What to Do If Your GLP-1 Provider Changes Pharmacies
By WPG Research Team · Published · Last verified:
This is a verification guide, not medical advice. For dosing, symptoms, diabetes management, missed doses, or whether to continue therapy, contact your prescriber or pharmacist. For emergency symptoms, call your local emergency number.
We re-check FDA, BeSafeRx, NABP, manufacturer pricing, and named-provider status monthly.
If you searched “what to do if your GLP-1 provider changes pharmacies,” here's the short answer: do not use the next shipment until you have done three things — verified the new pharmacy on your state's Board of Pharmacy license lookup, confirmed the concentration and dose in writing, and checked that the package arrived cold. A pharmacy change in 2026 can be a routine business or regulatory move, not a red flag by itself. But three things now decide whether the next dose is the medication you were prescribed.
| Type of change | What changed | What to verify |
|---|---|---|
| Dispensing pharmacy change | Same FDA-approved product (Wegovy, Zepbound, Ozempic, Mounjaro, Foundayo), different pharmacy filling it | Pharmacy license, label, cold shipping |
| Compounding pharmacy change | Compounded semaglutide or tirzepatide, source pharmacy changed | Everything in this guide — pharmacy license, 503A/503B status, active ingredient form, concentration, CoA, cold shipping |
| Product path change | Compounded → FDA-approved, or vice versa | Prescription details, new dose, new device, cost, refill timing |
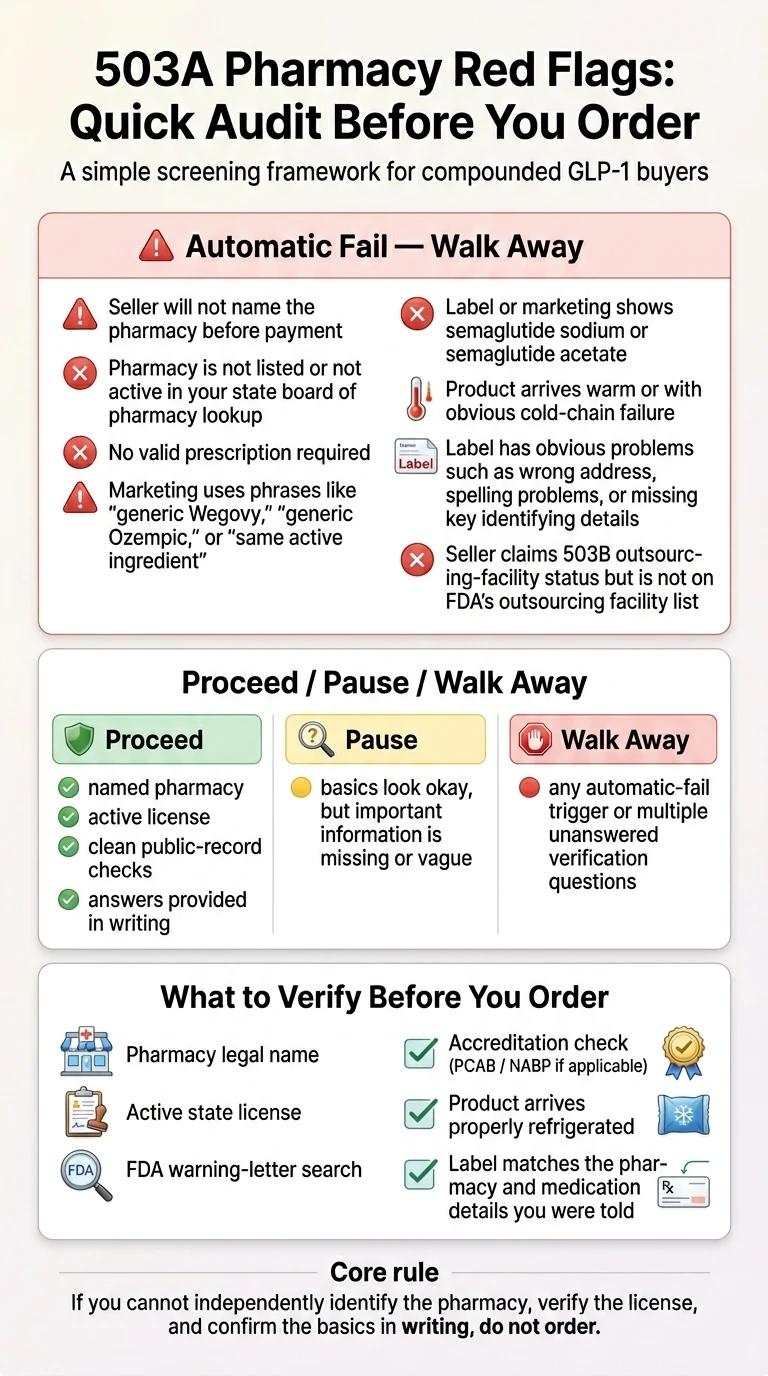
What to do if your GLP-1 provider changes pharmacies in the next 10 minutes
Quick answer: Before your next dose, run the package through a three-step triage — identify the new pharmacy by name, verify it on your state's Board of Pharmacy license lookup, and inspect the shipment for warm temperature, label errors, or a different concentration. Most failures show up at one of these three checkpoints.
| What you're seeing | What to do in the next 10 minutes |
|---|---|
You got an email or in-app notice that your pharmacy is changing, but no shipment yet | Reply asking for the legal name and state license of the new pharmacy before they ship. Don’t pay for the next month until you have it. |
Your new shipment arrived and the pharmacy name is one you don’t recognize | Look up the pharmacy in your state’s Board of Pharmacy license database (not their state — yours). Confirm an active license and, for injectable GLP-1s, an active sterile compounding authorization. |
| STOP The package arrived warm, or the ice packs were melted or missing | Do not inject. Take photos of the package, ice packs, and vial. Message your provider and pharmacy and request a replacement. The FDA has explicitly warned against using injectable GLP-1s that arrive warm or inadequately refrigerated. |
| STOP The vial size, concentration, or syringe units look different from your last refill | Do not draw a dose by guessing. Ask the pharmacy for your dose in mg, mL, and units in writing before you inject. The FDA has documented cases where patients gave themselves 5 to 20 times the intended dose because of mg/mL/unit confusion. |
| STOP The label has misspellings, a wrong address, or names a pharmacy that denies making it | This is a fraud red flag. Stop and call the named pharmacy directly using a phone number you find independently — not the number printed on the label. Report to FDA MedWatch. |
| STOP Your provider won’t tell you which pharmacy filled your prescription | Treat this as unresolved. The FDA’s March 2026 Warning Letters cited telehealth companies for obscuring product sourcing. Ask for the pharmacy’s legal name and license number in writing, and don’t inject until you have it. |
| STOP The label or your portal mentions retatrutide or cagrilintide | These cannot legally be used in compounding under federal law. Do not use. Contact your prescriber and consider reporting to the FDA. |
Everything matches your prescription, the pharmacy verifies on your state’s lookup, and the package arrived cold | You’re in good shape to continue as prescribed. Save the box, label, and instructions in case anything changes later. |
What we verified before publishing this guide
| What we checked | When | Source |
|---|---|---|
| FDA “Concerns with Unapproved GLP-1 Drugs Used for Weight Loss” | May 12, 2026 | fda.gov |
| FDA dosing-error alert on compounded injectable semaglutide | May 12, 2026 | fda.gov |
| FDA April 2026 compounding clarification | May 12, 2026 | fda.gov |
| FDA Registered Outsourcing Facilities table | May 12, 2026 | fda.gov |
| BeSafeRx state-licensed pharmacy guidance | May 12, 2026 | fda.gov |
| NABP Safe.Pharmacy directory and warning-sign criteria | May 12, 2026 | safe.pharmacy |
| NovoCare Pharmacy self-pay pricing for Wegovy and oral Wegovy | May 12, 2026 | novocare.com |
| LillyDirect self-pay pricing for Zepbound and Foundayo | May 12, 2026 | lilly.com |
| FDA Foundayo (orforglipron) approval (April 1, 2026) | May 12, 2026 | fda.gov press announcement |
Why your GLP-1 provider just changed pharmacies (2024\u20132026 enforcement timeline)
Quick answer: GLP-1 telehealth providers are changing pharmacies in 2025 and 2026 largely because the FDA declared the semaglutide and tirzepatide shortages over (semaglutide on February 21, 2025; tirzepatide on December 19, 2024), ended the period of enforcement discretion that had allowed compounding pharmacies to make near-copies of brand-name GLP-1s, and is actively warning and inspecting. There are seven common reasons a GLP-1 telehealth provider changes the pharmacy that fills your prescription. None of them are automatically bad — but the reason changes what you should verify.
- 1The previous pharmacy received an FDA Warning Letter or state board action. The most public recent example: the FDA’s Warning Letter to Boothwyn Pharmacy, LLC, issued January 16, 2026 and published February 10, citing insanitary conditions and strength, quality, or purity issues in its GLP-1 compounding.
- 2The previous pharmacy lacked the right license. In December 2024, Florida’s Department of Health filed an administrative complaint against Ousia Pharmacy over sterile compounding without the required license; Zappy, a telehealth company that had used Ousia, said it immediately ended that relationship.
- 3A manufacturer sued the telehealth provider over compounding practices. Eli Lilly sued Mochi Health, Fella Health, Willow Health, and Henry Meds on April 23, 2025 over compounded tirzepatide marketing. Novo Nordisk ended its collaboration with Hims & Hers in June 2025 and sued them in February 2026 over compounded semaglutide.
- 4The provider acquired a pharmacy or signed a new partnership. Eden Health acquired Contigo Compounding in August 2025 to bring 503A compounding in-house. On April 23, 2026, Hims & Hers said providers on its platform could send prescriptions for Zepbound and Foundayo to LillyDirect Pharmacy.
- 5The provider moved you from compounded to FDA-approved. With NovoCare Pharmacy and LillyDirect now offering self-pay pricing, more providers are routing patients to manufacturer-affiliated pharmacies.
- 6The compounding pharmacy stopped or changed its GLP-1 production. Several compounding pharmacies and outsourcing facilities have ceased or modified GLP-1 production after shortage and enforcement changes.
- 7Routine business reasons. Capacity, geography, shipping speed, or cost — unrelated to safety.
FDA and manufacturer enforcement timeline
| Date | Action | Source |
|---|---|---|
| Dec 19, 2024 | FDA declares tirzepatide shortage resolved | FDA Drug Shortages |
| Feb 21, 2025 | FDA confirms semaglutide shortage resolution | FDA Drug Shortages |
| Apr 23, 2025 | Eli Lilly sues Mochi Health, Fella Health, Willow Health, and Henry Meds over compounded tirzepatide | NPR; CNBC |
| Jun 2025 | Novo Nordisk ends collaboration with Hims & Hers over compounded semaglutide | Pharmacy Times |
| Aug 2025 | Eden Health acquires Contigo Compounding (in-house 503A) | Policy Labs |
| Sep 16, 2025 | FDA issues 55+ Warning Letters to online sellers of compounded GLP-1s for misleading marketing | FDA; Spencer Fane |
| Jan 16, 2026 | FDA Warning Letter to Boothwyn Pharmacy, LLC (published Feb 10) | FDA; Pharmacy Times |
| Feb 5, 2026 | TrumpRx launches direct-to-patient FDA-approved GLP-1 pricing | Spencer Fane |
| Feb 6, 2026 | FDA announces it will restrict GLP-1 active pharmaceutical ingredients in unapproved compounded products | FDA; McDermott+Plus |
| Feb 9, 2026 | Novo Nordisk sues Hims & Hers for patent infringement over compounded semaglutide | Pharmacy Times |
| Mar 3, 2026 | FDA issues 30 Warning Letters to telehealth companies in a single day over misleading compounded GLP-1 marketing | FDA press release; Venable LLP |
| Apr 1, 2026 | FDA approves Foundayo (orforglipron, Eli Lilly), a once-daily oral GLP-1 for weight management | FDA press announcement |
| Apr 1, 2026 | FDA reaffirms that enforcement discretion for routine 503A compounding of semaglutide injection has ended | FDA |
| Apr 23, 2026 | Hims & Hers integrates with LillyDirect Pharmacy for Zepbound and Foundayo | Hims & Hers newsroom |
| Apr 2026 | FDA proposes excluding semaglutide, tirzepatide, and liraglutide from 503B bulks list. Public comment through June 29, 2026 | FDA; US News |
Is the new GLP-1 pharmacy safe?
Quick answer: You cannot prove a new GLP-1 pharmacy is safe from a provider email alone. You can reduce risk by verifying the pharmacy's license in your state, checking sterile-compounding authority for injectables, confirming the prescription and concentration, and screening for the red flags the FDA has flagged. Verification reduces risk; it does not certify the product as safe, sterile, potent, effective, or legally compounded. Compounded drugs are not FDA-approved and are not reviewed by FDA for safety, effectiveness, or quality before marketing.
What “safe enough to continue” looks like
- The pharmacy is licensed in your state and reachable
- The label clearly identifies the compounder, not just the telehealth brand
- The active ingredient is semaglutide base or tirzepatide base
- The concentration and dose match your prescription, or any change is in writing
- The shipment arrived cold
- Nothing on the FDA’s red-flag list applies
What it does not look like
- A pharmacy that won’t return your call
- A telehealth provider that won’t disclose who compounded the drug
- A label that says “the same as” Wegovy or Zepbound, or implies generic status
- A vial that arrived warm or with melted ice packs
- Any mention of retatrutide or cagrilintide
How to verify the new pharmacy in 10 minutes
Quick answer: Look the pharmacy up by name on your state's Board of Pharmacy public license database, confirm an active sterile compounding authorization for injectable GLP-1s, check whether it has any disciplinary action on file, and cross-check using the FDA's BeSafeRx tools or the NABP Safe.Pharmacy directory. A pharmacy must be licensed in your state to legally ship to you, not just in its home state.
Run all 10 checks. If any item lands red and you can't resolve it before your next scheduled dose, don't take that dose until your prescriber or pharmacist answers it.
| # | Check | Green | Yellow | Red | Why it matters |
|---|---|---|---|---|---|
| 1 | Pharmacy is listed and active on your state's Board of Pharmacy license lookup | Active license, current | Active but expires in under 30 days | Not listed, expired, surrendered, or discipline on file | A pharmacy must be licensed in your state to legally ship to you. |
| 2 | Pharmacy carries a sterile compounding designation (or its non-resident equivalent) | Yes, on the license record | Pending | No, or not listed | Injectable compounded GLP-1s require sterile compounding authorization. The exact license name varies by state. |
| 3 | Pharmacy is identified as 503A or 503B | Clearly disclosed | Only on request | Won't say | Different rules apply. If they say 503B, cross-check against the FDA's Registered Outsourcing Facilities table. |
| 4 | Pharmacy will provide a lot-specific Certificate of Analysis (CoA) — third-party lab, covering identity, potency, sterility, endotoxins | Provided proactively or on request | "Available," with a slow timeline | Refuses, or only shows a generic example | A CoA can support quality documentation. Refusal to explain testing is a transparency problem. |
| 5 | Pharmacy holds PCAB accreditation (voluntary, not legally required) | PCAB accredited | Not PCAB but explains internal quality controls | Not accredited and won't explain quality process | Voluntary accreditation can be a quality signal. Its absence alone is not disqualifying. |
| 6 | Active ingredient is semaglutide base or tirzepatide base — not a salt form, retatrutide, or cagrilintide | Base form, confirmed in writing | Won't specify | Salt form, retatrutide, cagrilintide, or evasion | The FDA states semaglutide sodium/acetate are different active ingredients. Retatrutide and cagrilintide cannot lawfully be used in compounding. |
| 7 | Pharmacy has no recent FDA Warning Letter and no active state board discipline | Clean record | Older issue, since resolved | Active letter or pending action | Search the FDA Warning Letters database and your state board's public discipline records. |
| 8 | Label clearly names the actual compounder, not just the telehealth company's brand | Compounder named on the vial label | Compounder named only in paperwork | You can't tell who made it from the label | The FDA's March 2026 Warning Letters cited obscured product sourcing through telehealth-branded labels. |
| 9 | The compound is for an identified individual patient based on a valid prescription, not from a stock menu | Yes | Unclear | Sold as a stock product | Under 503A rules, compounds are for an identified individual patient. |
| 10 | The compounded formulation has a documented clinical reason to differ from the FDA-approved product | Documented in your chart | Vague | Marketed as "the same as" or "generic" Wegovy, Zepbound, Ozempic, or Mounjaro | Per FDA's April 2026 guidance, routine "essentially-a-copy" compounding of these GLP-1s is no longer allowed. Cost alone is not sufficient. |
How to use your state's Board of Pharmacy license lookup
Every U.S. state publishes a Board of Pharmacy license lookup, but the URLs, search fields, and license-type labels vary. Always look up the pharmacy in the state where you live, not the state where the pharmacy is based — a pharmacy shipping across state lines must hold a non-resident license in your state.
| State | How to look up | Sterile compounding label to look for |
|---|---|---|
| California | pharmacy.ca.gov/about/verify_lic.shtml | "Sterile Compounding" or "Non-Resident Sterile Compounding" |
| Texas | pharmacy.texas.gov/dbsearch/default.asp — use Pharmacy Search | "Class E-S (Non-Resident Sterile Compounding)" |
| Florida | Florida Department of Health MQA Search Portal | "Special Sterile Compounding" — also shows "Discipline on File: Yes/No" |
4 questions to ask your telehealth provider (and how to grade the answers)
Quick answer: Send your provider four written questions. Score each answer green, yellow, or red. Three or four greens means continue. Any unresolved red means pause until your prescriber or pharmacist answers it. Send these through your provider's patient portal or email — you want the answers in writing so you can verify and refer back.
Question 1: Which pharmacy is filling my prescription, and in which state is it licensed?
| A specific named pharmacy, with its home-state license number and a non-resident license in your state | Green |
| A "network" of pharmacies but they'll tell you which one before each shipment | Yellow |
| They can't or won't say until after you pay or until the package ships | Red |
Question 2: Is it a 503A pharmacy or a 503B outsourcing facility, and what's the documented clinical reason for compounding my prescription?
| 503A, patient-specific, with a documented clinical reason (excipient allergy, dose not commercially available, route change, etc.) | Green |
| 503A with a vague or generic reason | Yellow |
| "It's essentially the same as Wegovy / Zepbound at a lower price" — that exact framing is what the FDA has been enforcing against in 2025–2026 | Red |
Question 3: Can I see a Certificate of Analysis for my lot, from an independent third-party lab, covering identity, potency, sterility, and endotoxins?
| Yes, sent to your portal or emailed | Green |
| "Available on request," with a delay of more than a few days | Yellow |
| They've never heard of one, or only show a generic sample | Red |
Question 4: What is the active ingredient form — semaglutide base or a salt form?
| Base form — confirmed in writing | Green |
| They don't know and will get back to you | Yellow |
| Salt form (semaglutide sodium / semaglutide acetate), retatrutide, cagrilintide, or evasion | Red |
Copy-paste message template
Subject: Pharmacy change — verification before next dose
I received notice that my GLP-1 prescription is now being filled by a different pharmacy. Before my next dose, please confirm in writing: (1) the legal name and state license number of the new pharmacy, including its non-resident license in my state if it ships from out of state; (2) whether it operates as a 503A pharmacy or 503B outsourcing facility, and the documented clinical reason a compounded product is being used for me; (3) whether I can receive a Certificate of Analysis for my specific lot, from a third-party lab, covering identity, potency, sterility, and endotoxins; and (4) the active ingredient form — semaglutide base or tirzepatide base, versus a salt form or any other compound. I will not adjust my dose without written instructions from my prescriber or pharmacist. Thank you.
Save the response. If they push back on any of these, that is a signal in itself.
What to check on the vial, label, and prescription
Quick answer: Compare your new label to your old label field by field. If concentration or syringe units changed, do not convert the dose yourself. The FDA has documented cases where patients gave themselves 5 to 20 times the intended dose because of mg/mL/unit confusion with compounded semaglutide. Ask your prescriber or pharmacist for the new dose written out in mg, mL, and units.
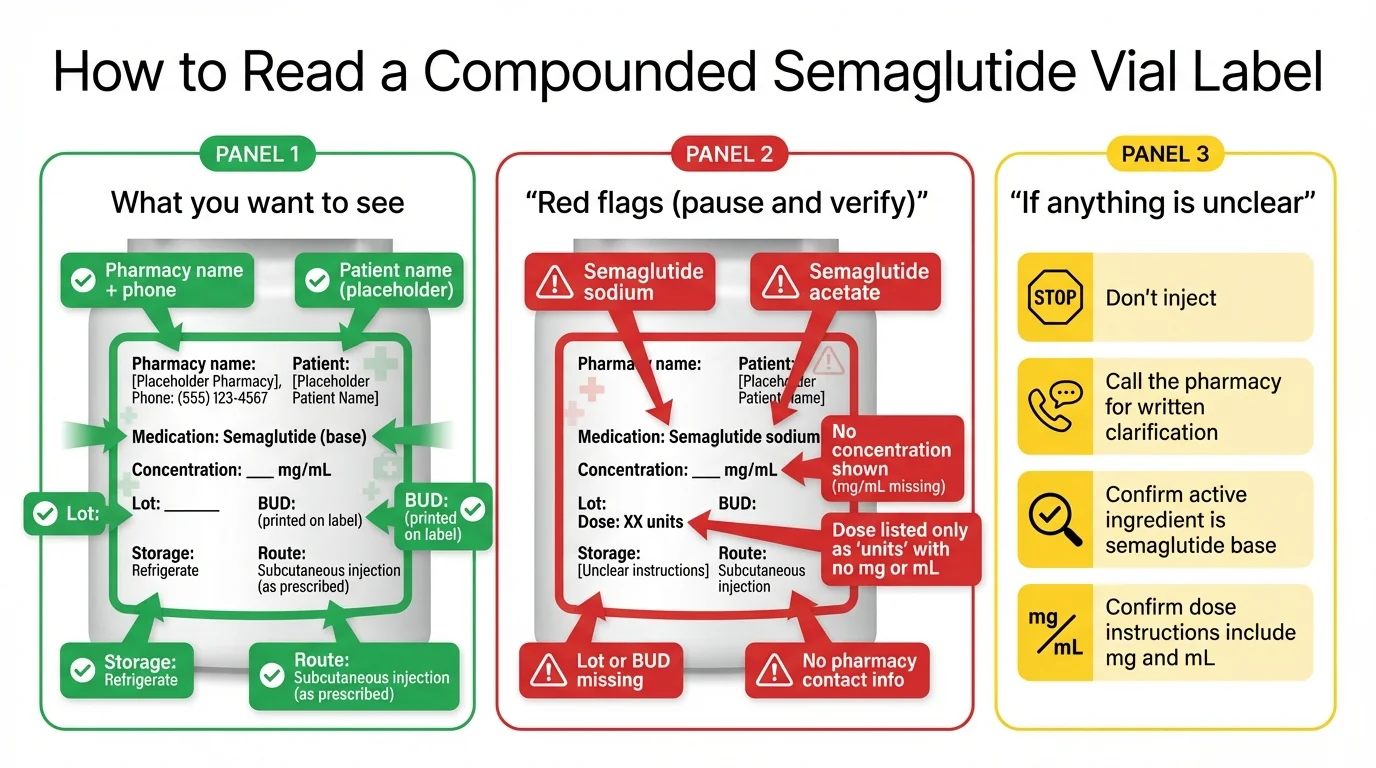
Side-by-side label check
Put your old vial and your new vial next to each other. Compare each row. If any row is different, don't inject until your prescriber or pharmacist confirms the new dose in writing.
| Field | Old vial | New vial | Same, changed, or unclear? | Action |
|---|---|---|---|---|
| Patient name | Must match yours exactly | |||
| Prescriber name | Should be your provider | |||
| Pharmacy name and address | Verify on your state's lookup | |||
| Drug | Confirm semaglutide / tirzepatide / other | |||
| Active ingredient form | Base, not salt or unapproved compound | |||
| Concentration (mg/mL) | If changed: do not self-convert | |||
| Dose | Get written instructions | |||
| Syringe units | Confirm against syringe markings | |||
| Beyond-use date (BUD) | Ask why if it changed | |||
| Lot number | Save a photo | |||
| Storage instructions | Refrigerate per label | |||
| Any added ingredients | Ask why anything is added |
Why concentration and unit changes are so dangerous
If your new vial has a different concentration, do not eyeball it. Do not use an online conversion tool. Get the new dose written out by your prescriber or pharmacist in three units at once: the dose in milligrams (mg), the volume in milliliters (mL), and the marking on your specific syringe.
What to do if your GLP-1 shipment arrived warm
Quick answer: If your injectable GLP-1 shipment arrived warm or with melted or missing ice packs, do not inject it.
The FDA recommends patients not use injectable GLP-1 products that arrive warm or inadequately refrigerated. Photograph the package, the ice packs, the vial, and any temperature strip. Message your provider and pharmacy, request a replacement, and ask in writing whether to delay the next dose or skip it.
What to do in the first 30 minutes
- 1Don't open the cap on the vial. Don't draw a dose.
- 2Photograph everything: the outer box, shipping label with delivery timestamp, ice packs (or where they should be), thermal liner, vial label, any temperature indicator strip.
- 3Put the vial in the refrigerator, clearly marked "do not use until pharmacy confirms." Don't throw it away — you may need it as evidence.
- 4Message your provider and pharmacy in writing (use the template below). Don't call alone — get it in text so you have a record.
- 5Wait for explicit instructions before doing anything else.
Copy-paste message for a warm shipment
Subject: GLP-1 shipment arrived warm — instructions needed before next dose
My GLP-1 shipment from [pharmacy name] arrived on [date and time]. The ice packs were [warm / melted / missing], and the vial felt [describe]. I have not used it. The shipping was [overnight / 2-day / other]. Photos attached.
Please confirm in writing: (1) whether this vial should be replaced and how; (2) whether I should skip, delay, or reschedule my next scheduled dose of [date]; (3) whether the next refill date will shift; and (4) any disposal instructions for the warm vial.
If you take GLP-1 for type 2 diabetes, not weight management
Red flags vs. routine changes: how to tell the difference
Quick answer: A routine pharmacy change comes with proactive notice, a named pharmacy you can verify, and an honest reason. Any one red-flag item is enough to pause and verify. Two or more unresolved red-flag rows — don't use the next shipment.
| Routine change | Red-flag change |
|---|---|
| You were notified in advance, by email or in-app, before any shipment | You only learned the pharmacy changed when the package arrived |
| The notice named the new pharmacy and gave a reason | The notice was vague or didn't name the pharmacy |
| The new pharmacy is searchable on your state's Board of Pharmacy lookup with an active license | The new pharmacy isn't listed in your state, or its license is expired or under discipline |
| The label clearly shows the compounder's name and address | The label shows only the telehealth company's brand, not who actually compounded the drug |
| The active ingredient is semaglutide base or tirzepatide base | The active ingredient is semaglutide sodium, semaglutide acetate, retatrutide, cagrilintide, or another flagged compound |
| The concentration and dose are unchanged, or you got written instructions for the new ones | The vial is a different size or concentration and nobody told you how to adjust |
| The package arrived cold, with intact ice packs | The package arrived warm or with melted or missing ice packs |
| The provider can answer the four verification questions in writing within a day | The provider dodges, delays, or refuses to identify the pharmacy |
| The provider's reason matches public information (a Warning Letter, lawsuit, partnership announcement) | The provider's reason is vague or contradicts what's publicly reported |
| The compounded product has a documented clinical reason to be different from the FDA-approved version | The marketing says the compounded product is "the same as," "generic," or "equivalent to" Wegovy, Zepbound, Ozempic, or Mounjaro |
How much do we actually know about compounded GLP-1 problems?
Can your telehealth provider change pharmacies without asking you?
Quick answer: Sometimes — it depends on your state, your provider agreement, and the pharmacy relationship. What does not change is your right to ask who is filling the prescription, verify the pharmacy before you use the shipment, and ask your prescriber about alternatives.
You can ask which pharmacy is filling each shipment, and you should expect an answer. A provider that refuses is operating against the spirit of the FDA’s 2026 Warning Letters about obscured sourcing.
You can refuse to accept a shipment from a pharmacy you don’t trust. Most telehealth providers will work with you on this, especially after you’ve documented a verification concern.
You can ask your prescriber to transfer or reissue your prescription. GLP-1s are not Schedule II–V controlled substances, but transfers still depend on state rules, pharmacy policy, refill status, and payer rules.
You can ask your prescriber about a manufacturer-direct option like NovoCare Pharmacy (for Wegovy or Ozempic) or LillyDirect (for Zepbound, Mounjaro, or Foundayo) if you want the FDA-approved product instead.
Should you continue, switch, or cancel? A decision tree
Quick answer: Your three options after a pharmacy change are continue (after running the 10-point checklist), switch providers (often to a manufacturer-direct channel or a different telehealth), or cancel and pause therapy. Find your situation below.
| Your situation | Best next move |
|---|---|
| You're titrating up your dose on compounded semaglutide or tirzepatide, the new pharmacy passes all 10 checks, and your dose and concentration didn't change | Continue. Request a CoA on your first shipment from the new pharmacy. Track any change in side effects or appetite control for the first 2–4 weeks. |
| You're titrating, the new pharmacy passes, but the concentration or syringe units changed | Continue only after your prescriber sends written dose instructions in mg, mL, and syringe units. Do not self-convert. |
| You're titrating and the new pharmacy fails one or more checks | Don't inject. Ask the provider for an alternative pharmacy. If they won't, switch providers. |
| You've reached your maintenance dose and the new pharmacy passes all 10 checks | Continue, with the same CoA-on-first-shipment check. Track effects for 2–4 weeks. |
| You've reached maintenance and want stability over price | Ask your prescriber about moving to an FDA-approved manufacturer-direct option. Current self-pay pricing is in the next section. |
| You can't afford brand-name and want to stay on compounded | The legal pathway is narrowing. Make sure the new pharmacy is a 503A, and that your prescriber has documented a patient-specific clinical reason beyond cost. Cost alone is not sufficient under the FDA's April 2026 guidance. |
| Your provider switched you to FDA-approved (Wegovy, Zepbound, Ozempic, Mounjaro, or Foundayo) via LillyDirect or NovoCare | Continue. Verify the new "pharmacy" is the manufacturer-affiliated one. This is the cleaner FDA-approved-product path. |
Not sure which GLP-1 provider or pharmacy is right for you?
Our 60-second path finder matches you to vetted providers that use licensed, transparent pharmacies — and tells you when to skip the telehealth model entirely.
Your real alternatives if you don't trust the new pharmacy
If the new pharmacy fails your checks, there are three paths. None of them are perfect; all of them are better than using a medication from a pharmacy you can't verify.
Option 1 — Manufacturer-direct (FDA-approved product, self-pay)
Both major GLP-1 manufacturers now have direct-to-patient pharmacy channels at known, fixed prices. These deliver FDA-approved finished products — not compounded versions — through licensed pharmacies affiliated with the manufacturer.
| Brand | Company | Channel | Form | Self-pay pricing | Offer notes |
|---|---|---|---|---|---|
| Wegovy injection | Novo Nordisk | NovoCare Pharmacy | Pen | $199/month for first 2 fills of 0.25 mg or 0.5 mg (new patient offer through June 30, 2026); then $349/month for 0.25 mg–2.4 mg; $399/month for Wegovy HD 7.2 mg | Eligibility and restrictions apply; verify at novocare.com |
| Wegovy oral | Novo Nordisk | NovoCare Pharmacy | Tablet | $149/month for 1.5 mg and 4 mg; 4 mg offer through August 31, 2026, then $199/month for 4 mg | Verify at novocare.com |
| Ozempic | Novo Nordisk | NovoCare Pharmacy | Pen | Self-pay tiers similar to Wegovy injection; offer terms vary | Verify at novocare.com |
| Zepbound | Eli Lilly | LillyDirect | Single-dose vials | Starts at $299/month for 2.5 mg; higher-dose pricing varies by dose and offer | Verify at lilly.com |
| Mounjaro | Eli Lilly | LillyDirect | KwikPen | Self-pay pricing varies by dose | Verify at lilly.com |
| Foundayo (orforglipron) | Eli Lilly | LillyDirect | Tablet | $149/month for 0.8 mg; $199/month for 2.5 mg; $299/month for 5.5 mg and 9 mg; $299/month for 14.5 mg and 17.2 mg if refilled within 45 days, otherwise $349/month | FDA-approved April 1, 2026. Verify at lilly.com |
Option 2 — Insurance and prior authorization
If you have commercial insurance, your prescriber can run a prior authorization for Wegovy or Zepbound. Coverage depends on your plan, diagnosis, the FDA-approved indication, prior-authorization criteria, and documentation. If your insurance denies the first time, your prescriber can appeal.
Option 3 — A different telehealth provider
If you want to stay in the telehealth model but switch to one whose pharmacy you've verified, the same 10-point checklist applies to whoever you move to. Don't pick a new telehealth on price alone. Check the pharmacy first. See our best GLP-1 telehealth provider comparisons for vetted options.
What to do if you already used the new pharmacy's product
Quick answer: If you already injected before reading this page, don't panic — most pharmacy changes go without incident. Document the vial, label, lot number, and any new symptoms. Watch for changes in side effects over the next 1 to 2 weeks.
What to track for the next 2 to 4 weeks
Don't escalate your own dose because the new product “feels weaker.” Talk to your prescriber instead. The FDA's dosing-error reports for compounded semaglutide came from exactly this kind of unsupervised adjustment. Use our free GLP-1 symptom diary to track changes systematically.
Call your prescriber or seek care if you have:
- \u2022Severe vomiting or signs of dehydration
- \u2022Severe abdominal pain
- \u2022Signs of an allergic reaction (rash, swelling of face or throat, trouble breathing)
- \u2022Symptoms of low blood sugar — sweating, shakiness, confusion — especially if you take other diabetes medications
- \u2022Any reason to suspect an accidental overdose, like drawing up a much larger volume than usual
How to report a problem
Quick answer: Report adverse events from any GLP-1 — compounded or FDA-approved — to the FDA's MedWatch program. Report unsafe online pharmacies through BeSafeRx resources. Report pharmacy license or compounding concerns to the state Board of Pharmacy. Report suspected fraud to MedWatch and to your state attorney general's consumer protection office. Save all documentation.
| What to report | Where to report it |
|---|---|
| Side effects or harm from a compounded or FDA-approved GLP-1 | FDA MedWatch (fda.gov/safety/medwatch). Your prescriber can file on your behalf. |
| An online pharmacy that fails BeSafeRx / NABP checks | Start with the FDA\u2019s BeSafeRx resources, then file with the NABP. |
| A pharmacy with license, sterility, or compounding concerns | The state Board of Pharmacy where the pharmacy is licensed, and your own state board if it ships from out of state. |
| Suspected fraud — a vial labeled with a pharmacy that doesn’t exist or denies making it | MedWatch, plus your state attorney general’s consumer protection office. The FDA has documented cases of fraudulent compounded GLP-1 labels. |
| Misleading telehealth marketing about compounded GLP-1s | The FDA. The agency issued 55+ Warning Letters in September 2025 and 30 Warning Letters on March 3, 2026. |
Frequently asked questions
What we actually verified for this page
We built this page by reviewing the FDA's current guidance on compounded GLP-1 drugs, the FDA's April 2026 clarification for 503A and 503B compounders, the FDA's Registered Outsourcing Facilities table, the FDA's Warning Letters to telehealth companies, the FDA's alert on dosing errors with compounded injectable semaglutide, the FDA's April 1, 2026 press announcement on Foundayo's approval, the FDA's BeSafeRx guidance, and the NABP's Safe.Pharmacy directory. We pulled current self-pay pricing from NovoCare and LillyDirect on May 12, 2026, and the named regulatory actions and lawsuits from primary-source legal write-ups (McDermott+Plus, Spencer Fane, Venable LLP, Foley & Lardner, Orrick) and major news coverage (NPR, CNBC, BioSpace, Pharmacy Times, Reuters).
We did not independently test any compounded product. We did not independently inspect any pharmacy. We did not verify your specific vial, your specific shipment, or your specific provider's relationship with any pharmacy. That verification is yours to do — this page gives you the framework to do it in 10 minutes. We update this page monthly.
Related guides
Compounded Semaglutide Safety Checklist
FDA-based red flags before your first fill.
Compounded Tirzepatide Safety Checklist
What to verify before your first tirzepatide vial.
503A vs 503B Pharmacies: What’s the Difference
Rules, inspection, and what each means for you.
GLP-1 Dose Units: mg vs mL vs Syringe Units
The math that prevents 5–20× overdoses.
Free GLP-1 Symptom Diary
28-day tracker for monitoring any product change.
Switching GLP-1 Providers
How to transfer your prescription safely.
Best GLP-1 Telehealth Providers
Vetted comparison with pharmacy transparency scores.
How to Report a GLP-1 Problem to FDA MedWatch
Step-by-step guide to filing a report.
GLP-1 Safety Overview
Everything you need to know about safe use.
GLP-1 Medication Guide
The full guide to how GLP-1s work.
Looking for a vetted GLP-1 provider with a transparent pharmacy?
Our path finder matches you to providers that meet our pharmacy transparency and compliance standards, and tells you when the right answer is the FDA-approved manufacturer-direct route.
Last verified: . We re-check FDA, BeSafeRx, NABP, manufacturer pricing, and named-provider status monthly.
Sources cited on this page:
- •FDA: "FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss" — fda.gov
- •FDA: Drug Safety Communication on dosing errors with compounded injectable semaglutide — fda.gov
- •FDA: April 1, 2026 reaffirmation of 503A enforcement discretion end for semaglutide injection — fda.gov
- •FDA: Registered Outsourcing Facilities table — fda.gov
- •FDA: BeSafeRx — fda.gov/drugs/buying-using-medicine-safely/besaferx
- •FDA: Warning Letters to telehealth companies (55+ on Sep 16, 2025; 30 on Mar 3, 2026) — fda.gov
- •FDA: Warning Letter to Boothwyn Pharmacy, LLC (issued Jan 16, 2026, published Feb 10) — fda.gov
- •FDA: Foundayo (orforglipron) approval, April 1, 2026 — fda.gov
- •NABP: Safe.Pharmacy directory — safe.pharmacy
- •NovoCare Pharmacy self-pay pricing — novocare.com (verified May 12, 2026)
- •LillyDirect self-pay pricing — lilly.com (verified May 12, 2026)
- •McDermott+Plus, Spencer Fane, Venable LLP, Foley & Lardner, Orrick: regulatory analysis of FDA GLP-1 enforcement, 2025–2026
- •NPR; CNBC; BioSpace; Pharmacy Times; Reuters: news coverage of manufacturer lawsuits and FDA actions, 2025–2026
Nothing on this page is medical advice. For decisions about your medication, contact your prescriber or pharmacist.