GLP-1 Dizziness: Why It Happens, When to Worry, and What to Do Tonight
By the Weight Loss Provider Guide Research Team ·
Verified against current FDA prescribing information on DailyMed, FDA compounding safety communications, MedlinePlus, Johns Hopkins emergency dizziness guidance, and CDC hypoglycemia guidance.
The bottom line, in one screen
GLP-1 dizziness is real, it’s documented on the FDA labels, and the right action depends on the pattern. It shows up at 8% on the Wegovy® (semaglutide) 2.4 mg label versus 4% on placebo, and at 4–5% across the Zepbound® (tirzepatide) doses versus 2% on placebo. Most non-emergency cases trace back to dehydration from reduced eating or GI side effects, low blood sugar (especially with insulin or a sulfonylurea), a drop in blood pressure on standing, vascular effects, reduced intake during rapid weight loss, or — in a small fraction of users — a vestibular disorder signal documented in a 2025 cohort study.
⚠️ Call 911 right now if dizziness comes with chest pain, fainting, swelling of your face or throat, slurred speech, weakness on one side of your body, severe headache, or trouble breathing. Everything else in this guide assumes you don’t have those symptoms.
Want a personalized version with a printable plan and a copy-paste message for your prescriber?
→ Match my GLP-1 dizziness pattern (free, ~60 seconds)No email gate. No provider sales pitch in the output.
What we actually verified for this page:
- Current FDA prescribing information (via DailyMed) for Wegovy®, Zepbound®, Ozempic®, Mounjaro®, and Rybelsus®
- The FDA’s July 26, 2024 alert on compounded semaglutide dosing errors and the FDA’s 2026 page on concerns with unapproved GLP-1 drugs
- FDA adverse event report counts as of July 31, 2025: 605 reports for compounded semaglutide and 545 for compounded tirzepatide
- A 2025 retrospective cohort study of 496,756 GLP-1 users on vestibular outcomes (PMC12109458)
- A 2019 network meta-analysis comparing dizziness across nine antidiabetic drug classes (PMC6844583)
- MedlinePlus, Johns Hopkins, and CDC guidance on dizziness and hypoglycemia
Quick triage: which level of help do you actually need?
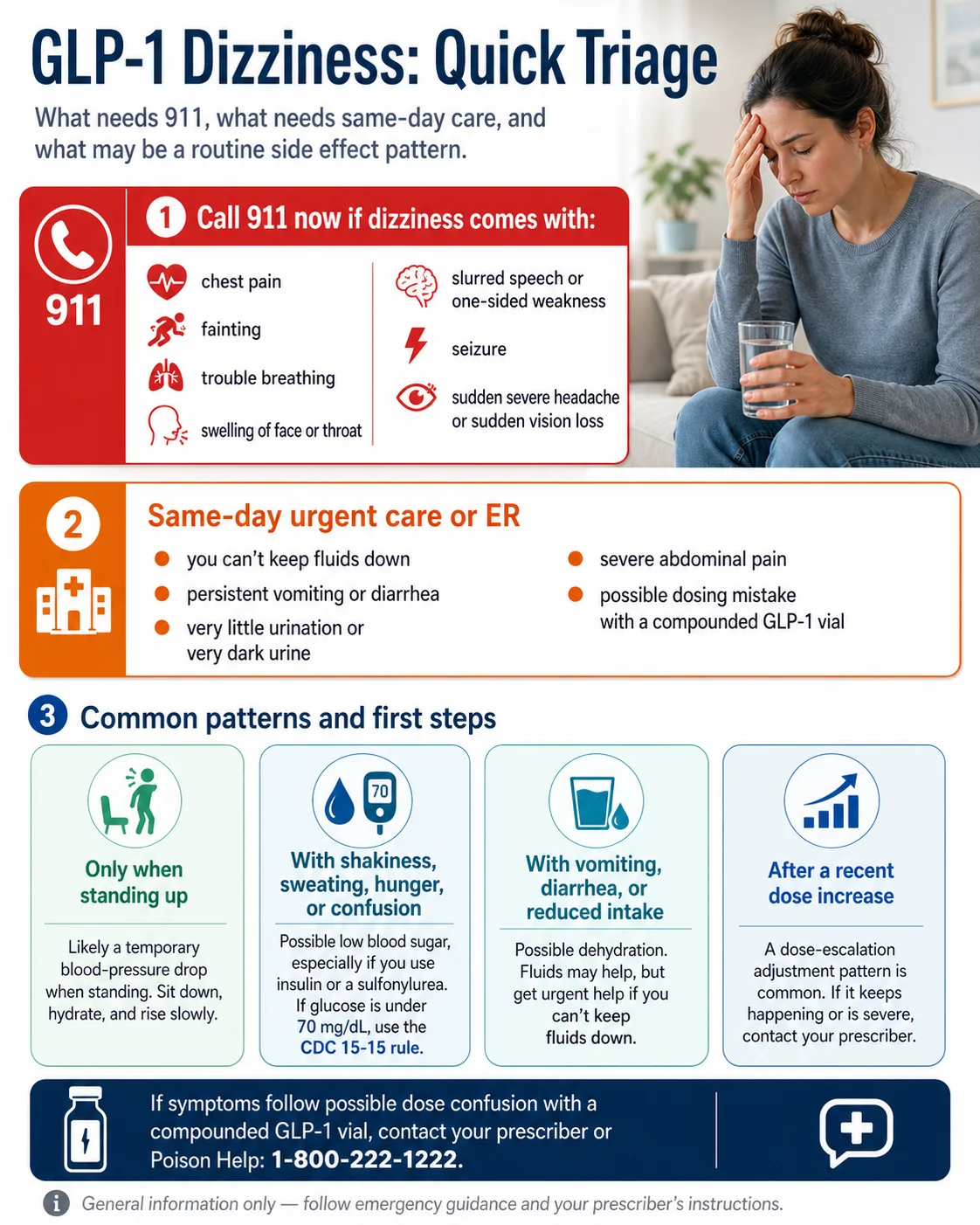
| What you’re feeling | What it might be | What to do right now |
|---|---|---|
| Dizziness plus chest pain, fainting, slurred speech, one-sided weakness, severe headache, swelling of face/throat, trouble breathing, or a seizure | Possible cardiac event, stroke, or severe allergic reaction — not “just” the medication | Call 911. Don’t drive yourself. |
| Dizziness plus persistent vomiting or diarrhea, can’t keep fluids down, very little urination, dark urine, or severe abdominal pain radiating to your back | Severe dehydration, possible kidney injury, or possible pancreatitis | Urgent care or ER today. Message your prescriber on the way. |
| Dizziness plus shaking, sweating, confusion, hunger, or a racing heart — and you take insulin or a sulfonylurea | Possible hypoglycemia (low blood sugar) | Check glucose if you can. If under 70 mg/dL, use the CDC’s 15-15 rule. Severe symptoms → call 911. |
| Dizziness only when standing up that resolves once you sit back down | Likely orthostatic hypotension (temporary blood-pressure drop on standing) | Sit down. Don’t drive. Read the management section below before changing anything. |
| Mild lightheadedness in the first weeks of treatment, or shortly after a dose increase | Possible adjustment-period side effect | Use the action plan below. Message your prescriber if not improving in 7 days. |
| Dizziness right after a compounded GLP-1 vial dose — especially if the syringe units or concentration confused you | Possible dosing error (FDA has documented errors of 5× to 20× the intended dose with compounded vials) | Contact your prescriber immediately. If symptoms are severe, call Poison Help at 1-800-222-1222 or go to the ER. |
Source basis: FDA prescribing information (DailyMed); CDC 15-15 rule; Johns Hopkins emergency dizziness guidance; FDA compounded semaglutide dosing-error alert (July 26, 2024).
Can GLP-1 medications cause dizziness?
Short answer: yes, dizziness is a recognized side effect on the FDA labels for the major GLP-1 medications. It’s most common in the first weeks of treatment and shortly after dose increases, and most non-emergency cases respond to fluids, consistent eating, and time. But “recognized side effect” doesn’t mean “ignore it” — the right response depends entirely on the pattern.
Dizziness wasn’t supposed to be the headline side effect of these drugs — that reputation belongs to nausea (44% of adults on Wegovy reported it versus 16% on placebo per the FDA prescribing information). But dizziness shows up often enough that the FDA listed it as one of the most common adverse reactions on multiple GLP-1 labels. The Wegovy label uses a ≥5% threshold for “common.” Zepbound defines common as ≥2%, and dizziness clears that bar at every dose tested.
The catch: “dizziness” can mean four different things. Lightheaded (like you might pass out). Vertigo (the room is spinning). Unsteady (your balance feels off). Foggy (disconnected from your body). Each one points to a different cause and a different fix. The next sections separate them out — because telling them apart is most of the work.
When GLP-1 dizziness is an emergency
If dizziness is paired with any of the symptoms below, the cause likely isn’t your GLP-1 — or it’s an extreme reaction that needs immediate attention. Don’t wait. Don’t research more. Don’t drive yourself.
📞 Call 911 immediately if dizziness comes with:
- ✕Chest pain, pressure, or pain radiating to your jaw, neck, or arm — possible cardiac event
- ✕Sudden severe headache or stiff neck — could be a stroke or hemorrhage
- ✕Slurred speech, facial drooping, or weakness on one side of your body — stroke symptoms
- ✕Sudden vision loss or double vision
- ✕Loss of consciousness or fainting
- ✕Seizure
- ✕Trouble breathing or shortness of breath at rest
- ✕Swelling of your face, lips, tongue, or throat, or widespread hives — possible anaphylaxis
- ✕Inability to stand or walk
- ✕New confusion or inability to speak normally
Johns Hopkins specifically warns that dizziness paired with neurologic symptoms can signal a stroke and requires emergency evaluation, regardless of what medications you take.
🏥 Same-day urgent care or ER (don’t sleep on these):
- Repeated vomiting that won’t stop — risk of severe dehydration and electrolyte loss
- Diarrhea you can’t get under control after 24 hours
- You can’t keep fluids down
- You haven’t urinated in 8+ hours, or your urine is dark brown
- Severe weakness or fatigue you’ve never had before
- Severe abdominal pain, especially radiating to your back — possible pancreatitis (FDA Medication Guides instruct patients to stop the medication and seek medical help right away)
- A possible compounded GLP-1 dosing error
🩸 Hypoglycemia red flags (especially if you take insulin or a sulfonylurea):
The Wegovy label reports clinically significant hypoglycemia (blood glucose under 54 mg/dL) in 6.2% of patients with type 2 diabetes versus 2.5% on placebo. If you have diabetes and take insulin or a sulfonylurea (drugs like glipizide, glimepiride, or glyburide), the dizziness might be your blood sugar dropping rather than the GLP-1 itself.
The CDC’s 15-15 rule: if your glucometer reads under 70 mg/dL, eat or drink 15 grams of fast-acting carbohydrates (glucose tablets, 4 oz juice, regular soda — not diet — or hard candies). Recheck in 15 minutes. Repeat if still under 70. Severe symptoms — confusion, seizure, loss of consciousness — are a 911 call.
The 6 real causes of GLP-1 dizziness
Most non-emergency GLP-1 dizziness traces back to one of six patterns, and they overlap. Dehydration is one of the first to rule out because the FDA labels warn that GI symptoms can cause fluid loss. Hypoglycemia is the most dangerous if you also take insulin or a sulfonylurea. Orthostatic hypotension (the standing-up pattern) is one of the easiest to recognize.
Cause #1: Dehydration and reduced fluid/food intake
GLP-1 receptor agonists slow how fast food leaves your stomach. But the same mechanism, plus the nausea, vomiting, or diarrhea that hits in the first weeks, can leave you mildly dehydrated. The Wegovy and Zepbound labels both flag that nausea, vomiting, and diarrhea can cause fluid loss and increase the risk of kidney problems.
How to recognize the pattern: dark urine, fewer bathroom trips, headache, dizziness specifically when you stand up, fatigue. Red flags requiring urgent medical evaluation: inability to keep fluids down, very little urination over 8+ hours, dark urine, or severe weakness.
The fix isn’t always just water. Plain water alone can fall short if you’ve been losing sodium through vomiting or diarrhea. Pair fluids with sodium and potassium — broth, electrolyte powder, oral rehydration salts. Caveat: if you have kidney disease, heart failure, fluid restriction, uncontrolled high blood pressure, or you take a diuretic, follow your clinician’s specific fluid instructions.
Cause #2: Low blood sugar (hypoglycemia)
GLP-1 medications lower blood glucose. But “lower” can become “too low,” especially if you also take insulin or a sulfonylurea. The Wegovy label specifically lists dizziness, lightheadedness, blurred vision, sweating, shakiness, hunger, fast heartbeat, and confusion as signs of low blood sugar.
The combination is what creates the risk. If you have diabetes and your dizziness comes with shakiness and sweating, the question isn’t “is the GLP-1 broken?” — it’s “are my other glucose-lowering medications now too strong because the GLP-1 is doing its job?” That’s a prescriber conversation.
Cause #3: Orthostatic hypotension (low blood pressure when standing)
Orthostatic hypotension is defined as a drop of 20 mmHg in your top blood pressure number (systolic) or 10 mmHg in the bottom number (diastolic) within three minutes of standing. The Wegovy label reports more hypotension and orthostatic hypotension events on Wegovy than on placebo.
How to recognize it: dizziness happens within seconds of standing up. It goes away within seconds of sitting back down. Usually worst first thing in the morning.
The fix: the 30-second standing rule. Sit up. Pause 30 seconds with your feet on the floor. Then stand. This single behavioral change resolves more orthostatic dizziness than any other tactic. If you take a blood pressure medication or diuretic, your antihypertensive may now be too strong because the GLP-1 has lowered your baseline — a prescriber conversation, not a stop-the-medication situation.
Cause #4: Blood-pressure and vascular effects
The Wegovy and Zepbound labels both flag hypotension-related signals. Dizziness when standing can overlap with lower blood pressure, dehydration, or interactions with antihypertensive medications. This often blurs with cause #3 — and the management is the same: hydration where appropriate, slow position changes, and a prescriber check on your blood pressure medications.
Cause #5: Reduced intake during rapid weight loss
If you’re losing weight quickly, the appetite suppression can push some people below their actual nutritional needs. Significantly under-eating, under-hydrating, or losing weight much faster than expected can make you feel weak, light, and dizzy — not from the medication directly, but from what’s happening to your intake.
How to recognize it: dizziness emerges later (weeks to months in), you’ve lost noticeable weight, you feel fine sitting but light when you stand or exercise, and you’ve been eating less than you probably should. The fix: make sure you’re eating enough total calories, getting adequate protein, and not skipping electrolytes.
Cause #6: Vestibular disorder signal in observational data
A 2025 retrospective cohort study using the TriNetX research network looked at 419,497 semaglutide users and 77,259 tirzepatide users between 2018 and 2024. The researchers compared them to matched controls and tracked who developed new vestibular disorders (like vertigo or balance problems) over up to three years.
| Medication | Hazard ratio at 6 months | Hazard ratio at 3 years | Cumulative incidence (3 yr) |
|---|---|---|---|
| Semaglutide | 4.02 (3.33–4.86) | 4.95 (4.51–5.43) | 0.41% |
| Tirzepatide | 3.19 (2.11–4.81) | 4.55 (3.43–6.03) | 0.19% |
Honest framing: the absolute risk is low. The signal is real. This is observational data — association, not proven causation. But if your dizziness is room-spinning vertigo (not lightheadedness) and it’s persistent, this is worth bringing to your prescriber so they can rule out a vestibular cause rather than assuming it’s a routine side effect. (PMC12109458)
How to figure out what’s actually causing your dizziness
The fastest way: pay attention to when the dizziness happens. The pattern points to the cause more reliably than the severity does. Walk through the questions below in order. This is not a medical diagnosis — it’s a starting point for your prescriber conversation.
→ "When I stand up, and it goes away after a few seconds."
Likely cause: Most likely: orthostatic hypotension (cause #3) or vascular effects (cause #4)
First step: Use the 30-second standing rule. If you take a blood pressure medication, message your prescriber.
Escalate if: You fall, you faint, or it's still happening after 4 weeks.
→ "When I haven't eaten in a while, or between meals."
Likely cause: Most likely: low blood sugar (cause #2)
First step: If you have a glucose meter, check during an episode. If under 70 mg/dL, treat with the 15-15 rule. If you take insulin or a sulfonylurea, contact your prescriber now.
Escalate if: You're shaky, sweaty, or confused.
→ "Constant or random — not really tied to standing or eating."
Likely cause: Most likely: dehydration (cause #1) or under-eating during rapid weight loss (cause #5)
First step: Track your fluid and food intake honestly for 3–5 days. Look for under-eating patterns or fluid loss.
Escalate if: Still happening at 2 weeks despite proper intake.
→ "Right after I started, or right after my dose went up."
Likely cause: Most likely: dose escalation period — your body is adjusting
First step: Hydrate, eat consistently, ride out 1–2 weeks. Many people adjust.
Escalate if: Not improving after 2 weeks at the new dose. Ask about pausing dose escalation.
→ "It's a spinning sensation — like the room is moving."
Likely cause: Most likely: vestibular involvement (cause #6) or something unrelated to the GLP-1
First step: Talk to your prescriber this week, not "next visit." This is different from lightheadedness and warrants its own evaluation.
Escalate if: Hearing changes, severe vertigo with vomiting, or any neurologic symptoms.
→ "Right after a compounded vial dose."
Likely cause: Most likely: possible dosing error
First step: Stop. Re-check the vial label, the syringe, the units, and the dose your prescriber gave you. Contact your prescriber or Poison Help (1-800-222-1222).
Escalate if: Any doubt about whether you measured correctly → contact Poison Help immediately.
Want a personalized version you can save? Run your specific symptoms through our pattern matcher — it returns the most likely cause, a printable plan tailored to your answers, and a copy-paste message for your prescriber.
→ Match my dizziness pattern (free, ~60 seconds)GLP-1 dizziness by drug: Wegovy vs Zepbound vs Ozempic vs Mounjaro
Per the FDA labels on DailyMed: Wegovy lists dizziness as a common adverse reaction (8% of adults vs 4% of placebo at 2.4 mg). The Zepbound label reports dizziness at 4%, 5%, and 4% across the 5 mg, 10 mg, and 15 mg doses (vs 2% on placebo). Ozempic and Mounjaro do not list dizziness among their most common (≥5%) adverse reactions. The directional pattern: dizziness reports are higher with the weight-management semaglutide doses.

| Medication | Active ingredient | FDA-label dizziness rate | Listed as “common” on its own label? | Hypotension on the label? |
|---|---|---|---|---|
| Wegovy® injection 2.4 mg | semaglutide | 8% (vs 4% placebo) | ✅ Yes (≥5% threshold) | Yes — orthostatic hypotension events more common than placebo |
| Wegovy® injection 7.2 mg | semaglutide | 6% (vs 5% placebo) | ✅ Yes | Same class effect |
| Wegovy® tablet 25 mg | semaglutide | Common adverse reactions similar to injection label; no separate tablet-specific dizziness % shown | ✅ Yes (per label statement) | Same class effect |
| Zepbound® | tirzepatide | 5 mg: 4% / 10 mg: 5% / 15 mg: 4% (vs 2% placebo) | ✅ Yes (≥2% threshold) | Yes — "associated with GI events, dehydration, and antihypertensive therapy" |
| Ozempic® | semaglutide (lower max dose) | Reported above 0.4%; not listed among most common (≥5%) | ❌ No | Listed under hypoglycemia symptoms |
| Mounjaro® | tirzepatide (T2D dose) | Not listed among common (≥5%) adverse reactions; mentioned as low-blood-sugar symptom | ❌ No | Not listed in the common-reactions table |
| Rybelsus® | oral semaglutide | Not common; listed as low-blood-sugar symptom | ❌ No | Listed under hypoglycemia symptoms |
| Foundayo™ (orforglipron) | orforglipron (oral) | Specific dizziness rate requires current label verification; FDA-approved April 1, 2026 | — | — |
Sources: FDA prescribing information via DailyMed (Wegovy, Zepbound, Ozempic, Mounjaro, Rybelsus). Last verified April 28, 2026. Important caveat: these rates come from different clinical trials with different designs, doses, populations, and indications. The FDA explicitly notes that adverse-reaction rates from one drug’s clinical trials cannot be directly compared to another drug’s. Use this as label context, not proof one drug will be safer for any specific person.
The directional signal
A 2025 retrospective cohort study of nearly 500,000 patients also found semaglutide users had roughly 1.5 to 2 times higher relative risk of new-onset vestibular disorders compared to tirzepatide users. The absolute risk in both groups was small (under 0.5% over 3 years), but the directional signal matches the label pattern.
If you’re on Wegovy (or compounded semaglutide) and your dizziness has been persistent for 6+ weeks despite hydration, eating consistency, and a stable dose — that’s a legitimate moment to ask your prescriber whether tirzepatide (Zepbound) might be a better fit. The label pattern and the cohort data both support the question. It’s not a fringe ask. But it’s not automatic either — switching is a clinical decision, not an algorithmic one. See also: Zepbound vs Wegovy.
The hidden risk most pages don’t talk about: compounded GLP-1 dosing errors
On July 26, 2024, the FDA issued an alert about dosing errors with compounded injectable semaglutide. The reports involved patients taking 5 to 20 times the intended dose — some of which required hospitalization. As of July 31, 2025, the FDA had received 605 adverse event reports for compounded semaglutide and 545 for compounded tirzepatide. If your dizziness happened right after a compounded vial dose and you have any doubt about whether you measured correctly, treat that as a different kind of safety situation than a standard side effect.
Compounded medications are drugs that licensed pharmacies prepare for individual patients. Compounding can be lawful when it meets the conditions of the FD&C Act, but compounded GLP-1 products are not FDA-approved, and the FDA does not review them for safety, effectiveness, or quality. The regulatory landscape changed meaningfully in 2025–2026: as the brand-name supply stabilized, the FDA clarified that compounders face restrictions on making “essentially copies” of commercially available FDA-approved drugs.
What the FDA actually documented
- ⚠Patients have administered 5× to 20× their intended dose of compounded semaglutide due to confusion measuring from vials
- ⚠Hospitalizations have occurred as a result
- ⚠Adverse effects reported included severe nausea, vomiting, fainting, headache, dehydration, acute pancreatitis, and gallstones
- ⚠Cases involved both patient errors and healthcare provider miscalculations when converting between milligrams, milliliters, and “units”
- ⚠In one case, a patient who couldn’t reach their telemedicine provider did an internet search and ended up taking five times the intended dose
- ⚠Fraudulent compounded products have also been identified — some labeled with pharmacy names that didn’t actually compound the medicine
Why compounded vials are confusing
With most prefilled, pre-dialed pens (Wegovy 2.4 mg, Zepbound pen, Ozempic, Mounjaro), you twist the dial to your prescribed dose, push the button, done. The math is the manufacturer’s problem. Compounded vials require you to do the math yourself:
- A vial labeled with a concentration (like “5 mg/mL” or “10 mg/mL”)
- A syringe marked in “units” or in milliliters (or both)
- Instructions that may say “take 0.25 mL” or “draw to 25 units” or “inject 1.7 mg”
Three different units. Different concentrations between pharmacies. Different syringes. This is the setup the FDA flagged.
What to do if you might have taken too much
- 1Call Poison Help: 1-800-222-1222. Free, available 24/7, they handle medication overdoses regularly. They will tell you whether to go to the ER or monitor at home.
- 2Call your prescriber’s after-hours line. Tell them the concentration on the vial, your prescribed dose in mg, and how many units or mL you actually drew. Photograph the vial label and syringe before you call.
- 3If you can’t reach either and symptoms are severe — go to the ER. Bring the vial.
- 4Do not take your next scheduled dose until you’ve spoken with your prescriber.
How to prevent this on future doses
- Get your prescriber to write the dose in two different units (e.g., “Draw 0.25 mL = 25 units on a U-100 insulin syringe = 1.25 mg”)
- Confirm your syringe matches what your prescriber expects (U-100 insulin syringes are most common)
- Take a photo of the vial, the syringe drawn up, and the prescriber’s instructions, every time, until it’s automatic
- If your provider is hard to reach for a dosing question — that’s a support gap that matters
Already had a serious side effect or a quality problem with a compounded GLP-1? Report it to the FDA’s MedWatch program — it’s how the FDA tracks these patterns and protects future patients. → Report to FDA MedWatch
How long does GLP-1 dizziness last?
The FDA labels don’t give a universal dizziness-resolution timeline. What the Zepbound label does say: nausea, vomiting, and diarrhea were “most frequently reported during dose escalation” and decreased with continued use. Dizziness often tracks with that GI side-effect curve. Below is the typical pattern most patients describe — not a medical guarantee.
First 1–2 weeks of treatment
The most common onset window. Often paired with nausea and reduced food intake.
Weeks 3–4
Body begins adapting; many people see improvement.
Each dose escalation step
Can re-trigger everything for 1–2 weeks. Each step up roughly resets the adaptation window.
3+ months at a stable dose
Persistent dizziness becomes uncommon. If you're still struggling, it's worth investigating with your prescriber rather than waiting it out.
The point most readers miss: each dose increase is essentially a new starting point. If you’re moving from 0.25 mg to 0.5 mg to 1.0 mg to 1.7 mg to 2.4 mg of semaglutide over 4–5 months, you’re going through this adjustment four times, not once. If your prescriber knows you’re struggling, they can hold a dose for an extra month or even reduce. The standard titration schedule is a guideline, not a sentence.
What to do right now if you’re dizzy on a GLP-1
The safest first move: sit down. If your symptoms aren’t on the emergency list, work through the action steps below and don’t drive until you’re steady. Save this checklist to your notes app.
Right now (next 5 minutes)
- ☐Sit down or lie down. Don't try to "push through it." Falling and hitting your head turns a manageable problem into an ER visit.
- ☐If you have diabetes — check your blood glucose if you can. If under 70 mg/dL, treat with the CDC's 15-15 rule unless your clinician gave you different instructions.
- ☐Sip fluids. If you have vomiting, diarrhea, low intake, or standing-related lightheadedness, fluids may help. Follow your clinician's instructions if you have kidney disease, heart failure, fluid restriction, uncontrolled high blood pressure, or take a diuretic.
- ☐A small snack may help if you haven't eaten and your dizziness fits a hunger pattern. Don't treat every dizziness episode as low blood sugar without checking.
- ☐Don't drive. Don't shower in hot water. Don't operate machinery.
Next 24 hours
- ☐Eat 4–6 small meals if your appetite allows it. Don't go more than 4–5 hours without something
- ☐Use the 30-second standing rule: feet on floor, pause 30 seconds, then stand
- ☐Avoid alcohol entirely
- ☐Avoid intense heat exposure (hot showers, saunas)
- ☐Avoid intense exercise — light walking is fine
- ☐Log every episode in a notes app: time, what you were doing, last meal, last fluid, how long the episode lasted, how severe (1–10)
When to escalate
- If symptoms are getting worse, recurring, or paired with red flags → use the triage table at the top of this page
- If they’re not improving and a week has passed → message your prescriber with your symptom log
- Don’t skip your next scheduled GLP-1 dose without prescriber direction
What to tell your prescriber about GLP-1 dizziness
Vague messages get vague responses. Specific messages get specific actions. Send your prescriber the medication name, dose, dose-change history, exact symptom pattern, your fluid and food intake, vomiting/diarrhea details, glucose or blood pressure readings (if you have them), and a complete list of your other medications.
Copy-paste prescriber message
Hi [provider name], I'm [N] weeks into [Wegovy / Zepbound / compounded semaglutide / etc.] at the [X mg] dose. I started/increased to this dose on [date]. Over the past [N] days I've had [N] episodes of dizziness. Pattern: - Type: [lightheaded / spinning / unsteady / faint feeling] - When: [when I stand up / between meals / random / right after my dose / morning only] - Duration of each episode: [seconds / minutes / hours] - Severity (1-10): [N] Context: - Fluid intake: [estimated oz/day] - Eating: [N meals/day, what kind] - Vomiting/diarrhea: [yes/no, frequency] - Last urination: [normal / reduced / very dark] - Other meds: [insulin / sulfonylurea / BP medications / diuretics / other] - Recent fingerstick glucose: [if available] - Recent home BP readings: [if available] I'm NOT having: [chest pain / fainting / slurred speech / one-sided weakness / severe abdominal pain] — those would have been a 911 call. My questions: 1. Should I hold or delay my next dose? 2. Do any of my other medications need adjusting? 3. Could we slow my titration if this doesn't improve? 4. Should I be evaluated in person? Thanks.
If you’re on a compounded GLP-1, add this
Compounded medication details: - Pharmacy: [name] - Concentration on vial: [X mg/mL] - Prescribed dose: [X mg] - Volume I drew up: [X mL or X units on a [U-100] syringe] - I've attached photos of the vial and the syringe before injection. - I have NO concerns about a possible overdose / I have concerns about a possible overdose [pick one and explain].
Should you skip, lower, or stop your GLP-1 dose?
Don’t self-adjust without your prescriber. Skipping doses, restarting after gaps, and stopping abruptly all have clinical consequences — and most non-emergency dizziness has a fixable cause that doesn’t require stopping. The exceptions are emergency-level symptoms (anaphylaxis, pancreatitis-like pain, suspected overdose), where the FDA Medication Guides instruct patients to stop the medication and seek medical help right away.
Why “just skip it” is rarely the right move:
- •Restarting a GLP-1 after a gap can require dose adjustment. Going more than 2 weeks without a dose may mean restarting at a lower dose to avoid worsened side effects. That’s a clinician decision, not a guess.
- •The dizziness usually has a cause that’s not the medication itself. Dehydration, low blood sugar, low blood pressure, polypharmacy — those are fixable without stopping.
- •The “other” medications are often the real lever. If you’re on a blood pressure med or a sulfonylurea, those doses likely need to come down now that the GLP-1 is doing work.
- •Stopping without a plan often leads to weight regain. In published semaglutide withdrawal data, participants regained roughly two-thirds of prior weight loss within one year after stopping treatment.
When the FDA labels point to immediate action:
- Suspected serious allergic reaction (face/throat swelling, hives, breathing problems): stop the medication and get medical help right away
- Severe abdominal pain that may radiate to your back, with or without vomiting (possible pancreatitis): stop the medication and get medical help right away
- Severe or ongoing vomiting/diarrhea, inability to keep fluids down, very little urination, or signs of severe dehydration: drink fluids if you can and contact a healthcare provider — escalate to urgent care or ER if severe
- Suspected compounded overdose: don’t take the next dose, call your prescriber and Poison Help at 1-800-222-1222
Outside of those, the rule is simple: message before you skip.
Should you switch GLP-1s or providers if dizziness won’t stop?
For most people, dizziness improves with the basics — fluids, eating consistency, slow position changes, and time. Switching the medication or the provider isn’t necessary. But if your dizziness has persisted past 8–12 weeks on a stable dose with proper management, three legitimate options exist: dose hold or reduction with the same drug, switching from semaglutide to tirzepatide, or moving to a provider with insurance support that can unlock brand-name FDA-approved options at copay pricing.
A candid admission from us: we have affiliate relationships with several GLP-1 telehealth providers. On a normal commercial page, we’d compare them and recommend one. On this page, we’re not going to. The topic is dizziness. The FDA has specifically warned about compounded dosing errors and has logged 605 adverse event reports for compounded semaglutide and 545 for compounded tirzepatide as of July 31, 2025. Pushing compounded providers in a guide explicitly about a symptom that compounded dosing errors can cause would be tone-deaf. So instead, here’s the actual decision framework.
Path 1: Stay where you are. Adjust dose or drug with your current provider.
If your current prescriber is responsive and willing to slow titration, hold a dose, or switch you from semaglutide to tirzepatide — stay. Switching providers doesn’t fix medication side effects. It just changes who’s writing the prescription.
Path 2: Switch from semaglutide to tirzepatide.
If your dizziness has been persistent on Wegovy, Ozempic, Rybelsus, or compounded semaglutide despite proper management, the FDA-label data and the 2025 cohort study both point toward tirzepatide (Zepbound or Mounjaro) showing lower dizziness signals. This is a clinical conversation with your prescriber, not a self-serve switch — but it’s a legitimate one to bring up. See: Zepbound vs Wegovy comparison.
Path 3: Switch providers if access is the bottleneck.
If your current provider can’t offer the medication you want, can’t help with insurance prior authorization, won’t pause your titration when you ask, or won’t return messages within 24 hours during a side effect — that’s a support problem, and you have other options.
Not sure which path is right for you? Take our free 60-second matching quiz — we’ll factor in your insurance situation, your formulation preference, your tolerance history, and your state availability, and route you to the providers most likely to fit your specific situation. No follow-up calls. No spam.
→ Find my GLP-1 pathDoes your GLP-1 program give you enough support?
A good GLP-1 program should give you clear urgent-symptom guidance, a same-day path to a clinician, dose-change support, screening for medication interactions, transparent pharmacy sourcing, and a side-effect follow-up process. If yours fails on more than two items, it’s worth looking elsewhere.
| Support standard | Why it matters for dizziness |
|---|---|
| Clear emergency-symptom guidance built into onboarding | You know when to call 911, urgent care, or message |
| Same-day clinician messaging path | Dizziness rarely waits for next week's appointment |
| Dose-change support without an extra fee | Many dizziness cases need a slower titration |
| Hypoglycemia screening for diabetes patients | Insulin/sulfonylurea interactions are the most dangerous setup |
| Blood pressure medication review | Standing dizziness often means your BP med is now too strong |
| Transparent pharmacy sourcing | You can verify what you got and from where |
| Clear compounded dosing instructions in writing | Vial/syringe confusion is a documented FDA safety risk |
| Side-effect follow-up beyond the first month | Dose escalation re-triggers side effects months later |
Affiliate disclosure: Weight Loss Provider Guide has an affiliate relationship with Ro and may earn a commission if you sign up after using our links. We mention Ro on this page because the use case below specifically fits what Ro offers, not because of the affiliate relationship. We’d write the same recommendation if there were no commission. This is the only provider link on this page.
When the right next move is to switch — and to where
If you specifically want to switch from semaglutide to FDA-approved Zepbound® (tirzepatide) with insurance support:
| What Ro states | What we verified | Last checked |
|---|---|---|
| FDA-approved formulary includes Zepbound® pen, Wegovy® pen, Wegovy® pill, Foundayo™ (orforglipron), and Ozempic® | Confirmed on ro.co/weight-loss/ pricing page | April 28, 2026 |
| Dedicated insurance concierge handling prior-authorization paperwork | Confirmed on ro.co | April 28, 2026 |
| Free GLP-1 Insurance Coverage Checker (no membership required to use) | Confirmed accessible without signup | April 28, 2026 |
| Ro Body membership: $39 first month, then $149/month — or as low as $74/month with annual prepay | Confirmed on ro.co/weight-loss/pricing/ | April 28, 2026 |
| Foundayo on Ro: starts at $149/month, varies by dose | Confirmed on Ro's Foundayo pricing page | April 28, 2026 |
| Medication billed separately at LillyDirect / NovoCare / TrumpRx-matched cash prices when not covered by insurance | Confirmed in Ro's pricing FAQ | April 28, 2026 |
Ro does NOT lead on compounded GLP-1s. If your priority is the lowest possible cash price on a compounded formulation, Ro is not your provider — a quiz match through us will route you to a better fit. But because Ro skips the compounded race-to-the-bottom, they can offer the strongest insurance navigation in the category and the cleanest path to FDA-approved brand-name medications.
→ Check your insurance coverage on Ro (free, no membership required to use)What the research says about GLP-1 dizziness
Peer-reviewed research on GLP-1 dizziness has accelerated since 2023. A 2019 network meta-analysis ranked GLP-1 receptor agonists as having the highest probability of causing dizziness and headache among nine antidiabetic drug classes studied. A 2025 retrospective cohort study of nearly 500,000 patients documented an elevated hazard ratio for new-onset vestibular disorders in both semaglutide and tirzepatide users — with a higher signal for semaglutide.
The 2019 network meta-analysis (PMC6844583)
Ranked nine antidiabetic drug classes by their probability of causing dizziness and headache. GLP-1 receptor agonists ranked highest for both — with a 22.5% probability of being the worst class for dizziness and 23.4% for headache. DPP-4 inhibitors had a lower dizziness risk than GLP-1 RAs (OR 0.76, 95% CI 0.67–0.87).
The 2025 retrospective cohort study (PMC12109458)
Used the TriNetX research network to follow 419,497 semaglutide users and 77,259 tirzepatide users over up to three years. Both medications showed elevated hazard ratios for new-onset vestibular disorders versus matched controls. Semaglutide had a higher relative risk than tirzepatide. Absolute risk was low (under 0.5% over 3 years), but the directional signal was clear.
The honest caveats: observational studies show association, not causation. There’s potential for surveillance bias (GLP-1 users see clinicians more often, so they may get diagnosed more often). Mechanisms aren’t fully settled. Both studies recommend further investigation rather than alarm.
What this tells us: GLP-1 dizziness isn’t anecdotal. It’s labeled by the FDA, replicated in observational research, and documented across multiple drug-class comparisons. It’s also, for almost everyone, manageable.
GLP-1 Dizziness FAQ
Does GLP-1 cause dizziness?
Yes. Dizziness is listed as a common adverse reaction on the Wegovy® FDA label at 8% in adults versus 4% on placebo at the 2.4 mg dose. The Zepbound® label reports dizziness at 4–5% across doses (versus 2% on placebo) and labels it common at the Zepbound label's own ≥2% threshold. Ozempic and Mounjaro do not list dizziness among their most common (≥5%) adverse reactions, though both labels mention it as a possible symptom of low blood sugar.
Why does GLP-1 make me dizzy?
The most common reasons trace to fluid loss (because GI side effects can reduce intake or cause vomiting and diarrhea), low blood sugar (especially with insulin or sulfonylureas), and a temporary drop in blood pressure when standing. Less common causes include vascular effects, reduced intake during rapid weight loss, and a vestibular disorder signal seen in observational data.
How long does GLP-1 dizziness last?
The FDA labels don't give a universal timeline. The pattern most patients describe: dizziness peaks in the first weeks of treatment and shortly after dose increases, then improves as the body adjusts. The Zepbound label notes that GI side effects were most reported during dose escalation and decreased over time. Dizziness still present at 6–8 weeks on a stable dose is worth a prescriber conversation.
Is dizziness a sign of low blood sugar on a GLP-1?
It can be — especially if you take insulin or a sulfonylurea, or if you've been eating much less than usual. Hypoglycemia symptoms include shakiness, sweating, hunger, confusion, fast heartbeat, and dizziness. If you have a glucometer, check during an episode. If your blood sugar is below 70 mg/dL, follow the CDC's 15-15 rule: eat 15 grams of fast carbs, recheck in 15 minutes.
Can I drive if I'm dizzy on a GLP-1?
Not during an active episode. If dizziness is recurrent — even if it feels manageable — avoid driving until you've been episode-free for at least 24–48 hours. Persistent or unpredictable dizziness on any medication is a reason to talk to your prescriber before driving routinely.
Should I stop my GLP-1 if I feel dizzy?
Don't stop without medical advice. Most non-emergency dizziness has a fixable cause that doesn't require stopping the medication. Message your prescriber with a detailed symptom log, and ask whether to hold the next dose, slow your titration, or adjust your other medications. The FDA Medication Guides do say to stop and get medical help right away for serious allergic-reaction symptoms or pancreatitis-like severe abdominal pain.
Is dizziness worse on Wegovy than on Ozempic?
Wegovy's label lists dizziness at 8% (2.4 mg dose) and 6% (7.2 mg dose) as a common adverse reaction. Ozempic's label reports dizziness above 0.4% but doesn't list it among the most common (≥5%) reactions. The labels report different rates, but they come from different trials with different doses and indications, so the FDA explicitly cautions that adverse-reaction rates from one drug's trials cannot be directly compared to another's.
Is Zepbound less likely to cause dizziness than Wegovy?
The Wegovy 2.4 mg label reports dizziness at 8%; the Zepbound label reports dizziness at 4–5% across doses. A 2025 real-world cohort study of nearly 500,000 patients also found tirzepatide had a lower hazard ratio for vestibular disorders than semaglutide. The directional signal is consistent, but these are not head-to-head trials. Individual response varies.
Can compounded semaglutide or tirzepatide cause dizziness?
Compounded GLP-1 products are not FDA-approved, and the FDA has received adverse event reports involving compounded semaglutide and tirzepatide products — 605 and 545 reports respectively as of July 31, 2025. The FDA also notes that compounded adverse events are likely underreported. Many reported reactions appear consistent with reactions for FDA-approved versions, but the FDA cannot verify the safety, effectiveness, or quality of non-FDA-approved compounded GLP-1 drugs.
How do I report a serious side effect or compounded product problem to the FDA?
Use the FDA's MedWatch Adverse Event Reporting Program. The FDA specifically encourages patients, healthcare professionals, and compounders to report adverse events or quality problems with compounded GLP-1 products through MedWatch, because reporting drives the FDA's ability to track patterns and protect future patients.
Will electrolytes help with GLP-1 dizziness?
Often, yes — especially if dizziness is related to fluid loss or low blood pressure on standing. If you've been losing fluids through vomiting or diarrhea, electrolytes may help more than plain water. Important caveat: if you have kidney disease, heart failure, fluid restriction, uncontrolled high blood pressure, or you take a diuretic, follow your clinician's specific instructions instead of generic electrolyte advice.
What if my dizziness only happens when I stand up?
That pattern strongly suggests orthostatic hypotension — a temporary drop in blood pressure when you go from sitting or lying down to standing. The most effective behavioral fix is the 30-second standing rule: sit up, pause 30 seconds with your feet on the floor, then stand. If you also take a blood pressure medication or a diuretic, your prescriber may need to adjust those doses.
What if my dizziness is room-spinning (vertigo) instead of lightheaded?
True spinning vertigo points more toward a vestibular cause than toward the typical GLP-1 dizziness pattern of fluid loss or low blood pressure. The 2025 cohort study documented elevated hazard ratios for new-onset vestibular disorders in GLP-1 users, but the absolute risk is low. New, severe, or persistent vertigo — especially with hearing changes, severe headache, or any neurologic symptoms — warrants prompt evaluation rather than home management.
Bottom line: your next step
GLP-1 dizziness is real and documented on FDA labels. It shows up at 8% on the Wegovy 2.4 mg label and 4–5% on the Zepbound label across doses. It peaks in the first weeks of treatment and after dose increases. Most non-emergency cases trace back to fluid loss, low blood sugar, or a drop in blood pressure on standing.
The first move for most people: hydration where appropriate, eating consistency, the 30-second standing rule, and time. The next move for the rest: a prescriber conversation about dose, drug, or — in a small number of cases — switching to a different molecule or a provider with better access.
Red flags first. Then the basics. Then your prescriber. In that order.
Still not sure which GLP-1 program is right for you? Take our free 60-second matching quiz. We’ll factor in your medication preference (FDA-approved vs. compounded, injection vs. oral), your insurance situation, your tolerance history, your budget, and your state availability — and route you to the providers that fit your specific situation. No follow-up calls. No spam.
→ Take the GLP-1 Path QuizSources and verification
FDA prescribing information (verified via DailyMed): Wegovy® (semaglutide) — including 2.4 mg, 7.2 mg, and 25 mg tablet labeling; Zepbound® (tirzepatide); Ozempic® (semaglutide); Mounjaro® (tirzepatide); Rybelsus® (oral semaglutide)
FDA July 26, 2024 alert on dosing errors with compounded injectable semaglutide products
FDA’s “Concerns with Unapproved GLP-1 Drugs Used for Weight Loss” page — current as of February 2026, including 605 adverse event reports for compounded semaglutide and 545 for compounded tirzepatide as of July 31, 2025
FDA’s “Clarifies Policies for Compounders as the National GLP-1 Supply Begins to Stabilize” statement
Peer-reviewed research: PMC12109458 (Risk of Vestibular Disorders with Semaglutide and Tirzepatide, 2025 retrospective cohort, n = 496,756); PMC6844583 (Neurological Manifestation of Incretin-Based Therapies, 2019 network meta-analysis)
Clinical guidance: MedlinePlus (Dizziness and Vertigo); Johns Hopkins Department of Neurology and Neurosurgery (emergency dizziness evaluation); CDC (Treatment of Low Blood Sugar / Hypoglycemia, 15-15 rule)
Provider information: Ro pricing and formulary verified at ro.co/weight-loss/pricing/ on April 28, 2026.
Last verified: . This page is general health information, not medical advice. It does not establish a clinician-patient relationship. Always consult your prescriber before changing your dose or your medication.
If you found a factual error or have a verified update we should incorporate, contact us — we maintain a documented update log for our commercial and health-decision pages.