GLP-1 Injection Site Reaction: What's Normal, What's Not, and What to Do
By the Weight Loss Provider Guide editorial team — an independent comparison resource for GLP-1 telehealth providers.
· · Last verified: April 30, 2026
A GLP-1 injection site reaction can show up as a red, itchy, swollen, bruised, or lumpy spot where your shot went in — and most of the time, it's a normal local skin response, not a sign you need to stop your medication. In FDA clinical trials, injection site reactions occurred in about 1.4% of Wegovy patients and 0.2% of Ozempic patients at standard doses. Tirzepatide (Zepbound, Mounjaro) runs higher: 6–8% at therapeutic doses.
So if you just looked at your stomach, thigh, or arm and saw a small angry-looking patch, take a breath. It's almost certainly not the end of your treatment.
But "almost certainly" isn't "definitely." Spreading redness, increasing pain, drainage, fever, hives spreading across your body, or any swelling around your face or throat? That's a different conversation, and it needs a clinician now, not Google.
First-screen triage: is your GLP-1 injection site reaction normal?
| What you're seeing | What it likely is | What to do |
|---|---|---|
| Localized redness, itching, soreness, bruise, or firm bump where the needle went in | Common local injection site reaction | Cool compress, document it, rotate to a different spot next dose. |
| Reaction is getting bigger, hotter, or hasn't improved after a week | Worth a clinician's eye | Message your prescriber within 24–48 hours. Send a photo. |
| Pus or drainage, red streaks moving outward, fever or chills, severe and worsening pain | Possible infection (cellulitis) | Contact your prescriber today, or go to urgent care if you can't reach them. |
| Hives away from the injection site, swelling of face/lips/tongue/throat, trouble breathing, dizziness, or feeling faint | Possible serious allergic reaction | Call 911 or go to the ER now. This is rare, but it's an emergency. |
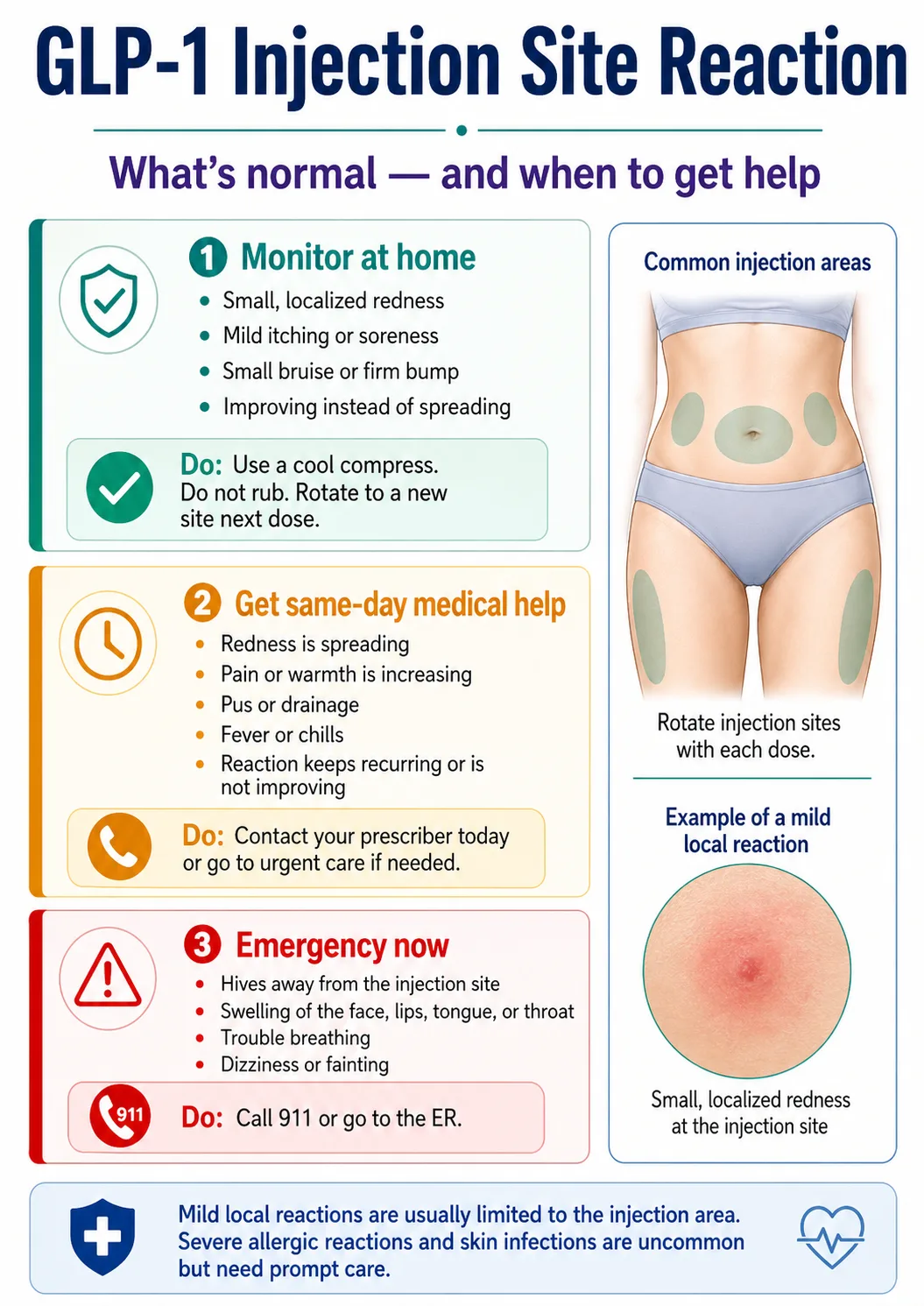
Is a GLP-1 injection site reaction usually serious?
Most GLP-1 injection site reactions are mild, localized, and improve on their own without stopping the medication. The exceptions — true allergic reactions, infections, and persistently worsening reactions — are uncommon but real, and they have specific signs that separate them from routine irritation.
The simple rule:
- Local and improving: monitor it, document it, keep going.
- Local but worsening or not getting better after a week: message your prescriber.
- Spreading, hot, painful, draining, or feverish: that's an urgent medical concern, not a wait-and-see.
- Hives, facial swelling, throat tightness, trouble breathing, dizziness: that's an emergency.
When should you get medical help for a GLP-1 injection site reaction?
Get emergency care for trouble breathing, swelling of the throat, tongue, lips, or face, faintness, or widespread hives. Contact a clinician urgently for spreading redness, increasing pain, pus, red streaks, or fever. Message your prescriber within 24–48 hours for a reaction that's lasting longer than expected, recurring with each dose, or growing instead of shrinking.
Tier 1: Emergency — call 911 or go to the ER
Per the American Academy of Dermatology, emergency care is needed for trouble breathing or swallowing and swelling around the eyes or lips. For GLP-1 medications specifically:
- Trouble breathing or wheezing
- Swelling of the lips, tongue, throat, or face
- Widespread hives or rash spreading away from the injection site
- Severe dizziness, faintness, or feeling like you might pass out
Serious hypersensitivity reactions, including anaphylaxis and angioedema, are documented in GLP-1 prescribing information for liraglutide, semaglutide, and tirzepatide. They're rare. They're real. Don't try to wait one out.
Tier 2: Urgent — contact your prescriber today, or go to urgent care
These are signs of a possible skin infection. MedlinePlus describes cellulitis as a bacterial skin infection with pain, tenderness, expanding redness, warmth, and possible fever:
- Redness expanding outward from the injection site
- Increasing warmth and pain over several days
- Pus, drainage, or weeping fluid
- Red streaks moving away from the site
- Fever above 100.4°F (38°C)
- Skin that looks tight, glossy, or rapidly worsening
Tier 3: Non-urgent — message your prescriber within 24–48 hours
- Reaction lasts longer than 7 days
- Same reaction returns every week with each dose
- Lump or hardness keeps growing
- Itching or rash worsens after each injection rather than getting better
- You changed dose, pharmacy, or product and the reaction started or got worse
- You use a compounded vial and aren't sure if dosing, sterility, or source could be a factor
What does a normal GLP-1 injection site reaction look like?
A normal reaction is localized to the injection area: redness, mild swelling, itching, a light bruise, or a small firm bump. It appears within hours, peaks at 24–48 hours, and starts improving within a few days. It doesn't spread far past the injection point, doesn't drain, doesn't cause fever, and doesn't hurt severely.
The actual phrases people use to describe it:
- A red itchy welt
- A small raised bump
- A hard knot under the skin
- A bruise or "needle mark"
- A burning or stinging spot
- A sore patch the size of a quarter
- A swollen pink circle
- A rash where the shot went in
The American Academy of Allergy, Asthma & Immunology notes that GLP-1 injection site reactions can include pain, redness, itching, and raised rubbery lumps, and that mild reactions usually improve on their own.
One important note about skin tone
Most medical content describes reactions as "redness," but redness reads differently on different skin. On medium and darker skin, a local reaction can look purple, brown, gray, or simply darker than the surrounding skin — or raised and shiny rather than visibly red.
Track warmth, swelling, tenderness, and whether it's spreading — not just color. The triage rules above don't change with skin tone.
How to photograph a reaction (so you can show your clinician)
- Same lighting each time. Natural daylight near a window is ideal. Don't use flash — it washes out color changes.
- Same distance. About 8–12 inches away. Close enough to see texture, far enough to show the surrounding skin.
- Include a scale. A coin or a finger next to the area gives your clinician size context.
- Same angle and same time of day. A morning photo and an evening photo of the same spot can look like two different things.
- Don't draw on the area with a marker unless your clinician asks you to.
How long should a GLP-1 injection site reaction last?
Most mild reactions appear within hours of injection, peak around 24–48 hours, and start improving within a few days, often resolving within a week. Reactions that worsen after day 3, persist past day 7, or progress to drainage, spreading redness, or fever are not normal and should be evaluated.
| Time since injection | What's typical | What to do |
|---|---|---|
| First few hours | Mild stinging, small bruise, slight redness or puffiness | Document. Don't rub. |
| 24–48 hours | Itching, redness, small welt may peak here | Cold compress if it bothers you. Avoid scratching. |
| Days 3–4 | A normal reaction is stable or improving | If it's worse, message your prescriber. |
| Days 5–7 | Most reactions are gone or close to it | If it's still there or growing, contact your prescriber. |
| Beyond 7 days | A small firm bump may still be palpable but should not be painful or growing | If painful, growing, or recurring, get it checked. |
Duration depends on the medication, your skin, the dose, the site, and your individual immune response. Weekly GLP-1s like Wegovy and Zepbound are absorbed gradually — Zepbound reaches peak concentration at a median of about 24 hours, with a range of 8–72 hours. Your skin's local response can mirror that timeline.
What causes a lump, welt, or itch after a GLP-1 injection?
A local reaction is usually caused by needle trauma, mild local immune response, or a technique issue. A smaller subset are linked to anti-drug antibodies, which can amplify local responses for some patients over time.
The common, fixable causes:
- Reusing the same exact spot week after week
- Injecting into bruised, red, tender, hard, or scarred skin
- Alcohol swab not fully dry before injection (causes stinging and irritation)
- Rubbing the site afterward
- Tight clothing or waistband friction over a fresh injection site
The less-discussed cause: anti-drug antibodies. The FDA labels are explicit about this:
- The Zepbound label reports injection site reactions in 11.3% of patients with anti-tirzepatide antibodies versus 1% without — a more-than-tenfold difference.
- The Mounjaro label reports 4.6% of patients with anti-tirzepatide antibodies vs 0.7% without.
- The Wegovy label reports allergic reactions in 16% of patients with anti-semaglutide antibodies vs 7% without (allergic reactions broadly, not just ISRs).
How common are injection site reactions with each GLP-1 medication? (FDA-label data)
Injection site reaction rates vary widely by medication, dose, and trial design — so they shouldn't be read as a clean head-to-head safety ranking. The useful takeaway: these reactions are recognized in every approved GLP-1 label, but the frequency, dominant symptoms, and dose-response patterns differ meaningfully.
We assembled this comparison directly from current DailyMed prescribing information. Last verified April 30, 2026.
| Medication | Active ingredient | Dosing | ISR rate (drug) | Placebo rate | Notable subgroup data | Source |
|---|---|---|---|---|---|---|
| Wegovy® | Semaglutide 2.4 mg | Weekly | 1.4% | 1.0% | 16% allergic reactions in patients with anti-semaglutide antibodies vs 7% without | DailyMed |
| Ozempic® | Semaglutide 0.5–2 mg | Weekly | 0.2% | — | Lower rate than weight-loss-dose semaglutide | DailyMed |
| Zepbound® | Tirzepatide 2.5–15 mg | Weekly | 6% (5 mg), 8% (10 mg), 8% (15 mg) | 2% | 11.3% with anti-tirzepatide antibodies vs 1% without | DailyMed |
| Mounjaro® | Tirzepatide 2.5–15 mg | Weekly | 3.2% | 0.4% | 4.6% with anti-tirzepatide antibodies vs 0.7% without | DailyMed |
| Saxenda® | Liraglutide 3.0 mg | Daily | 13.9% | 10.5% | Anti-liraglutide antibodies in 2.8%; may be associated with higher ISR rates | DailyMed |
| Victoza® | Liraglutide 1.2–1.8 mg | Daily | ~2% | — | Fewer than 0.2% discontinued due to ISR | DailyMed |
| Trulicity® | Dulaglutide 0.75–4.5 mg | Weekly | 0.5% | 0% | Lower than tirzepatide and exenatide | DailyMed |
| Bydureon BCise® | Exenatide ER 2 mg | Weekly | 23.9% (incl. nodules); 10.5% nodules; 3.2% pruritus; 2.3% erythema | — | 0.5% discontinued due to injection-site nodules | DailyMed |
Daily-dosed medications have higher overall rates because patients receive seven times more injections per week. Saxenda's 13.9% looks scary next to Wegovy's 1.4%, but the Saxenda placebo arm also reported 10.5% — most of those were normal injection-related events.
Tirzepatide runs higher than semaglutide at therapeutic doses. Zepbound's rate climbs from 6% at 5 mg to 8% at 10–15 mg, suggesting a dose-response relationship the FDA label calls out directly.
Bydureon BCise is in a different category. Its 23.9% rate is dramatically higher than modern weight-loss GLP-1s. If you're on Wegovy or Zepbound, Bydureon data isn't your data.
Compounded GLP-1s aren't on this table. Compounded preparations are not FDA-approved and have no published RCT data on injection site reaction rates. See the compounded section below.
How do you treat a mild GLP-1 injection site reaction at home?
For a mild local reaction, the AAAAI specifically supports cold compresses and rotating injection sites. Most reactions improve within a few days without anything else. For itching or inflammation that's bothering you, an over-the-counter antihistamine or 1% hydrocortisone cream can help — but check with your prescriber or pharmacist first if you take other medications.
- Apply a cool compress for 10–15 minutes, two to three times during the first 24–48 hours. Wrap ice in a thin cloth. Don't put ice directly on skin.
- Take an oral antihistamine if itching is bothering you. Cetirizine (Zyrtec), loratadine (Claritin), and diphenhydramine (Benadryl) are common options — check with your pharmacist if you take other medications.
- Consider 1% hydrocortisone cream, applied as a thin layer once or twice daily. Don't use on broken or bleeding skin.
- Don't scratch, rub, or "pop" anything. This is the single most common way a mild reaction turns into an infection.
- Take a photo each day. If you end up needing to message your prescriber, the photo timeline is the most useful thing you can send.
- Keep your medication on schedule unless you're seeing red-flag symptoms or your prescriber has told you otherwise.
What not to do:
- Don't reuse the exact same spot for your next injection
- Don't inject into skin that's still red, tender, bruised, or hardened from the last shot
- Don't rub the site after injecting
- Don't take an extra dose to "make up for" a reaction
- Don't ignore spreading redness, drainage, or fever
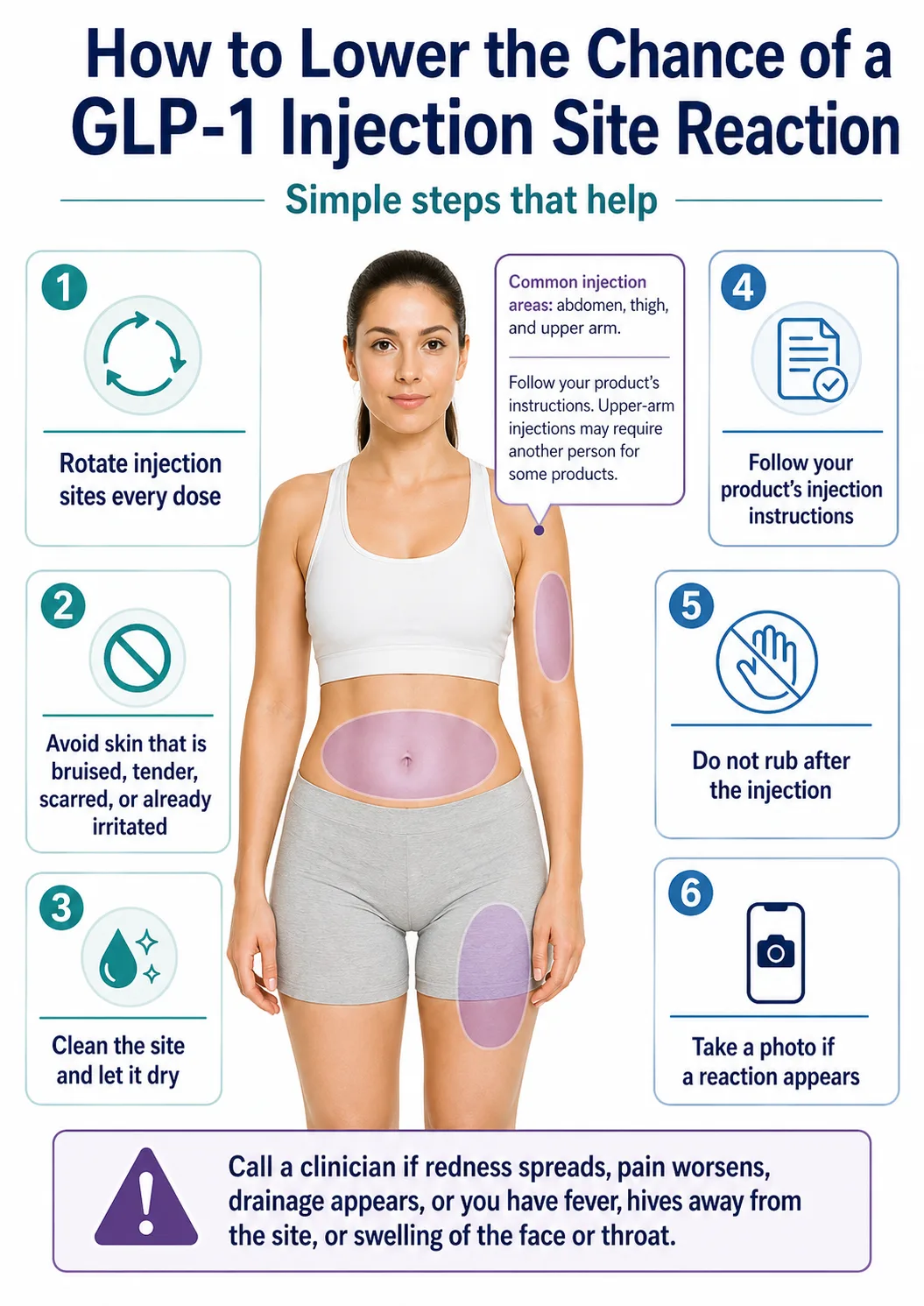
How do you prevent reactions on your next injection?
The biggest prevention levers are rotating injection sites with every dose, following your specific product's Instructions for Use, avoiding tender or bruised skin, cleaning with alcohol and letting it fully dry, and not rubbing afterward.
- Pick a fresh site. Stay at least an inch from your last spot. If you injected your right thigh last week, go left thigh, abdomen, or arm this week.
- Avoid bad real estate. Skip skin that's still tender, bruised, red, hard, or scarred.
- Follow your product's Instructions for Use. The Wegovy IFU allows warming to room temperature; the Mounjaro IFU explicitly says it is not necessary. Read your specific product's IFU rather than following a generic rule.
- Wash your hands. This sounds obvious. Most people skip it.
- Clean the site with alcohol. Then let it fully dry. Injecting through wet alcohol stings.
- Use the technique your prescriber or pharmacy trained you on.
- Don't rub afterward. Light pressure with a cotton ball is fine.
- Log the site. A simple grid you tick off each week prevents you from accidentally reusing your favorite spot.
Site rotation differs by product — here's what the labels say
| Product | Self-injection sites | Notes |
|---|---|---|
| Wegovy® | Abdomen, thigh, or upper arm | Rotate each injection. Avoid 2 inches around the navel. |
| Ozempic® | Abdomen, thigh, or upper arm | Rotate. Don't share pens. |
| Zepbound® | Stomach or thigh for self-injection | Another person can inject in the upper arm. Rotate weekly. |
| Mounjaro® | Stomach or thigh for self-injection | Another person can inject in the upper arm. Rotate weekly. |
| Trulicity® | Stomach, thigh, or upper arm | Rotate weekly. |
| Saxenda® | Abdomen, thigh, or upper arm | Daily — rotate aggressively. |
Rethinking whether your current GLP-1 is the right fit?
Take our 60-second matching quiz. We'll send you a personalized GLP-1 path based on your goals, what you've tried, your insurance situation, and what's actually available in your state. Free, no commitment, no spam.
See your match in 60 seconds →Is it a local reaction, an allergy, or an infection?
A local injection site reaction stays near the shot site and improves within days. An infection (most commonly cellulitis) spreads outward, gets warmer and more painful over several days, and can come with drainage or fever. A true allergic reaction can include hives spreading away from the injection site, swelling of the face or throat, or trouble breathing — and is a medical emergency. The patterns are usually distinct.
| Sign | Normal local reaction | Possible allergic reaction | Possible infection (cellulitis) |
|---|---|---|---|
| Redness | Localized | May include hives spreading across body | Spreading outward from injection site |
| Onset | Hours after injection, peaks 24–48 hr | Minutes to hours after injection | Develops over 1–4 days, gets worse |
| Itching | Mild, localized | Often intense, often widespread | Less common; pain dominates |
| Pain | Mild tenderness | Variable | Increasing, throbbing |
| Swelling | Mild, localized | May involve face, lips, tongue, throat | Spreading firmness |
| Drainage | None | None | Pus or fluid possible |
| Fever | No | Possible | Yes — over 100.4°F |
| Breathing | Normal | Difficulty breathing, throat tightness | Normal |
| What to do | Self-care, rotate, watch | Emergency care — call 911 | Call prescriber today, possibly urgent care |
The two patterns to memorize: Stays small and gets better → local reaction. Spreads, worsens, or pulls in your whole body → not a local reaction, get help.
Should you take your next dose if you had a reaction?
For a mild, local, improving reaction, many people continue after rotating sites — the FDA labels don't require discontinuation for mild local reactions. Contact your prescriber before your next dose if the reaction was severe, spreading, recurring across multiple sites, worsening rather than improving, or paired with allergy or infection symptoms.
What to send your prescriber if you're not sure:
Hi — I had a reaction after my GLP-1 injection and want to confirm whether to monitor it or change anything.
Medication and dose: ____
Injection date and time: ____
Injection site (abdomen / thigh / upper arm, left or right): ____
Reaction started: ____
Symptoms: redness / itching / swelling / lump / bruise / pain / warmth / drainage / other ____
Size (inches or cm): ____
Is it spreading? ____
Fever or chills? ____
Hives or swelling away from the injection site? ____
Photo attached: yes / no
Have I had this reaction before? ____
Did anything change recently — new dose, new vial, new pharmacy, new site? ____
That message takes 90 seconds to write and gives your clinician everything they need. Most telehealth GLP-1 programs handle these messages asynchronously and respond within a business day.
Why did I start getting injection site reactions after months on a GLP-1?
A reaction that appears after months of smooth treatment usually has one of a few causes: a recent dose increase, repeatedly using the same exact spot, a new vial or pharmacy, a new product lot, or — for tirzepatide and liraglutide specifically — the development of anti-drug antibodies.
The most likely triggers, in order:
- You titrated up. Tirzepatide ISR rates are dose-dependent — Zepbound's rate climbs from 6% at 5 mg to 8% at 10–15 mg. A reaction that shows up the week you go up a dose is a known pattern, not a coincidence.
- You've been hitting the same spot. Even with conscious rotation, most people have a "default" zone they use 70% of the time. Cumulative tissue irritation builds slowly.
- A new vial, pen, or pharmacy. If you switched recently, that change is the variable. Document the lot number and date.
- Anti-drug antibodies. If you've been on tirzepatide or liraglutide for several months and reactions are getting worse rather than settling, antibody development is a documented possibility worth raising with your prescriber.
Does the injection site matter — stomach, thigh, or upper arm?
Yes — but mostly because rotating between sites is what prevents reactions, not because one site is universally "better." Wegovy and Ozempic let you self-inject in the abdomen, thigh, or upper arm. Zepbound and Mounjaro restrict self-injection to the stomach or thigh.
- Can I use the same body area? Often yes — same area is fine, same exact spot is not. Move at least an inch from the previous shot.
- Is the thigh better than the stomach? Not universally. Pick what's comfortable and where you can use correct technique.
- Can I inject near a stretch mark or scar? Avoid scarred or damaged tissue — absorption is unpredictable.
- Should I rub it afterward? No. Light pressure with cotton or gauze if needed.
What if this happened with compounded semaglutide or tirzepatide from a vial?
A local reaction can happen with any subcutaneous injection, including compounded GLP-1 products. But compounded preparations come with extra questions that brand-name pens don't: the source pharmacy, sterility, dose accuracy, syringe measurement, label clarity, and whether the product is actually the active ingredient you think it is.
As of April 30, 2026, the FDA is clarifying its compounded GLP-1 policy now that brand-name shortages have largely resolved, and Reuters reported that the FDA proposed excluding the active ingredients in Novo Nordisk's and Eli Lilly's GLP-1 weight-loss drugs from the 503B bulks list — meaning the window for legally compounding these specific medications is narrowing.
The FDA's stated position is that compounded GLP-1 products are not reviewed for safety, effectiveness, or quality before being marketed — and the FDA has received reports of adverse events tied to dosing errors with compounded injectable semaglutide.
The compounded GLP-1 vial safety checklist
| Check | Why it matters |
|---|---|
| Was it prescribed by a licensed clinician after a real medical evaluation? | This separates legitimate compounding from gray-market "research peptide" sales. |
| Did it come from a state-licensed pharmacy with a verifiable address? | The FDA has warned consumers to buy only from state-licensed pharmacies and to check for spelling errors or odd addresses on labels. |
| Are the dose instructions written clearly in both medication amount (mg) and syringe units? | Compounded injectable semaglutide dosing errors have been reported to FDA — including patients drawing up 5x or 10x their intended dose. |
| Does the vial look clean — clear, no particles, no discoloration, intact seal? | Cloudy, leaking, or discolored product is a stop-everything signal. Contact your pharmacy. |
| Does the label reference "semaglutide sodium" or "semaglutide acetate"? | The FDA has stated that semaglutide salt forms are different active ingredients than what's in approved drugs, and that the agency is not aware of a lawful basis for their use in compounding. |
| Did the reaction start with a new vial, new pharmacy, new dose, or new lot? | That's a pattern your clinician needs to know. |
| Is the lot number documented and visible? | If you ever need to report a product issue, you'll need this. |
Serious adverse events with compounded products can be reported to the FDA's MedWatch program.
We feature several compounded GLP-1 providers on this site. But the regulatory environment in 2026 is materially different from 2024. If you're getting a reaction and you're on a compounded product, the question "is this me, or is this the product?" is a fair one to ask your prescriber, your pharmacy, or both. See our guide to FDA-approved GLP-1 medications and best GLP-1 telehealth providers for your full range of options.
Worried your vial, pharmacy, or sourcing might be the bigger issue?
Take our 60-second matching quiz. We'll show you which FDA-approved brands, oral GLP-1s, and clinician-supported telehealth programs fit your situation, your state, and your budget. You can also keep your current setup if it's working — the goal is information, not pressure.
Get your personalized GLP-1 path →Can switching GLP-1s — or switching to a pill — help if reactions keep coming back?
Persistent reactions are a clinical conversation worth having before you change anything. For some patients, switching from one GLP-1 to another, reducing the dose temporarily, or moving to an oral GLP-1 may help. The right answer depends on your medical history, what's available in your state, your insurance situation, and the reason for the reactions.
When a switch is worth asking about:
- You've had reactions every single week for over a month
- Reactions are getting larger, more uncomfortable, or more inflamed over time
- You've tried better technique and rotation and it hasn't helped
- You're afraid of needles and the anxiety is making your technique worse
- You're on a compounded product and the reactions started with a new vial or pharmacy
- You've previously had allergic reactions to other injectable medications
Oral GLP-1 options have gotten more relevant in 2026. Oral GLP-1 options now include Wegovy as an oral tablet (dosing starts at 1.5 mg daily) and Foundayo (orforglipron), FDA-approved April 1, 2026, as the first non-peptide oral GLP-1. They're not a magic fix — both have their own side effects, mostly gastrointestinal — but they remove the injection site reaction question entirely.
One legitimate option for FDA-approved brand-name GLP-1s with insurance support
If your reactions have you considering FDA-approved brand-name medication with insurance support, Ro is one option worth knowing about. Ro Body offers FDA-approved Zepbound® and Foundayo™, with insurance support and a free GLP-1 Insurance Coverage Checker. Not the right fit if you're on Medicare, Medicaid, or specifically looking for compounded products.
| Element | Detail |
|---|---|
| Membership pricing | $39 first month, then $149/month, or as low as $74/month with annual prepay |
| Medication cost | Separate from membership. Foundayo cash-pay starts around $149 first month, then $199–$299/month. Zepbound KwikPen cash-pay starts around $299 first month, then $399–$449/month. |
| Insurance support | Insurance Coverage Checker and concierge support included |
| Government insurance | Ro currently does not coordinate GLP-1 medication coverage for Medicare, Medicaid, or other government insurance plans |
| FDA-approved only | Ro carries FDA-approved brand-name GLP-1s only. For compounded options, see our GLP-1 provider comparison. |
Ro Body
$39 first month
Then $149/mo or $74/mo with annual prepay. Medication cost separate.
Zepbound® + Foundayo™ available · Insurance Coverage Checker included · Not available for Medicare/Medicaid
See if Ro is right for youComparing injection vs. oral, branded vs. compounded, telehealth vs. in-person?
Take our 60-second matching quiz to see your options side by side. We use your medication route, insurance status, state availability, budget, and brand-name preference to match you to a fit-based path.
Compare your options in 60 seconds →What we actually verified for this guide
Verified April 30, 2026.
Show primary sources
- DailyMed — Wegovy® prescribing information
- DailyMed — Ozempic® prescribing information
- DailyMed — Zepbound® prescribing information
- DailyMed — Mounjaro® prescribing information
- DailyMed — Saxenda® prescribing information
- DailyMed — Victoza® prescribing information
- DailyMed — Trulicity® prescribing information
- DailyMed — Bydureon BCise® prescribing information
- FDA postmarket safety information on unapproved GLP-1 drugs
- FDA April 2026 statement on compounded GLP-1 policy
- FDA MedWatch reporting program
- AAAAI — GLP-1 receptor agonist allergy explainer
- AAD — rash red-flag guidance
- MedlinePlus — cellulitis information
Frequently asked questions about GLP-1 injection site reactions
Is a GLP-1 injection site reaction an allergic reaction?
Usually no. A typical reaction is a local inflammatory response, not a systemic allergy. True allergic reactions involve symptoms beyond the injection site — hives spreading across your body, swelling of the face, lips, tongue, or throat, or trouble breathing — and are medical emergencies. Local reactions are not.
Why is my Zepbound injection site itchy days later?
Tirzepatide is absorbed gradually after subcutaneous injection — Zepbound reaches peak concentration at a median of about 24 hours, with a range of 8 to 72 hours. Itching can persist for several days while the medication is absorbing locally. Use cool compresses if it's bothersome. If it lasts beyond 7 days, gets larger, or worsens, message your prescriber. The Zepbound label reports injection site reactions in 6–8% of patients depending on dose.
Can I be allergic to Wegovy or Ozempic (semaglutide)?
Yes, rarely. Postmarketing reports include cases of anaphylaxis and angioedema with semaglutide. The Wegovy label notes that allergic reactions occurred in 16% of patients with anti-semaglutide antibodies versus 7% without. If you have hives spreading away from the injection site, facial swelling, or trouble breathing, stop the medication and seek emergency care.
What does an infected GLP-1 injection site look like?
Cellulitis — the most common skin infection complication — typically presents with spreading redness, increasing warmth, increasing pain, sometimes drainage or pus, and often a fever above 100.4°F. It develops over 1 to 4 days, not within hours. If your symptoms match this pattern, contact your prescriber today or go to urgent care.
Should I use ice or heat on a GLP-1 injection site reaction?
A cold compress for the first 24–48 hours is the AAAAI-supported comfort step for mild local reactions. Do not apply ice directly to skin — wrap it in a thin cloth.
Should I rub my GLP-1 injection site?
No. The Ozempic, Mounjaro, and Zepbound Instructions for Use support light pressure if needed, but not rubbing.
Can I take Benadryl before my GLP-1 injection to prevent reactions?
Some patients do this with their prescriber's guidance. Talk to your prescriber before adopting it as a routine — for some patients it's reasonable, for others it's unnecessary or interacts with other medications.
Why did I start getting reactions after months on a GLP-1?
The most common triggers are a recent dose increase, repeatedly using the same exact spot, a new vial or pharmacy, or — for tirzepatide and liraglutide — the development of anti-drug antibodies. Document the change and message your prescriber.
Are injection site reactions more common with compounded GLP-1s than brand-name products?
There's no published clinical-trial comparison, because compounded preparations aren't FDA-approved and don't generate the same trial-based safety reporting. Excipient profiles can differ from brand-name products. The more useful question is whether your compounded product came from a verifiable, state-licensed pharmacy with clear dose instructions and good documentation. The FDA has warned about dosing errors and adverse events tied to compounded injectable semaglutide products.
Did I lose the dose if a bubble or lump formed at the injection site?
Probably not, but don't guess. If you're worried the medication didn't go in correctly, document what happened (photo, what you saw, where it leaked if it leaked) and ask your prescriber or pharmacist. Don't take an extra dose to "make up for it" without specific instructions.
How do I know if I'm developing antibodies to my GLP-1?
There's no routine outpatient test. The clinical signal is persistent reactions across multiple injection sites over multiple weeks, often getting worse rather than better. If that pattern fits you, raise it with your prescriber — they can decide whether testing or a medication switch is appropriate.
Should I stop my GLP-1 because of a reaction?
For a mild, local, improving reaction — generally no. The FDA labels list injection site reactions as an adverse reaction but don't require discontinuation for mild local cases. For severe, persistent, recurring, or systemic reactions, that's a clinical decision your prescriber needs to make, not the internet.
The bottom line
A GLP-1 injection site reaction is, almost always, a small local skin response that improves on its own and doesn't require stopping your medication. Cool compress, rotate sites, take a photo, keep going. The exceptions are real and worth knowing — spreading redness, severe pain, drainage, fever, hives spreading across your body, swelling of the face or throat — and they have specific signs that don't look like a normal welt.
Across Wegovy, Ozempic, Zepbound, Mounjaro, and Saxenda, discontinuation specifically because of injection site reactions was uncommon in the trials where it was reported: Victoza reports fewer than 0.2% discontinuing for ISRs; Bydureon BCise reports 0.5%. The medication is doing its job. Your skin is doing what skin does when something gets injected into it.
Still working out which GLP-1 program is the right fit for your situation?
Take our free 60-second matching quiz. We'll send you a personalized plan based on your goals, your insurance, what's available in your state, and your tolerance for injections versus oral options. Free, no commitment, no spam.
Take the 60-second quiz →