GLP-1 and Oral Contraceptives: Which Birth Control Pills Need Backup?
Last verified against FDA prescribing information on DailyMed: · By the WPG Research Team · ~15-min read · Published

The short version
If you are here because you just read the warning on your prescription leaflet — take a breath. The answer is more specific than the internet makes it sound. Below we walk through every commonly prescribed GLP-1 drug, exactly what its current FDA label says about birth control pills, and exactly what to do today. Every rule on this page was pulled from the live prescribing information on DailyMed, the National Library of Medicine database that mirrors current FDA labeling.
We also handle the things most other guides skip: what happens when you vomit on a GLP-1, what to do if you missed pills, the mini-pill difference, the "Ozempic babies" rumor versus what is actually in the label, and the planning-a-pregnancy washout timing.
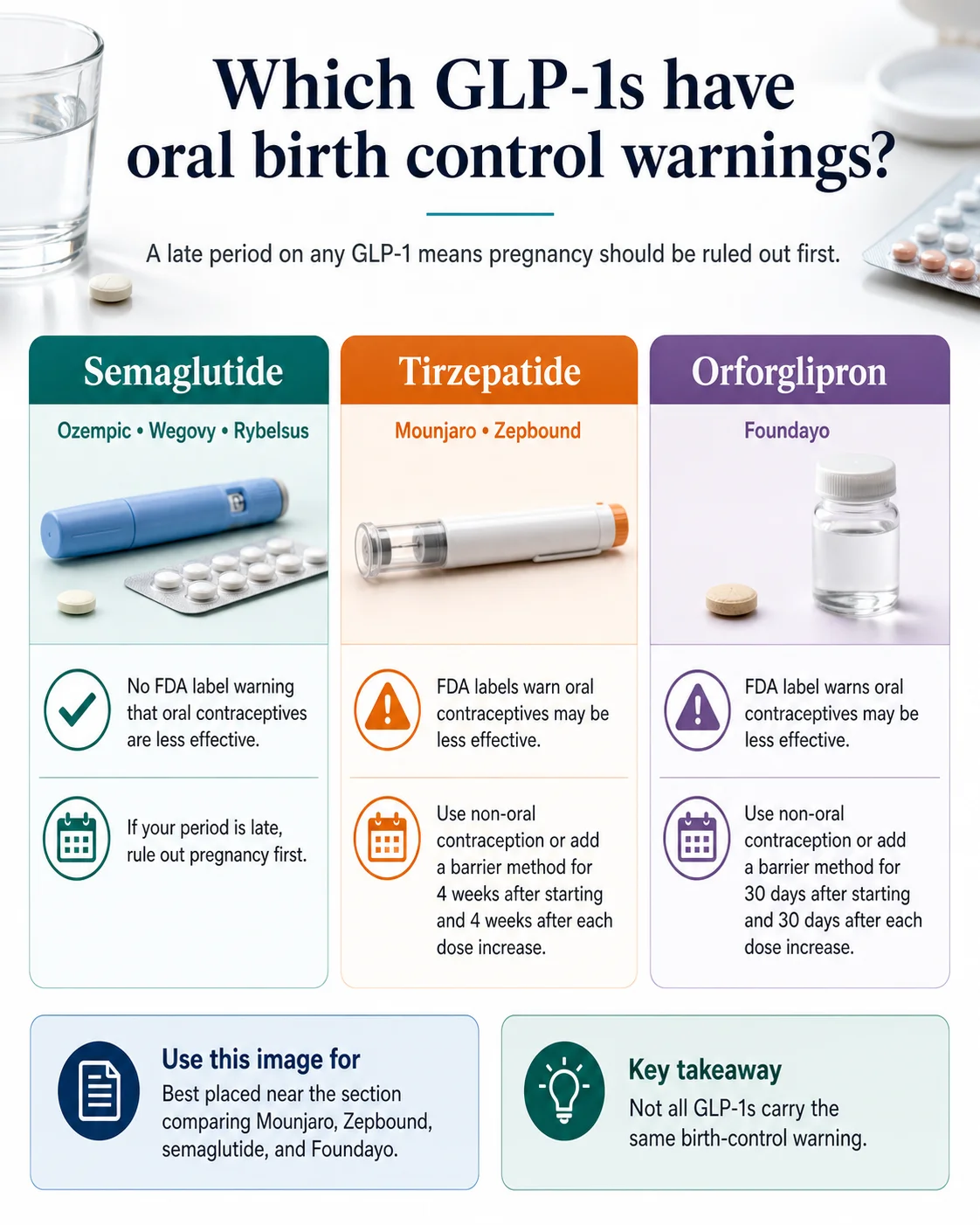
Which GLP-1 needs what: the 60-second answer
The action you take depends entirely on which GLP-1 you take, whether your birth control is a pill, and whether you started recently or just changed dose.
| If you take… | What your FDA label tells you to do today |
|---|---|
| Mounjaro (tirzepatide) | If you take an oral hormonal contraceptive: switch to a non-oral method, or add a barrier method (condoms) for 4 weeks after you start and 4 weeks after every dose increase. |
| Zepbound (tirzepatide) | Same as Mounjaro. 4 weeks after start, 4 weeks after every dose increase. |
| Foundayo (orforglipron) | Switch to a non-oral method or add a barrier method for 30 days after start and 30 days after every dose increase. |
| Ozempic (semaglutide injection) | No tirzepatide-style backup window in the label. Standard missed-pill rules still apply if you vomit or have diarrhea. |
| Wegovy (semaglutide injection or tablet) | No tirzepatide-style backup window. If you take Wegovy tablets, wait 30 minutes before food or other oral medicines. |
| Rybelsus (oral semaglutide) | No tirzepatide-style backup window. Take on an empty stomach with up to 4 oz of water; wait 30 minutes before food, beverages, or other oral medicines. |
| Saxenda or Victoza (liraglutide) | No tirzepatide-style backup window in the label. |
| Trulicity (dulaglutide) | No tirzepatide-style backup window in the label. |
| Byetta (exenatide immediate-release) | Take your oral contraceptive at least 1 hour before Byetta. |
| Bydureon BCise (exenatide extended-release) | Ask your prescriber about timing. The label warns that delayed gastric emptying can slow absorption of oral medicines. |
| Adlyxin (lixisenatide) | Take your oral contraceptive at least 1 hour before or at least 11 hours after Adlyxin. |
| Hormonal IUD, copper IUD, implant, patch, ring, or shot | Not affected by the delayed-gastric-emptying absorption issue. The tirzepatide and Foundayo labels both state non-oral hormonal contraceptives should not be affected. |
Backup-window calculator: figure out your exact dates
You do not need an app. Use the dose dates already in your calendar — or enter them below to get your exact backup-through date.
Backup-Window Date Calculator
Enter your drug and key dates — see your backup-through date instantly
4 weeks (28 days) after start and after each dose escalation. Write the backup-through date on your pen box or pill pack. The clock restarts at every dose increase.
- •Mounjaro or Zepbound: add 28 days to the day you started, and 28 days to the day of any dose increase.
- •Foundayo: add 30 days to your start date, and 30 days to any dose increase.
- •Vomiting, diarrhea, missed pills, or unprotected sex inside a window? Use the stricter rule and call your pharmacist the same day.
- •Semaglutide, liraglutide, dulaglutide: no GLP-1-specific backup window from the label.
What we actually verified for this page
Most "GLP-1 and birth control" articles are one source paraphrasing another. Here is the full source trail.
| What we verified | Source we checked | Last checked |
|---|---|---|
| Mounjaro oral contraceptive warning | DailyMed FDA label (sections 7.2, 8.3, 12.3) | May 14, 2026 |
| Zepbound oral contraceptive warning | DailyMed FDA label (sections 7.2, 8.3, 12.3) | May 14, 2026 |
| Foundayo 30-day warning and half-life | DailyMed FDA label (sections 7.3, 8.3, 12.3) | May 14, 2026 |
| Semaglutide PK with oral contraceptives | DailyMed FDA labels for Wegovy, Ozempic, Rybelsus | May 14, 2026 |
| Liraglutide and dulaglutide label language | DailyMed FDA labels for Saxenda, Victoza, Trulicity | May 14, 2026 |
| Byetta and Adlyxin timing rules and PK values | DailyMed FDA labels and Medication Guides | May 14, 2026 |
| Vomiting and diarrhea pill guidance | CDC U.S. Selected Practice Recommendations for Contraceptive Use | May 14, 2026 |
| Paragard duration | DailyMed FDA label | May 14, 2026 |
| Plan B over-the-counter status | FDA Plan B One-Step labeling and consumer information | May 14, 2026 |
| Compounded GLP-1 status | FDA drug alerts and statements, 2025–2026 | May 14, 2026 |
| PCOS evidence | 2026 systematic review, European Journal of Endocrinology | May 14, 2026 |
We did not use Reddit, TikTok, or anecdotal stories as evidence. Medical claims on this page come from FDA labels, CDC guidance, and peer-reviewed studies.
Why do some GLP-1s affect birth control pills and others don't?
The reason is mechanical, not magical. GLP-1 receptor agonists slow down how fast your stomach empties into your intestines — this is called delayed gastric emptying, and it is part of how the drugs make you feel full and lose weight.
Most pills you swallow — including combined oral contraceptives — are absorbed in the upper small intestine. If your stomach takes longer than normal to push the pill through, the Cmax (peak level of the drug in your blood) drops and the tmax (time to reach that peak) shifts later.
For most GLP-1s, the slowdown is mild enough that the contraceptive ends up in your bloodstream at clinically similar levels overall. Studies of semaglutide and liraglutide found no clinically relevant reduction in oral contraceptive exposure. Trulicity's label tested oral medications at the 1.5 mg dose and reported no clinically relevant effect.
Why tirzepatide is different
Foundayo (orforglipron) is an FDA-approved oral GLP-1 receptor agonist for chronic weight management, approved in 2026. Its label states that the effect on oral contraceptive absorption has not been evaluated in a clinical trial — but because the drug delays gastric emptying, the FDA put a precautionary 30-day backup window in the label. The half-life of ~29–49 hours means it clears relatively quickly, hence a 30-day window rather than a longer one.
One more biology point that matters: tirzepatide's gastric-emptying effect undergoes tachyphylaxis — the drug's effect on this specific system gets weaker each time you take it. The slowdown is biggest after the very first dose, smaller after the second, and minimal by week 4 at a steady dose. That is exactly why the FDA window is 4 weeks — and exactly why the window restarts when you escalate: a new dose level resets the tachyphylaxis clock.
Which GLP-1 drugs affect oral contraceptives? The full action matrix
Every entry below traces to the current FDA label or a primary pharmacokinetic study. Source numbers correspond to the numbered sources list at the bottom of this page.
Tirzepatide drugs (Mounjaro, Zepbound)
| Drug class | Dual GIP / GLP-1 receptor agonist |
| Route | Subcutaneous injection, once weekly |
| FDA indication | Mounjaro: type 2 diabetes. Zepbound: chronic weight management and obstructive sleep apnea in adults with obesity. |
| Contraceptive warning? | YES — backup required |
| Specific PK finding (single 5 mg dose + combined OC) | Ethinyl estradiol: Cmax −59%, AUC −20%. Norgestimate: Cmax −66%, AUC −21%. Norelgestromin (active metabolite): Cmax −55%, AUC −23%. tmax delayed 2.5–4.5 hours. |
| Backup requirement | Switch to non-oral contraception OR add a barrier method |
| Duration | 4 weeks after first dose; 4 weeks after every dose escalation |
| Non-oral methods affected? | No — the label states non-oral hormonal contraceptives should not be affected |
| Source | Mounjaro USPI; Zepbound USPI (DailyMed, current revision) — sources 1, 2 |
For a side-by-side comparison of Zepbound and Foundayo for weight management, see our Foundayo vs. Zepbound guide.
Foundayo (orforglipron)
| Drug class | Oral non-peptide GLP-1 receptor agonist |
| Route | Oral tablet, once daily |
| FDA indication | Chronic weight management in adults with obesity or overweight with at least one weight-related comorbidity |
| Contraceptive warning? | YES — precautionary backup required |
| Specific finding | Effect on oral contraceptive absorption has not been evaluated in a clinical trial. Precautionary window based on delayed gastric emptying. |
| Backup requirement | Switch to non-oral contraception OR add a barrier method |
| Duration | 30 days after initiation; 30 days after each dose escalation |
| Elimination half-life | ~29–49 hours |
| Non-oral methods affected? | No |
| Source | Foundayo USPI (DailyMed, 2026 initial U.S. approval) — source 3 |
Semaglutide drugs (Ozempic, Wegovy, Rybelsus)
| Drug class | GLP-1 receptor agonist |
| Route | Subcutaneous injection (Ozempic, Wegovy injection) or oral tablet (Rybelsus, Wegovy tablet) |
| Contraceptive warning? | No backup window required |
| Specific finding | A drug interaction study of semaglutide with a combined oral contraceptive (ethinyl estradiol / levonorgestrel) found no clinically significant differences in contraceptive exposure. tmax for ethinyl estradiol was delayed by about 1 hour — not clinically relevant. |
| Backup requirement | None routinely required from the label. Standard missed-pill rules apply for vomiting, diarrhea, or missed doses. |
| Special note for oral semaglutide | Tablet must be taken on an empty stomach with up to 4 ounces of water. Wait at least 30 minutes before food, beverages, or other oral medicines. |
| Source | Wegovy, Ozempic, Rybelsus USPIs (DailyMed); Kapitza et al. 2015 — sources 4, 5, 6, 16 |
Liraglutide (Saxenda, Victoza)
| Drug class | GLP-1 receptor agonist |
| Route | Subcutaneous injection, once daily |
| FDA indication | Saxenda: chronic weight management. Victoza: type 2 diabetes. |
| Contraceptive warning? | No backup window required |
| Specific finding | Lower peak concentration of ethinyl estradiol observed, but no reduction in overall ethinyl estradiol AUC; levonorgestrel AUC was increased. No clinically significant effect on contraceptive efficacy identified. |
| Backup requirement | None routinely required from the label |
| Source | Saxenda USPI; Victoza USPI (DailyMed); Jacobsen et al. 2011 — sources 7, 8, 17 |
Dulaglutide (Trulicity)
| Drug class | GLP-1 receptor agonist |
| Route | Subcutaneous injection, once weekly |
| Contraceptive warning? | No backup window required |
| Specific finding | At the 1.5 mg dose tested, oral medication absorption rate was reduced but not to a clinically relevant degree. The label does not give a specific oral contraceptive conclusion and higher-dose experience is more limited. |
| Backup requirement | None routinely required from the label |
| Source | Trulicity USPI (DailyMed) — source 9 |
Exenatide (Byetta, Bydureon BCise)
| Drug class | GLP-1 receptor agonist |
| Route | Subcutaneous injection — twice daily (Byetta) or once weekly (Bydureon BCise) |
| Contraceptive warning? | Timing rule — not a multi-week backup window |
| Specific finding | When a combined OC was given 30 minutes after Byetta: ethinyl estradiol Cmax −45%, levonorgestrel Cmax −27%. When given 1 hour before Byetta: ethinyl estradiol Cmax −15% only, levonorgestrel not significantly changed. |
| Backup requirement | Take oral birth control at least 1 hour before Byetta. For Bydureon BCise (extended-release), ask your prescriber for product-specific timing. |
| Source | Byetta and Bydureon BCise USPIs (DailyMed) — sources 10, 11 |
Lixisenatide (Adlyxin)
| Drug class | GLP-1 receptor agonist |
| Route | Subcutaneous injection, once daily |
| Contraceptive warning? | Timing rule |
| Specific finding | Lixisenatide delays gastric emptying enough to reduce oral contraceptive absorption when taken close to the injection. |
| Backup requirement | Take oral birth control at least 1 hour before Adlyxin or at least 11 hours after. |
| Source | Adlyxin Medication Guide and USPI (DailyMed) — source 12. Note: U.S. availability has been limited in recent years; confirm with your pharmacy. |
Compounded GLP-1 products (semaglutide, tirzepatide, liraglutide)
| What it is | A compounded medication is mixed by a pharmacy rather than mass-produced by an FDA-approved manufacturer. Compounded GLP-1 products are not FDA-approved drugs. |
| Contraceptive guidance | Use the active ingredient as your starting point. If your compounded medication contains tirzepatide, follow the tirzepatide 4-week backup rule unless your prescriber says otherwise. If it contains semaglutide, the brand semaglutide guidance is the closest reference point — but formulation, dose accuracy, and pharmacokinetic behavior may differ. |
| What we are not saying | We are not saying compounded products are equivalent to FDA-approved Mounjaro, Zepbound, Wegovy, or Ozempic in safety or efficacy. They are different products under different oversight. |
| Source | FDA drug alerts and statements on compounded GLP-1 policies (2025–2026), including the proposed exclusion of semaglutide, tirzepatide, and liraglutide from the 503B bulks list. — source 20 |
"GLP-1s make birth control 20% less effective" — what that claim actually means
- •The number is real, but it is not what you think. The "20%" figure comes from the Mounjaro and Zepbound FDA label, where the area under the curve (AUC — total hormonal exposure over 24 hours) was reduced by roughly 20–23% after a single 5 mg dose of tirzepatide.
- •Lower exposure is not the same as a 20% drop in pregnancy protection. Contraceptive failure is a complicated function of hormone levels, ovulation suppression, cervical mucus changes, and consistency of use. The FDA does not say "your pill is 20% less effective." It says the absorption change is large enough that backup or a method switch is warranted during a defined window.
- •The practical instruction is what matters. For tirzepatide: 4 weeks of backup, repeating after every dose increase. For Foundayo: 30 days. For semaglutide, liraglutide, and dulaglutide: no extra backup window from the label.
- •This claim does not apply to all GLP-1s. The single biggest mistake online is people on Ozempic, Wegovy, or Rybelsus assuming the 20% number applies to them. It does not. The Mounjaro/Zepbound numbers come from a tirzepatide-specific study.
Which contraceptive methods bypass the absorption issue entirely
Any method that does not depend on being swallowed and absorbed through the gut bypasses the delayed-gastric-emptying issue. The Mounjaro, Zepbound, and Foundayo labels all explicitly state that non-oral hormonal contraceptives should not be affected.
| Method | What it is | FDA-labeled duration | Bypasses absorption issue? |
|---|---|---|---|
| Hormonal IUD — Mirena | Levonorgestrel-releasing IUD | Up to 8 years | Yes |
| Hormonal IUD — Liletta | Levonorgestrel-releasing IUD | Up to 8 years | Yes |
| Hormonal IUD — Kyleena | Levonorgestrel-releasing IUD | Up to 5 years | Yes |
| Hormonal IUD — Skyla | Levonorgestrel-releasing IUD | Up to 3 years | Yes |
| Copper IUD — Paragard | Hormone-free | Up to 10 years; also used as emergency contraception | Yes |
| Implant — Nexplanon | Etonogestrel subdermal rod | Up to 3 years | Yes |
| Patch — Xulane, Twirla | Transdermal estrogen/progestin | Replaced weekly | Yes |
| Vaginal ring — NuvaRing | Releases ethinyl estradiol/etonogestrel | 3 weeks in, 1 week out | Yes |
| Vaginal ring — Annovera | Releases ethinyl estradiol/segesterone | Reusable for 1 year | Yes |
| Shot — Depo-Provera | Medroxyprogesterone injection | Every 3 months | Yes |
| Male and female condoms | Barrier | Per use | Yes |
| Diaphragm, cervical cap | Barrier | Per use | Yes |
If you are comparing oral GLP-1 pills specifically — Foundayo vs. Wegovy — see our best GLP-1 pill for women guide for a detailed comparison including contraceptive considerations.
Which scenario describes you?
Most readers fit one of four situations. Find yours and follow the plan.
"I'm starting a GLP-1 next week and I'm on the pill"
If your medication is Mounjaro, Zepbound, or Foundayo:
- Keep taking your pill as prescribed.
- Add condoms (or another barrier method) the day you start the GLP-1.
- Use that backup for 4 weeks (30 days if Foundayo).
- Or switch to a non-oral method now and skip the backup-window game entirely.
If your medication is Ozempic, Wegovy, Rybelsus, Saxenda, Victoza, or Trulicity, no backup is required from the label. Continue your pill as prescribed.
If your medication is Byetta or Adlyxin, adjust the timing of your pill instead of adding backup: pill at least 1 hour before the injection (or at least 11 hours after, for Adlyxin).
"I just had a dose increase on Mounjaro, Zepbound, or Foundayo"
The backup-window clock just restarted.
- • Mounjaro/Zepbound: 4 weeks of backup from the day of the new dose.
- • Foundayo: 30 days from the day of the new dose.
The clock resets because each new dose level produces a fresh round of stronger gastric-emptying slowdown before tachyphylaxis settles in. Every dose escalation gets its own window — this is not optional in the label.
"I had unprotected sex on Mounjaro, Zepbound, or Foundayo within the last 5 days and I'm worried"
This one needs a clinician, not a webpage. But here is what you need to know so you do not go in blind.
- • Time matters. Levonorgestrel-based emergency contraception (Plan B) is most effective within 72 hours and loses efficacy quickly after. Ulipristal acetate (ella, prescription) works up to 120 hours.
- • The copper IUD is the most effective form of emergency contraception — over 99% effective, insertable up to 5 days after unprotected sex. It is not affected by any GLP-1 absorption issue.
- • Whether oral emergency contraception is affected by tirzepatide has not been directly studied. Because levonorgestrel is absorbed through the stomach, there is reason to suspect reduced absorption during a tirzepatide window. Ask a pharmacist or clinician whether the copper IUD makes more sense.
- • Plan B is available over the counter in the U.S. without age restriction.
"I want to get pregnant and need to know when to stop my GLP-1"
Different drugs leave the body at different speeds. The standard recommendation is to stop your GLP-1 well before trying to conceive.
| Drug | Half-life | Approx. time to clear | Pre-pregnancy stop guidance |
|---|---|---|---|
| Mounjaro / Zepbound (tirzepatide) | ~5 days | 25–30 days (5–6 half-lives) | U.S. labeling does not give one universal preconception stop interval; UK NHS guidance suggests stopping at least 1 month before trying. Ask your prescriber. |
| Wegovy / Ozempic / Rybelsus (semaglutide) | ~1 week | 5–6 weeks | Novo Nordisk advises discontinuing at least 2 months before planned pregnancy. |
| Foundayo (orforglipron) | ~29–49 hours | ~6–12 days | Follow current label and prescriber guidance; the label instructs discontinuation when pregnancy is recognized. |
| Saxenda / Victoza (liraglutide) | ~13 hours | 2–3 days | Follow prescriber guidance. |
| Trulicity (dulaglutide) | ~5 days | 25–30 days | Follow prescriber guidance. |
A 2024 analysis published in the BMJ of first-trimester GLP-1 exposure did not find a clear signal of increased major birth defects — but human safety data remain limited. Contact your prescriber and obstetric provider promptly if you become pregnant while on a GLP-1.
What to do if you vomit, have diarrhea, or miss a pill while on a GLP-1
Nausea, vomiting, and diarrhea are common side effects of every GLP-1, especially in the first weeks and after dose increases. These can affect pill reliability regardless of whether your specific GLP-1 has the contraceptive warning. The rules below come from the CDC's U.S. Selected Practice Recommendations for Contraceptive Use — the official guideline doctors and pharmacists rely on.
If you take a combined oral contraceptive (estrogen + progestin pill)
- Vomiting or diarrhea lasting less than 24 hours, or less than 48 hours total: Keep taking your pills on schedule. No backup is typically needed.
- Vomiting or diarrhea lasting 48 hours or more: Keep taking your pills, and use a backup method (or avoid sex) until you have taken 7 hormonal pills in a row after the symptoms stop.
- Vomiting or diarrhea during the first week of a new pack + unprotected sex in the previous 5 days: Consider emergency contraception.
Source: CDC, U.S. Selected Practice Recommendations for Contraceptive Use — Combined Hormonal Contraceptives section.
If you take a progestin-only pill (mini-pill)
- Norethindrone or norgestrel mini-pill: If you vomit or have severe diarrhea within 3 hours of taking your pill, treat it as a missed pill. Take another dose if possible, and use backup or avoid sex until 2 days after symptoms resolve.
- Drospirenone-only pill (Slynd): If vomiting or diarrhea continues for more than 24 hours, use backup or avoid sex for 7 days after the symptoms stop.
Source: CDC, U.S. Selected Practice Recommendations for Contraceptive Use — Progestin-Only Pills section.
If you simply missed a pill while on a GLP-1
The standard missed-pill rules from your pill's package insert still apply. Take the missed pill as soon as you remember; if it has been more than 24 hours, follow the brand-specific missed-pill instructions. If you are inside a tirzepatide or Foundayo backup window when this happens, the stricter rule wins — keep using barrier protection regardless.
"Ozempic babies" — what's actually happening
Women are reporting surprise pregnancies on GLP-1s, sometimes after years of infertility. It is a real phenomenon — but it gets reported as a single story when it is actually three different stories.
Story 1
Metabolic fertility restoration
Weight loss and improved insulin sensitivity can restart ovulation in women whose cycles were irregular or absent due to obesity, polycystic ovary syndrome (PCOS), or metabolic syndrome. This is the mechanism that matters on any GLP-1 that leads to meaningful weight loss — including semaglutide.
Story 2
Oral contraceptive absorption (tirzepatide and Foundayo only)
This is the mechanism the FDA labels address directly. It applies to tirzepatide and, precautionarily, to Foundayo. It does not apply to semaglutide drugs.
Story 3
GI side effects
Vomiting and severe diarrhea can reduce pill effectiveness on any GLP-1. This is the same risk as if you got the stomach flu — it has nothing to do with which GLP-1 you take.
Many viral "Ozempic baby" stories use Ozempic as shorthand for GLP-1-related surprise pregnancies, even when the drug involved is Mounjaro, Zepbound, or another GLP-1. If you are on a GLP-1 and you do not want to get pregnant, you need contraception that works through all three stories, not just Story 2. A non-oral method handles all three.
What if I have PCOS and take oral contraceptives on a GLP-1?

If you have polycystic ovary syndrome and started a GLP-1 for weight management or insulin resistance, your situation is a little different. GLP-1 medications can lead to weight loss and improved insulin sensitivity, which may improve cycle regularity or restore ovulation for some people with PCOS.
A 2026 systematic review in the European Journal of Endocrinology identified 11 randomized trials of GLP-1 receptor agonists in PCOS. It found modest short-term BMI reduction and low-certainty evidence for reproductive outcomes. Brand-specific PCOS data for semaglutide and tirzepatide remains limited.
For a comprehensive look at GLP-1 medications specifically for PCOS, see our GLP-1 and PCOS guide and our comparison of best GLP-1 pill for PCOS.
Questions to ask your prescriber or pharmacist
Bring this to your appointment. Or screenshot it for your phone.
For your prescriber
- Which specific GLP-1 am I taking — the generic name and the brand?
- Does my GLP-1's FDA label include an oral contraceptive warning?
- If yes, how long is the backup window?
- Does the backup window restart at every dose increase, and when are my dose increases planned?
- If I take the mini-pill instead of a combined pill, does the same advice apply?
- Would switching to a non-oral method be simpler for my situation?
- Is there a different GLP-1 you would consider if avoiding contraceptive interactions matters to me?
For your pharmacist
- Can you confirm the timing rule for my specific medication?
- What should I do if I vomit within an hour of taking my pill?
- Are there any other medications I take that need their own timing changes?
- If I had unprotected sex within the last 5 days, what are my emergency contraception options given my GLP-1?
- Can I get emergency contraception here without a prescription?
What should I do next if I'm on a GLP-1 and the pill?
The honest answer is short.
- •On tirzepatide (Mounjaro, Zepbound): Follow the 4-week rule, restart at every dose increase, or switch to a non-oral method.
- •On Foundayo (orforglipron): Follow the 30-day rule, restart at every dose increase, or switch to a non-oral method.
- •On semaglutide (Ozempic, Wegovy, Rybelsus): Your label does not add a backup window. Standard missed-pill rules apply for vomiting and diarrhea.
- •On liraglutide (Saxenda, Victoza) or dulaglutide (Trulicity): No backup window from the label.
- •On Byetta or Adlyxin: Separate your pill from the injection by the timing rules above.
- •On any non-oral hormonal method (IUD, implant, patch, ring, shot): The absorption issue does not apply.
If you only do three things after reading this page: write your backup-through date on your medication box, save the missed-pill section for the next stomach bug, and pick the questions from the prescriber checklist that apply to you for your next visit.
Frequently asked questions about GLP-1 and oral contraceptives
How we built this guide
This page exists because the "GLP-1 and birth control" answer most people find online is too broad to be useful. We wanted one place that gives a real, drug-specific answer.
Our source hierarchy
- 1Current U.S. FDA prescribing information and Medication Guides, as published on DailyMed (the National Library of Medicine database that mirrors current FDA labeling).
- 2CDC U.S. Selected Practice Recommendations for Contraceptive Use, for vomiting, diarrhea, and missed-pill triage.
- 3Peer-reviewed pharmacokinetic studies and systematic reviews, cited where they add specificity beyond the label.
- 4FDA drug alerts and statements for compounded GLP-1 status.
What we deliberately did not do
- We did not use Reddit, Facebook groups, or TikTok as evidence for medical claims.
- We did not invent a medical reviewer credit. This page was written by the WPG Research Team and verified against the primary sources listed above.
- We did not equate compounded GLP-1 products with FDA-approved versions.
- We did not present any single drug's data as if it applied to the entire GLP-1 class.
Sources
- 1.Mounjaro (tirzepatide) Prescribing Information. Eli Lilly. DailyMed: dailymed.nlm.nih.gov
- 2.Zepbound (tirzepatide) Prescribing Information. Eli Lilly. DailyMed: dailymed.nlm.nih.gov
- 3.Foundayo (orforglipron) Prescribing Information. Eli Lilly. DailyMed: dailymed.nlm.nih.gov
- 4.Wegovy (semaglutide injection and tablets) Prescribing Information. Novo Nordisk. DailyMed: dailymed.nlm.nih.gov
- 5.Ozempic (semaglutide injection) Prescribing Information. Novo Nordisk. DailyMed: dailymed.nlm.nih.gov
- 6.Rybelsus / Ozempic Tablets (oral semaglutide) Prescribing Information. Novo Nordisk. DailyMed: dailymed.nlm.nih.gov
- 7.Saxenda (liraglutide) Prescribing Information. Novo Nordisk. DailyMed: dailymed.nlm.nih.gov
- 8.Victoza (liraglutide) Prescribing Information. Novo Nordisk. DailyMed (most recent revision on file).
- 9.Trulicity (dulaglutide) Prescribing Information. Eli Lilly. DailyMed: dailymed.nlm.nih.gov
- 10.Byetta (exenatide) Prescribing Information and Medication Guide. AstraZeneca. DailyMed: dailymed.nlm.nih.gov
- 11.Bydureon BCise (exenatide extended-release) Prescribing Information. AstraZeneca. DailyMed: dailymed.nlm.nih.gov
- 12.Adlyxin (lixisenatide) Prescribing Information and Medication Guide. Sanofi. DailyMed (most recent revision on file).
- 13.Paragard (copper IUD) Prescribing Information. CooperSurgical. DailyMed: dailymed.nlm.nih.gov
- 14.CDC. U.S. Selected Practice Recommendations for Contraceptive Use — Combined Hormonal Contraceptives and Progestin-Only Pills sections. cdc.gov/contraception/hcp/usspr/
- 15.FDA. Plan B One-Step (levonorgestrel) consumer information — over-the-counter availability without age restriction.
- 16.Kapitza C, Nosek L, Jensen L, et al. Semaglutide, a Once-Weekly Human GLP-1 Analog, Does Not Reduce the Bioavailability of the Combined Oral Contraceptive, Ethinylestradiol/Levonorgestrel. J Clin Pharmacol. 2015;55(5):497–504.
- 17.Jacobsen LV, Vouis J, Hindsberger C, Zdravkovic M. Treatment with liraglutide — a once-daily GLP-1 analog — does not reduce the bioavailability of ethinyl estradiol/levonorgestrel taken as an oral combination contraceptive drug. J Clin Pharmacol. 2011;51(12):1696–1703.
- 18.Skelley JW, Swearengin K, York AL, Glover LH. The impact of tirzepatide and glucagon-like peptide 1 receptor agonists on oral hormonal contraception. J Am Pharm Assoc. 2024;64(1):204–211.
- 19.Systematic review of GLP-1 receptor agonists in PCOS. European Journal of Endocrinology. 2026;194(3):S25.
- 20.FDA. Drug alerts and statements on compounded GLP-1 products, including the proposed exclusion of semaglutide, tirzepatide, and liraglutide from the 503B bulks list. 2025–2026.