. Texas State Board of Pharmacy can tell you. Source: Texas State Board of Pharmacy |
| Florida | If you can't reach anyone online, the physical sign (or the board's records) will tell you. Source: Florida Board of Pharmacy |
| Utah | The transfer destination is required to be named publicly. Source: Utah Admin Code R156-17b-604 |
8. Will You Need a New Prescription, a New Consult, or a Dose Restart?
Likely carries forward with minimal friction
- Your original prescriber is still practicing and reachable
- Your gap will be short and the new pharmacy will honor the same formulation
- Your titration history and last dose are clearly documented
Fresh prescription likely needed
- Your original telehealth provider is gone or unresponsive
- You're switching from compounded to brand-name (Wegovy, Zepbound, Foundayo)
- Your state's closure rules didn't result in a clean records transfer
- You're switching routes of administration (injection to tablet, or vice versa)
Patients who arrive with a photo of their last label and their titration history have a much stronger case for continuity than those who say “I was on some semaglutide but I don’t remember the dose.”
9. Refunds and What to Do If the Pharmacy Won’t Respond
Start with a written refund request — this template often works
Subject: Refund request and prescription records — Account [your account number]
I am writing to request a refund for the unfulfilled portion of my subscription/prepaid order dated [date of last payment], in the amount of $[amount], and access to my medication records, including transfer of my active prescription for [medication name, dose, strength] to a pharmacy of my choice.
Please confirm receipt in writing. If I do not receive a response, I will review my options, which may include a credit card chargeback, a complaint to the [your state] Attorney General’s consumer protection division, and a complaint to the [your state] Board of Pharmacy.
Thank you.
Save that email. Send it from an email account you’ll keep. Copy yourself.
If the pharmacy doesn’t respond — your escalation ladder
- 1Credit card chargeback. Chargeback rules vary by card issuer; common windows range from 60 to 120 days after the charge. Document the missing service and include your refund request email.
- 2State Attorney General consumer protection complaint. Every state has an online complaint form. These carry more weight when enough patients file.
- 3State Board of Pharmacy complaint. Your state board has authority over prescription transfer failures, closure compliance, and licensing issues.
- 4Small claims court or a consumer-protection attorney for amounts above chargeback limits.
10. The 2024–2026 GLP-1 Pharmacy Enforcement Tracker
| Entity | Action |
|---|---|
| Boothwyn Pharmacy, LLC (PA) — 503A | FDA warning letter |
| Boothwyn Pharmacy | Class II recall |
| MEDVi, LLC (telehealth) | FDA warning letter #721455 |
| Hims & Hers (telehealth) | Novo Nordisk patent lawsuit |
| Fullerton Wellness (CA) | Voluntary cease after complaint + state inspection |
| Pharm D Solutions, LLC (TX) | Federal consent decree |
| ProRx | Recall + FDA warning letter |
| Industry-wide — 503A GLP-1 | End of shortage-based compounding authority |
| FDA mass enforcement | 55+ warning letters to online sellers |
| FDA mass enforcement Round 2 | 30+ warning letters to telehealth companies |
| FDA clarification | "Essentially a copy" guidance with GLP-1 examples |
| Novo Nordisk petition | DDC list nomination for semaglutide |
| Eli Lilly petition | DDC list nomination for tirzepatide |
11. Your 4 Safest Backup Paths (with Current April 2026 Pricing)
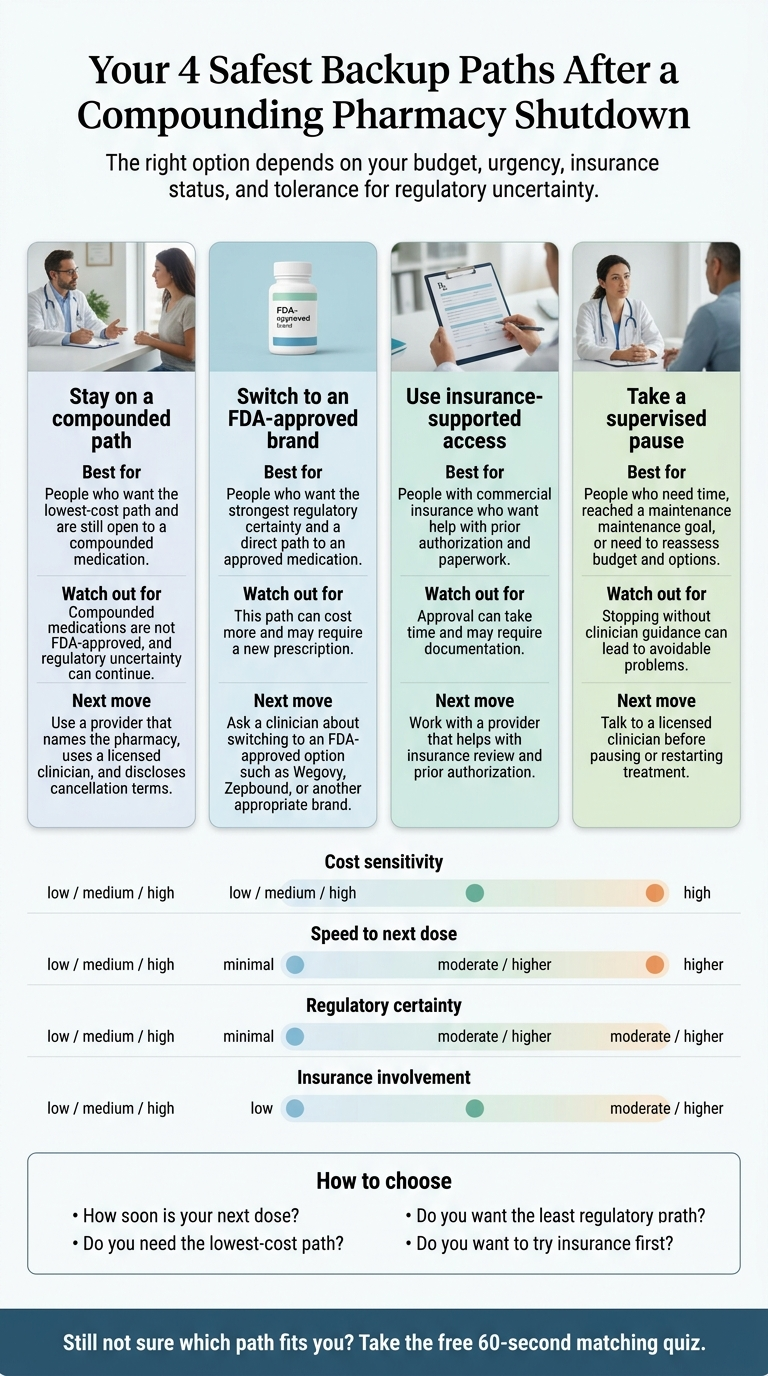
Still not sure which path fits you? Take the free 60-second matching quiz → it asks where you live, what you were on, and how urgent your next dose is, then routes you to the single best option.
Affiliate disclosure: we may earn a commission if you start through the provider links below. Pricing, medication, and fit came first. We verified pricing on each provider’s own website on April 22, 2026.
Path 1: Transfer to Another Compliant Compounded Provider
Best for: lowest monthly cost, understand compounded is not FDA-approved, meet the narrow patient-specific exception.
What to look for in any replacement provider:
- Provider names the compounding pharmacy (not "our pharmacy partner" — the actual name)
- The pharmacy does not appear on any active FDA warning letter, recall, or import alert
- Prescribing is done by a licensed U.S. clinician, not automated intake
- Marketing language does not claim the compounded product is "the same as" or "equivalent to" Wegovy, Zepbound, or any FDA-approved drug
- Cancellation terms are disclosed before you pay
Eden is one of the broad compounded telehealth providers we have previously verified and that has not appeared on the 2025–2026 FDA warning letter list we tracked. For cost-conscious self-pay shoppers specifically, Yucca Health offers async low-friction approval with installment options. For oral, sublingual, or needle-free compounded alternatives, SHED is the specialist. Verify current details on each provider’s own pages before enrolling.
Path 2: Switch to an FDA-Approved Brand-Name Medication
Best for: you want the regulatory certainty of an FDA-reviewed product. Current 2026 pricing cuts mean this is finally accessible at $149–$449/month — not the $1,000+ it used to be.
This is the cleanest bridge in 2026. Both Novo Nordisk and Eli Lilly cut their self-pay prices under a White House pricing agreement in late 2025 and early 2026. Verified April 22, 2026:
| Medication | Program | Self-pay price |
|---|---|---|
| Foundayo™ (orforglipron) — oral daily GLP-1 | LillyDirect | $149 (0.8 mg), $199 (2.5 mg), $299 (5.5 mg / 9 mg), $349 (14.5 mg / 17.2 mg) |
| Zepbound® (tirzepatide) single-dose vials | LillyDirect Self Pay Journey Program | $299/mo (2.5 mg); $399/mo (5 mg); $449/mo (7.5–15 mg) |
| Zepbound® KwikPen | LillyDirect Self Pay Journey Program | $299/mo (2.5 mg); $399/mo (5 mg); $449/mo (7.5–15 mg) |
| Wegovy® pen | NovoCare Pharmacy | $199/mo (first 2 fills, new patients through Jun 30, 2026); then $349/mo; $399/mo for HD 7.2 mg |
| Wegovy® pill (oral) | NovoCare Pharmacy | $149/mo (1.5 mg and 4 mg starter through Aug 31, 2026); higher maintenance doses priced per NovoCare guide |
| Ozempic® — FDA-approved for type 2 diabetes | NovoCare Pharmacy | $199/mo (first 2 fills), then $349/mo (0.25–1 mg); $499/mo (2 mg) |
Who we’d route you to for the brand-name path. Ro is the telehealth provider we’d send you to for FDA-approved GLP-1 access with insurance support. Ro publicly carries Zepbound® and the newly-approved Foundayo™, offers a free GLP-1 Insurance Coverage Checker, and includes a dedicated insurance concierge that handles prior authorization paperwork on your behalf — genuinely useful if you’re switching from compounded to brand-name and want to try insurance before paying cash. Ro’s Body membership is $39 for the first month, $149/month ongoing, or as low as $74/month with the annual plan paid upfront, with medication priced separately.
Path 3: Use Your Insurance (with Concierge Help)
Best for: commercial insurance, BMI ≥30 (or ≥27 with a comorbidity), willing to spend 2–6 weeks navigating prior authorization.
What changed in 2026 that matters here
- Wegovy now has FDA approval for MASH (metabolic dysfunction-associated steatohepatitis) and for reducing major cardiovascular event risk in people with obesity and heart disease.
- Zepbound is FDA-approved for moderate-to-severe obstructive sleep apnea in adults with obesity.
- These are new coverage pathways. If your prior insurance denial was "weight loss is not a covered indication," and you have MASH, cardiovascular disease, or OSA, a new prescription tied to one of those indications may be approved when weight-loss alone wasn't.
Our full prior authorization GLP-1 guide walks through the PA packet, denial fixes, and appeal templates. If you’re starting from scratch, read that first.
Path 4: Supervised Pause
Best for: you’ve reached or exceeded your maintenance weight goal, you want time to evaluate options, or your financial situation has changed.
12. What NOT to Do If Your Compounded GLP-1 Gets Cut Off
Don't buy from unverified online or overseas sources. FDA is aware of fraudulent compounded semaglutide and tirzepatide in the U.S., including products with false label information, pharmacy identities that don't exist, or the names of real licensed pharmacies that didn't actually compound the product. Patients have been hospitalized.
Don't assume "same active ingredient" means same regulatory status or same quality. FDA specifically flagged this type of marketing language in its February and March 2026 warning letter waves.
Don't buy from any source selling GLP-1s without requiring a prescription or a real medical evaluation. That's a bright-line safety signal.
Don't stockpile a year of vials from a questionable source to 'get ahead of regulation.' Beyond-use dates exist for a reason. Sterility degrades.
Don't wait until your last dose is gone to ask for help. Patients who wait two weeks hoping the problem resolves itself, then panic on the day they'd be injecting, have the worst outcomes. You have more runway than you feel like you do — use it.
Don't confuse Reddit reassurance with a refill plan. One message to your prescriber and one matching quiz will do more for you than four hours of doom-scrolling.
A few real things people have said (community voice, not medical evidence)
"What if they just stop selling after the date? What then?"
"I'm left stranded."
"Any chatter on a compounding pharmacy shutting down?"
Source: r/SemaglutideFreeSpeech
If you’ve thought any of the above in the last week — you’re not alone and you’re not overreacting. The path forward exists. Most of the uncertainty you’re feeling right now comes from not knowing which of the four scenarios in section 2 happened. Name that, and the next move becomes clear.
13. Frequently Asked Questions
Still not sure which GLP-1 program is right for you?
Take our free 60-second matching quiz. Tell us your state, your medication history, and how urgent your next dose is — we’ll route you to the single best option. Whether you need a transfer, a new prescription, a brand-name bridge, or a supervised pause, we’ll match you to one. No signup. No commitment.
Methodology & sources (verified April 22, 2026)
We built this page by:
- Reading FDA’s April 1, 2026 “essentially a copy” clarification and the underlying Section 503A guidance
- Reviewing every entry on FDA’s Compounding: Inspections, Recalls, and other Actions page dated 2024–2026
- Pulling current self-pay pricing from LillyDirect, NovoCare Pharmacy, and Ro.co on April 22, 2026
- Cross-referencing warning letter coverage from Pharmacy Times, Spencer Fane, Foley & Lardner, Buchanan Ingersoll, and Partnership for Safe Medicines
- Opening official state pharmacy closure rules for California, New York, Texas, Florida, and Utah
- Reviewing patient-voice language from public forums
What we could not verify:
- The exact current enforcement status of every 503A pharmacy in the U.S. (FDA enforcement is continuous — check FDA.gov directly)
- Every state’s specific closure rules (we spot-checked five)
- The outcome of pending litigation (Novo v. Hims & Hers; Novo and Lilly DDC list petitions)
- State-specific consumer protection refund timelines
Update cadence: Monthly — FDA warning letters, recalls, compounding actions, manufacturer pricing; Quarterly — state closure rule table, enforcement tracker; Immediately — any new FDA statement or major action that materially affects patient continuity.
Related guides
- Best GLP-1 online programs — full comparison including compounded and brand-name options
- Cheapest Wegovy online (2026) — verified self-pay paths by situation
- Cheapest Zepbound online (2026) — LillyDirect Journey Program and all alternatives
- Wegovy cost without insurance — pen, pill, NovoCare, and telehealth all-in totals
- GLP-1 prior authorization guide — appeal scripts, PA criteria, and denial response templates
- Cheapest FDA-approved GLP-1 — every brand-name option compared by monthly cost
About Weight Loss Provider Guide: We are an independent comparison resource for GLP-1 telehealth providers. We may earn a commission if you start a plan through links on this page. On this specific disruption/continuity topic, our routing prioritizes the reader’s regulatory safety and medical continuity over affiliate priority — which is why MEDVi (currently under a February 2026 FDA warning letter over marketing language) is not a featured recommendation on this page even though we cover it in our broader provider reviews. Advertising disclosure. Editorial standards.
Last verified: April 22, 2026. Next scheduled re-verification: May 22, 2026.
Medical & legal disclaimer: This article is for informational purposes only and does not constitute medical or legal advice. Compounded medications are not FDA-reviewed for safety, effectiveness, or quality. Always consult a licensed clinician before stopping, starting, or switching any medication. If you are in a medical emergency, call 911 or your local emergency number.