GLP-1 Insomnia: Why You Can't Sleep on Semaglutide or Tirzepatide — and What to Do
By Weight Loss Provider Guide Editorial Team · Last verified: April 30, 2026
Weight Loss Provider Guide is an independent comparison resource for GLP-1 telehealth providers. We may earn a commission from some links on this page. Our medical and regulatory claims are sourced separately from any commercial recommendations. Nothing on this page replaces advice from the clinician who prescribed your medication.
You took your shot. You went to bed exhausted. Now it's 3 AM and you're staring at the ceiling — wired, frustrated, wondering if your GLP-1 is the reason you can't sleep.
Your Situation Right Now
| If this is you… | Most likely next step |
|---|---|
| First or second night after shot day, otherwise normal sleep | Track it for 3 nights and watch for the pattern to fade |
| Waking at 3–4 AM, can't get back to sleep | Check the glucose-symptom pattern and the reflux pattern |
| Heart racing, sweating, shaking — especially if on insulin or a sulfonylurea | Call your prescriber promptly. This isn't plain insomnia. |
| Symptoms started right after a dose increase | Run the 4-week reset protocol |
| Vivid dreams or nightmares but you're sleeping through them | Read the dreams section — usually fades on its own |
| Snoring, gasping, daytime sleepiness | This may be sleep apnea — see our sleep apnea guide |
| On a compounded GLP-1 and unsure what's in your vial | See the compounded section — verify your formulation first |
| Severe vomiting, dehydration, or unsafe daytime fatigue | Contact a clinician promptly. This is past the "tips" stage. |
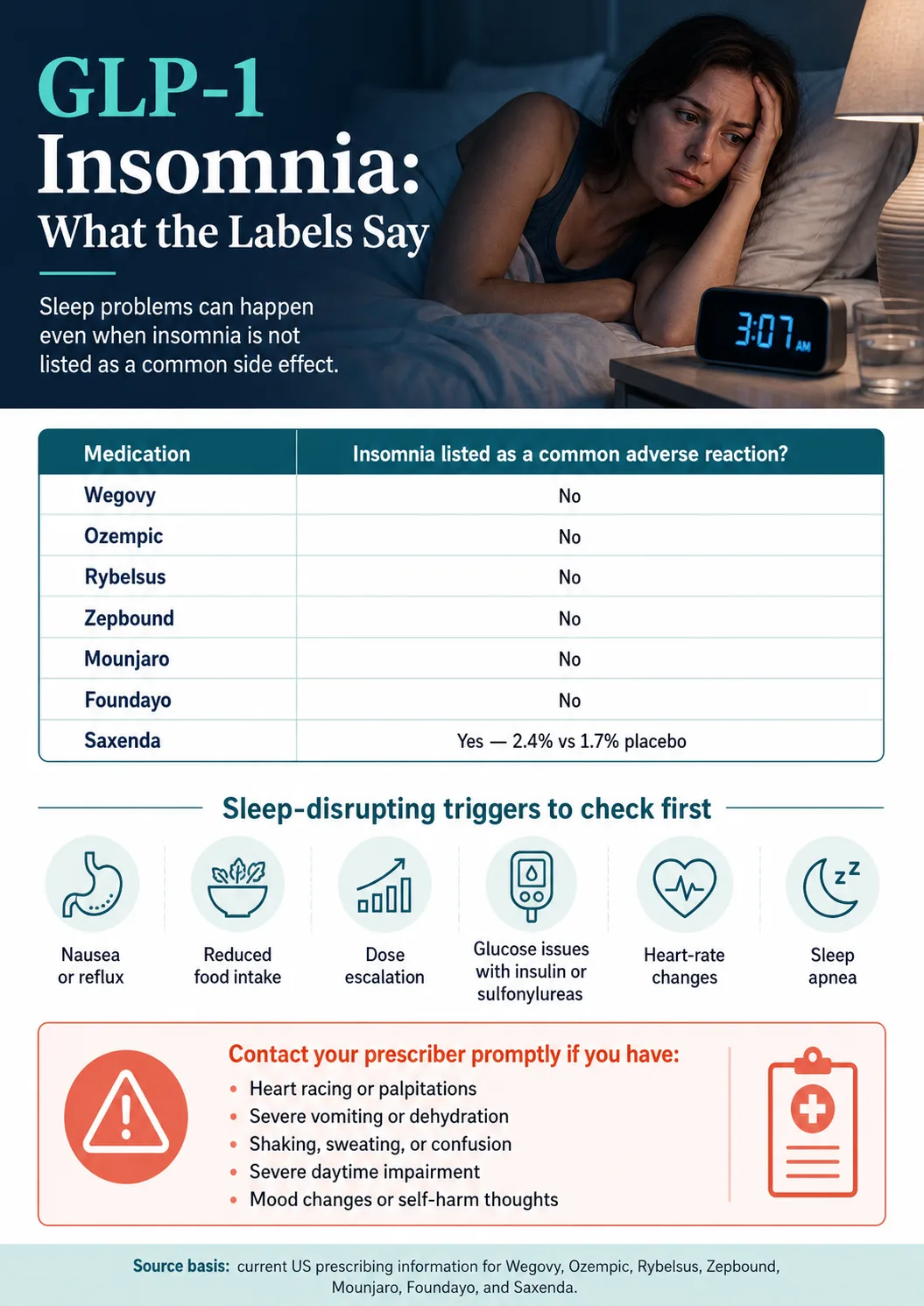
Does GLP-1 Cause Insomnia? Here's What the Evidence Actually Says
The FDA labels and the real-world reports tell different parts of the same story.
If you check the DailyMed prescribing information for Wegovy, Ozempic, Mounjaro, or Zepbound right now, you won't find "insomnia" in the list of common adverse reactions. The labels are correct. And the real-world reports are also correct. Both can be true.
The Chen et al. 2024 paper in Frontiers in Endocrinology ran a disproportionality analysis on GLP-1 medications using FAERS data. Two findings matter:
- Within the psychiatric adverse-event reports for GLP-1s, insomnia was the most-reported category — 1,198 reports, accounting for 11.7% of psychiatric reports.
- The narrower term "sleep disorder due to general medical condition — insomnia type" had a reporting odds ratio of 2.01 (95% CI 1.60–2.52). That means it shows up more often in GLP-1 reports than statistical expectation for this database.
A separate 2023 paper in Brain Sciences (Arillotta et al.) used a mixed-methods analysis of social media posts. Sleep-related issues were the largest category captured, with insomnia accounting for around 620 matches.
Our damaging admission — we'd rather lead with it
FAERS shows reporting signals, not proven causation. Anyone can submit a FAERS report. The database doesn't control for everything else happening in someone's life. Chen et al. explicitly note this limitation. So a 2x reporting signal is real and worth taking seriously — but it's not the same as a randomized trial showing "GLP-1 causes insomnia in X% of users."
The GLP-1 Insomnia Evidence Matrix (Label Check + Real-World Signal)
Labels pulled from DailyMed. FAERS class signal from Chen et al. 2024. Verified April 30, 2026.
| Medication | Active ingredient | Insomnia in current US label as common adverse reaction? | Sleep-relevant label effects | Heart-rate effect on label | Half-life |
|---|---|---|---|---|---|
| Wegovy (injection) | Semaglutide 2.4 mg | No | Nausea, diarrhea, vomiting, constipation, abdominal pain, headache, fatigue, dyspepsia, dizziness, eructation, GERD | Not flagged as common AE | ~7 days |
| Wegovy tablets (oral) | Semaglutide oral | No | Same GI profile as injection; must be taken morning, empty stomach | Not flagged as common AE | ~7 days |
| Ozempic | Semaglutide 0.25–2.0 mg | No | Nausea, vomiting, diarrhea, abdominal pain, constipation; delayed gastric emptying; hypoglycemia risk with insulin/sulfonylurea | Not flagged as common AE | ~7 days |
| Rybelsus | Semaglutide oral 7–14 mg | No | Nausea, abdominal pain, diarrhea, decreased appetite, vomiting, constipation | Not flagged as common AE | ~7 days |
| Zepbound | Tirzepatide 2.5–15 mg | No | Nausea, diarrhea, vomiting, constipation, abdominal pain, dyspepsia, fatigue, GERD, eructation, hair loss | Mean HR increase 1–3 bpm vs placebo | ~5 days |
| Mounjaro | Tirzepatide 2.5–15 mg | No | Nausea, diarrhea, decreased appetite, vomiting, constipation, dyspepsia, abdominal pain | Mean HR increase 2–4 bpm vs 1 bpm placebo; more sinus tachycardia events than placebo | ~5 days |
| Saxenda | Liraglutide 3.0 mg | Yes — 2.4% vs 1.7% placebo in pivotal trials | GI effects, fatigue, anxiety | Not flagged at the same level | ~13 hours (daily injection) |
| Foundayo | Orforglipron (oral, daily) | No exact "insomnia" match in current label | Nausea, constipation, diarrhea, vomiting, dyspepsia, headache, fatigue, GERD, flatulence, hair loss | Tachycardia-related terms 3% vs 0.9% placebo; mean HR increase 4–5 bpm vs 0.5 bpm placebo | ~29–49 hours (oral, daily) |
| Compounded semaglutide / tirzepatide | Compounded — not FDA-approved as finished products | N/A — no FDA-approved label for compounded finished products | Depends on specific preparation, dose, pharmacy, and any additives | N/A | N/A |
Sources: DailyMed prescribing information linked throughout; Chen et al. 2024 FAERS analysis. All percentages are reported adverse-event rates from labels — not direct head-to-head trial comparisons unless noted. Foundayo data was not part of Chen et al. (the analysis predates its 2026 approval).
Note on Foundayo's heart-rate effect
Foundayo's current label reports a noticeably larger mean heart-rate increase than the weekly injectables (4–5 bpm vs 0.5 bpm placebo). For some users that may correspond to the "wired at night" pattern. Foundayo is also too new for long-term real-world post-market data.
A compliance note on compounded products: Compounded semaglutide and compounded tirzepatide are not FDA-approved finished medications. Formulation, concentration, pharmacy source, additives, and dosing instructions can differ between compounding pharmacies. We won't blur that line.
6 Reasons GLP-1 Medications Mess With Your Sleep (and How to Tell Which One Is You)
1. The "Wired but Tired" Pattern
You're physically exhausted. Your body wants to sleep. But your brain is racing, your heart feels a little faster than it should, and lying still makes everything worse. Patients consistently describe this as "wired but tired."
What might be going on: Tirzepatide products (Zepbound, Mounjaro) report small mean heart-rate increases on label — 1–3 bpm for Zepbound, 2–4 bpm for Mounjaro versus 1 bpm for placebo. Foundayo reports a larger mean increase (4–5 bpm). A small average increase can feel disproportionately big at bedtime when everything else is quiet.
What's worth trying
- Move the shot to morning instead of evening (weekly GLP-1 labels permit any time of day)
- Cut caffeine after noon — many users find they're more sensitive to it now
- Consider magnesium glycinate before bed (confirm dose with your prescriber or pharmacist)
- Brain-dump on paper for 5 minutes before lights out
2. The "3 AM Wake-Up" Pattern
You fall asleep fine. Then 3:30 AM hits and you're wide awake — sometimes sweating, sometimes hungry, sometimes shaky.
What might be going on: If you take insulin or a sulfonylurea alongside your GLP-1, hypoglycemia overnight is a real risk. The Ozempic, Mounjaro, and Zepbound labels all warn about increased hypoglycemia risk in this combination. Even outside that combination, lower food intake from suppressed appetite can cause overnight glucose to dip enough to wake you.
What's worth trying (only if you're not on insulin or a sulfonylurea)
- Small protein-and-fat snack about an hour before bed: handful of nuts, a hard-boiled egg, plain Greek yogurt
- Don't go to bed underfed if you've barely eaten that day
- If you're diabetic and use a glucose meter or CGM, check overnight readings
3. The "Vivid Dreams" Pattern
You're sleeping. You're just having the most intense, weird, sometimes alarming dreams of your life. Nightmares. Dreams so vivid you wake up unsure what was real.
What might be going on: Vivid dreams aren't formally listed as a side effect in the labels we checked. The phenomenon has been widely reported in patient communities, but the mechanism isn't established in current peer-reviewed evidence. For most people, this pattern fades within weeks.
What's worth trying
- Reduce alcohol, which disrupts REM (the dreaming stage of sleep) on its own
- No screens in the 30 minutes before bed
- This pattern alone is rarely a reason to stop the medication
4. The "Nausea or Reflux at Night" Pattern
You lie down. Your stomach feels too full. You're a little nauseous. You're burping. By the time you finally drift off, you wake up at 1 AM with reflux.
What might be going on: GLP-1 medications slow gastric emptying — that's part of how they work. Food stays in the stomach longer than it used to. Lying flat makes reflux worse. The Zepbound label explicitly lists GERD among common adverse reactions; Wegovy lists eructation and GERD-type symptoms. See our GERD-focused provider guide for more.
What's worth trying
- Smaller dinner, eaten 3+ hours before bed
- Avoid spicy, fatty, and very high-fiber foods at dinner specifically
- Elevate the head of your bed by 4–6 inches (a wedge pillow works)
- If you inject in the evening, talk to your prescriber about shifting to morning
5. The "Shot-Night Insomnia" Pattern
You sleep fine most nights. But the night you took your shot — or the next night — you didn't sleep. And next week, same thing.
What might be going on: Many users find side effects feel most noticeable in the first 1–3 days after a weekly injection, when drug levels are rising. Most people don't notice the pattern until they look at it on a calendar.
What's worth trying
- Shift injection to a morning instead of evening (weekly GLP-1 labels permit any time of day)
- Move shot day to mid-week so the bad night doesn't fall on a workday
- Don't change days more than once every 2–3 weeks — drug levels need time to stabilize
6. The "Post-Dose-Escalation Crash" Pattern
You were sleeping fine. Then you stepped up to the next dose and the insomnia came back.
What might be going on: Each dose increase can re-trigger the early side-effect window your body had already adapted to. Both the Zepbound and Wegovy labels include explicit language about delaying the next dose escalation if a patient doesn't tolerate the current step — that's a prescriber-directed adjustment.
What's worth trying
- Ask your prescriber about holding the current dose for an extra 4 weeks before escalating
- Some labels include lower maintenance dose options — your prescriber decides
- Sleep often re-stabilizes within a few weeks of holding
Patient-reported quotes from Arillotta et al. 2023 — peer-reviewed social media analysis. These are patient-reported language, not medical evidence.
"Anyone noticed that tirz has affected their sleep? I have a really hard time falling asleep and when I sleep I usually toss and turn and wake up over and over, then wake up in the morning feeling literally hungover."
"After the initiation of GLP-1 RAs, losing weight was associated with either a marked improvement or, in some cases, a deterioration, in mood; increase/decrease in anxiety/insomnia."
The point: people experience this differently. Some get better sleep. Some get worse sleep. Many fall in the middle and adapt over weeks.
Find Your Pattern: 5-Question GLP-1 Insomnia Decoder
Answer these 5 questions in your head (or on paper). Each answer narrows you toward the most likely pattern.
Question 1: When does the sleep problem hit you most?
- A) Almost always at bedtime — can't fall asleep
- B) I fall asleep fine but wake at 3–4 AM
- C) Mostly on shot night or the next night
- D) Right after my last dose increase, regardless of which night
- E) I'm sleeping but the dreams are intense
Question 2: What does it feel like in your body?
- A) Wired but tired — heart a little faster, brain racing
- B) Sweaty, shaky, or hungry on waking
- C) Full, nauseous, or refluxy when I lie down
- D) Fine physically, just can't get comfortable
- E) Vivid or strange dreams, but I wake up rested-ish
Question 3: Are you on insulin or a sulfonylurea?
- A) Yes
- B) No
- C) I'm not sure
Question 4: When did the symptoms start?
- A) Within the first 2 weeks of starting the medication
- B) Within 2 weeks of my last dose increase
- C) They've come and gone for more than 2 months
Question 5: Do you snore, gasp during sleep, or feel exhausted no matter how long you sleep?
- A) Yes, frequently
- B) No
- C) I don't know — I sleep alone
If you answered Yes to Q5, the issue may be sleep apnea. See our sleep apnea guide.
Your most likely pattern
| Mostly answered | Most likely pattern |
|---|---|
| 1A + 2A | Wired but tired → Pattern 1 above |
| 1B + 2B | 3 AM glucose-symptom pattern — call prescriber if on insulin/sulfonylurea → Pattern 2 above |
| 1A or 1B + 2C | Reflux / nighttime stomach pattern → Pattern 4 above |
| 1C + any | Shot-night peak pattern → Pattern 5 above |
| 1D + 4B | Dose-escalation pattern → Pattern 6 above |
| 1E + 2E | Vivid dreams pattern → Pattern 3 above |
| Mostly Q5 = Yes | Possible obstructive sleep apnea — see sleep apnea section below |
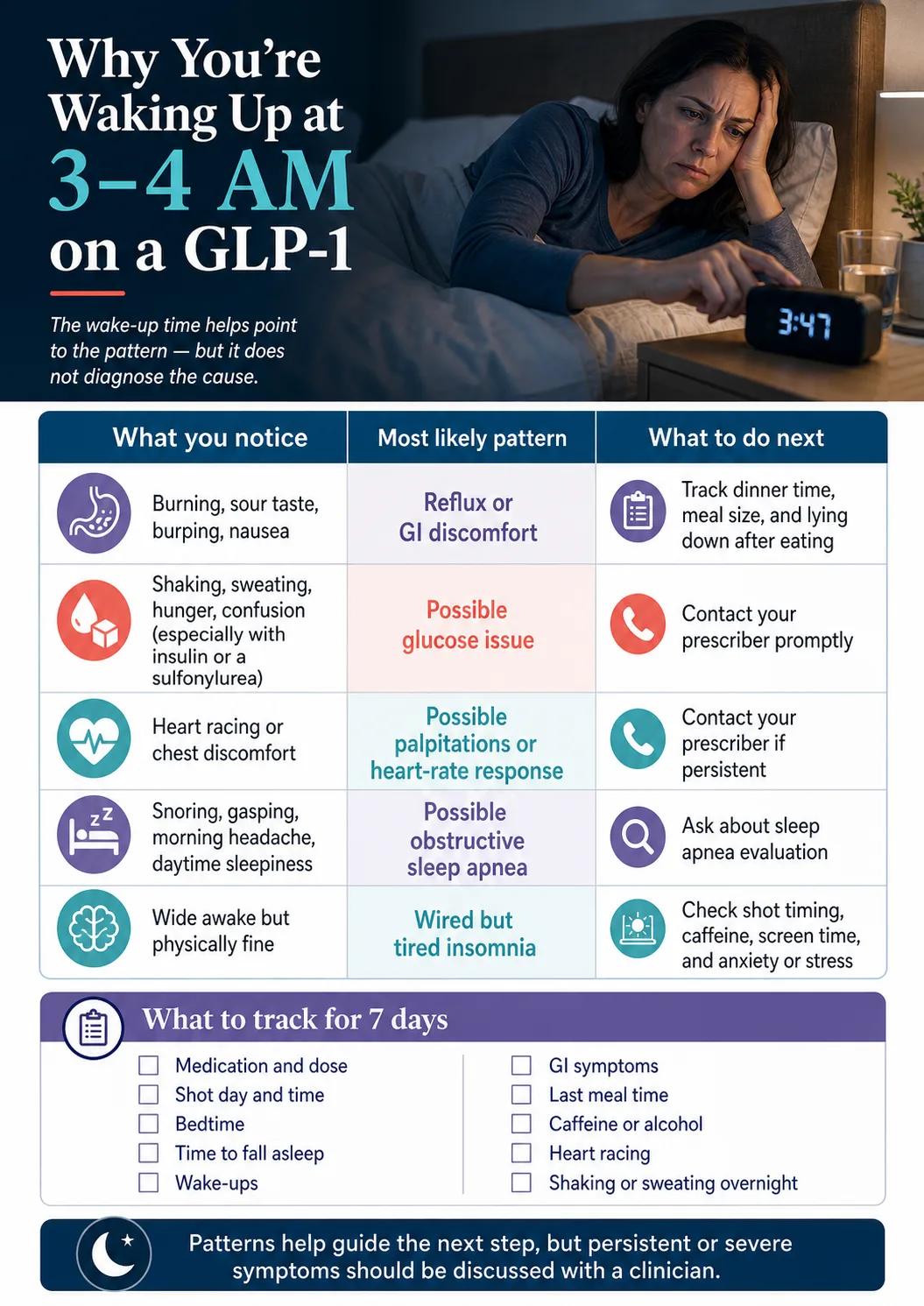
How Long Does GLP-1 Insomnia Last?
| Window | What typically happens |
|---|---|
| Days 0–14 | Adjustment window. Sleep can be disrupted on shot nights specifically. Don't panic-stop the medication this early. |
| Day 31 (median onset) | Midpoint of when GLP-1-related psychiatric AEs were reported in Chen et al. If you're going to develop symptoms, this is the typical window. |
| Weeks 4–8 | For many people, a small fix — timing change, lighter dinner, dose hold — resolves it. |
| Weeks 8+ | If you're still struggling, run the 4-week reset protocol below if you haven't, then escalate to your prescriber with data. |
| Beyond 12 weeks | Persistent insomnia despite a structured approach is uncommon and warrants a clinician conversation. |
Is GLP-1 Insomnia Dangerous?
What Should I Do If I Can't Sleep on a GLP-1? Tonight's 5 Same-Night Moves
1. Small protein + fat snack about an hour before bed (with a caveat)
The best non-pharmaceutical guard against the '3 AM hungry wake-up' pattern. Examples: small handful of nuts, a hard-boiled egg, a tablespoon of peanut butter, plain Greek yogurt. If you take insulin or a sulfonylurea and you wake with sweating, shaking, or confusion, this isn't a snack problem — it's a medication conversation. Call your prescriber.
2. Magnesium glycinate
The glycinate form is generally better tolerated than magnesium oxide for evening use and less likely to cause GI upset. Confirm an appropriate dose with your prescriber or pharmacist, especially if you take other medications.
3. No screens for 30 minutes before bed
Blue light suppresses your evening melatonin. Even paper reading for 20 minutes is better. This is one of the cheapest interventions in the entire sleep playbook.
4. Cool bedroom
Sleep onset depends on a small drop in core body temperature. A cool, comfortable room makes that drop easier. CDC guidance supports cool, quiet, dark, and comfortable.
5. 5-minute brain-dump on paper before lights-out
The "tired but wired" mental loop is the most common complaint we see. Externalizing whatever your mind is racing on — onto paper — quiets the loop. Your brain stops re-running thoughts when it knows they're written down.
The 4-Week GLP-1 Sleep Reset Protocol
Week 1 — Same-Night Tactics + Sleep Symptom Journal
Run the five same-night moves above. Keep a simple log. Each night, record:
Don't change anything else this week. The goal is data.
Week 2 — Dose-Timing Adjustment
Look at the journal. If you see a clear shot-night pattern, ask about moving your shot to morning, or moving shot day mid-week. If you see an "after dinner" pattern, move dinner earlier and lighter. Wait 7 days. Don't change anything else.
Week 3 — Dose Hold
If you're due for a dose escalation in this window, ask your prescriber to hold at the current dose for an additional period. The Wegovy and Zepbound labels both include explicit language about delaying the next dose increase if you're not tolerating the current one — this is normal practice, not an unusual ask.
Week 4 — Evaluate and Decide
GLP-1 Insomnia by Medication: Wegovy, Ozempic, Zepbound, Mounjaro, Saxenda, Foundayo, Compounded
Wegovy (semaglutide 2.4 mg) and Insomnia
The current Wegovy prescribing information does not list insomnia among common adverse reactions. Most Wegovy-related sleep disruption traces to one of those listed effects (nighttime nausea, reflux, fatigue) rather than a direct sleep effect. Half-life is approximately 7 days, so the medication clears slowly — which is why "just stop and see" isn't a clean one-week experiment.
A note on the January 2026 label change: the previous Wegovy label included a monitoring statement on suicidal behavior and ideation. That section was removed in early 2026 based on accumulated clinical-trial evidence that did not show a clear causal link. Any new mood changes should still always be reported to your prescriber regardless of label status.
Ozempic (semaglutide 0.25–2.0 mg) and Insomnia
Same active ingredient as Wegovy at lower doses. Insomnia is not in the labeled common adverse reactions. The big sleep-relevant consideration: Ozempic's label warns that combining it with insulin or a sulfonylurea increases hypoglycemia risk, including overnight. If you're on that combination and waking up shaky, sweating, or confused — that is a glucose event, not "Ozempic insomnia" — and your prescriber needs to know.
Zepbound and Mounjaro (tirzepatide) and Insomnia
Insomnia was not identified as significant versus placebo in the SURPASS or SURMOUNT trials. Heart-rate effects worth knowing: Zepbound's label reports a mean HR increase of 1–3 bpm. Mounjaro's label reports 2–4 bpm vs 1 bpm placebo, with more sinus tachycardia events.
Saxenda (liraglutide 3.0 mg) and Insomnia
Saxenda is the exception. Its US prescribing information does list insomnia in the adverse-event table at 2.4% Saxenda versus 1.7% placebo. Saxenda is a daily injection with a short ~13-hour half-life — the side-effect curve repeats every day rather than peaking once per week. If you switched from Saxenda to weekly Wegovy or Zepbound, your side-effect timing genuinely changed, which can be confusing.
Foundayo (orforglipron) and Insomnia
Foundayo is FDA-approved once-daily oral GLP-1, approved in 2026. Insomnia is not an exact match in the current label. The label fact most relevant to sleep: tachycardia-related terms were reported in 3% of Foundayo-treated patients versus 0.9% placebo, and mean HR increase was 4–5 bpm versus 0.5 bpm placebo — noticeably larger than the weekly injectables. Half-life is approximately 29–49 hours.
Compounded Semaglutide and Compounded Tirzepatide and Insomnia
Compounded GLP-1 products are not FDA-approved finished medications. There's no FDA-approved label for any specific compounded preparation, no published clinical trial data on the finished compounded product.
For your sleep symptoms specifically: formulation, concentration, additives (some compounded preparations include B12, B6, or other adjuncts), and dose-titration schedules can differ between compounding pharmacies. If you switched between brand and compounded and your symptoms changed at the switch, those formulation variables are worth raising with your prescriber.
The single most useful thing you can do if you're on a compounded product: Know your pharmacy's name, the exact concentration, the dose units (mg vs mL vs "units"), and whether anything besides the GLP-1 is in your vial.
Why Do People Wake Up at 3–4 AM on GLP-1s?
| What you notice when you wake | Most likely pattern | What to track | Where to go next |
|---|---|---|---|
| Burning, sour taste, burping, nausea | Reflux / GI discomfort | Dinner time, meal size, lying-down posture | Reflux pattern |
| Shaking, sweating, hunger, confusion | Possible glucose dip — especially if on insulin or a sulfonylurea | Diabetes medications, glucose readings | Call your prescriber |
| Heart racing, chest discomfort | Possible palpitations / heart-rate response | Pulse, timing, dose, caffeine | Call your prescriber if persistent |
| Snoring, gasping, morning headache, daytime sleepiness | Possible obstructive sleep apnea | Snoring, witnessed pauses, daytime fatigue | Sleep apnea guide |
| Wide awake but physically fine | "Wired but tired" insomnia | Bedtime routine, screen time, caffeine, anxiety | Tonight's 5 moves |
If you have a smart watch or fitness tracker that records heart rate during sleep, the data on those wake-ups is gold. Bring it to your prescriber.
Should You Take Your GLP-1 Shot in the Morning or at Night for Better Sleep?
Move shot to morning if:
- Your worst sleep is shot night specifically
- You inject in the evening and feel "wired" within a few hours
- Your nausea or reflux peaks in the first 4–8 hours after dosing
Keep evening dosing if:
- You don't have shot-night insomnia
- Peak nausea during the day after morning dosing — some people would rather sleep through the worst nausea than work through it
Move shot day mid-week if:
- You're sleeping fine on shot night but next-day fatigue wrecks workdays
- Your shot day currently lands on a Sunday or Monday
Sleep Aids and GLP-1s: What's Generally Safe, What to Avoid
Generally well-tolerated alongside GLP-1s
- Magnesium glycinate — well-tolerated, less GI upset than magnesium oxide
- Low-dose melatonin — higher doses often work worse; talk to a pharmacist about dose
- L-theanine — promotes calm without sedation
- Glycine before bed — some evidence for sleep quality, generally well-tolerated
Use with caution / talk to prescriber first
- Diphenhydramine (Benadryl, ZzzQuil, PM formulations) — adds to next-day grogginess that overlaps with GLP-1 fatigue
- Doxylamine (Unisom) — same caution
- CBD products — variable quality, may interact with other medications
Prescription decisions, not OTC fixes
- Z-drugs (zolpidem, eszopiclone) — your prescriber decides
- Trazodone — your prescriber decides
- Any prescription sleep medication — your prescriber decides
Avoid combining sleep aids with alcohol. Alcohol disrupts REM sleep on its own. Many GLP-1 users find their alcohol tolerance is meaningfully lower than it was — both the GI effects and the next-day fatigue stack badly.
When to Call Your Prescriber: 7 Red Flags
- Persistent insomnia beyond 8 weeks despite a structured protocol
- New or worsening mood changes — depression, anxiety, irritability, intrusive thoughts that weren't there before
- Signs of overnight hypoglycemia — sweating, shaking, confusion on waking, severe hunger — especially if you take insulin or a sulfonylurea
- Severe daytime impairment — falling asleep at the wheel, missing work, cognitive impairment that affects your safety
- Heart racing or chest discomfort at night, especially across multiple nights
- Severe overnight nausea or vomiting that prevents you from staying hydrated
- Any thoughts of self-harm or suicide. This is always urgent. Call 988 (US Suicide & Crisis Lifeline) or your local emergency number first, then your prescriber.
What to say when you call — copy this script
"I started [medication name] on [date]. My current dose is [dose]. I'm also taking [list other medications]. Sleep disruption started around day [X]. The pattern is [pick one or more: can't fall asleep / waking at 3 AM / shot-night insomnia / vivid dreams / heart racing / nausea or reflux at night / dose-escalation crash]. I've already tried [list interventions from the 4-week protocol]. I'd like to discuss [pick one or more: dose timing / holding the dose / switching class / evaluating for sleep apnea / sleep aid options]."
This script frames you as someone who's tracked the problem, which gets you better answers.
Should You Lower the Dose, Switch GLP-1s, or Stop?
When dose adjustment may be the conversation
Talk to your prescriber about a dose hold or slower escalation if: insomnia started right after a dose increase; your current side-effect profile is intolerable but was fine at the previous dose; you haven't held a dose for the labeled minimum before escalating. The labels for Wegovy, Ozempic, Zepbound, and Mounjaro all include language permitting delayed escalation if the current dose isn't tolerated.
When switching may be the conversation
A class switch (semaglutide → tirzepatide or vice versa) may be discussed if symptoms are severe and didn't respond to dose-timing or dose-hold adjustments, or if you want to try Zepbound (the only GLP-1 FDA-approved for OSA) because you suspect sleep apnea is part of the picture. Honest framing: there's no head-to-head trial showing one GLP-1 is better than another for sleep.
When stopping may be the conversation
Only when: symptoms are severe enough to outweigh the metabolic benefit; a red flag from the list above is present; or you've completed a structured protocol with prescriber guidance and the medication isn't tolerable. Do not self-stop as a routine insomnia experiment. GLP-1s have a long half-life (5–7 days for weekly injectables), so the "I'll skip a week and see" experiment doesn't give you a clean read for 2–3 weeks. That said, if you have urgent symptoms per the labels — suspected pancreatitis, serious allergic reaction, severe dehydration, or self-harm thoughts — follow the label instructions and contact a clinician.
GLP-1s and Sleep Apnea: The Counterintuitive Good News
If you're searching "GLP-1 insomnia" but you also snore, wake up unrefreshed, or feel constantly sleepy, you may have undiagnosed obstructive sleep apnea — a condition where breathing repeatedly stops and starts during sleep, jolting you out of deep stages all night without you realizing it.
Talk to your prescriber about a sleep study if any of these are true:
- Loud snoring most nights
- Witnessed pauses in breathing (your partner notices)
- Gasping or choking awake
- Morning headaches
- Heavy daytime sleepiness despite "enough" hours in bed
- Waking up feeling unrefreshed
What to Track Before Your Next Prescriber Appointment
| Date | Med & dose | Shot day? | Bedtime | Time to fall asleep | Wake-ups | Final wake time | GI symptoms | Last meal time | Caffeine after noon? | Alcohol? | Heart racing? | Shaking/sweating? | Notes |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Copy this into your notes app or print it. Fill in one row per night for at least a week before your appointment. | |||||||||||||
The five questions worth asking your prescriber
- Could this be tied to my dose escalation schedule?
- Does shifting injection timing help my pattern based on what I tracked?
- Should I hold this dose longer before the next increase?
- Could reflux, undereating, or hypoglycemia be the real driver based on this log?
- Should I be evaluated for sleep apnea?
If You're Considering Switching Programs
Some readers conclude — fairly — that the issue isn't the medication itself. It's the program around it: a prescriber who isn't responsive to side-effect concerns, a program that escalates doses on a fixed schedule regardless of tolerance, or one that doesn't include enough clinician contact to actually troubleshoot.
The criteria that matter most for tolerability: clinician access (24/7 messaging beats once-a-month video), dose-escalation flexibility (can they hold the dose? can they step down?), and clear cancellation terms.
| Provider | What they offer | Sleep-relevant note | Verified |
|---|---|---|---|
| Eden | FDA-approved Wegovy and Zepbound (HSA/FSA-eligible). Zepbound at $1,399/month and Wegovy at $1,695/month. No membership fee. Same-price-at-every-dose. | Strong fit if you want FDA-approved branded medication and predictable pricing as your dose changes | April 2026 |
| Ro | FDA-approved Foundayo, Wegovy pill, Wegovy pen, Zepbound pen, and Zepbound KwikPen. Wegovy pill from $149 first month, Foundayo from $149 first month, Zepbound KwikPen from $299 first month. Ro Body membership separate: $39 first month, then $149/month (or $74/month annual). Insurance concierge handles prior-authorization; free GLP-1 Insurance Coverage Checker. | Strong fit if you want FDA-approved brand-name medication and want a clinical team to handle insurance | April 2026 |
| SHED | Specializes in compounded oral and sublingual semaglutide and tirzepatide. Compounded products are not FDA-approved as finished medications. | Worth considering only if you specifically want a non-injection compounded option and have discussed it with a clinician | April 2026 |
These are descriptions of what each program currently offers, not endorsements. Verify all pricing and policies on the provider's site before enrolling.
GLP-1 Insomnia FAQs
Does GLP-1 cause insomnia?
Insomnia is not listed as a common adverse reaction in the US FDA prescribing information for most GLP-1 medications. But the Chen et al. 2024 FAERS analysis identified insomnia as the most-reported psychiatric adverse-event category in this drug class (1,198 reports, 11.7% of psychiatric reports), and the narrower 'sleep disorder — insomnia type' category showed a disproportional reporting signal (ROR 2.01). The honest answer: it's a real-world reporting signal, not a labeled side effect, and not proof of causation.
Does Wegovy cause insomnia?
Wegovy's current US prescribing information does not list insomnia among common adverse reactions. The most common reactions are GI symptoms — nausea, diarrhea, vomiting, constipation, abdominal pain — plus headache, fatigue, dyspepsia, dizziness, and GERD. Sleep disruption on Wegovy most often traces to one of those listed effects rather than a direct sleep effect.
Does Ozempic cause insomnia?
Ozempic's listed common adverse reactions include nausea, vomiting, diarrhea, abdominal pain, and constipation — not insomnia. The biggest sleep-relevant consideration is hypoglycemia risk when Ozempic is combined with insulin or a sulfonylurea. Overnight shaking, sweating, or confusion in that combination is a glucose event, not insomnia, and warrants a prescriber call.
Does Zepbound cause insomnia?
Insomnia was not identified as significant versus placebo in the SURMOUNT trials, and Zepbound's prescribing information does not list it among common adverse reactions. The label notes a mean heart-rate increase of 1–3 bpm versus placebo, which can correspond to the 'wired' sensation some users describe. Notably, Zepbound is FDA-approved to treat obstructive sleep apnea.
Does Mounjaro cause insomnia?
Mounjaro's listed common adverse reactions include nausea, diarrhea, decreased appetite, vomiting, constipation, dyspepsia, and abdominal pain. Insomnia is not on the list. The label reports a mean heart-rate increase of 2–4 bpm versus 1 bpm placebo, with more sinus tachycardia events than placebo.
Does semaglutide cause insomnia?
The same answer applies across the semaglutide products (Wegovy, Ozempic, Rybelsus, Wegovy oral): insomnia is not in their US labels at the common-adverse-reaction threshold, but the FAERS class signal applies. Indirect mechanisms — nighttime nausea, reflux, glucose dips — explain most reports.
Does tirzepatide cause insomnia?
Tirzepatide's labels (Mounjaro and Zepbound) do not list insomnia at the common-adverse-reaction threshold. Mean heart-rate increases of 1–4 bpm versus placebo are reported and may correspond to the 'wired' sensation some users report. Tirzepatide is also FDA-approved to treat obstructive sleep apnea.
Does Foundayo cause insomnia?
Foundayo's current label does not list 'insomnia' as an exact match. The label does report tachycardia-related terms in 3% of Foundayo-treated patients versus 0.9% placebo, and a mean heart-rate increase of 4–5 bpm versus 0.5 bpm placebo — a noticeably larger effect than the weekly injectables.
How long does GLP-1 insomnia last?
In Chen et al.'s 2024 FAERS analysis, the median time to onset for GLP-1-related psychiatric adverse-event reports was 31 days (interquartile range 7–145 days). Many people adapt over weeks. Persistent insomnia beyond 8 weeks despite a structured protocol warrants a prescriber conversation.
Should I take my GLP-1 in the morning or at night?
The FDA prescribing information for weekly injectable GLP-1s permits administration at any time of day, with or without meals. For sleep specifically, many users find that morning injection or shifting injection day to mid-week reduces shot-night insomnia. Consistency week-to-week matters more than the specific hour. (Note: Wegovy oral tablets and Rybelsus must be taken in the morning on an empty stomach.)
Is melatonin safe to take with semaglutide or tirzepatide?
Most over-the-counter sleep aids haven't been formally studied alongside GLP-1 medications. Confirm any new sleep aid or supplement with the clinician who prescribed your GLP-1, especially if you take other medications.
Will Ozempic give me weird dreams?
Vivid or unusual dreams are not officially listed as a side effect in the labels we checked. The phenomenon has been widely reported in patient communities and in mainstream coverage, but the mechanism isn't established in current peer-reviewed evidence. For most users this self-limits within a few weeks.
Can compounded GLP-1s cause insomnia?
Compounded products are not FDA-approved as finished medications and don't carry approved labels. There's no published trial data specific to a compounded finished product, and formulation variables can differ between compounding pharmacies. The most useful step if you're on a compounded product is to verify your pharmacy, exact concentration, dose units, and any additives directly with your provider.
Is GLP-1 insomnia a sign I should stop the medication?
Almost never as a first move. Stopping usually means losing the metabolic benefit you started for, and the side effect often resolves with dose-timing or dose-hold adjustments. Consider stopping (in conversation with your prescriber) only after a structured protocol has failed for 8+ weeks, or if red-flag symptoms like new mood changes, severe daytime impairment, or thoughts of self-harm appear.
Still Not Sure What to Do Next?
If you're earlier in the journey — you haven't started a GLP-1 yet, or you've decided the issue isn't the medication but the program around it — the cleanest next step is our free 60-second matching quiz. It walks you through your situation: insurance, medication preference, brand vs compounded, budget, and what kind of clinician support you actually need.
We don't ask for your email to use it.
References
- Wegovy Prescribing Information. DailyMed (current revision).
- Ozempic Prescribing Information. DailyMed (current revision).
- Rybelsus / Wegovy Tablets Prescribing Information. DailyMed.
- Zepbound Prescribing Information. DailyMed.
- Mounjaro Prescribing Information. DailyMed.
- Foundayo (orforglipron) Prescribing Information. DailyMed.
- Chen W., et al. (2024). Psychiatric adverse events associated with GLP-1 receptor agonists: a real-world pharmacovigilance study based on the FDA Adverse Event Reporting System database. Frontiers in Endocrinology.
- Arillotta D., et al. (2023). GLP-1 Receptor Agonists and Related Mental Health Issues: Insights from a Range of Social Media Platforms Using a Mixed-Methods Approach. Brain Sciences (PMC10669484).
- FDA. FDA Approves First Medication for Obstructive Sleep Apnea.
- FDA. FDA Intends to Take Action Against Non-FDA-Approved GLP-1 Drugs.
- CDC. About Sleep.
- 988 Suicide and Crisis Lifeline. Available 24/7 by call or text in the United States.
Last verified: April 30, 2026. This page is informational and is not medical advice. It does not replace the relationship with the clinician who prescribed your medication. If you are experiencing severe symptoms, signs of hypoglycemia, or any thoughts of self-harm, contact your prescriber or call 988 (US Suicide & Crisis Lifeline) immediately.
Weight Loss Provider Guide is an independent comparison resource for GLP-1 telehealth providers. We may earn a commission from some links on this page. This does not affect our rankings or analysis.