🚨 If you have severe upper-belly pain right now:
Stop your GLP-1. Do not take your next dose. Go to the ER now — or call 911 if pain is severe. Do not drive yourself.
GLP-1 Pancreatitis Symptoms: Warning Signs and the ER Red Flags You Shouldn’t Ignore
By Weight Loss Provider Guide Editorial Team ·
Sources: FDA/DailyMed prescribing information for Wegovy, Ozempic, Zepbound, Mounjaro, Saxenda, Trulicity, Rybelsus, and Byetta; NIDDK; Mayo Clinic; UK MHRA January 2026 safety communication; 2024 FAERS pharmacovigilance data.
Medical disclaimer: This page is for education and triage support only. It does not diagnose pancreatitis or replace emergency medical care. If your symptoms match the warning signs below, stop reading and go to the ER.
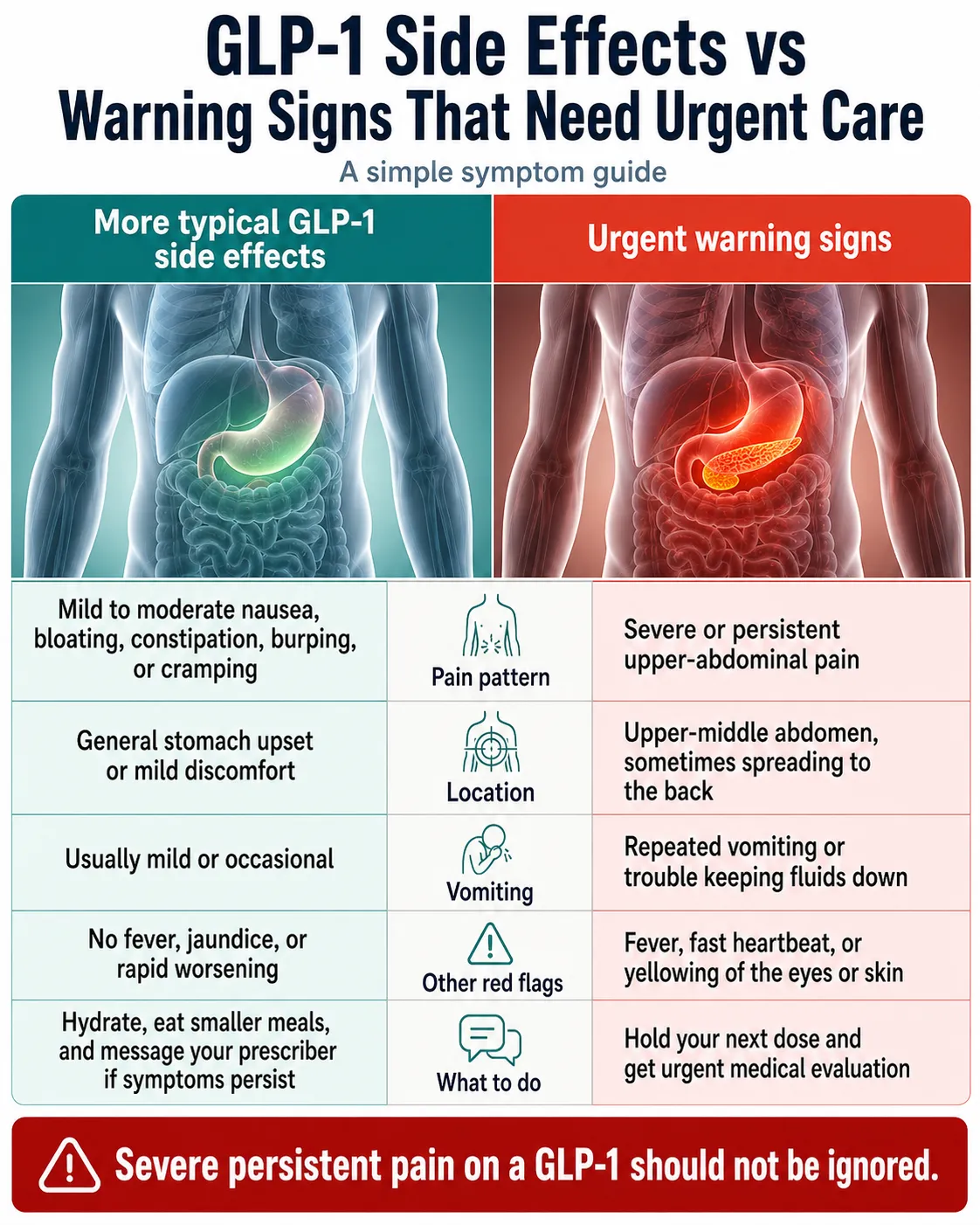
If you’re on a GLP-1 — Wegovy, Ozempic, Zepbound, Mounjaro, Saxenda, Trulicity, Rybelsus, Byetta, the new Foundayo (orforglipron), or a compounded semaglutide or tirzepatide — and you have severe upper-belly pain that won’t go away, especially if it spreads through to your back, comes with repeated vomiting, fever, fast heartbeat, or yellowing of your eyes or skin — stop your GLP-1, don’t take your next dose, and get to the ER now.
Most GLP-1 stomach trouble is the ordinary kind: nausea, mild cramping, constipation, burping, and the occasional bad day after a dose increase. This page helps you tell the difference in 60 seconds — and tells you exactly what to do next.
The Decision Table — Read This First
| What you’re feeling | How urgent | What to do |
|---|---|---|
| Severe upper-belly pain that won’t quit, especially radiating to your back | 🚨 Emergency | Stop your GLP-1. Go to the ER. Don’t drive yourself if pain is severe. |
| Severe pain plus repeated vomiting, fever, fast pulse, shortness of breath, jaundice, or you can’t keep fluids down | 🚨 Emergency | Call 911 or have someone drive you to the ER. |
| Moderate pain that’s new, worsening, or not going away after a dose change | ⚠ Same-day | Call your prescriber today. Don’t take the next dose until they say so. |
| Mild nausea, mild cramping, burping, constipation, brief upset after a dose | ✓ Usually fine | Smaller meals, more water. Message your prescriber if it doesn’t ease in a few days. |
| You’re not sure where you fit | → Triage needed | Use the symptom differentiator in Section 2, then message your prescriber if any red flag appears. |
1. The Four Pancreatitis Warning Signs on GLP-1 Medications
The four warning signs every GLP-1 prescribing label tells doctors to act on are (1) severe, persistent upper-abdominal pain — often radiating through to the back, (2) vomiting that won’t stop, (3) fever or rapid heartbeat with the pain, and (4) yellowing of the skin or whites of the eyes. If any of these appear — especially the first one — stop the medication and seek emergency care. The FDA labels for Wegovy, Ozempic, Zepbound, Mounjaro, and the rest of the class all say the same thing.
Warning sign #1: Severe upper-belly pain that radiates to your back
This is the hallmark. NIDDK describes the main pancreatitis symptom as upper-abdominal pain that may spread to the back, and Mayo Clinic uses almost identical language. Here’s what makes it different from a cranky stomach:
- •It’s severe. Patients describe it as “the worst pain of my life” or so intense they can’t sit still or find a comfortable position.
- •It's constant, not crampy. Routine GLP-1 nausea fluctuates with meals and hydration. Pancreatitis pain doesn't ease and come back — it stays.
- •It often goes through to the back. Pain in the upper-middle abdomen that you can also feel in your mid-back, like it's drilling through.
- •It doesn't respond to position changes or OTC meds. Lying still, sitting up, ibuprofen, antacids — nothing touches it.
If your pain checks any three of those four boxes, treat it as pancreatitis until a clinician proves otherwise.
Warning sign #2: Vomiting that won’t stop
GLP-1 nausea is common — Wegovy clinical trials report nausea in roughly 44% of patients early on. That’s not the kind of vomiting we’re talking about here. Pancreatitis vomiting is repeated, severe, and often comes with the inability to keep fluids down at all. If you’re throwing up every time you sip water, that’s a red flag.
Warning sign #3: Fever or fast heartbeat with the pain
A body temperature over 100.4°F (38°C) or a resting heart rate over 100 bpm — alongside abdominal pain — signals systemic inflammation. NIDDK specifically lists fever and tachycardia as features of acute pancreatitis that warrant immediate care.
Warning sign #4: Yellowing of skin or whites of the eyes (jaundice)
Less common but very specific. Jaundice on a GLP-1 with abdominal pain often points to a gallstone blocking the bile duct — which can trigger gallstone pancreatitis, the most common cause of acute pancreatitis in the U.S. If your eyes look yellow, this is not the morning to “see how it goes.”
From the Wegovy FDA prescribing information (current label): “After initiation of WEGOVY, observe patients carefully for signs and symptoms of acute pancreatitis, which may include persistent or severe abdominal pain (sometimes radiating to the back), and which may or may not be accompanied by nausea or vomiting. If pancreatitis is suspected, discontinue WEGOVY and initiate appropriate management.”
The same language, almost word-for-word, appears in the Ozempic, Zepbound, Mounjaro, Saxenda, Trulicity, Rybelsus, and Byetta labels.
2. Pancreatitis Pain vs Normal GLP-1 Side Effects: The 60-Second Differentiator
Normal GLP-1 GI side effects are mild to moderate, come and go with meals, ease with hydration and smaller portions, and improve as your body adjusts to the dose. Pancreatitis pain is sudden, severe, constant, located in the upper-middle abdomen, often radiates straight to the back, and doesn’t respond to position changes or OTC remedies. If your pain pattern fits the third or fourth columns below, treat it as an emergency.
| Symptom pattern | Normal GLP-1 effects | Pancreatitis (ER NOW) | Gallbladder attack | Gastroparesis flare |
|---|---|---|---|---|
| Pain location | Generalized stomach discomfort, mild epigastric | Severe upper-middle, often radiating to back | Right upper abdomen, may radiate to right shoulder | Upper abdomen, fullness, bloating |
| Pain quality | Mild–moderate cramping, comes and goes with meals | Severe, constant; not relieved by position or OTC pain relievers | Sharp, episodic, often after fatty meals; eases over 1–6 hours | Heavy, full, persistent |
| Onset timing | Worst in first 2–4 weeks and after dose increases; improves over time | Sudden or rapidly worsening; common in first 60 days or after dose escalation | After fatty meal; episodic | Gradual; chronic with episodes |
| Vomiting | Occasional; eases with hydration & smaller meals | Persistent, won't stop; may include inability to keep fluids down | Possible during the attack | Possible, often after meals |
| Fever | Rare or none | May be present (>100.4°F / 38°C) | May be present | Rare |
| Jaundice | None | Possible (bile-duct involvement) | Possible if a stone blocks the duct | None |
| Response to time + hydration | Improves over hours | Doesn't improve; often worsens | Eases over hours | Variable |
| What to do | Smaller lower-fat meals; hydrate; tell prescriber if persistent | STOP your GLP-1, go to ER immediately | Urgent care or ER same day, especially with fever or jaundice | Call prescriber; may need a dose hold |
What’s not in the table: Bloating, burping, mild reflux, occasional cramping, appetite loss, and brief queasy spells after a dose are all common GLP-1 effects. They’re annoying. They aren’t pancreatitis on their own. If you have those symptoms without any of the four warning signs in Section 1, the answer is usually “smaller meals, more water, give it another week” — not the ER.
3. How Common Is Pancreatitis on Ozempic, Wegovy, Mounjaro, Zepbound, and Other GLP-1s?
Pancreatitis on GLP-1 medications is uncommon. Clinical trials report rates of roughly 0.2 to 0.4 cases per 100 patient-years across the major weight-loss and diabetes GLP-1s — similar to or only modestly above placebo in most studies. A 2025 meta-analysis of more than 102,000 participants across multiple cardiovascular outcome trials found no statistically significant increased risk of acute pancreatitis with GLP-1 therapy versus placebo. The risk is real and documented — but it’s uncommon, and understanding the actual numbers puts the risk in context.
Drug-by-drug pancreatitis rates from FDA labels
| Medication | Active ingredient | Pancreatitis rate in pivotal trials | vs comparator |
|---|---|---|---|
| Wegovy (injection) | semaglutide 2.4 mg | 0.2 cases / 100 patient-years (4 cases adjudicated in adult weight-loss trials) | <0.1 / 100 patient-years on placebo |
| Wegovy (oral, 25 mg tablet) | semaglutide oral | Class warning applies | Class warning applies |
| Ozempic | semaglutide ≤2 mg | 0.3 cases / 100 patient-years (7 cases adjudicated in glycemic control trials) | 0.2 / 100 patient-years on comparator |
| Mounjaro | tirzepatide | 0.23 patients / 100 years exposure (T2D trials) | 0.11 / 100 years on comparator |
| Zepbound | tirzepatide | 0.14 patients / 100 years (pooled weight-reduction trials) | 0.15 / 100 years on placebo (i.e., not elevated) |
| Saxenda | liraglutide 3 mg | Reported in trials; class warning applies | — |
| Trulicity | dulaglutide | Reported in trials; class warning applies | — |
| Rybelsus | oral semaglutide | Class warning applies | — |
| Byetta | exenatide | Reported in trials; class warning applies | — |
| Foundayo | orforglipron (FDA-approved April 2026) | Class warning applies; long-term real-world data still emerging | — |
| Compounded semaglutide / tirzepatide | varies | Not measured in RCTs (compounded products are not FDA-evaluated) | See Section 6 for FAERS data |
Sources: FDA accessdata.fda.gov labels for each medication, current as of April 2026; Cleveland Clinic Journal of Medicine 2025 review.
How to read these rates
A “0.2 cases per 100 patient-years” rate means that if 1,000 people took the drug for one year, you’d expect about two cases of acute pancreatitis. Most of those cases would be people with at least one independent risk factor — gallstones, hypertriglyceridemia, prior pancreatitis, heavy alcohol use — that would have raised their pancreatitis risk regardless of the medication.
The strongest historical FAERS pancreatitis-reporting signal is liraglutide, followed by exenatide, semaglutide, dulaglutide, and lixisenatide. Tirzepatide (Mounjaro/Zepbound) shows the lowest GLP-1-class signal in the same FAERS analysis. FAERS reports reflect prescribing volume, reporting bias, and media attention as much as true risk — but the relative ordering is consistent with clinical-trial data.
4. When in Treatment Is Pancreatitis Most Likely to Show Up?
Most reported pancreatitis cases on GLP-1 medications appear within the first 60 days of starting treatment or shortly after a dose escalation. A pharmacovigilance case-series analysis of 39 GLP-1-associated pancreatitis cases found a median time to onset of about 2.5 months from drug initiation, with a quarter of cases occurring within three months of a dose increase.
Why the early-treatment window is riskier
- ⚠Dose escalation is when GI side effects peak. Nausea, vomiting, and slowed gastric emptying are most pronounced during titration — a setup for bile sludge and gallstone formation, which is the leading cause of acute pancreatitis in the U.S.
- ⚠Rapid weight loss in the first 8–12 weeks is itself an independent pancreatitis risk factor. Sustained weight loss faster than about 2 lb per week is associated with gallstone formation.
- ⚠Hypertriglyceridemia takes time to improve. For someone who started with very high triglycerides (>500 mg/dL), the early weeks remain a higher-risk zone.
5. What the Drug Labels Actually Say — The 8-Medication Crosswalk
Every major GLP-1 and dual GIP/GLP-1 medication carries near-identical pancreatitis warning language in its FDA prescribing information. The labels differ in indication, population, and dose — but the patient-facing red-flag rule is the same across all eight. Below is a side-by-side crosswalk so you don’t have to open eight DailyMed tabs.
| Medication | Active ingredient | What the label tells patients | What it means for you |
|---|---|---|---|
| Wegovy | semaglutide (injectable & oral 25 mg tablet) | Stop using and call your provider right away if you have severe stomach pain that won't go away — with or without vomiting; pain may radiate from abdomen to back. | Severe upper-belly pain that persists is the trigger. Don't normalize it. |
| Ozempic | semaglutide injection | Observe for persistent severe abdominal pain, sometimes radiating to the back, with or without vomiting. Discontinue if suspected. Do not restart if confirmed. | The "do not restart" language is firm — Ozempic is off the table after a confirmed episode. |
| Zepbound | tirzepatide (KwikPen and vials) | Acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis, has been observed; warning signs include persistent or severe abdominal pain sometimes radiating to the back. | Same red-flag rule. Restart guidance is more nuanced than Ozempic — see Section 9. |
| Mounjaro | tirzepatide injection | Stop using and call your provider right away if severe stomach-area pain won't go away, with or without nausea/vomiting; pain may radiate to the back. | Same as Zepbound. The label specifies that continuation should be individually determined by a clinician. |
| Saxenda | liraglutide 3 mg | Acute pancreatitis has been observed; signs may include persistent or severe abdominal pain sometimes radiating to the back. Discontinue if suspected. | Liraglutide carries the strongest historical FAERS pancreatitis-reporting signal of the GLP-1 class. |
| Trulicity | dulaglutide | Persistent or severe abdominal pain sometimes radiating to the back, with or without nausea/vomiting. Discontinue if suspected. | Trulicity is for type 2 diabetes — diabetes itself is an independent pancreatitis risk factor. |
| Rybelsus | oral semaglutide | Discontinue promptly and contact physician if pancreatitis is suspected; severe abdominal pain may radiate to the back, with or without nausea/vomiting. | Oral form, same red flags as injectable semaglutide. |
| Byetta | exenatide | Acute pancreatitis has been observed with GLP-1 receptor agonists including Byetta; persistent severe abdominal pain sometimes radiating to the back, with or without vomiting. | Older GLP-1; same warning class. |
| Foundayo | orforglipron (FDA-approved April 2026) | GLP-1 class warning applies; first oral once-daily small-molecule GLP-1; long-term real-world data still emerging. | Treat as GLP-1 class for symptom vigilance purposes. |
6. Compounded GLP-1 vs FDA-Approved: Does the Pancreatitis Risk Change?
The pancreatitis warning signs are identical for compounded and FDA-approved GLP-1 medications. The difference isn’t in the symptom rule — it’s in the manufacturing oversight. A 2024 FAERS pharmacovigilance analysis of 707 compounded-product reports found compounded GLP-1s had higher reporting odds for abdominal pain (2.84×) and cholecystitis (3.39×) compared with FDA-approved branded products, plus dramatically higher rates of dosing and preparation errors.
| Event | Reporting odds ratio (vs branded) | 95% CI |
|---|---|---|
| Abdominal pain | 2.84× | 2.29–3.49 |
| Cholecystitis (gallbladder inflammation) | 3.39× | 1.61–6.31 |
| Diarrhea | 1.59× | — |
| Nausea | 1.27× | — |
| Preparation errors | 48.9× | 12.6–189.6 |
| Prescribing errors | 4.46× | — |
| Contamination reports | 19.0× | — |
| Compounding/manufacturing issues | 8.51× | — |
What to bring to the ER if you’re on a compounded GLP-1
- Tell the team it’s compounded — the ER’s first instinct is to assume FDA-approved medication. Correct that immediately.
- Bring the bottle, vial, or syringe — compounder’s name, lot number if you have it, concentration, and the dose you’ve been taking.
- Be specific about how you measured your dose — “I drew up 0.25 mL on the insulin syringe to the 25-unit line” is more useful than “I took 2.5 mg.”
- Mention any additives — some compounded products include B12, lipotropic compounds, or other ingredients that can complicate the workup.
- If you suspect a dosing error, say so — even uncertainty is useful clinical information.
7. What to Do Right Now If You Think It Might Be Pancreatitis
Stop your GLP-1 immediately, do not take your next dose, and get to an emergency room. If pain is severe, call 911 or have someone drive you — don’t drive yourself. Bring your medication container. Don’t eat or drink anything until you’ve been evaluated.
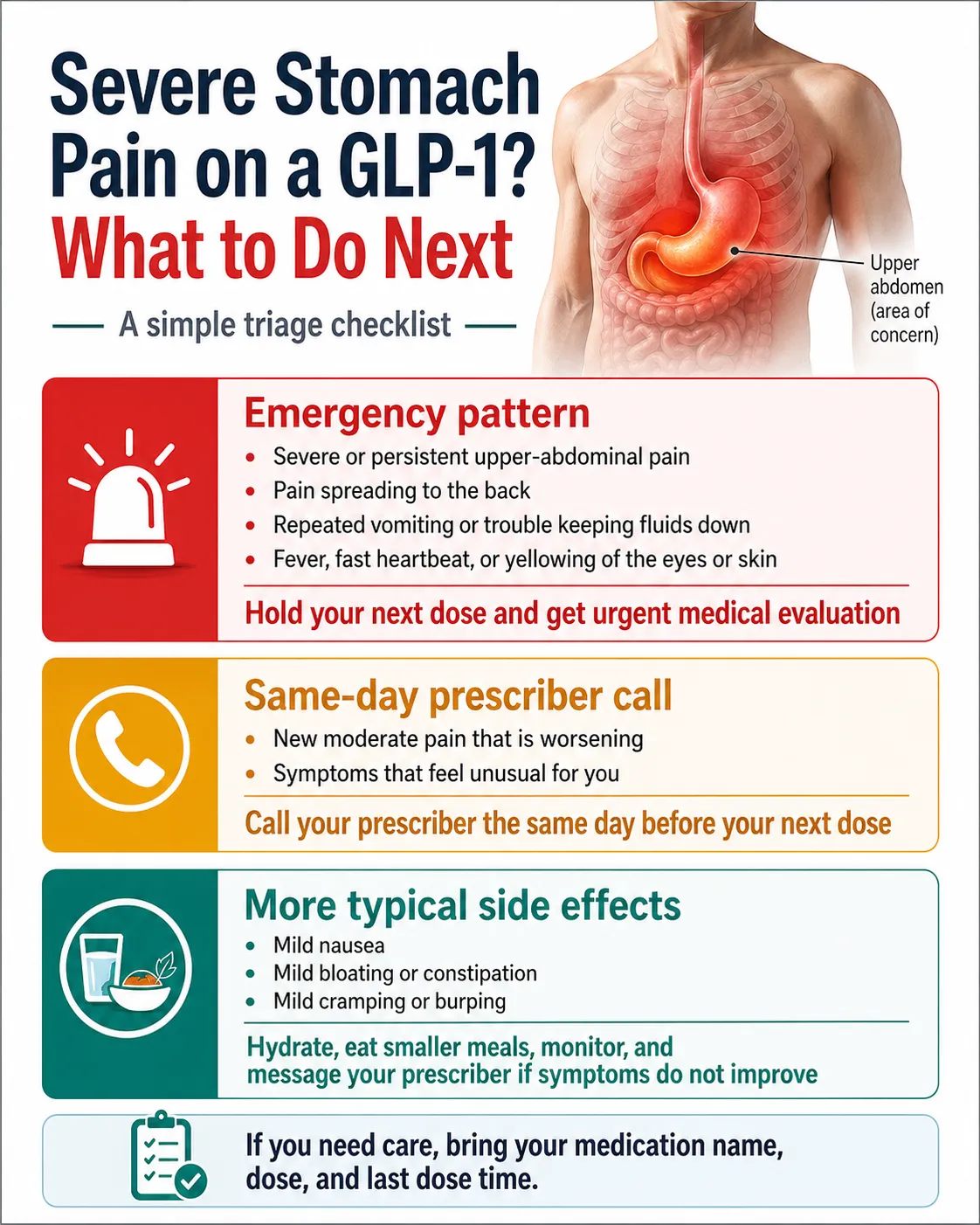
The 6-step action guide
- 1Stop the medication. Don't take your next scheduled dose. Don't try to "wait it out."
- 2Note the time, severity (1–10), and location of pain. Note any vomiting, fever, color changes (skin, eyes, stool, urine), and how long this has been going on.
- 3Bring the medication container with you — pen, vial, prefilled syringe, compounded bottle, whatever it is. Bring the prescription label too.
- 4Bring a list of every other medication, supplement, and recent alcohol intake. Pancreatitis has many causes; the ER will work through a differential.
- 5Tell the ER team upfront that you're on a GLP-1. Ask them to check lipase and amylase. The Atlanta classification requires lipase or amylase ≥3× the upper limit of normal for diagnosis.
- 6If you're on a compounded product, bring the compounder's name and lot number. See Section 6 above.
The copyable symptom note
If your symptoms are concerning but not at the “call 911” level — moderate pain, persistent but not severe, vomiting that’s manageable — message your prescriber today before your next dose. Copy this template:
Hi, I’m currently on [medication name and dose]. I’m having symptoms I’m worried about and want to check before my next dose.
Symptom timeline: Started [time/date], current severity [1–10].
Pain location: [upper-middle abdomen / right upper / radiating to back / other].
Vomiting: [yes/no, how often, can I keep fluids down].
Fever or chills: [yes/no, temperature if measured].
Heart rate: [if you’ve checked it].
Last dose: [date and time].
Recent dose change: [yes/no, when].
Other risk factors: [history of pancreatitis, gallstones, high triglycerides, alcohol use, compounded vs FDA-approved source].
Should I take my next dose, or wait until I’ve been seen?
8. What to Expect at the ER
The ER team will draw blood to check pancreatic enzymes (lipase and amylase), order imaging — usually a CT scan or abdominal ultrasound — to look for inflammation or gallstones, and likely admit you for IV fluids, pain management, and a temporary “nothing by mouth” period to rest the pancreas. About 80% of acute pancreatitis cases are mild; about 20% are severe.
What they’ll test
- •Lipase — the more specific pancreatic enzyme. A level ≥3× the upper limit of normal is a key diagnostic criterion.
- •Amylase — older marker, less specific, often run alongside lipase.
- •Complete metabolic panel — kidney function, liver function, calcium, electrolytes.
- •Triglycerides — to check for hypertriglyceridemic pancreatitis.
- •CBC and inflammation markers — white blood cell count, sometimes CRP.
- •Glucose — pancreatic inflammation can affect blood sugar.
Imaging they may order
- •Abdominal ultrasound — first-line for looking at the gallbladder and bile ducts.
- •CT scan with contrast — gold standard for imaging the pancreas itself. Identifies inflammation, fluid collections, and necrosis.
- •MRCP (magnetic resonance cholangiopancreatography) — used when bile-duct stones are suspected.
- •Endoscopic ultrasound — sometimes used in selected cases.
How they’ll diagnose it — Revised Atlanta classification
Two of three criteria are required:
- Abdominal pain consistent with pancreatitis
- Lipase or amylase elevated to at least 3× the upper limit of normal
- Imaging findings consistent with acute pancreatitis (CT, MRI, or ultrasound)
Mild pancreatitis (~80% of cases)
- •Hospital admission for 2–5 days
- •IV fluids — often aggressive in the first 24 hours
- •Pain control (typically IV opioids)
- •Bowel rest (nothing by mouth) initially, advancing to clear liquids and a low-fat diet
- •Treat the underlying cause (gallstones, high triglycerides)
Severe pancreatitis (~20% of cases)
- •Longer hospitalization, sometimes ICU
- •Possible necrotizing pancreatitis, organ failure (kidney, lung)
- •Infected pancreatic fluid collections
- •May need surgical or endoscopic intervention
9. Can You Restart Your GLP-1 After Pancreatitis?
It depends on which medication you were on. The Wegovy and Ozempic labels say not to restart if pancreatitis is confirmed. The Zepbound and Mounjaro labels are slightly more permissive — restart should be individually determined by your prescriber based on benefit-risk analysis. In every case, the decision is between you and your clinician — not a self-restart.
| Medication | What the label says about restarting |
|---|---|
| Wegovy | "Do not restart if pancreatitis is confirmed." |
| Ozempic | "Do not restart if pancreatitis is confirmed." |
| Rybelsus | Same as injectable semaglutide. |
| Mounjaro | "Continuation should be individually determined by the prescriber's clinical judgment." |
| Zepbound | Same as Mounjaro. |
| Saxenda | Discontinue if suspected; clinician judgment for continuation. |
| Trulicity | Discontinue if suspected; clinician judgment for continuation. |
| Byetta | Discontinue if suspected; clinician judgment for continuation. |
| Foundayo | GLP-1 class warning applies; clinician-led decision. |
When a clinician might consider GLP-1 therapy again
Recent meta-analyses suggest that in carefully selected patients — those whose original pancreatitis had a clear, modifiable cause that’s now been corrected (gallstones since removed, hypertriglyceridemia controlled, alcohol use stopped) — GLP-1 therapy may eventually be revisited. This is a clinician-led conversation that includes:
- →What caused the original episode and whether it's been treated
- →Whether the original cause is reversible (gallstones, lipids, alcohol) or not (idiopathic, genetic)
- →The severity of the original episode (mild vs necrotizing)
- →Your overall benefit-risk balance for the underlying condition (diabetes, obesity, cardiovascular risk)
If your prescriber decides GLP-1s are off the table, alternatives exist. For weight management: lifestyle and nutrition counseling; non-GLP-1 medications (phentermine-topiramate, naltrexone-bupropion, orlistat); bariatric surgery referral for those who qualify. For type 2 diabetes: metformin, SGLT2 inhibitors, DPP-4 inhibitors, sulfonylureas, insulin.
If you came to this site comparing GLP-1 programs and now realize you may not be a candidate, our GLP-1 Contraindications guide covers who shouldn’t take these medications and what alternatives make sense.
10. Who’s at Higher Risk for GLP-1-Related Pancreatitis?
Most reported pancreatitis cases on GLP-1 medications occur in patients with at least one independent risk factor: a prior history of pancreatitis, triglycerides above 500 mg/dL, gallstones, heavy alcohol use, rapid weight loss, or active dose escalation. Type 2 diabetes and obesity are themselves independent baseline risk factors regardless of medication.
Personal risk scorecard
Educational tool, not a diagnostic instrument. Built from peer-reviewed risk-factor research and current FDA labeling. Does not replace your prescriber’s judgment.
| Risk factor | Weight | Why it matters |
|---|---|---|
| History of acute pancreatitis (any cause) | +3 | Excluded from most GLP-1 RCTs; closest monitoring required |
| Triglycerides >500 mg/dL within last 12 months | +3 | Independent pancreatitis trigger; >1,000 mg/dL is acute risk |
| Known gallstones / current gallbladder symptoms | +2 | Gallstones cause about half of acute pancreatitis cases |
| Heavy or daily alcohol use | +2 | Second leading cause of acute pancreatitis |
| Currently in dose-escalation phase (last titration <30 days ago) | +1 | Peak GI side-effect window |
| Rapid weight loss (>2 lb/week sustained for >4 weeks) | +1 | Independent gallstone-formation risk |
| Type 2 diabetes | +1 | Baseline elevated risk regardless of medication |
| Family history of pancreatitis or hereditary pancreatitis genes | +1 | Genetic susceptibility |
Score 0–1 — Low baseline risk
Your absolute risk on a GLP-1 is in the same range as the general population. Stay aware of warning signs but don't lose sleep.
Score 2–4 — Moderate
Your risk is meaningfully elevated. Symptom vigilance matters more for you. Discuss the picture with your prescriber.
Score 5+ — Elevated
A clinician conversation is needed before continuing. At this risk level, the tradeoffs depend on your specific situation, and a generic "GLP-1s are usually safe" doesn't apply cleanly.
11. Real Patient Voices: Why People Wait Too Long
People often delay seeking care for pancreatitis-pattern symptoms for human reasons — they want the medication to work, they don’t want to seem dramatic, they think the pain isn’t “bad enough yet.” These quotes are not medical evidence; they capture the exact emotional pattern that keeps people out of the ER when they should be going. Quotes are from r/Mounjaro public posts, used for language and emotional context only.
"I wanted so bad for Mounjaro to work for me, that I ignored my symptoms."— r/Mounjaro user, 2024. Used for context only, not as medical evidence.
"I've been concerned bc I've been having stomach pains but it hasn't been severe enough…"— r/Mounjaro user, 2024. Used for context only, not as medical evidence.
"If you ever have stomach pain while on mounjaro and the pain doesn't go away, get to the hospital before it becomes unbearable."— r/Mounjaro user, 2024. Used for context only, not as medical evidence.
The pattern: people normalize escalating pain because GLP-1 side effects are common, because the medication is finally helping, or because they don’t want to bother their doctor. Pancreatitis doesn’t care about your motivation. If your pain matches the warning signs, the right call is the early one.
12. How to Lower Your Pancreatitis Risk Without Stopping Your GLP-1
Most pancreatitis cases on GLP-1 medications trace back to modifiable factors — too-fast weight loss, alcohol use, untreated hypertriglyceridemia, dehydration, or dose-escalation strain. You can meaningfully lower your risk by following the titration schedule, eating smaller lower-fat meals, staying hydrated, avoiding alcohol, treating high triglycerides before they climb above 500 mg/dL, and disclosing any history of pancreatitis or gallstones to your prescriber.
The 7 practical steps
- 1Follow the titration schedule. Don't push to a higher dose ahead of plan because progress feels slow. Most clinical-trial pancreatitis cases happened during dose escalation.
- 2Limit fat intake to roughly 20–35% of daily calories during dose escalation. A high-fat meal in a stomach that's emptying slowly is a setup for both nausea and biliary stress.
- 3Stay hydrated. Aim for 64 oz of water daily; more in heat or with exercise. Dehydration concentrates bile and can contribute to gallstone formation.
- 4Avoid alcohol. Heavy alcohol use is the second leading cause of acute pancreatitis. If you're going to drink, keep it minimal.
- 5Get a baseline lipid panel — especially triglycerides. If your triglycerides are above 500 mg/dL, treat them. If they're above 1,000 mg/dL, treat them aggressively before starting or escalating a GLP-1.
- 6Report any persistent upper-abdominal pain to your prescriber within 24 hours. Same-day if it's severe. The early window is when intervention matters most.
- 7If you have new gallstone-suspicious symptoms — right-upper-belly pain after fatty meals, especially with nausea or jaundice — get a gallbladder ultrasound. Untreated gallstones are the leading cause of acute pancreatitis in the U.S.
If you’re losing weight faster than 2 lb per week consistently, that’s a flag — faster isn’t better in this context. Talk to your prescriber about whether a dose hold or a slower titration would make sense. For the broader picture on what’s normal versus concerning across all GLP-1 side effects, our companion guide GLP-1 Side Effects: Normal vs ER-Worthy covers gallbladder symptoms, bowel obstruction, kidney concerns, allergic reactions, and dosing errors in the same triage-first format.
13. After You’ve Recovered — What’s Next?
If you’ve had a confirmed pancreatitis episode, your priority is following your prescriber’s discharge plan, identifying and treating the underlying cause, and not restarting any medication on your own.
A direct admission, because you deserve one
Weight Loss Provider Guide is an independent comparison resource for GLP-1 telehealth providers. Most pages on our site help people choose between programs. This page does not. If you’re here because you’re worried about pancreatitis, we don’t want you choosing a provider today. We want you safe today.
The provider links below exist for one specific reader: someone who has fully recovered, spoken with their prescriber, and is making an informed decision about whether and how to continue GLP-1 therapy. If that’s not you yet — if you’re still in the acute window, still triaging symptoms, or still working through the underlying cause — ignore them. They’ll still be here when you’re ready.
The primary next step (no affiliate involved)
Not a provider link. A free internal tool that asks about your medical history, what was actually causing your symptoms, what your prescriber said, and what your goals are. The output is a personalized action plan — including non-GLP-1 alternatives if those are now a better fit.
Get Your Personalized Post-Recovery Plan →No login. No upsell. Built for the reader who’s been through something hard and wants a clear next step.
For the reader who is cleared to consider an FDA-approved GLP-1 again:
Ro Body — for insurance navigation and FDA-approved medications
Ro Body carries FDA-approved GLP-1s: Wegovy (pill and pen), Zepbound (pen and KwikPen), Foundayo, Mounjaro, and Ozempic. Ro includes a dedicated insurance concierge that handles prior-authorization paperwork and a free GLP-1 Insurance Coverage Checker. Membership is $39 for the first month, then $149/month ongoing — or as low as $74/month with the annual plan paid upfront (verify current pricing before signing up). Note: Ro does not lead in compounded GLP-1s — this is specifically for FDA-approved branded medication paths.
Ro does not coordinate GLP-1 coverage for government plans (Medicare, Medicaid, TRICARE). Exception: FEHB members.
Check Insurance Coverage with Ro →Affiliate link. WPG earns a commission. Pricing verified April 2026.
Eden — for FDA-approved brand-name GLP-1s without a membership fee
Eden carries branded Wegovy and Zepbound, flags them as HSA/FSA eligible, charges no membership fee, and prices branded medications consistently across doses. For a reader who doesn’t need an insurance concierge — say, you’re paying cash or using HSA/FSA funds — Eden pairs FDA-approved medication with the simplest cash-pay structure.
See Eden’s FDA-Approved GLP-1 Options →Affiliate link. WPG earns a commission.
14. Frequently Asked Questions
What We Actually Verified for This Page
Last verified: .
We pulled the current FDA prescribing information for Wegovy (injection and 25 mg oral tablet), Ozempic, Rybelsus, Mounjaro, Zepbound, Saxenda, Trulicity, and Byetta from accessdata.fda.gov and DailyMed. We confirmed the Foundayo (orforglipron) FDA approval date (April 1, 2026) and class-warning status. We cross-referenced clinical-trial pancreatitis rates against current label data for LEADER, SCALE, STEP (semaglutide), and SURMOUNT-1 (tirzepatide) trials. We pulled FAERS pharmacovigilance reporting odds for compounded versus brand-name GLP-1 products from the 2024 Daoud et al. analysis. We confirmed NIDDK and Mayo Clinic acute pancreatitis symptom criteria match the Revised Atlanta classification used in U.S. emergency medicine. We checked the UK MHRA’s January 2026 strengthened pancreatitis warning communication. We reviewed the September 9, 2025 FDA warning letter to Eli Lilly regarding Zepbound and Mounjaro pancreatitis-risk minimization.
What we did not do: We did not generate this page from a single AI prompt. We did not fabricate any clinical credentials. We are not your medical provider. This page is informational; it is not a diagnosis or a substitute for emergency care. We do not claim a clinician medically reviewed this page because we don’t have a real clinician on the byline — and we’d rather be honest about that than fake it.
Conflict of interest disclosure: Weight Loss Provider Guide earns commissions from some GLP-1 providers. The provider links on this page are limited to two affiliates (Ro and Eden), placed only in the post-recovery section, after a clear disclosure, and routed only to FDA-approved branded medication paths. The medical and safety guidance does not change based on affiliate relationships.
Reverification cadence: Quarterly, plus immediate updates on any FDA label revision, new GLP-1 approval, or new safety communication. Next scheduled review: July 2026.
Sources
- —FDA prescribing information (current as of April 2026): Wegovy, Ozempic, Mounjaro, Zepbound, Rybelsus, Saxenda, Trulicity, Byetta, Foundayo. accessdata.fda.gov and DailyMed.nlm.nih.gov.
- —Cleveland Clinic Journal of Medicine. "Glucagon-like peptide-1 receptor agonists and pancreatitis: A reconcilable divorce." August 2025.
- —NIDDK. Symptoms & Causes of Pancreatitis; Diagnosis of Pancreatitis. niddk.nih.gov.
- —Mayo Clinic. Pancreatitis — Symptoms and causes. mayoclinic.org.
- —Revised Atlanta classification of acute pancreatitis (Banks et al.).
- —Liu et al. "Association between different GLP-1 receptor agonists and acute pancreatitis: case series and real-world pharmacovigilance analysis." PMC11600108. 2024.
- —Daoud et al. "Safety analysis of compounded GLP-1 receptor agonists: a pharmacovigilance study using the FDA adverse event reporting system." PubMed 40285721. 2024.
- —Lomeli et al. "The incidence of acute pancreatitis with GLP-1 receptor agonist therapy in individuals with a known history of pancreatitis." Diabetes Research and Clinical Practice. 2024.
- —Postlethwaite et al. Risk-factor study, GLP-1 receptor agonist initiation in obesity. 2022.
- —LEADER trial (liraglutide); SCALE trial (liraglutide weight reduction); STEP trials (semaglutide); SURMOUNT-1 (tirzepatide).
- —MHRA. "GLP-1 receptor agonists and dual GLP-1/GIP receptor agonists: strengthened warnings on acute pancreatitis, including necrotising and fatal cases." January 2026. gov.uk/drug-safety-update.
- —FDA Warning Letter to Eli Lilly. September 9, 2025.
- —FDA. "Concerns with Unapproved GLP-1 Drugs Used for Weight Loss." fda.gov.
- —Patient-voice quotes: r/Mounjaro Reddit posts, used for language and emotional context only.
Weight Loss Provider Guide is an independent comparison resource for GLP-1 telehealth providers. This page is for education and triage support; it does not diagnose pancreatitis or replace urgent medical care. If your symptoms match the warning signs in Section 1, stop reading and go to the emergency room.
Last verified: · Quarterly re-verification cadence