GLP-1 Ileus: Symptoms, Risk Factors, and Exactly When to Get Help
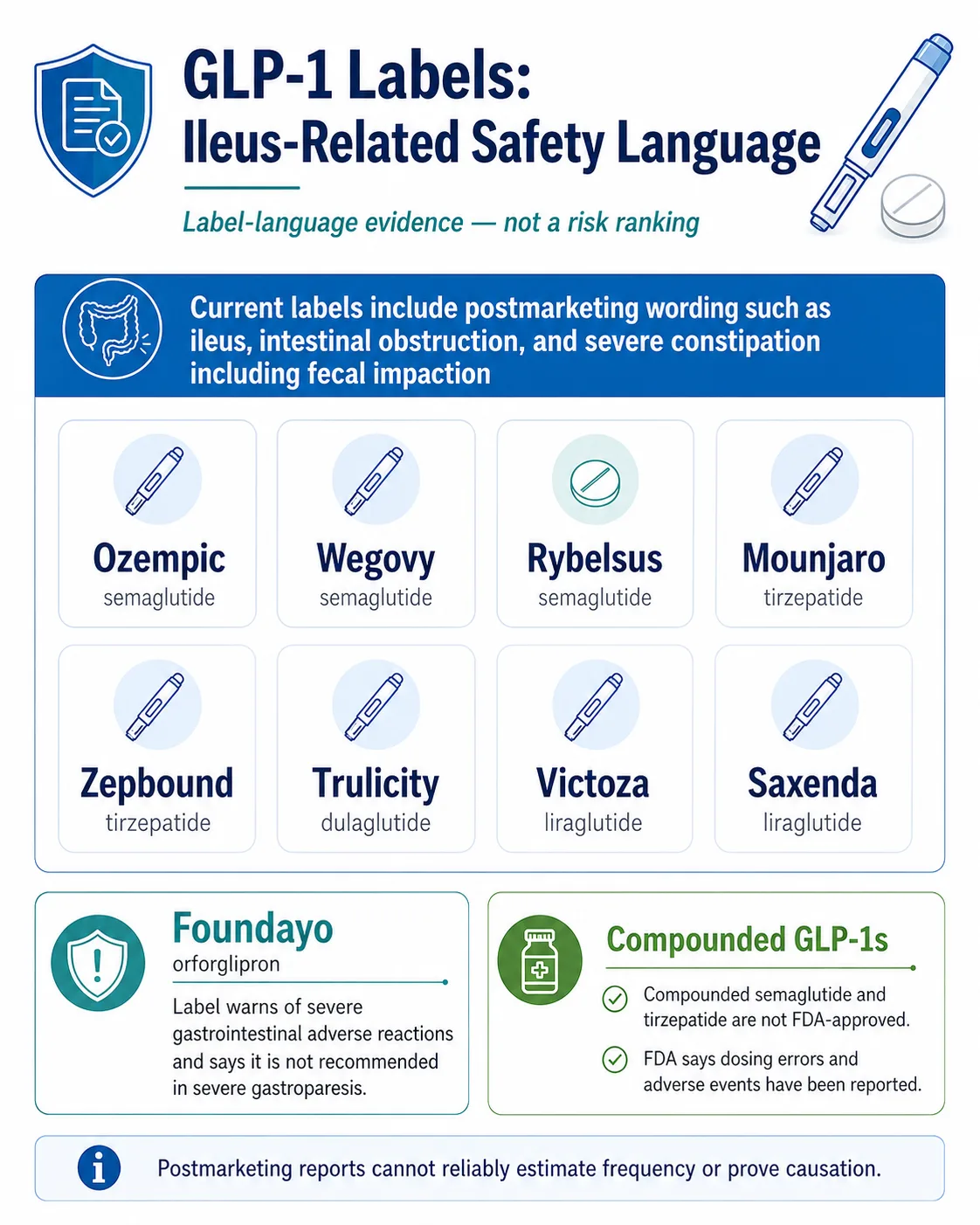
GLP-1 ileus is a rare but real slowdown of the intestines that has been reported in people taking semaglutide, tirzepatide, liraglutide, dulaglutide, and orforglipron. Every major FDA-approved GLP-1 — Ozempic, Wegovy, Rybelsus, Zepbound, Mounjaro, Trulicity, Victoza, Saxenda, and the newly approved Foundayo — now lists ileus, intestinal obstruction, or severe constipation in postmarketing safety language on its FDA label. The absolute risk is low. The pattern that matters is the combination of symptoms — not a single symptom in isolation.
Quick triage: where do you fit right now?
| Your situation | What to do |
|---|---|
| Severe / worsening belly pain + vomiting + swollen or hard abdomen + no gas or stool | ER or urgent evaluation now |
| Constipation that's worsening despite usual measures, plus pain, vomiting, or dehydration | Call your prescriber or urgent care today |
| Mild constipation, still passing gas, no vomiting, drinking fluids fine | Monitor, hydrate, contact prescriber if it persists |
| Just learned the word “ileus” and want to understand the risk before your next dose | Read on — start with the FDA label matrix |
What is GLP-1 ileus, exactly?
The plain-English version
Your intestines are a one-way conveyor belt. Food and waste move along it because the muscles in the bowel wall squeeze in coordinated waves — peristalsis. The underlying rhythm is called the migrating motor complex.
Ileus is what happens when the conveyor belt slows to a crawl or stops. Stuff doesn't move. Pressure builds. Fluid backs up. The belly swells. Vomiting starts. If it goes on long enough, the bowel can lose blood supply, which is the dangerous endgame nobody wants to reach.
GLP-1 medications — semaglutide, tirzepatide, liraglutide, dulaglutide, orforglipron — work in part by slowing this whole system down on purpose. That's why they reduce appetite. The food sits longer, you feel full longer, you eat less. Most people get away with this fine. A small minority slow down too much.
Ileus vs. ileum vs. bowel obstruction vs. gastroparesis
These get confused constantly. Worth pinning down:
| Term | What it actually means | Why people confuse it |
|---|---|---|
| Ileus | The intestines aren't moving normally (functional problem) | Sounds like "ileum" |
| Ileum | The last part of the small intestine (anatomy, not a disease) | Some legal pages say "ileal obstruction" |
| Bowel obstruction | Something physically blocks the intestine | Symptoms overlap with ileus |
| Gastroparesis | The stomach itself empties too slowly | GLP-1 labels use "delayed gastric emptying" — it's related but not the same as ileus |
| Severe constipation / fecal impaction | Stool becomes hard or impossible to pass | Can feel like obstruction; can become urgent |
How GLP-1 medications affect gut motility
Glucagon-like peptide-1 (GLP-1) is a hormone your gut already makes after meals. It tells your pancreas to release insulin, tells your brain you're full, and tells your stomach to empty more slowly. GLP-1 receptor agonists are drugs that mimic this hormone — at higher concentrations and for much longer than your body would naturally produce.
Slowed gastric emptying is on every GLP-1 label. It's not a bug; it's how the drugs work. The migrating motor complex in the small bowel is also affected — published research from Hellström and colleagues at the Karolinska Institute showed that GLP-1 inhibits the fasting-state contractions that sweep the small intestine clean between meals. (Hellström et al., Neurogastroenterology & Motility, 2008)
Stack that on top of dehydration, low fiber intake, opioids, anticholinergic medications, prior abdominal surgery, or pre-existing motility problems, and the math can occasionally tip into trouble.
Can GLP-1 medications cause ileus or bowel obstruction?
The FDA tracks adverse events that show up after a drug is approved through a system called FAERS — the FDA Adverse Event Reporting System. It's voluntary. Patients report what happened; clinicians report; manufacturers report. By mid-2023, FAERS had logged 33 reports of ileus from GLP-1 users with two reported deaths. (FDA FAERS data, via Fierce Pharma)
That signal triggered a label update. On September 20, 2023, the FDA required Novo Nordisk to add ileus to Ozempic's postmarketing experience section. (HCPLive coverage of FDA labeling change) Wegovy and Mounjaro already had similar language on their labels. By 2026, every major GLP-1 label includes ileus or intestinal obstruction language. The matrix below shows you exactly what each one says.
The label is what it is: a postmarketing safety signal from voluntary reports. The FDA itself cautions that these reports cannot reliably estimate frequency or establish causation. We'll cover what the actual research shows — and where it disagrees — in a section below.
When is GLP-1 ileus an emergency?
ER now: the red-flag cluster
Any of these on their own warrants urgent evaluation. Together, they're an emergency:
| Symptom pattern | Why it matters | Action |
|---|---|---|
| Severe or worsening abdominal pain | Can signal obstruction, ischemia, inflammation, or something else serious | ER / urgent evaluation |
| Persistent vomiting (or vomiting bile/feculent material) | Dehydration risk; classic obstruction sign | ER / urgent evaluation |
| Hard, swollen, or very tender abdomen | Concerning when paired with pain, vomiting, or no gas/stool | ER / urgent evaluation |
| Cannot pass gas and cannot pass stool | Especially with pain, vomiting, or distension | ER / urgent evaluation |
| Fever, fainting, confusion, severe weakness | Could indicate systemic illness or significant dehydration | Emergency care |
| Bloody or black stool | Not a typical "normal GLP-1 side effect" | Urgent medical care |
Same-day: call your prescriber or go to urgent care
Reach out today, not next week, if:
- ▸Constipation is worsening despite fluids, fiber, and OTC measures
- ▸New or persistent abdominal pain
- ▸More than one episode of vomiting
- ▸Trouble keeping fluids down
- ▸Severe GI symptoms after a recent dose increase
- ▸You have a history of bowel obstruction, severe gastroparesis, abdominal surgery, adhesions, hernia, or severe chronic constipation
Monitor at home — but don't ignore
Reasonable to wait and watch only if all of the following are true:
- ✓Mild constipation
- ✓Still passing gas
- ✓No vomiting
- ✓No severe pain
- ✓Able to hydrate
- ✓Trajectory is stable or improving
If you're constipated but the rest of the picture is calm, our GLP-1 constipation relief guide covers fiber timing, hydration, and the gentle laxative protocol most providers actually recommend.

What happens at the ER for possible ileus or bowel obstruction?
Knowing what to expect lowers the panic. A few things to internalize before you walk in:
- →
The exam is fast. A trained ER clinician can usually tell within minutes whether your abdomen is a "watch carefully" abdomen or a "needs imaging right now" abdomen.
- →
Imaging is targeted. They're not going to scan you for everything. They're going to look for the specific things ileus and obstruction show up as on the films.
- →
Most cases get better with time and IV fluids. Bowel rest means no food or drink for a stretch (usually 24–48 hours), with IV fluids replacing what you're losing. The bowel often resumes normal function on its own once the offending input — in this case, possibly the GLP-1 — is paused.
- →
Tell them you're on a GLP-1 immediately. It changes the differential and may change the imaging or sedation plan if they need to do a procedure.
Which GLP-1 drugs have ileus on the label? (FDA Label Evidence Matrix)
Verify each cell against the live label PDFs at accessdata.fda.gov before relying on it for medical decisions.
GLP-1 Ileus Label Evidence Matrix — verified April 30, 2026
| Drug / brand | Active ingredient | Maker | FDA-approved indication | Postmarketing label includes | Other relevant safety language |
|---|---|---|---|---|---|
| Ozempic | Semaglutide (injection) | Novo Nordisk | Type 2 diabetes; CV risk reduction; reducing risk of sustained eGFR decline, end-stage kidney disease, and CV death in adults with T2D and chronic kidney disease | Ileus, intestinal obstruction, severe constipation including fecal impaction (added Sep 20, 2023) | Delays gastric emptying; pulmonary aspiration during anesthesia/sedation noted in postmarketing |
| Wegovy (injection) | Semaglutide | Novo Nordisk | Chronic weight management in adults and adolescents 12+; CV risk reduction in adults with established CVD and obesity/overweight; MASH with F2–F3 fibrosis (accelerated approval) | Acute pancreatitis and necrotizing pancreatitis (sometimes resulting in death); ileus, intestinal obstruction, severe constipation including fecal impaction | Delays gastric emptying |
| Wegovy (oral tablet) | Semaglutide | Novo Nordisk | Chronic weight management; CV risk reduction in adults with established CVD and obesity/overweight | Same class postmarketing language as semaglutide injection | Delays gastric emptying |
| Rybelsus | Semaglutide (oral tablet) | Novo Nordisk | Type 2 diabetes; reducing risk of major adverse CV events in adults with T2D at high CV risk | Ileus, intestinal obstruction, severe constipation including fecal impaction | Severe GI adverse reaction language |
| Zepbound | Tirzepatide (injection) | Eli Lilly | Chronic weight management; obstructive sleep apnea in adults with obesity | Ileus, intestinal obstruction, severe constipation including fecal impaction | Severe GI adverse reaction warning; constipation rates listed; delayed gastric emptying |
| Mounjaro | Tirzepatide (injection) | Eli Lilly | Type 2 diabetes | Ileus, intestinal obstruction, severe constipation including fecal impaction | Delays gastric emptying |
| Foundayo | Orforglipron (oral) | Eli Lilly | Chronic weight management in adults with obesity, or overweight with weight-related comorbidity (FDA-approved April 1, 2026) | GLP-1 receptor agonist class postmarketing language; postmarketing data still accumulating given recent approval | Severe GI adverse reaction language; delayed gastric emptying |
| Trulicity | Dulaglutide (injection) | Eli Lilly | Type 2 diabetes; CV risk reduction | Ileus, intestinal obstruction, severe constipation including fecal impaction | Not recommended in severe gastroparesis; delays gastric emptying |
| Victoza | Liraglutide (injection) | Novo Nordisk | Type 2 diabetes | Ileus, intestinal obstruction, severe constipation including fecal impaction | Delays gastric emptying |
| Saxenda | Liraglutide (injection) | Novo Nordisk | Chronic weight management | Acute pancreatitis; hemorrhagic and necrotizing pancreatitis (sometimes resulting in death); ileus, intestinal obstruction, severe constipation including fecal impaction; nausea, vomiting and diarrhea leading to dehydration | Delays gastric emptying; not recommended in severe gastroparesis |
| Bydureon BCISE (discontinued Oct 28, 2024) | Exenatide ER (injection) | AstraZeneca | Was approved for type 2 diabetes; permanently discontinued by manufacturer | Historical label included ileus, plus nausea, vomiting, and/or diarrhea resulting in dehydration; abdominal distension; abdominal pain; eructation; constipation; flatulence | Delayed gastric emptying |
| Compounded semaglutide / tirzepatide | Semaglutide / tirzepatide (compounded) | Various 503A/503B pharmacies | Not FDA-approved | Not FDA-reviewed. Adverse-event reporting is less complete because federal law does not require state-licensed compounding pharmacies that are not outsourcing facilities to submit adverse-event reports to FDA. | FDA has issued specific concerns about compounded GLP-1 dosing errors, sourcing, and inappropriate salt forms |
Sources: DailyMed labels for each drug (cross-checked April 2026); FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss.
What we searched: Each label for the exact strings “ileus,” “intestinal obstruction,” “severe constipation,” “fecal impaction,” “gastric emptying,” and “pulmonary aspiration.”
What “postmarketing” actually means
When a drug is approved, the clinical trials are over. Real-world use surfaces things trials never could — events too rare to show up in 5,000-person studies but real enough to matter when 10 million people take the drug.
Postmarketing experience sections are how those signals get on the label. The FDA's standard caveat — printed on every postmarketing section — is essentially this: we cannot reliably estimate frequency or establish causation from these reports. That doesn't mean the events are fake. It means the label is being honest about what it can and can't prove.
What this matrix does not mean
- ▸It does not mean every drug above has the same ileus risk. We don't have head-to-head incidence data.
- ▸It does not mean compounded versions have been FDA-reviewed. They haven't.
- ▸It does not mean you should stop your medication based on this page alone. That's a clinician decision unless symptoms are urgent.
- ▸It does not replace seeing a doctor when something feels wrong.
How common is GLP-1 ileus, really?
The four major studies side by side
| Study | Year | Journal | Population | Sample | Compared to | Finding | What this can / can't prove |
|---|---|---|---|---|---|---|---|
| Sodhi et al. | 2023 | JAMA | Adults using GLP-1 for weight loss | 613 semaglutide + 4,144 liraglutide users | Bupropion-naltrexone (different weight-loss drug) | 4.22× higher bowel obstruction risk (HR 4.22, 95% CI 1.02–17.40) | Real signal but very small obstruction event count; CI 1.02–17.40 means precision is poor; comparator isn't a neutral reference |
| Faillie et al. | 2022 | Clin Pharmacol Ther | Type 2 diabetes patients | 25,617 GLP-1 users | SGLT-2 inhibitors | ~3.5× higher intestinal obstruction, risk peaking around 1.6 years of use | UK CPRD database; small event count limits power; longer-duration finding is the most novel insight |
| Ueda et al. | 2024 | Clin Gastroenterol Hepatol | T2D patients in Sweden, Denmark, Norway | 300,000+ | SGLT-2 inhibitors | No significant difference | Largest registry-based comparison to date; T2D-only population |
| Gao et al. | 2025 | Acta Diabetologica | T2D patients in US (TriNetX EHR) | 1.2 million (181,795 GLP-1 users) | 6 anti-diabetic drug classes | No increased risk vs. any class; reduced vs. insulin | Largest US study to date; T2D-only, no obesity-specific weight-loss arm |
Sources: Sodhi 2023 JAMA; Faillie 2022; Ueda 2024; Gao 2025 via NCBI PMC.
Why the studies disagree (and what to actually believe)
The Sodhi JAMA study had a small absolute number of obstruction events. The 95% confidence interval — 1.02 to 17.40 — tells you the precision was poor. A confidence interval that wide means the “true” effect could be anywhere from “barely above 1” to “17×.” Bupropion-naltrexone is also not a neutral reference drug; the comparison choice affects the relative risk calculation.
The Ueda Scandinavian study and the Gao 2025 TriNetX study compared GLP-1s to other diabetes drugs in much larger samples. In those comparisons, the relative risk shrinks to nothing or near nothing.
The honest synthesis: GLP-1s probably don't increase ileus risk much compared to similar diabetes drugs in similar patients. The relative-risk numbers from weight-loss-focused studies are imprecise and based on small event counts. The absolute rate is low across all of these — we're talking events that are rare enough that very large studies still struggle to count them precisely.
When the risk is highest
Dose escalation
GI symptoms — constipation, nausea, vomiting — are often worst in the first few days after starting a GLP-1 or after every dose increase. The first 12 weeks of treatment are when most people experience the strongest motility effects.
Cumulative exposure
The Faillie analysis found that intestinal obstruction risk peaks around 1.6 years of cumulative GLP-1 use — longer than the duration of most original clinical trials. That's part of why the signal showed up in postmarketing data.
So if you're either currently in an escalation phase OR you've been on the drug for over a year, those are the two windows where extra vigilance makes the most sense.
A note on FAERS reports
As of September 2023, the FDA had received over 8,500 reports of GI disorders associated with semaglutide products, including 33 reports specifically of ileus from GLP-1 users and 2 reported deaths. That number has continued to accumulate.(FDA FAERS via Fierce Pharma)
The serious caveat: FAERS is voluntary reporting from a denominator of millions of users. Reports cannot establish causation, they undercount real events, and they don't tell you per-person risk. They're a signal, which is exactly what the FDA used them for to update the label.
How to tell normal GLP-1 constipation from possible ileus
Side-by-side: what's normal vs. what's concerning
| Symptom | Likely normal GLP-1 side effect | Possible ileus / obstruction |
|---|---|---|
| Bowel movement | Less frequent; passes eventually with fiber, fluid, or gentle laxative | None at all even with intervention |
| Passing gas | Yes | No — this is the key tell |
| Abdomen | Soft, maybe uncomfortable, mild bloating | Hard, distended, tender to touch |
| Nausea | Worse after eating; improves between meals | Persistent, paired with vomiting |
| Vomiting | Occasional, especially after dose increase | Repeated; can't keep fluids down |
| Pain pattern | Mild, intermittent cramping | Crampy waves that escalate |
| Response to OTC laxatives | Improves within 24–48 hours | No improvement |
| Trajectory | Improves as body adapts | Worsens or persists |
| Hydration | Can drink fluids | Dehydration; reduced urination |
Why “days since last bowel movement” alone isn't enough
People fixate on the BM count. It's the wrong number to fixate on alone.
A more reliable indicator is whether you can still pass gas. The intestines move gas before they move stool. If you're still passing gas, the bowel is still moving, even if it's slow. If you've stopped passing gas and stool and your belly is hard and getting more bloated and you're nauseated — that combination is the pattern that warrants urgent evaluation.
A common mistake to avoid
Stacking laxatives at home when you can't pass gas, your belly is hard, and you're vomiting is dangerous. If the bowel is mechanically obstructed, more laxative isn't going to push the contents through — it's going to increase pressure on a system that can't relieve it. If you have severe pain, vomiting, distension, or no flatus, do not double-dose Miralax and ride it out. Get evaluated.
For mild constipation that doesn't fit the warning pattern, our GLP-1 constipation relief guide covers fiber timing, hydration math, and the gentle laxative protocol most providers actually recommend.
Who is most at risk for GLP-1 ileus?
Higher-caution history checklist
Each item raises baseline risk. The more that apply to you, the more important close monitoring and prescriber screening become:
The October 2024 ASA/AGA multi-society guidance specifically flags dose escalation, higher dose, weekly dosing, current GI symptoms, and conditions known to delay gastric emptying as the variables that move someone from “low-risk” to “elevated-risk” for GLP-1-related GI complications. These don't add up to a numeric score — they're risk factors a clinician weighs together.
Medication interactions that compound the risk
Several common medication classes slow gut motility on their own. Stacking them with a GLP-1 multiplies the effect:
- ▸Opioids (oxycodone, hydrocodone, tramadol, codeine) — even occasional use
- ▸Anticholinergics — diphenhydramine (Benadryl), oxybutynin, scopolamine, some tricyclic antidepressants
- ▸Iron supplements at high doses
- ▸Calcium-channel blockers (verapamil especially)
- ▸Aluminum-containing antacids
- ▸Some antiemetics — ironically, some of the drugs prescribed for GLP-1 nausea also slow the gut
If you take any of these regularly, mention them at every prescriber visit. A pharmacist consultation is genuinely worth the 10 minutes.
Five questions to ask before starting or escalating a GLP-1
“Given my history, what should I watch for in the first 12 weeks?”
“How many days without a bowel movement should trigger a call to your office?”
“Which symptoms mean urgent care vs. ER vs. monitor at home?”
“Who do I call after hours, on weekends, on holidays?”
“If I get severe GI symptoms, do I hold the next dose or come in first?”
A provider who can't answer these is a provider whose support infrastructure isn't built for the medication they're prescribing.
GLP-1s and surgery: the perioperative rules
Surgery is where GLP-1 problems most often go wrong. Postoperative ileus is already common — about 9% of patients develop it after even routine hysterectomy without any GLP-1 in the picture. (Cureus 2025 perioperative case report) Add a drug that delays gastric emptying and slows the small bowel, and the postoperative course gets more complicated.
The other risk surgery surfaces is pulmonary aspiration. If your stomach hasn't emptied before anesthesia, the contents can come up and into your lungs. That can be fatal. The current Ozempic DailyMed label notes pulmonary aspiration has occurred in GLP-1 receptor agonist patients undergoing elective surgeries or procedures requiring general anesthesia or deep sedation.
What the October 2024 multi-society guidance actually says
Five major medical societies — ASA, AGA, ASMBS (bariatric surgery), ISPCOP (perioperative care of obese patients), and SAGES (gastrointestinal/endoscopic surgeons) — issued joint guidance on October 29, 2024. The headline: most patients can continue their GLP-1 before elective surgery.
(stable dose, no GI symptoms, no other risk factors) can continue and follow standard fasting protocols.
(dose escalation, weekly injection, current GI symptoms, motility-slowing comedications) should follow a 24-hour clear liquid diet before the procedure with an adjusted anesthesia plan.
the procedure should be delayed.
(nausea, vomiting, abdominal pain, distension, constipation) should have those symptoms resolved before elective surgery.
The full guidance is on the ASA website and the AGA news page. If you're scheduling surgery, send your surgical team a link.
Endoscopy and colonoscopy
Same risk profile, slightly different concern. Retained gastric contents under sedation increase aspiration risk. The American Society for Gastrointestinal Endoscopy issued its own 2025 position statement — most platforms now ask about GLP-1 use in pre-procedure intake. Answer truthfully. If you've had your dose this week, say so.
Restarting after surgery
The lesson from published case reports
A 2025 Cureus case study describes a woman who restarted her GLP-1 on postoperative day one after a hysterectomy without specific instructions. Ten days later she presented with nausea, vomiting, and abdominal pain — concerning enough that exploratory laparotomy was scheduled before clinical improvement led to its cancellation. (Cureus 2025 perioperative case study)
The lesson: do not restart your GLP-1 after surgery without explicit instructions from your surgical team. “Resume your normal medications” is not specific enough.
What to do if you think your GLP-1 caused ileus
What to tell the ER (copy-ready)
If you're heading to the emergency department, this is what you say at triage:
— Copy and read this at triage —
“I'm taking [Ozempic / Wegovy / Zepbound / Mounjaro / Rybelsus / Foundayo / compounded semaglutide / compounded tirzepatide] at [dose]. My last dose was [date and time]. I [did / did not] recently increase my dose. I've had [list symptoms — pain, vomiting, bloating, no bowel movement, no gas] for [duration]. I'm worried about ileus or bowel obstruction because GLP-1 labels include serious GI warnings. My medical history includes [hernia / abdominal surgery / prior obstruction / gastroparesis / IBD / none of these].”
What to message your prescriber (copy-ready)
If you're not in immediate danger but you need a same-day or next-day touchpoint, send this through your portal:
— Copy and send through your patient portal —
“I'm having [list symptoms] while taking [drug] at [dose]. My last bowel movement was [date]. I last passed gas [date or time]. I [am / am not] vomiting. I [can / cannot] keep fluids down. My abdomen is [normal / bloated / swollen / hard / tender]. Pain level is [0–10]. I have a history of [list — or 'no relevant history']. Should I be evaluated today, and should I hold my next dose until I hear back from you?”
What to bring to the ER or appointment
| Item | Why |
|---|---|
| Medication pen, box, vial, or bottle | Exact name, dose, lot number |
| Dose and dose schedule | Most recent dose increase date |
| Pharmacy name | Especially important if compounded |
| List of recent meds taken | Laxatives, fiber, anti-nausea, opioids |
| Medical history list | Surgeries, prior obstructions, current conditions |
| Insurance card and ID | Standard |
| Phone with this page open | Reference for the FDA label language |
FDA MedWatch reporting
If a clinician confirms a serious adverse event tied to your GLP-1, ask whether the event should be reported through FDA MedWatch. You can submit your own report — it takes about 15 minutes. These reports are how postmarketing surveillance works. They're how the September 2023 label change happened in the first place. This is not the same as filing a lawsuit.
Should you stop your GLP-1 after a possible ileus?
The typical clinical course
In the published case literature, the pattern is reassuring: GLP-1 is held, conservative treatment (NPO, IV fluids, sometimes nasogastric decompression) is started, and within days the bowel function returns. (AJG 2024 case report); (Cureus 2025 case report)
The dangerous progression is when ileus tips into bowel ischemia — loss of blood supply to the gut. That requires emergency surgery. It's rare in the published reports, but it's the reason “let me ride this out at home” is a bad strategy when the warning signs are present.
Questions to ask before restarting
If you've had an ileus episode (or a near-miss that involved an ER visit, imaging, or holding doses), don't restart on autopilot. Take this list to your prescriber and a GI specialist:
- 1
Was ileus, obstruction, fecal impaction, or gastroparesis confirmed by imaging or other testing?
- 2
Did you think the GLP-1 contributed, or was something else more likely?
- 3
Was there a dose increase right before symptoms started?
- 4
Should I restart at a lower dose, slow the titration, switch medications, or stop?
- 5
What constipation prevention plan should I follow if I restart?
- 6
Which symptoms mean I should not take the next dose?
- 7
Who do I call after hours if symptoms return?
- 8
Should I see a gastroenterologist before restarting?
This is also a fair moment to evaluate whether your current provider is the right level of support. A provider who says “just take it again, you'll probably be fine” after a bowel-related ER visit is not a provider with the medical depth this medication requires.
Does compounded semaglutide or tirzepatide change the picture?
What FDA actually says about compounded GLP-1s
The FDA's own Concerns with Unapproved GLP-1 Drugs Used for Weight Loss page lays it out directly:
- ▸Compounded versions are not FDA-approved.
- ▸The FDA does not review them for safety, effectiveness, or quality before marketing.
- ▸The FDA has received adverse-event reports tied to compounded versions, including dosing errors that resulted in hospitalization. As of July 31, 2025, FDA had received 605 reports involving compounded semaglutide and 545 involving compounded tirzepatide.
- ▸Adverse events with compounded products are likely underreported because federal law does not require state-licensed compounding pharmacies that are not outsourcing facilities to submit adverse-event reports to FDA.
- ▸Some compounded products use unapproved salt forms of semaglutide that are not the same molecule as FDA-approved Ozempic or Wegovy.
- ▸Storage, shipping, and labeling vary by source.
What to verify if you're using a compounded GLP-1
The biggest practical failure mode in compounded GLP-1 programs is the support gap. Medication arrives on schedule; clinical guidance is thin; when something goes wrong, the patient doesn't know who to call.
If safety is your priority: a note on FDA-approved options
This is the only commercial routing block on this page. We held it until now on purpose. If you're not in the higher-risk category and you're happy with your current program, ignore this section — it's not for you.
When FDA-approved is genuinely the better path
Two practical advantages:
1. FDA-reviewed labeling and postmarketing surveillance
What's on the label has been through the regulatory process; what's reported afterward gets aggregated into the national database.
2. Manufacturing quality controls
Are inspected and verified by FDA. Dosing, storage, and labeling are regulated.
If you're low-risk, healthy, on a stable dose, and your current provider answers messages within hours, you're probably fine where you are. Don't switch for the sake of switching.
Insurance coverage and prior authorization: where to actually start
This is the only specific provider we're going to name on this page, because it's the only one we believe fits the specific reader who got this far on a safety page.
Ro
Ro is a telehealth platform that carries an FDA-approved GLP-1 lineup including Wegovy® (pill and pen), Foundayo™, Zepbound®, Zepbound® KwikPen, Ozempic®, and Saxenda®. (Ro weight-loss product page) What makes Ro relevant for a risk-aware reader specifically:
- ✓Dedicated insurance concierge — handles prior authorization paperwork — the part most people give up on
- ✓Free GLP-1 Insurance Coverage Checker — public and takes 60 seconds
- ✓Pricing transparency — Ro Body membership: $39 first month, then $74/month annual prepay or $149/month monthly. Medication cost is separate and depends on insurance.
60 seconds. We may earn a commission.
The damaging admission: Ro is not the cheapest option. Ongoing membership at standard pricing is $149/month — meaningfully more than the cheapest compounded programs — and your medication cost is on top of that. If your priority is the absolute lowest cash-pay cost and you're low-risk, our GLP-1 matching quiz and our best FDA-approved GLP-1 providers comparison route to better-fit options. But if you're a higher-risk reader who reached the end of a safety page, the insurance route is usually the right starting point — and Ro is the cleanest insurance route in the FDA-approved lane.
What we actually verified for this page
Source hierarchy
| Type of claim | Source we accept |
|---|---|
| FDA label language (ileus, obstruction, severe constipation) | DailyMed.nlm.nih.gov labels and accessdata.fda.gov label PDFs |
| Compounded GLP-1 regulatory facts | FDA's official compounded GLP-1 concerns page |
| Drug discontinuation status | FDA discontinuation notifications and DailyMed |
| Symptom and urgent-care guidance | Mayo Clinic, NIDDK, NCBI PMC peer-reviewed case reports |
| Incidence and risk research | Peer-reviewed primary literature in JAMA, Acta Diabetologica, Clin Gastroenterol Hepatol, Clin Pharmacol Ther |
| Perioperative guidance | Direct ASA / AGA / ASMBS / ISPCOP / SAGES press releases |
| Provider commercial claims (Ro pricing) | Ro's own pricing page, verified directly |
What we do not claim
- ▸We do not claim GLP-1s commonly cause ileus.
- ▸We do not diagnose anyone's symptoms.
- ▸We do not rank drugs by ileus risk based on postmarketing labels — postmarketing language doesn't support that comparison.
- ▸We do not claim compounded medications are equivalent to FDA-approved medications.
- ▸We do not use testimonials to prove medical safety or efficacy.
- ▸We do not have a "Medically Reviewed by Dr. _____" line on this page because we did not have a physician sign off on it. We will not invent one.
What needs re-verification before our next update
| Element | Cadence | Verification |
|---|---|---|
| FDA / DailyMed label language for each drug | Quarterly | Direct check of each DailyMed entry |
| Ro pricing and product list | Monthly | Direct check of Ro membership and product pages |
| New peer-reviewed studies | Quarterly | PubMed search for "GLP-1 ileus" / "GLP-1 bowel obstruction" |
| ASA/AGA perioperative guidance | Biannually | Society press release feeds |
| FDA safety communications | Monthly + alert-based | FDA safety communications page |
| FAERS adverse event totals | Annually | FDA FAERS dashboard |
Last verified: .
GLP-1 Ileus FAQ
Can Ozempic cause ileus?
Can Wegovy cause ileus?
Can Zepbound or Mounjaro cause bowel obstruction?
Is ileus the same as bowel obstruction?
Is ileus the same as gastroparesis?
How long without a bowel movement is dangerous on a GLP-1?
Should I take laxatives if I can't pass gas?
Should I skip my next GLP-1 dose if I have severe constipation or vomiting?
Can I restart a GLP-1 after an ileus episode?
Are compounded semaglutide and tirzepatide FDA-approved?
How do I report a suspected GLP-1 ileus event?
Do I really have to stop my GLP-1 before surgery?
Still not sure which GLP-1 program is right for you?
We built a free 60-second matching quiz that asks about your medication preference, budget, insurance status, risk factors, and support needs — then recommends the GLP-1 path that fits. It's the same logic we'd apply if a friend asked.
If you're having severe or worsening symptoms, please get medical care first. The quiz can wait.
Take our free 60-second matching quiz →Related guides on Weight Loss Provider Guide
- GLP-1 side effects: normal vs. ER-worthy — the broader triage hub
- GLP-1 constipation relief that actually works — for mild constipation without red flags
- GLP-1 matching quiz — for readers who want a personalized provider recommendation
- Compare FDA-approved GLP-1 provider options — for readers who want the insurance / approved medication path